Actinomyces Naeslundii and Aggregatibacter Aphrophilus Brain Abscess in an Adolescent
Article Information
Michael Croix1, Christopher Schwarz2, Ryan Breuer3,4, Amanda B. Hassinger3,4, Kunal Chadha5, Mark Daniel Hicar4,6
1Division of Internal Medicine and Pediatrics, University at Buffalo. Buffalo, New York, USA
2Division of Emergency Medicine, University at Buffalo. Buffalo, New York, USA
3Division of Pediatric Critical Care, John R. Oishei Children’s Hospital. Buffalo, New York, USA
4Department of Pediatrics, University at Buffalo. Buffalo, New York, USA
5Division of Pediatric Emergency Medicine, University at Buffalo. Buffalo, New York, USA
6Division of Pediatric Infectious Diseases, University at Buffalo. Buffalo, New York, USA
*Corresponding Authors: Dr. Mark Daniel Hicar, Department of Pediatrics, Jacobs School of Medicine and Biomedical Sciences, University at Buffalo, 1001 Main Street, Buffalo, NY, 14203 USA
Dr. Michael Croix, Division of Internal Medicine and Pediatrics, 300 Linwood Ave, Buffalo, NY, 14209 USA
Received: 20 July 2019; Accepted: 02 August 2019; Published: 04 November 2019
Citation: Michael Croix, Christopher Schwarz, Ryan Breuer, Amanda B. Hassinger, Kunal Chadha, Mark Daniel Hicar. Actinomyces Naeslundii and Aggregatibacter Aphrophilus Brain Abscess in an Adolescent. Archives of Clinical and Medical Case Reports 3 (2019): 409-413.
View / Download Pdf Share at FacebookAbstract
We report the case of a child with a brain abscess from which Actinomyces naeslundii and Aggregatibacter aphrophilus were isolated. The is the first case describing A. naeslundii causing a brain abscess. This case highlights the association of these two organisms which may affect antibiotic choice and therapy length.
Keywords
Brain; Abscess; Actinomyces; Aggregatibacter
Article Details
1. Case Report
A 13 year old male with no known past medical history initially presented to the Emergency Department with one week of headache, nausea, and vomiting. He had a three day long upper respiratory infection (URI) approximately one month prior to this visit that was treated with over-the-counter symptomatic relief medication. He denied fever or persistence of the respiratory symptoms in the interim. He was diagnosed with tension headaches and discharged home with ibuprofen for pain relief.
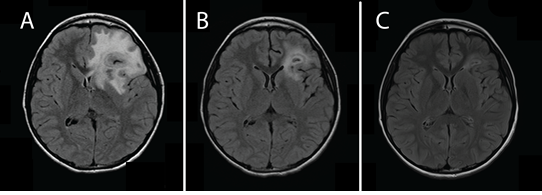
Two weeks later, he presented to his pediatrician’s office with persistent daily headaches, nausea, and vomiting, though notably still lacking fever. An outpatient computed tomography (CT) scan of the head without contrast revealed a 2.5 cm ring enhancing spherical mass with surrounding edema and midline shift concerning for malignancy or abscess. He was referred to the Emergency Department for further evaluation. Upon arrival, the patient remained afebrile and his physical exam, including a full neurological exam, remained unremarkable. Initial laboratory studies revealed a total white blood cell count of 8200/uL with 61% neutrophils, 25% lymphocytes, and 13% monocytes. The patient’s erythrocyte sedimentation rate was noted to be elevated (50 mm/hr), but his electrolytes, liver function studies, urinalysis, and C-reactive protein (7.73 mg/L) were within normal limits. Magnetic resonance imaging (MRI) of the brain confirmed the presence of a left frontal mass with features most consistent with an abscess (Figure 1A). He was administered Vancomycin, ceftriaxone, and metronidazole and admitted to the pediatric intensive care unit for observation.
Additional history revealed that the patient had emigrated to the United States from Yemen one year prior to presentation. There was no history of prior personal or familial tuberculosis exposure. Notably, a dental exam was performed approximately six months earlier and was unremarkable. Apart from his URI symptoms noted one month prior, he denied any known sick contacts, travel since emigration, dental issues, or recent head trauma.
On hospital day two, the neurosurgical service aspirated ten milliliters of purulent drainage from the abscess through a burr hole. The initial stains showed many white blood cells but were negative for organisms. Aerobic, anaerobic, fungal, and mycobacterial cultures were all performed. The patient had a negative response to purified protein derivative (PPD) skin testing and a chest X-ray that revealed no evidence of pulmonary disease. On hospital day four, a gram negative bacilli was isolated from the aspirate aerobic culture and vancomycin was discontinued. This culture was also noted to have a second slower growing organism. By hospital day seven his initial symptoms of headache, nausea, and vomiting had all resolved. Repeat MRI of the brain at the time demonstrated no new intracranial lesions, a decompressed (attributed to the procedure) multilobulated rim-enhancing lesion, and stable to slightly decreased midline shift. The patient was briefly noted to have premature ventricular contractions secondary to hypomagnesemia that resolved with magnesium supplementation. The patient was transferred out of intensive care on hospital day thirteen with further management on the pediatric ward.
The two organisms isolated were described as facultative anaerobes with one forming a white, dry colony that stained gram negative and the second forming a grey slow growing colony that stained gram positive. By hospital day eight, these were identified as Aggregatibacter aphrophilus and Actinomyces naeslundii respectively. Anaerobic, fungal, and acid fast bacteria cultures taken from the fluid aspirate all remained negative. During his hospitalization, cardiac echocardiography did not show any valvular defects and multiple blood cultures were negative. With lack of bacteremia, a CT of the sinuses was obtained to evaluate sinus disease as a possible source. The CT showed a small retention cyst in the right sphenoid sinus and bubbly debris in the left maxillary sinus, but no bony erosions or defects. The lack of relative proximity to the lesion made direct extension from the sinuses highly unlikely per multiple radiologist’s opinions. The patient underwent a full dental exam in the hospital that was unrevealing. He was discharged to home with a total six week course of IV ceftriaxone and oral metronidazole.
Outpatient follow up at the completion of the initial six weeks of antibiotics showed the patient continuing to do well without fever or recurrent symptoms. A repeat MRI showed a decrease in size of the left frontal lesion with decreased midline shift (Figure 1B). Based on a review of the pertinent literature, we chose to continue therapy specifically targeting the slow-growing Actinomyces naeslundii by using high-dose oral amoxicillin for a planned additional five months. A repeat of the brain MRI at four months from his initial presentation showed marked improvement with only a small area of rim enhancement (Figure 1C). He was doing well throughout the numerous follow-up visits and tolerating his amoxicillin without sequelae. We discontinued his oral amoxicillin after completion of the planned six months of outpatient antibiotic treatment.
Figure 1: Magnetic resonance imaging T2 flair sequence axial imaging. (A) The patient’s initial imaging upon presentation to the Emergency Department. (B) Repeat imaging following aspiration of abscess and initial six weeks of treatment. (C) Repeat imaging four months after initial treatment while solely on high dose amoxicillin for treatment of Actinomyces naeslundii.
Herein, we present the first case report of Actinomyces naeslundii identified in a brain abscess. Brain abscesses in children are rare. Among all age groups the incidence rates are between 0.4 to 0.9 cases per 100,000 population and are more common in immunosuppressed patients [1]. In children, direct extension of infection from the sinuses, middle ear, or teeth is the most common origin [2]. Hematogenous spread of microbes from distant infections (i.e. endocarditis, pulmonary infection) or in patients with underlying congenital heart disease may also lead to abscess formation. Polymicrobial infections, particularly from direct extension, are not uncommon. Brain abscess formation associated with sinusitis or dental abscess often grows Streptococcus viridans and anaerobic organisms, while those resulting from otitis media or mastoiditis commonly include Streptococci and gram negative organisms. Direct trauma and endocarditis are frequently associated with Staphyloccocal species.
In 2006, the former Actinobacillus actinomycetemcomitans, Hemophilus aphrophilus, and Haemophilus segnis were reclassified into the same genus, Aggregatibacter [3]. Aggregatibacter aphrophilus was first isolated in 1940 from a case of infective endocarditis. The organism has since been identified in several cases of periodontal disease, brain abscess, and other infections. Nearly 40% of invasive cases are associated with a preceding dental procedure [4]. Similar to our cultured microbe, reported susceptibilities include a number of antibiotics; including penicillins with and without beta-lactamase inhibitors, cephalosporins, carbapenems, and fluoroquinolones [5].
Actinomyces species cause a wide array of infections. They are slow-growing microaerophilic to anaerobic gram positive organisms often found in polymicrobial infections. They are most commonly identified as causing cervicofacial actinomycosis, but have been identified in respiratory infections, osteomyelitis, genitourinary infections, and digestive tract infections [6]. Brain abscess is the most common form of central nervous system (CNS) infection caused by Actinomyces. However, overall, CNS infections are a relatively rare presentation, and, like other organisms, tend to result from direct extension or hematogenous spread [6, 7]. The most commonly identified cause of CNS actinomycosis is Actinomyces israelii correlating with it being the most common cause of cervicofacial actinomycosis.
Actinomyces naeslundii is considered a commensal oral organism. Prior reports have identified it in dental abscesses, an infected knee following total knee arthroscopy, and a cause of bacteremia in a diabetic patient [8-10]. Generally, Actinomyces species are very susceptible to a broad range of antibiotics including penicillin G and amoxicillin. Third generation cephalosporins and carbapenems are likewise active and macrolides have been used as alternative treatment. Unlike Aggregatibacter aphrophilus, these organisms do not respond well to fluoroquinolones. An important consideration for treatment of actinomycosis is the slow growth of the organism. which necessitates a longer treatment period often for several months to prevent recurrence [7].
2. Conclusion
This is the first case report of a brain abscess containing Actinomyces naeslundii. Although there was initial surprise in also culturing Aggregatibacter aphrophilus, association between members of these two genuses has been previously described. Actinomyces species have been isolated in peridontitis with Aggregatibacter aphrophilus and Aggregatibacter actinomycetemcomitans, the latter notably named for it’s initial characterization as growing in lesions with actinomyces. Pairing of members of these two genera may even be able to survive a blood borne phase, as we saw no evidence of direct extension or trauma to explain the source of brain abscess in our case. We theorize an oral source was the cause of a polymicrobic bacteremia, likely related to his period of preceding URI.
Clinicians should remain mindful of the predilection of Actinomyces and Aggregatibacter species to cause polymicrobial infections as both choice of antimicrobial therapy and duration of therapy may be affected.
3. Sources of Support
No additional sources of support were provided for this article.
4. Disclosure Statement
The authors have no conflicts or perceived conflicts of interest to disclose.
References
- Brouwer MC, Tunkel AR, McKhann GM, et al. Brain Abscess. NEJM 371 (2014): 447-456.
- Saez-LLorens, Xavier. Brain abscess in children. Seminars in Pediatric Infectious Disease 14 (2018): 108-114.
- Nørskov-Lauritsen N, Kilian M. Reclassification of Actinobacillus actinomycetemcomitans, Haemophilus aphrophilus, Haemophilus paraphrophilus and Haemophilus segnis as Aggregatibacter actinomycetemcomitans gen. nov., comb. nov., Aggregatibacter aphrophilus comb. nov. and Aggregatibacter segnis comb. nov., and emended description of Aggregatibacter aphrophilus to include V factor-dependent and V factor-independent isolates. International Journal of Systematic and Evolutionary Microbiology 56 (2006): 2135-2146.
- Huang ST, Lee HC, Lee NY, et al. Clinical characteristics of invasive Haemophilus aphrophilus infections. J Microbiol Immunol Infect 38 (2005): 271-276.
- Coburn B, Toye B, Rawte P, et al. Antimicrobial Susceptibilities of Clinical Isolates of HACEK Organisms. Antimicrob. Agents Chemother 57 (2005): 1989-1991.
- Valour F, Sénéchal A, Dupieux C, et al. Actinomycosis: etiology, clinical features, diagnosis, treatment, and management. Infect Drug Resist 7 (2014): 183-197.
- Smego RA Jr. Actinomycosis of the Central Nervous System. Rev Infect Dis 9 (1987): 855-865.
- Coleman RM, Georg LK, Rozzell AR. Actinomyces naeslundii as an Agent of Human Actinomycosis. Appl. Environ. Microbiol 18 (1969): 420-426.
- Wüst J, Steiger U, Vuong H, et al. Infection of a hip prosthesis by Actinomyces naeslundii. J Clin Microbiol 38 (2000): 929-930.
- Bhandari K, Gonzales Z, Armando J. Actinomyces naeslundii Bacteremia in an Elderly Woman With Type 2 Diabetes Mellitus. Infectious Diseases in Clinical Practice 25 (2017): e23.


 Impact Factor: * 3.1
Impact Factor: * 3.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
