Acute Aortic Thrombosis Related to Adjuvant Capecitabine-Oxaliplatin in a Chinese Patient with Resected Adenocarcinoma of Sigmoid Colon
Article Information
Henry C. Y. Wong*, Fiona M. Y. Lim, Ashley C. K. Cheng
1Department of Oncology, Princess Margaret Hospital, Kwai Chung, Kowloon, Hong Kong
*Corresponding Author: Henry C. Y. Wong, 6th Floor, Block H, Department of Oncology, Princess Margaret Hospital, 2-10 Princess Margaret Hospital Road, Lai Chi Kok, Kowloon, Hong Kong
Received: 08 July 2019; Accepted: 22 July 2019; Published: 25 September 2019
Citation: Henry C. Y. Wong, Fiona M. Y. Lim, Ashley C. K. Cheng. Acute Aortic Thrombosis Related to Adjuvant Capecitabine-Oxaliplatin in a Chinese Patient with Resected Adenocarcinoma of Sigmoid Colon. Archives of Clinical and Medical Case Reports 3 (2019): 342-348.
View / Download Pdf Share at FacebookAbstract
Aortic thrombosis is a rare disease entity which has been reported to be related to malignancy and chemotherapeutic agents. Here we present the first case in literature about a colon cancer patient with well-controlled vascular risk factors who developed acute aortic thrombosis after the first cycle of adjuvant chemotherapy with capecitabine-oxaliplatin. Risk factors and causal relationship between systemic anti-cancer agents and this condition are reviewed and discussed.
Keywords
Aortic thrombosis; Arterial thromboembolism; Capecitabine; Oxaliplatin
Article Details
1. Introduction
Aortic thrombosis in the absence of atherosclerosis, dissection and aneurysm is extremely rare but potentially devastating due to embolic events to the brain, coronary vessels, intra-abdominal organs and extremities. Cancer therapeutic agents including chemotherapy and vascular endothelial growth factor inhibitors were reported as risk factors [1, 2]. We herein present a case to describe the relationship between acute aortic thrombus and adjuvant capecitabine-oxaliplatin in a colon cancer patient without any predisposing factors.
2. Case Presentation
A 64-year-old Chinese lady presented with acute left lower quadrant abdominal pain. She had history of well-controlled hypertension on amlodipine 10 mg daily and hyperlipidemia on dietary control. Computerized tomography (CT) of the abdomen and pelvis showed irregular mural thickening at sigmoid colon with uterine and small bowel invasion alongside presacral and left iliac lymph node metastasis. Sigmoidoscopy revealed an obstructing tumor 25 cm from anal verge with biopsy confirmed adenocarcinoma. Preoperative bloods including blood counts, organ function tests and coagulation profile were normal. Carcinoembryonic antigen (CEA) was 85 ng/L (normal range <5 ng/L).
She underwent low anterior resection, total abdominal hysterectomy and bilateral oophorectomy with a defunctioning ileostomy created. Pathology showed moderately differentiated adenocarcinoma of the sigmoid colon with uterine invasion. Nine out of forty-two resected lymph nodes were involved. Overall, she had T4bN2M0, stage III colon cancer (American Joint Committee on Cancer Staging Manual 8th Edition) [3]. Her postoperative recovery was uneventful. One month after surgery, her CEA normalized to 3.7 ng/L and loopogram showed no evidence of anastomotic leak.
Six weeks post-operation, she was started on adjuvant chemotherapy with oral capecitabine at 1000 mg/m2 on days one to fourteen and intravenous oxaliplatin at 130 mg /m2 on day one. Her Eastern Cooperative Oncology Group (ECOG) performance status was zero.
On the fifteenth day of the first cycle, she was admitted for malaise, epigastric pain and high output stoma. She reported no suspicious food intake. She was clinically stable and afebrile, but dehydrated. Abdominal examination showed mild tenderness over epigastrium without any peritoneal signs or organomegaly. Greenish loose stool was noted from her ileostomy.
Blood results showed dehydration picture with elevated white cell count to 9.8 x 109 /L (normal 3.7 - 9.2 x 109 /L), raised hemoglobin to 17.1 g/dL (normal 11.7-14.9 g/dL) and acute kidney injury with serum creatinine at 232 umol/L (normal 40-90 umol/L). Liver function was normal. Electrocardiogram showed sinus rhythm with no acute ischemic changes. Chest and abdominal X-Ray were unremarkable.
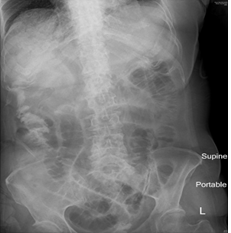
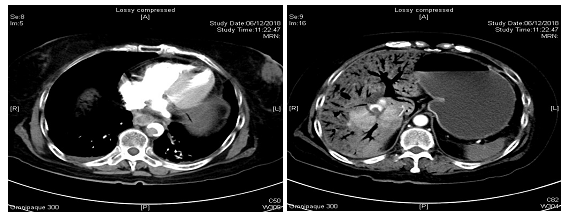
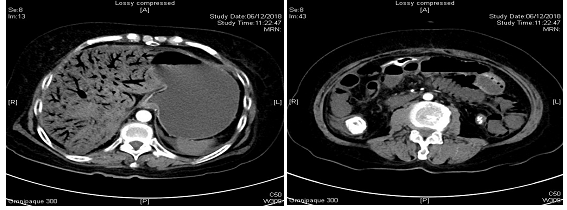
She was treated with intravenous fluid replacement. On the second day, she had severe abdominal pain, shock and hypothermia. Abdominal examination found generalized guarding and fresh blood from ileostomy. Abdominal X-Ray demonstrated “stack-of-coin appearance” suspicious of ischemic bowel (Figure 1). Bloods showed picture of disseminated intravascular coagulation with pancytopenia and deranged clotting profile, and metabolic acidosis with pH of 7.20. She was immediately started on intravenous pantoprazole and broad-spectrum antibiotics. An urgent CT of the abdomen and pelvis demonstrated a thrombus in the descending thoracic aorta above the celiac axis and right portal vein (Figure 2), associated with extensive ischemic involvement of the small bowel, both kidneys, spleen and liver (Figure 3).
Exploratory laparotomy and resection of ischemic organs were offered. The family opted for conservative management after discussion due to expected high risk of “open-and-close” and intraoperative mortality. The patient developed rapid deterioration and succumbed on the same day.
3. Discussion
The cause of death in this patient was extensive ischemia of the abdominal viscera secondary to an aortic thrombus. This condition is a rare life-threatening event more commonly associated with an atherosclerotic aorta, large embolus, or acute occlusion of an abdominal aortic aneurysm [4]. Although our patient had hypertension and hyperlipidemia, they were under control. Besides, neither did she have other risk factors of atherosclerosis such as smoking and diabetes, nor risk factors for embolic events including valvular heart disease or arrhythmias. Pre-operative imaging also did not reveal any evidence of aortic calcifications, atherosclerotic plaques or aneurysmal changes.
Without the above-mentioned factors, thrombosis in a non-aneurysmal, non-atherosclerotic aorta is exceedingly uncommon. Other reported risk factors for arterial thromboembolism include hypercoagulable disorders such as Protein C or S deficiency, antithrombin III deficiency, Factor V Leiden and underlying malignancy [5]. However, patient’s age being older than the typical onset of thrombotic events in familial disorders (before 50 years old) [6] and absence of active cancer makes these factors less likely to be the explained cause of our patient.
Without other apparent underlying causes of thrombotic tendency, chemotherapy is postulated to be the cause based on the timing of the event in relation to the initiation of therapy. This is the first report to describe a possible relationship between capecitabine-oxaliplatin and acute aortic thrombosis.
One large population based matched-cohort study using Surveillance, Epidemiology, and End Results (SEER) data identified a two-fold higher risk of arterial thromboembolism six months after cancer diagnosis (4.7% versus 2.2%; hazard ratio 2.2; 95% confidence interval 2.1 to 2.3), which generally resolved by one year [7]. Some respondents raised the importance of vascular toxicities of cancer therapies in contributing the higher arterial thromboembolism [8].
Cisplatin is the most commonly reported culprit [1, 9-12], with an 8.3% incidence noted in a retrospective analysis of 932 patients [13]. Underlying mechanisms include hypomagnesemia causing arterial spasm, endothelial injury, and increased von Willebrand factors [14]. Our patient was treated with oxaliplatin, which is another platinum-based chemotherapy with similar mechanism of action. Karabacak et al. reported a metastatic colon cancer patient who developed upper limb ischemia after three cycles of 5-fluorouracil and oxaliplatin [15] but not when previously given 5-fluorouracil alone, demonstrating a possible causal relationship between oxaliplatin and arterial thrombosis. A study on advanced gastric cancer also described an 1.1% incidence of arterial thrombosis in patients treated with oxaliplatin-based triplets, but was less compared to cisplatin-based triplets with incidence of 2.9% (p=0.044) [16].
Capecitabine, which is a prodrug of 5-fluorouracil, is more reported to cause coronary artery thrombosis resulting in cardiotoxicity; vascular complications in large vessels are less described. Potential mechanisms hypothesized for capecitabine/ 5-fluorouracil induced cardiotoxicity include vasospasm, endothelial injury, and an increase in thrombogenic substances such as fibrinopeptide A [17]. These postulations could theoretically result in large vessel thrombosis as well. This was illustrated by a recently published case report involving a Korean patient with no known vascular risk factors who developed aortic thrombosis after receiving adjuvant single agent capecitabine for resected colon cancer [18]. From literature review, it is interesting to note that most reported cases were given platinum based combination chemotherapy, most commonly with 5-fluorouracil. Whether there exist any synergistic effects between these two chemotherapeutic agents in generating a pro-thrombotic state warrants further study.
The clinical presentation, severity of symptoms and prognosis of acute aortic thrombosis is vast depending on the site, size of the arterial thrombus and presence of emboli. For example, patients with thoracic aortic thrombosis may present with myocardial infarction or cerebrovascular events from clot embolism, while thrombosis at the descending aorta may result in small bowel or lower limb ischemia. An immediate CT angiography of the aorta should be arranged to confirm the diagnosis when there is clinical suspicion.
The optimal management remains debated. Some authors promote anticoagulation as first line treatment and to consider surgical intervention only if recurrent thrombosis, contraindications to anticoagulation or mobile thrombus [5, 19]. On the other hand, some suggest immediate surgical revascularization [4]. Endovascular thrombectomy or stent graft insertion is preferred over open thrombectomy or extra-anatomical bypass because of lower surgical risks, but feasibility depends on size and location of thrombus [5]. Successful cases of early treatment have been described [20, 21]. Boon et al. reported a 70-year-old metastatic gastric cancer patient who developed acute abdominal and lumbar pains five days after first cycle of epirubicin, oxaliplatin and capecitabine (EOX) due to acute thrombosis within the abdominal aorta, lumbar and right common iliac artery. His condition improved after low molecular weight heparin without surgical intervention, and subsequently switched to second-line chemotherapy with docetaxel uneventfully [20]. Kim et al. also reported two cases of successful treatments with systemic anticoagulation and another two cases with endovascular management [21].
Due to the potentially serious consequences, assessment for risk factors of arterial thrombosis while on chemotherapy is warranted. Any pre-existing cardiovascular diseases (e.g. Hypertension, arrhythmias, aortic aneurysm) have to be optimally managed before the start of chemotherapy. Besides, caution should be exercised when using potentially pro-thrombotic supportive measures during chemotherapy, including indwelling vascular catheters, high dose dexamethasone as antiemetic (greater or equal to 80 mg per cycle) [22, 23], and granulocyte colony-stimulating factor [24, 25]. Clinicians’ awareness of this differential diagnosis in patients presenting with sudden onset of severe abdominal pain or limb paralysis is paramount to allow early recognition and timely management. Switching to other chemotherapy regimes should be carefully considered after the patient recovers from the acute episode.
4. Conclusion
Acute aortic thrombosis could be a rare complication of chemotherapy with capecitabine and oxaliplatin. Further studies are required to understand the mechanism and predictive factors for this life threatening complication, especially in patients without preexisting vascular risk factors.
5. Conflicts of Interest StatementThere are no conflicts of interest with writing this manuscript.
References
- Fernandes DD, Louzada ML, Souza CA, et al. Acute aortic thrombosis in patients receiving cisplatin-based chemotherapy. Curr Oncol 18 (2011): e97-e100.
- Herrmann J, Yang EH, Iliescu C, et al. Vascular Toxicities of Cancer Therapies: The Old and the New-An Evolving Avenue. Circulation 134 (2016): e466-e467.
- Amin MB, Greene F, Byrd DR, et al. AJCC Cancer Staging Manual (8th edition). 8 Edn: Springer International Publishing (2017).
- Kaschwich M, Behrendt CA, Tsilimparis N, et al. Management of acute aortic thrombosis. J Cardiovasc Surg (Torino) 58 (2017): 313-320.
- Tsilimparis N, Hanack U, Pisimisis G, et al. Thrombus in the non-aneurysmal, non-atherosclerotic descending thoracic aorta--an unusual source of arterial embolism. Eur J Vasc Endovasc Surg 41 (2011): 450-457.
- Murin S, Marelich GP, Arroliga AC, et al. Hereditary thrombophilia and venous thromboembolism. Am J Respir Crit Care Med 158 (1998): 1369-1373.
- Navi BB, Reiner AS, Kamel H, et al. Risk of Arterial Thromboembolism in Patients With Cancer. J Am Coll Cardiol 70 (2017): 926-938.
- Frere C, Martin-Toutain I, Thuny F, et al. Risk of Arterial Thrombosis in Cancer Patients: Which Role for Cancer Therapies Vascular Toxicities? J Am Coll Cardiol 71 (2018): 260.
- Apiyasawat S, Wongpraparut N, Jacobson L, et al. Cisplatin induced localized aortic thrombus. Echocardiography 20 (2003): 199-200.
- Chin SO, Lee JJ, Hwang YH, et al. Aortic thrombosis resolved with enoxaparin in a patient treated with cisplatin-based regimen for small cell lung cancer. Int J Hematol 91 (2010): 892-896.
- Doganci S, Kadan M, Kaya E, et al. Acute arterial thrombosis following chemotherapy in a patient with a gastric carcinoma. Cardiovasc J Afr 24 (2013): e7-e9.
- Ito S, Nakamura Y, Noumi T, et al. Acute aortic thrombosis during cisplatin based chemotherapy for gastric cancer. Intern Med 52 (2013): 973-975.
- Moore RA, Adel N, Riedel E, et al. High incidence of thromboembolic events in patients treated with cisplatin-based chemotherapy: a large retrospective analysis. J Clin Oncol 29 (2011): 3466-3473.
- Doll DC, List AF, Greco FA, et al. Acute vascular ischemic events after cisplatin-based combination chemotherapy for germ-cell tumors of the testis. Ann Intern Med 105 (1986): 48-51.
- Karabacak K, Kadan M, Kaya E, et al. Oxaliplatin Induced Digital Ischemia and Necrosis. Case Rep Vasc Med (2015): 248748.
- Starling N, Rao S, Cunningham D, et al. Thromboembolism in patients with advanced gastroesophageal cancer treated with anthracycline, platinum, and fluoropyrimidine combination chemotherapy: a report from the UK National Cancer Research Institute Upper Gastrointestinal Clinical Studies Group. J Clin Oncol 27 (2009): 3786-3793.
- Sorrentino MF, Kim J, Foderaro AE, et al. 5-fluorouracil induced cardiotoxicity: review of the literature. Cardiol J 19 (2012): 453-458.
- Lee MK, et al. Acute Aortic Thrombosis Following Capecitabine Chemotherapy in a Patient with Colon Cancer. Journal of the Korean Society of Radiology 79 (2018).
- Choukroun EM, Labrousse LM, Madonna FP, et al. Mobile thrombus of the thoracic aorta: diagnosis and treatment in 9 cases. Ann Vasc Surg 16 (2002): 714-722.
- Boon IS, Boon CS. In the nick of time: arterial thrombosis on starting combination chemotherapy in metastatic gastric adenocarcinoma. BMJ Case Rep (2016).
- Kim JH, Jeon YS, Cho SG. Successful management of four unusual cases of acute aortic thrombus induced by chemotherapy. Clin Imaging 40 (2016): 224-227.
- Mathews J, Goel R, Evans WK, et al. Arterial occlusion in patients with peripheral vascular disease treated with platinum-based regimens for lung cancer. Cancer Chemother Pharmacol 40 (1997): 19-22.
- Weijl NI, Rutten MF, Zwinderman AH, et al. Thromboembolic events during chemotherapy for germ cell cancer: a cohort study and review of the literature. J Clin Oncol 18 (2000): 2169-2178.
- Kawachi Y, Watanabe A, Uchida T, et al. Acute arterial thrombosis due to platelet aggregation in a patient receiving granulocyte colony-stimulating factor. Br J Haematol 94 (1996): 413-416.
- Spiel AO, Bartko J, Schwameis M, et al. Increased platelet aggregation and in vivo platelet activation after granulocyte colony-stimulating factor administration. A randomised controlled trial. Thromb Haemost 105 (2011): 655-662.


 Impact Factor: * 3.1
Impact Factor: * 3.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
