An Unusual Case of Primary Cardiac Large B-cell Lymphoma Presenting as Cardiac Tamponade A in Chronically Immunosuppressed Patient
Article Information
Daneshvar M1*, Khalid Y1, Kaur L1, Dasu N1, Willard P2, Gabler S3
1Rowan School of Medicine, Jefferson Health System, Stratford, NJ, USA
2Rowan School of Medicine, Stratford, NJ, USA
3Associated Cardiovascular Consultants, Voorhees NJ, USA
*Corresponding Author: Maziyar Daneshvar, Rowan School of Medicine, Jefferson Health System, One Medical Center Dr, UDP Suite 2300, Stratford, NJ, USA
Received: 24 June 2020; Accepted: 01 July 2020; Published: 08 July 2020
Citation: Daneshvar M, Khalid Y, Kaur L, Dasu N, Willard P, Gabler S. An Unusual Case of Primary Cardiac Large B-cell Lymphoma Presenting as Cardiac Tamponade A in Chronically Immunosuppressed Patient. Cardiology and Cardiovascular Medicine 4 (2020): 299-304.
View / Download Pdf Share at FacebookAbstract
A primary cardiac lymphoma (PCL) is an extremely rare form of a non-Hodgkin's lymphoma that involves the heart. It is a diffuse large B-cell lymphoma (DLBCL) that arises from the heart, pericardium, or both. Most PCLs are located in the right atrium. The mass alters the structure of the heart which creates electrical abnormalities. Pericardial involvement usually presents as dyspnea secondary to effusion [1].
We report a unique and rare case of an 82-year-old female who was on chronic immunosuppression for psoriatic arthritis that presented with shortness of breath secondary to cardiac tamponade requiring an emergent pericardial window. She was then diagnosed with primary cardiac lymphoma with primarily pericardial involvement. Subsequent cytology from the pericardial tissue and fluid revealed that she had stage 4 DLBCL. Finally, she developed recurrent pleural effusions in both lungs with rapid decompensation and respiratory failure which lead to death before further evaluation or treatment. Patients typically have a median survival of 7 months.
Keywords
B-cell Lymphoma; Immunosuppressed Patient
Article Details
Introduction
A primary cardiac lymphoma is a rare form of non-Hodgkin’s lymphoma that accounts for only 1% of primary cardiac tumors and less than 0.01% of all cardiac tumors [1]. Most commonly, diagnostic evaluation reveals a diffuse large B-cell lymphoma (DLBCL). For PCL, the most common location of involvement is the right atrium and ventricle. Although pericardial involvement can occur, it is extremely uncommon [1]. The clinical presentation is nonspecific and varies depending on the size and location of this neoplasm. The presentation can include heart rhythm disturbance, syncope, dyspnea, superior vena cava syndrome, and/or restrictive cardiomyopathy [2]. Early diagnosis and treatment is essential as this is a very fatal and rapidly progressive form of lymphoma with extremely poor prognosis and survival.
Case Presentation
The patient was an 82-year-old Caucasian female with a past history of sick sinus syndrome status-post St. Jude pacemaker implantation, hypertension and psoriatic arthritis on methotrexate who presented from her cardiologist’s office for evaluation of a large pericardial effusion and shortness of breath. She was having a routine follow-up with cardiology with a diagnostic transthoracic echocardiogram (TTE) to monitor her aortic stenosis. At the time of presentation, she reported mild dyspnea with exertion, intermittent lightheadedness and mild weight loss. Her family history revealed that her mother died at the age of 72 from an unspecified type of lymphoma. Her vitals were all within normal limits. Her physical exam revealed jugular venous distention and distant heart sounds.
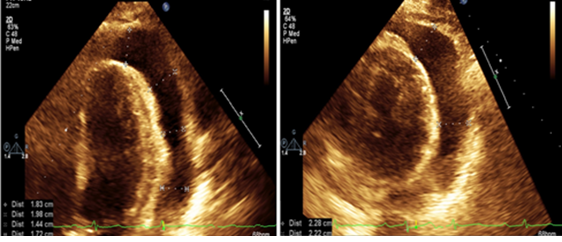
The initial TTE on presentation to the hospital revealed a circumferential pericardial effusion with movement of the ventricle in the cavity, diastolic collapse and systolic atrial collapse which confirmed tamponade physiology (Figure 2). Her initial electrocardiogram showed a normal sinus rhythm with a first-degree AV block, abnormal R wave progression and low voltage in the anterior leads. Computed tomography (CT) of her chest demonstrated bilateral pleural effusions. The patient was taken by cardiothoracic surgery for an emergent subxiphoid pericardial window that drained 600cc of fluid. Fluid cytology and cardiac tissue biopsy were sent to pathology. On the following day, a repeat TTE confirmed the resolution of the cardiac tamponade (Figure 2).
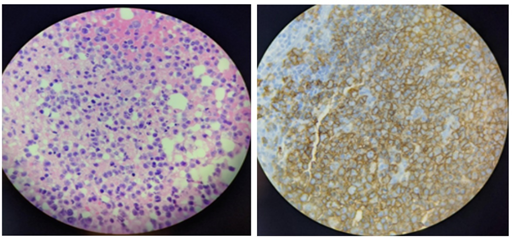
During the pericardial window, a pericardial biopsy was performed, and the tissue was characterized as purple-tan, rubbery, and fibrous-connective tissue. The pericardial tissue under microscopy showed pericardial adipose tissue infiltrated by large neoplastic cells (Figure 1). Immunocytochemistry stains were positive for CD20 (Figure 1), CD10, BCL-2, BCL-6, and C-myc as well as a Ki67 proliferative activity that increased at 80 to 90%. These findings confirmed the diagnosis of a DLBCL of the germinal center type.
On postoperative day two, she had a significant increase in her work of breathing, wheezing and increased shortness of breath. She was diagnosed with acute hypoxemic respiratory failure leading to intubation. The patient’s respiratory status continued to worsen, and she was treated for a bacterial pneumonia. Daily chest x-rays revealed persistent bilateral pulmonary vascular congestion and moderate bilateral pleural effusions greater on the left side. The critical care team then performed a thoracentesis with the removal of 1.6L of fluid from the patient's left pleural space. She was extubated 9 days later. On day 10, she once again began to decompensate with acute hypoxemic failure. A repeat CT of the chest demonstrated persistent bilateral pleural effusions.
Finally, the oncology service decided that the patient was not a good candidate for R-CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone plus the monoclonal antibody rituximab Rituxan) therapy, so palliative rituximab therapy was planned to begin once she became more stable. However, due to the patient's overall tenuous status and poor prognosis, the family opted for comfort care and the patient expired on day 10.
Discussion
PCL is an extremely rare primary malignant cardiac neoplasm. PCL accounts for only 1% of primary cardiac tumors and has a predominance for male patients around 60 years old [1]. However, our patient was a female with a slightly older age. This patient was chronically taking methotrexate, making her immunocompromised. Although some primary cardiac lesions are seen more often in immunocompromised patients, most cases of PCL are seen in people with normal functioning immune systems [3]. Ninety-three percent of these tumors are located in the right heart and they are rarely found in the left side of the heart [1]. The most common type of lymphoma that is found histologically in PCL cases is DLBCL, as seen with our patient [2].
The most common presenting symptoms are dyspnea (64%), constitutional symptoms (26%), and chest pain (24%) [4]. Forty seven percent of the patients have concomitant acute congestive heart failure as observed in our patient [4]. Various degrees of pericardial effusion can be seen with PCLs, but cardiac tamponade as the initial presenting symptom of an underlying PCL is extraordinarily rare and there are only a few reported cases in the current literature, reported in only one other case [5]. Additionally, pericardial involvement in a patient diagnosed with PCL is very infrequent having only been reported in a study of 13 patients from France [3].
Additionally, pericardial disease is commonly a complication of PCL [6]. The underlying pathology of these pericardial effusions is either secondary to seeding of the tumor to the pericardium or secondary to mass effect from mediastinal lymphadenopathy caused by the tumor [6]. Pleural effusions at the time of presentation are an indicator of extremely poor prognosis and disease relapse whether primary or otherwise [6]. This was seen in our patient, presenting with bilateral rapidly evolving pleural effusions that would eventually lead to her progressive decline.
Even though still quite rare, PCL is being diagnosed more frequently now either via advanced imaging or on autopsy following death. The electrocardiogram findings noted are atrial arrhythmias, less commonly ventricular arrhythmias, and sometimes heart block [1]. Although there is no set criteria for diagnosing PCL, the diagnostic evaluation usually involves an transesophageal echocardiogram, cardiac directed imaging using MRI or CT, as well as a biopsy [7]. CT scans can help initially identify the mass and show the degree of involvement of cardiac mass. However, MRI and TEE have 90% sensitivity and specificity, but MRI is better than TEE for identifying myocardial and pericardial thickening [8]. Unfortunately, our patient was unable to have the cardiac imaging due to her hemodynamic instability and rapid decline. Diagnosis can be established with tissue biopsy of the heart and cytology of the pericardial fluid for 67% of patients which was obtained for our patient [8]. The remaining cases that cannot be diagnosed with tissue biopsy and cytology will require more invasive techniques such as cardiac catheterization and echocardiography-guided transvenous biopsy [8]. If all else fails, sternotomy with excision can be used for diagnosis [8].
Furthermore, there is no standardized therapy for PCL. Most treatment data is anecdotal and experimental. Following guidelines for DLBCL in the elderly, a fit patient would be directed towards an anthracycline based chemotherapy like R-CHOP [9]. Radiation would most likely not added for these patients due to the risk radiation induced cardiac injury [10]. However, patients with LV systolic dysfunction, which can be many patients with PCL who present with heart failure as well, may benefit from less intense regimens such as bendamustine or rituximab alone [9].
The prognosis of PCL is poor due to the low rate of early detection and treatment. Our patient was diagnosed at a very advanced stage, so it was difficult to establish even a palliative treatment plan due to her rapid decline. She also developed pleural effusions which are associated with an increased mortality in PCL [6]. One of the key adverse prognostic factors include immunocompromised patients, such as our patient who was on chronic immunosuppression for her psoriatic arthritis. Due to the high mortality rate of PCL, suspicion for this rare disease can potentially lead to an early diagnosis in patients presenting with cardiac symptoms including effusions, arrhythmias, and heart failure.
Conclusion
Our patient demonstrates an extremely rare case of primary cardiac DLBCL manifesting with tamponade physiology on presentation. The rapid decline and eventual demise of this patient demonstrates the significance of early immunopathological diagnosis and surgical and medical intervention in patients with PCL to improve survival. This can only be accomplished by incorporating PCL in the differential diagnosis of patients presenting with sudden decompensated CHF symptoms, arrhythmias and heart blocks.
References
- Gowda RM. Khan IA. Clinical Perspectives of Primary Cardiac Lymphoma. Angiology 54 (2003): 599-604.
- Jonavicius K, Salcius K, Meskauskas R, Valeviciene N, Tarutis V, Sirvydis V. Primary cardiac lymphoma: two cases and a review of literature. Journal of cardiothoracic surgery 10 (2015): 138.
- Carras S, Berger F, Chalabreysse L, Callet?Bauchut E, Cordier JF, Salles G, Girard N. Primary cardiac lymphoma: diagnosis, treatment and outcome in a modern series. Hematological oncology. 35 (2017): 510-9.
- Jeudy J, Burke AP, Frazier AA. Cardiac lymphoma. Radiologic Clinics 54 (2016): 689-710.
- Magalhães S. Ferreira I. Themudo, R. Primary Pericardial Lymphoma: A Rare Entity. Acta Médica Port 29 (2016): 762-762.
- Das DK. Serous effusions in malignant lymphomas: a review. Diagn Cytopathol 34 (2006): 335-347.
- Maleszewski J, Anavekar N. Neoplastic Pericardial Disease. Cardiology Clinics 35 (2017): 589-600.
- Ceresoli GL, Ferreri AJ, Bucci E, Ripa C, Ponzoni M, Villa E. Primary cardiac lymphoma in immunocompetent patients: diagnostic and therapeutic management. Cancer: Interdisciplinary International Journal of the American Cancer Society 80 (1997): 1497-506.
- Chiappella A, Castellino A, Nicolosi M, Santambrogio E, Vitolo U. Diffuse large B-cell lymphoma in the elderly: standard treatment and new perspectives. Expert Review of Hematology 10 (2017): 289-97.
- Atkins KM, Rawal B, Chaunzwa TL, Lamba N, Bitterman DS, et al. Cardiac radiation dose, cardiac disease, and mortality in patients with lung cancer. Journal of the American College of Cardiology 73 (2019): 2976-87.


 Impact Factor: * 3.5
Impact Factor: * 3.5 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 14.80%
Acceptance Rate: 14.80%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 