A Case Control Study of Gene Environment Interaction in Pre-Eclampsia with Special Reference to Organochlorine Pesticides
Article Information
Saloni Kamboj1*, Kiran Guleria2, Tusha Sharma3, Richa Sharma4, Bannerjee BD 5
1Senior Resident, Department of Obstetrics & Gynecology, UCMS >BH, Delhi, India
2Director Professor, Department of Obstetrics & Gynecology, UCMS >BH, Delhi, India
3Research Associate, Department of Biochemistry, UCMS >BH, Delhi, India
4Associate Professor, Department of Obstetrics & Gynecology, UCMS >BH, Delhi, India
5Director Professor, Department of Biochemistry, UCMS >BH, Delhi, India
*Corresponding Author: Saloni Kamboj, Senior Resident, Department of Obstetrics & Gynecology, UCMS >BH, Dilshad Garden, Delhi - 110095, India
Received: 04 July 2020; Accepted: 13 July 2020; Published: 16 July 2020
Citation:
Saloni Kamboj, Kiran Guleria, Tusha Sharma, Richa Sharma, Bannerjee BD. A Case Control Study of Gene Environment Interaction in Pre-Eclampsia with Special Reference to Organochlorine Pesticides. Obstetrics and Gynecology Research 3 (2020): 161-171.
View / Download Pdf Share at FacebookAbstract
Objectives: To study and compare the frequency of GSTM1 & T1 gene polymorphism in cases (Pre-eclampsia) and controls (normal pregnancy), to estimate and compare serum levels of Glutathione-S-Transferase enzyme in cases and controls, to quantify and compare Organochlorine pesticides levels in maternal blood of cases and controls and to evaluate the correlation; if any; amongst GSTM1 & T1 gene polymorphism, GST activity and Organochlorine pesticides levels in cases.
Methodology: Maternal blood samples of 66 Pre-eclampsia cases and equal number of matched control were collected. Samples were analyzed for OCPs levels, GST activity and GST M1 and T1 gene polymorphism.
Results: β-HCH, Heptachlor, Dieldrin, Endosulphan-I levels were found in higher amounts in cases as compared to controls. GSTT1-/GSTM1- (double null polymorphism) was significantly high in cases as compared to controls. GSTT1-(null) genotype was exclusively associated with severe pre-eclampsia. When GST activity was correlated with genotypic variation of GSTT1/M1 gene, null genotypes significantly downregulated GST activity, lowest being with double null genotype.
Conclusions: There is a possible role of gene-environment interaction in the pathogenesis of pre-eclampsia. An interaction of increased levels of pesticides with GST polymorphisms (null type) causes low levels of GST enzyme levels (an anti-oxidant) resulting in increased oxidative stress causing pre-eclampsia.
Keywords
Pre-eclampsia, Organochlorine, Pesticides, Hypertension, Polymorphism, Glutathione-S-Transferases, Oxidative Stress
Pre-eclampsia articles Pre-eclampsia Research articles Pre-eclampsia review articles Pre-eclampsia PubMed articles Pre-eclampsia PubMed Central articles Pre-eclampsia 2023 articles Pre-eclampsia 2024 articles Pre-eclampsia Scopus articles Pre-eclampsia impact factor journals Pre-eclampsia Scopus journals Pre-eclampsia PubMed journals Pre-eclampsia medical journals Pre-eclampsia free journals Pre-eclampsia best journals Pre-eclampsia top journals Pre-eclampsia free medical journals Pre-eclampsia famous journals Pre-eclampsia Google Scholar indexed journals Organochlorine articles Organochlorine Research articles Organochlorine review articles Organochlorine PubMed articles Organochlorine PubMed Central articles Organochlorine 2023 articles Organochlorine 2024 articles Organochlorine Scopus articles Organochlorine impact factor journals Organochlorine Scopus journals Organochlorine PubMed journals Organochlorine medical journals Organochlorine free journals Organochlorine best journals Organochlorine top journals Organochlorine free medical journals Organochlorine famous journals Organochlorine Google Scholar indexed journals Pesticides articles Pesticides Research articles Pesticides review articles Pesticides PubMed articles Pesticides PubMed Central articles Pesticides 2023 articles Pesticides 2024 articles Pesticides Scopus articles Pesticides impact factor journals Pesticides Scopus journals Pesticides PubMed journals Pesticides medical journals Pesticides free journals Pesticides best journals Pesticides top journals Pesticides free medical journals Pesticides famous journals Pesticides Google Scholar indexed journals Hypertension articles Hypertension Research articles Hypertension review articles Hypertension PubMed articles Hypertension PubMed Central articles Hypertension 2023 articles Hypertension 2024 articles Hypertension Scopus articles Hypertension impact factor journals Hypertension Scopus journals Hypertension PubMed journals Hypertension medical journals Hypertension free journals Hypertension best journals Hypertension top journals Hypertension free medical journals Hypertension famous journals Hypertension Google Scholar indexed journals Polymorphism articles Polymorphism Research articles Polymorphism review articles Polymorphism PubMed articles Polymorphism PubMed Central articles Polymorphism 2023 articles Polymorphism 2024 articles Polymorphism Scopus articles Polymorphism impact factor journals Polymorphism Scopus journals Polymorphism PubMed journals Polymorphism medical journals Polymorphism free journals Polymorphism best journals Polymorphism top journals Polymorphism free medical journals Polymorphism famous journals Polymorphism Google Scholar indexed journals Glutathione-S-Transferases articles Glutathione-S-Transferases Research articles Glutathione-S-Transferases review articles Glutathione-S-Transferases PubMed articles Glutathione-S-Transferases PubMed Central articles Glutathione-S-Transferases 2023 articles Glutathione-S-Transferases 2024 articles Glutathione-S-Transferases Scopus articles Glutathione-S-Transferases impact factor journals Glutathione-S-Transferases Scopus journals Glutathione-S-Transferases PubMed journals Glutathione-S-Transferases medical journals Glutathione-S-Transferases free journals Glutathione-S-Transferases best journals Glutathione-S-Transferases top journals Glutathione-S-Transferases free medical journals Glutathione-S-Transferases famous journals Glutathione-S-Transferases Google Scholar indexed journals Oxidative Stress articles Oxidative Stress Research articles Oxidative Stress review articles Oxidative Stress PubMed articles Oxidative Stress PubMed Central articles Oxidative Stress 2023 articles Oxidative Stress 2024 articles Oxidative Stress Scopus articles Oxidative Stress impact factor journals Oxidative Stress Scopus journals Oxidative Stress PubMed journals Oxidative Stress medical journals Oxidative Stress free journals Oxidative Stress best journals Oxidative Stress top journals Oxidative Stress free medical journals Oxidative Stress famous journals Oxidative Stress Google Scholar indexed journals pregnancies articles pregnancies Research articles pregnancies review articles pregnancies PubMed articles pregnancies PubMed Central articles pregnancies 2023 articles pregnancies 2024 articles pregnancies Scopus articles pregnancies impact factor journals pregnancies Scopus journals pregnancies PubMed journals pregnancies medical journals pregnancies free journals pregnancies best journals pregnancies top journals pregnancies free medical journals pregnancies famous journals pregnancies Google Scholar indexed journals DNA articles DNA Research articles DNA review articles DNA PubMed articles DNA PubMed Central articles DNA 2023 articles DNA 2024 articles DNA Scopus articles DNA impact factor journals DNA Scopus journals DNA PubMed journals DNA medical journals DNA free journals DNA best journals DNA top journals DNA free medical journals DNA famous journals DNA Google Scholar indexed journals polymorphism articles polymorphism Research articles polymorphism review articles polymorphism PubMed articles polymorphism PubMed Central articles polymorphism 2023 articles polymorphism 2024 articles polymorphism Scopus articles polymorphism impact factor journals polymorphism Scopus journals polymorphism PubMed journals polymorphism medical journals polymorphism free journals polymorphism best journals polymorphism top journals polymorphism free medical journals polymorphism famous journals polymorphism Google Scholar indexed journals
Article Details
1. Introduction
Pre-eclampsia is a multi-system disorder characterized by hypertension (blood pressure of ≥ 140/ 90 mm Hg) and proteinuria (≥ 300 mg/day) [1, 2]. Hypertensive disorders complicate 5-10% of pregnancies [3]. Recent studies have investigated the role of genetic susceptibility and gene-environment interactions in pre-eclampsia [4]. However, despite the strong correlation between oxidative stress and pre-eclampsia, only a handful of genes have been investigated. A family of enzymes, Glutathione-S-Transferases, play a critical role in providing protection against electrophiles and products of oxidative stress [5]. Genetic polymorphisms in genes encoding GST have been defined [6, 7]. Association of hypertension in general with pesticides has been studied a lot but association with pre-eclampsia has recently received some attention.
However, despite being universal exposure of antenatal population to organochlorine pesticides, only some women are susceptible to adverse effects of pesticides as a result of certain gene polymorphisms (GSTM1 & T1) which lead to decreased excretion and thus accumulation of greater amounts of pesticides in their systems; thus manifesting adverse effects. This is how the gene environment interaction comes into play in pathogenesis of pre-eclampsia. The present study aimed at estimating and comparing the frequency of GSTM1 & T1 gene polymorphism, serum levels of Glutathione-S-Transferase enzyme and organochlorine pesticides levels in cases (pre-eclampsia) and controls (normal pregnancy), and to evaluate the correlation; if any.
2. Materials and Methods
This was a hospital based case control study in which 66 pre-eclampsia cases (according to ACOG Criteria [8] Blood pressure of 140 mmHg systolic or higher or 90 mmHg diastolic or higher that occurs after 20 weeks of gestation on two occasions 6 hours apart in a woman with previously normal blood pressure & Proteinuria, defined as urinary excretion of 0.3 g protein or higher in 24 hour urinary specimen or ≥ 1+ dipstick) were recruited. For each case, a gestational age, parity and BMI matched control was recruited. Females with Age <18 years or >35 year, women with occupational exposure to pesticides, Chronic hypertension, thyroid disorders, chronic liver disease, heart disease, Pre-existing or gestational diabetes mellitus, Chronic renal disease, Thrombophilias –familial or acquired, Systemic lupus erythematosus, Hydramnios, pregnancies complicated by foetal abnormalities and women in active labor were excluded.
A questionnaire survey of the women was done to collect general demographical information in order to define the inclusion/exclusion criteria. Women confirmed their participation by signing a consent form and this study was approved by the Institutional Ethical Committee for Human Research. Five ml of maternal blood was drawn from each recruited subject. Out of 5 ml, 3 ml of blood was collected in an EDTA vial & 2 ml was collected in plain vial. Two ml blood in EDTA vial was used for DNA extraction for polymorphism study. Two ml of blood in plain vial was used to study the GST activity in serum and one ml of blood in EDTA vial was used for organochlorine pesticides residues analysis.
2.1 Estimation of genetic polymorphism
DNA extraction was done using blood sample, deionized water, centrifugation & extracted DNA was used for PCR analysis. A single assay using a multiplex PCR was performed for simultaneous gene amplification. 50 ng of DNA was amplified in a 50-µl multiplex reaction mixture containing 30pmol of each of the following GSTM1 primersin a medium consisting of 1.5 mM MgCl2, 200 µmol dNTPs, 5µl 10X PCR buffer (10X 500 mM KCl), 100 mM Tris-HCl, (pH 9.0) and 2 U TaqDNA polymerase. The PCR protocol included an initial melting temperature of 94°C for 5 minutes followed by 35 cycles of amplification (2 min at 94°C,1 min at 59°C, and extension for 1 min at 72°C). A final 10 min extension step at 72°C terminated the process. The final PCR products from the co- amplification of GSTM1(215 bp) and GSTT1(480 bp) were visualized on an Etidium Bromide- stained 2.0% agarose gel to look for gene polymorphism present / absent.
2.2 Gst activity levels
The estimation of GST activity is based on principle that GST catalyzes the conjugation of L-glutathione to CDNB through the thiol group of the glutathione .The reaction product, GS-DNB Conjugate, absorbs at 340 nm. The rate of increase in the absorption is directly proportional to the GST activity in the sample. The linearity of the reaction was determined by plotting the absorbance values against time. The change in absorbance (DA340)/minute, in the linear range of the
plot, for the sample and for the blank was calculated by subtracting A340 (initial reading) from A340 (final reading) and dividing the result by reaction time (min.). Equation used:
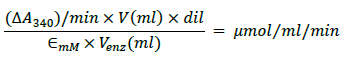
Where
V = the reaction volume (for test in 96-well plate = 0.2 ml)
dil = the dilution factor of the original sample
Venz= volume of the enzyme sample tested
∈ mM = extinction coefficient for CDNB
Conjugate at 340 nm = 5.3 mM-1 (path length - 0.552 cm)
2.3 Organochlorine pesticide levels
OCP residue extraction was done using hexane and acetone (2:1) according to the method of Bush et al. Clean-up was done by the USEPA method using Florisil (Sigma) by column chromatography. Quantification of organochlorine residue levels was done by a Perkin Elmer Gas chromatography system equipped with 63Ni selective electron capture detector. Limits of detection were <0.05 pg perchloroethylene with nitrogen. Quantitative analysis of OCP residues in each sample was done by comparing the peak heights with those obtained from a chromatogram of a mixed organochlorines standard of known concentration. Ten samples of each maternal and cord blood in triplicate were spiked with a mixed standard of organochlorine pesticides. Furthermore, a quality check sample was always run with each set of samples for pesticides analysis.
2.4 Statistical analysis
Data was analyzed by SPSS Software (version 20.0), binary logistic regression was performed to compare genotypic variation. Pearson’s coefficient of correlation was used to test correlation between levels of GST and pesticides. To correlate if high levels of organochlorine pesticides are associated with gene polymorphism and rise in blood pressure (SBP/DBP) in pre-eclampsia, multiple linear regression analysis was employed. P-value of <0.05 was considered significant.
3. Results
Six thousand three hundred women were screened during the study period from labor wards of GTB hospital. Out of these, 550 pregnancies were complicated with pre-eclampsia. The incidence of pre-eclampsia in the present study was 8.73%. Out of recruited cases, 21% were severe pre-eclampsia and 78% were pre-eclampsia without severe features. While estimating genetic polymorphism, women with double null genotype of GSTT1 & M1 were found to be at six times greater risk of development of pre-eclampsia (Table 1). While comparing the distribution of genetic polymorphism between non-severe & severe pre-eclampsia groups, GSTT1 null type was significantly associated with severe pre-eclampsia inferring thereby that GSTT1 null genotype may indicate the severity of disease in an individual. In other words, women with single null genotype of GSTT1 have eighteen times greater risk of development of pre-eclampsia (Table 2). GST acts as an antioxidant in body quenching all ROS produced in the body. There was no significant difference in GST values between cases and controls & between two subgroups of pre-eclampsia.
However when GST activity was correlated with genotypic variation of GSTT1/M1 gene, null genotypes down-regulated GST activity significantly, lowest activity observed with double null genotype (Figure 1) Significantly higher levels of β-HCH, Heptachlor, Dieldrin and Endosulphan were found in maternal blood of pre-eclampsia cases as compared to controls (Table 3). Significantly low levels of GST activity were associated with four pesticides (β- HCH, Endosulphan-I & II and Heptachlor); so pesticides were found to have an association with decreased antioxidant levels, hence increased oxidative stress. The interaction of GSTM1 genotype (null) and increased levels of DDT in blood resulted in significant increase in DBP levels (Table 4). The interaction of GSTT1 genotype (null) and high levels of β-HCH and heptachlor in blood also resulted in significant increase in DBP level (Table 4). The interaction of GSTM1 genotype (null) and increased levels of β-HCH in blood resulted in significant increase of in SBP levels (Table 5). Similarly the interaction of GSTT1 (null) genotype and increased levels of β-HCH in blood resulted in significant increase in SBP level (Table 5).
|
Genotypes |
Cases (n=66) |
Controls (n=66) |
OR |
95% CI |
p-value |
|
GSTT1+ / GSTM1+ |
14 |
29 |
|||
|
GSTT1+ / GSTM1- |
11 |
14 |
1.62 |
0.590-4.492 |
0.347 |
|
GSTT1- / GSTM1+ |
12 |
13 |
1.91 |
0.696-5.256 |
0.209 |
|
GSTT1- / GSTM1- |
29 |
10 |
6.00 |
2.298-15.704 |
0.000 |
Table 1: Genotypic distribution of Glutathione-S-Transferase (GSTT1/GSTM1) genotypes in study groups.
|
Genotypes |
Pre-eclampsia without severe features (n=52) |
Severe pre-eclampsia (n=14) |
OR |
95% CI |
p-value |
|
GSTT1+ / GSTM1+ |
13 |
1 |
|||
|
GSTT1+ / GSTM1- |
11 |
0 |
0.00 |
0.00- not det |
0.99 |
|
GSTT1- / GSTM1+ |
5 |
7 |
18.20 |
1.76-188.06 |
0.015 |
|
GSTT1- / GSTM1- |
23 |
6 |
3.39 |
0.36-31.36 |
0.282 |
Table 2: Correlation of genotypic distribution of GST with severity of pre-eclampsia.
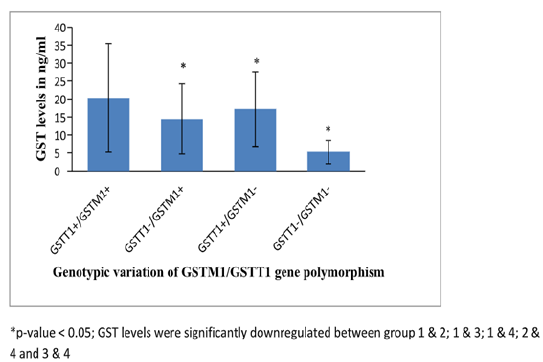
Figure 1: GST activity and correlation with GST gene polymorphism.
|
Organochlorine pesticides (ppb) |
Cases n=66 (mean ± SD) |
Controls n=66 (mean ± SD) |
p-value |
|
α HCH |
2.02 ± 2.02 |
1.63 ± 1.81 |
0.251 |
|
β HCH |
6.32 ± 2.23 |
4.79 ± 2.41 |
0.000 |
|
γ HCH |
1.98 ± 1.66 |
1.84 ± 1.46 |
0.612 |
|
Heptachlor |
3.93 ± 2.36 |
2.61 ± 1.54 |
0.000 |
|
Aldrin |
1.60 ± 1.57 |
1.58 ± 1.43 |
0.956 |
|
Endo-I |
3.08 ± 2.82 |
1.27 ± 1.87 |
0.000 |
|
Dieldrin |
3.07 ± 2.18 |
1.49 ± 1.64 |
0.000 |
|
DDE |
3.13 ± 2.19 |
3.65 ± 3.35 |
0.294 |
|
Endo-II |
1.21 ± 1.38 |
1.46 ± 3.01 |
0.533 |
|
DDT |
0.64 ± 0.99 |
0.41 ± 0.81 |
0.146 |
*p value≤0.05 has been considered as significant.
Table 3: Estimation & comparison of maternal blood organochlorine pesticide levels in study groups.
Significantly higher levels of β-HCH (6.32 ± 2.23 ppb, p=0.00), Heptachlor (3.93 ± 2.36 ppb, p=0.000), Dieldrin (3.07 ± 2.18 ppb, p=0.000), Endosulphan I (3.08 ± 2.82 ppb, p=0.000) were found in maternal blood of pregnancies with pre-eclampsia as compared to controls.
|
GSTM1/GSTT1 interaction |
Blood sample of study subjects (GSTM1) |
Blood sample of study subjects (GSTT1) |
||
|
B-value |
p-value |
B-value |
p-value |
|
|
GSTM1/GSTT1 (null vs present type) |
-3.692 |
0.432 |
-4.257 |
0.373 |
|
α-HCH |
0.419 |
0.329 |
0.391 |
0.359 |
|
Interaction term of GSTM1/GSTT1-α-HCH levels |
-0.737 |
0.248 |
-0.599 |
0.354 |
|
GSTM1/GSTT1 (null vs present type) |
0.044 |
0.983 |
24.871 |
0.001 |
|
β-HCH |
2.343 |
0.062 |
3.127 |
0.001 |
|
Interaction term of GSTM1/GSTT1-β-HCH levels |
-4.339 |
0.025 |
-5.875 |
0.001 |
|
GSTM1/GSTT1 (null vs present type) |
1.159 |
0.784 |
1.889 |
0.661 |
|
ϒ-HCH |
1.035 |
0.101 |
0.753 |
0.214 |
|
Interaction term of GSTM1/GSTT1-ϒ-HCH levels |
-2.172 |
0.011 |
-2.331 |
0.008 |
|
GSTM1/GSTT1 (null vs present type) |
0.197 |
0.963 |
-1.644 |
0.697 |
|
Aldrin |
1.497 |
0.052 |
1.627 |
0.040 |
|
Interaction term of GSTT1/GSTM1-Aldrin levels |
-2.978 |
0.046 |
-2.276 |
0.101 |
|
GSTM1/GSTT1 (null vs present type) |
2.451 |
0.575 |
3.173 |
0.468 |
|
Dieldrin |
1.794 |
0.221 |
1.644 |
0.258 |
|
Interaction term of GSTM1/GSTT1-dieldrin levels |
-5.648 |
0.006 |
-5.868 |
0.005 |
|
GSTM1/GSTT1 (null vs present type) |
0.034 |
0.993 |
0.877 |
0.822 |
|
Endosulfan-I |
2.333 |
0.060 |
2.527 |
0.040 |
|
Interaction term of GSTM1/GSTT1-Endosulphan-I levels |
-4.039 |
0.015 |
-4.311 |
0.010 |
|
GSTM1/GSTT1 (null vs present type) |
-1.644 |
0.697 |
0.197 |
0.963 |
|
Endosulfan-II |
1.627 |
0.040 |
1.497 |
0.052 |
|
Interaction term of GSTM1/GSTT1-Endosulphan-II levels |
-2.276 |
0.101 |
-2.978 |
0.046 |
|
GSTM1/GSTT1 (null vs present type) |
12.398 |
0.001 |
1.107 |
0.810 |
|
pp-DDT |
8.833 |
0.001 |
0.711 |
0.131 |
|
Interaction term of GSTM1/GSTT1-ppDDT levels |
-10.187 |
0.001 |
-1.581 |
0.025 |
|
GSTM1/GSTT1 (null vs present type) |
4.623 |
0.418 |
5.376 |
0.321 |
|
PpDDE |
5.991 |
0.005 |
6.256 |
0.002 |
|
Interaction term of GSTM1/GSTT1-ppDDE levels |
-5.367 |
0.033 |
-5.677 |
0.020 |
|
GSTM1/GSTT1 (null vs present type) |
0.735 |
0.871 |
12.433 |
0.001 |
|
Heptachlor |
0.822 |
0.085 |
8.869 |
0.001 |
|
Interaction term of GSTM1/GSTT1-Heptachlor levels |
-1.516 |
0.026 |
-10.248 |
0.001 |
*p value <0.005 is significant; B=regression coefficient
Table 4: Regression model testing an interactive effect of OCPs (ppb) and GSTM1/GSTT1 genotype on diastolic BP (mmHg) in study subjects.
|
GSTM1/GSTT1 interaction |
Blood sample of study subjects (GSTM1) |
Blood sample of study subjects (GSTT1) |
||
|
B-value |
p-value |
B-value |
p-value |
|
|
GSTM1/GSTT1 (null vs present type) |
-4.383 |
0.427 |
2.306 |
0.349 |
|
α-HCH |
-0.388 |
0.516 |
2.421 |
0.537 |
|
Interaction term of GSTM1/GSTT1-α-HCH levels |
0.772 |
0.304 |
-0.456 |
0.364 |
|
GSTM1/GSTT1 (null vs present type) |
11.398 |
0.001 |
15.536 |
0.001 |
|
β-HCH |
8.833 |
0.001 |
5.334 |
0.007 |
|
Interaction term of GSTM1/GSTT1-β-HCH levels |
-10.286 |
0.001 |
-9.769 |
0.001 |
|
GSTM1/GSTT1 (null vs present type) |
-5.253 |
0.274 |
1.371 |
0.654 |
|
ϒ-HCH |
-0.744 |
0.343 |
0.325 |
0.919 |
|
Interaction term of GSTM/GSTT1-ϒ-HCH levels |
1.391 |
0.158 |
-0.271 |
0.665 |
|
GSTM1/GSTT1 (null vs present type) |
-6.532 |
0.250 |
19.501 |
0.008 |
|
Aldrin |
-3.580 |
0.110 |
8.046 |
0.018 |
|
Interaction term of GSTT1/GSTT1-Aldrin levels |
3.748 |
0.164 |
-4.187 |
0.005 |
|
GSTM1/GSTT1 (null vs present type) |
-1.221 |
0.767 |
2.349 |
0.685 |
|
Dieldrin |
0.861 |
0.555 |
-0.756 |
0.774 |
|
Interaction term of GSTM1/GSTT1-dieldrin levels |
0.984 |
0.587 |
-0.163 |
0.890 |
|
GSTM1/GSTT1 (null vs present type) |
-6.367 |
0.132 |
4.980 |
0.309 |
|
Endosulfan-I |
0.131 |
0.902 |
1.012 |
0.738 |
|
Interaction term of GSTM1/GSTT1-Endosulphan-I levels |
2.679 |
0.047 |
-0.640 |
0.509 |
|
GSTM1/GSTT1 (null vs present type) |
6.402 |
0.133 |
3.799 |
0.198 |
|
Endosulfan-II |
5.107 |
0.023 |
1.556 |
0.663 |
|
Interaction term of GSTM1/GSTT1-Endosulphan-II levels |
-3.616 |
0.126 |
-0.384 |
0.522 |
|
GSTM1/GSTT1 (null vs present type) |
-3.719 |
0.456 |
-4.017 |
0.571 |
|
pp-DDT |
2.814 |
0.155 |
-4.715 |
0.116 |
|
Interaction term of GSTM1/GSTT1-ppDDT levels |
2.455 |
0.303 |
1.823 |
0.221 |
|
GSTM1/GSTT1 (null vs present type) |
0.961 |
0.654 |
4.054 |
0.099 |
|
PpDDE |
0.986 |
0.591 |
3.169 |
0.321 |
|
Interaction term of GSTM1/GSTT1-ppDDE levels |
-6.467 |
0.189 |
-0.700 |
0.166 |
|
GSTM1/GSTT1 (null vs present type) |
-9.974 |
0.110 |
3.044 |
0.099 |
|
Heptachlor |
0.431 |
0.654 |
3.176 |
0.344 |
|
Interaction term of GSTM1/GSTT1-Heptachlor levels |
1.992 |
0.069 |
-0.699 |
0.167 |
*p value <0.005 is significant; B=regression coefficient
Table 5: Regression model testing an interactive effect of OCPs (ppb) and GSTM1/GSTT1 genotype on systolic BP (mmHg) in study subjects.
4. Discussion
Pre-eclampsia is a pregnancy related disease with an unpredictable course that can have serious consequences for the mother and the fetus. It is very essential to analyze the etiological factors and carry out further research in targeted interventions to prevent pre-eclampsia. In this prospective case-control study we made an effort to find an association among genetic polymorphism in GST gene (genetic factors), GST enzyme activity and organochlorine pesticides (environmental insult) in pre-eclampsia and the possible mechanisms underlying this association. The incidence of pre-eclampsia in the present study was 8.73%, which is consistent with those reported from most of the studies in developing countries. One of the most important systems involved in metabolism and detoxification of ROS, xenobiotics and carcinogens is that of Glutathione-S-Transferase (GST) [5]. Null genotypes of GSTT1 and GSTM1 have been linked with an increased risk of several multifactorial traits including cancers and cardiovascular diseases [9]. In present study, the frequency of GSTM1/GSTT1 genotypic variation was studied and it was found that GSTM1-/T1- (both null deletion) genotype was significantly higher in subjects with pre-eclampsia as compared to controls. This is in concordance with a previous study by Sahar S et al. [10] where GSTM1- / GSTT1- (both null type) genotype was a potential genetic factor to predict development of essential hypertension and permit early therapeutic interventions. Human beings are constantly being exposed to variety of environmental contaminants like organochlorine pesticides. The problem of pesticide residue is very high in India due to poor safety measures during use and implementation despite low average consumption of pesticide than many other developed countries. The result in our study has also shown higher levels of various pesticides residues in blood samples of women with pre-eclampsia. The findings of the present study are in agreement with previous studies, where higher levels of OCPs in pregnant women may cause adverse reproductive outcomes as these pesticides can act as xenoestrogens as well as create oxidative stress [11-13]. Women are exposed to a variety of reproductive toxins, but not all who are exposed have adverse reproductive outcomes. It is speculated that an individual’s reproductive risk associated with these toxins may be modified by genetic variation in metabolic detoxification activity. Mustafa et al. [14] in 2013 reported that when GSTM1 genotype was absent (null), increasing levels of β-HCH increased the risk of preterm delivery significantly. Ours study is probably the first human study where attempts have been made to show that increase in pesticide levels in body result in pre-eclampsia in the presence of double null GST genotype due to decrease in antioxidant levels. Pre-eclampsia is a significant health problem both in developed and in developing world despite enhanced standard of antenatal care. Newer etiologies like environmental factors (pesticides) and specially gene-environment interactions need to be established to set appropriate preventive strategies in order to bring incidence and mortality under control. The results of our study reconfirmed that there is a possible role of gene-environment interaction in the pathogenesis of pre-eclampsia. An interaction of increased levels of pesticides with GST polymorphisms (null type) caused low levels of GST enzyme levels (an anti-oxidant) resulting in increased oxidative stress, thus causing pre-eclampsia. Thus we can conclude from the present study that continuous indirect exposure of pesticides in general population are detrimental to health and may result in adverse reproductive outcome like pre-eclampsia. Our study has proven the hypothesis that environmental factors (pesticides) interact at genetic level (gene polymorphism) to ultimately cause pre-eclampsia by altering the oxidative stress pathway. We recommend further such studies on a larger population to prove gene environment interactions causing pre-eclampsia. We notice that even after being banned by various organizations, use of these pesticides is still prevalent as shown by increased blood levels. Therefore, use of such harmful chemicals should be banned with immediate effect. Food safety and standards authority of India should implement stringent regulations to keep a check on the levels of pesticides found in food samples. Pregnant population in India should be encouraged to consume more antioxidants (natural & vitamins) to combat the oxidative damage caused by environmental exposure.
Author Contributions
S.K. – Patient recruitment and did the study
K.G. - Conceptualised the study and data analysis
T.S. – Biochemical Analysis
R.S. – Helped in literature search
B.B. – Provided inputs in methodology
Declaration of Interests
None
Funding
This study was funded by an intra-mural grant from University College of Medical Sciences, Delhi, India.
References
- Sibai BM, Caritis S, Hauth J. National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. What we have learned about preeclampsia. SeminPerinatol 27 (2003): 239-246.
- Walker JJ. Pre-eclampsia. Lancet 356 (2000): 1260-1265.
- Wallis AB, Saftlas AF, Hsia J, et al. Secular trends in the rates of preeclampsia, eclampsia and gestational hypertension, United States 1987-2004. Am J Hypertens 21 (2008): 521-526.
- Cummings AM, Kavlock RJ. Gene-environment interactions: a review of effects on reproduction and development. Crit Rev Toxicol 34 (2004): 461-485.
- Hayes JD, Strange RC. Glutathione S-Transferase polymorphism and their biological consequences. Pharmacology 61 (2000): 154-166.
- Hayes JD, Pulford DJ. The glutathione-S-transferase supergene family: regulation of GST and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit Rev Biochem Mol Biol 30 (1995): 445-600.
- Beckett GJ, Hayes JD. Glutathione S Transferases: biomedical applications. Adv Clin Chem 30 (1993): 281-380.
- Gestational Hypertension and Preeclampsia. ACOG Practice Bulletin No. 202. Obstet Gynecol 133 (2019): e1-e25.
- Beckett GJ, Hayes JD. Glutathione S Transferases: biomedical applications. Adv Clin Chem 30 (1993): 281-380.
- Bessa SS, Ali EM, Hamdy SM. The role of glutathione S- transferase M1 and T1 gene polymorphisms and oxidative stress-related parameters in Egyptian patients with essential hypertension.Eur J Intern Med 20 (2009): 625-630.
- Sharma N, Guleria K, Banerjee BD, et al. Role of oxidative stress and anti-oxidant gene in pre-eclampsia with reference to organochlorine pesticides: a case-control study (Masters Thesis). Delhi: University of Delhi (2015).
- Saldana TM, Basso O, Baird DD, et al. Pesticide exposure and hypertensive disorders during pregnancy. Environ Health Perspect 117 (2009): 1393-1396
- Nugteren JJ, Snijder CA, Hofman A, et al. Work-Related Maternal Risk Factors and the Risk of Pregnancy Induced Hypertension and Pre-eclampsia during Pregnancy. The Generation R Study. PLoS ONE 7 (2012): e39263.
- Mustafa MD, Banerjee BD, Ahmed RS, et al.
- Gene environment interaction in preterm delivery with special reference to organochlorine pesticides. Mol Hum Reprod 19 (2013): 35-42.


 Impact Factor: * 3.2
Impact Factor: * 3.2 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 