Endocrine Adjuvant Therapy might Impair Cardiac Autonomic Regulation in Breast Cancer Survivors
Article Information
Daniela Lucini1,2, Mara Malacarne1,2, Gianluigi Oggionni1, Wolfgang Gatzmeier3, Armando Santoro3, Massimo Pagani1
1University of Milan, Biometra Department, Milan, Italy
2Exercise Medicine Unit, Humanitas Clinical and Research Center, Rozzano, Italy
3Cancer Center, Humanitas Clinical and Research Center, Rozzano, Italy;
*Corresponding Author: Daniela Lucini, BIOMETRA University of Milano and Exercise Medicine Unit, Humanitas Clinical and Research Center, Via Alessandro Manzoni, 56, 20089 ROZZANO (Milano) Italy
Received: 19 February 2019; Accepted: 13 March 2019; Published: 15 March 2019
Citation: Daniela Lucini, Mara Malacarne, Gianluigi Oggionni, Wolfgang Gatzmeier, Armando Santoro, Massimo Pagani. Endocrine Adjuvant Therapy might Impair Cardiac Autonomic Regulation in Breast Cancer Survivors. Cardiology and Cardiovascular Medicine 3 (2019): 034-049.
View / Download Pdf Share at FacebookAbstract
Introduction: Cardiovascular diseases and mortality represent a major issue in breast cancer survivors (BCS). Unhealthy lifestyle and some side effects of long term adjuvant therapy may contribute to increase cardiovascular risk. Autonomic Nervous System (ANS) impairment may contribute to initial cardiovascular alteration and early detection of its malfunction in clinical settings could help foster cardiovascular health in these patients.
Methods: This observational study involved 229 outpatient subjects subdivided into three groups: 1) BCS under long term Adjuvant Endocrine Therapy (AET), 2) BCS not on AET, 3) matched healthy controls. Autoregressive spectral analysis of cardiovascular variabilities furnished markers of ANS control, considering simple time and frequency domain classifiers (such as RR variance), and meta-classifiers (such as the index alpha, combining RR and arterial pressure oscillatory information, or ANSI, a percent ranked multivariate unitary index of Autonomic control). Patients’ history, medical examination, blood tests and a lifestyle questionnaire, focusing on exercise, nutrition, and stress, were also employed.
Results: Amplitude indices of ANS regulation (in particular RR variance) appeared progressively (p<0.001) reduced from Controls to Patients not receiving (AET-) and to those receiving (AET+) endocrine therapy; similar changes were observed for meta-classifiers (particularly ANSI: 65.2 ± 21.7; 56.9 ± 27.7; 43.1 ± 26.2, p<0.001)).
Conclusion: Endocrine therapy is associated with impaired autonomic regulation. Autoregressive spectral analysis of heart rate variability may be an affordable and convenient tool to furnish, in a clinical setting, early markers of cardiovascular dysregulation, which may possibly contribute to an overall increased cardiovascular risk.
Keywords
Endocrine therapy; Autonomic nervous system; Stress; Prevention; Survival; Breast cancer
Article Details
1. Introduction
Breast cancer survival has improved during the last decades [1] as a result of early diagnosis and advances in cancer treatment. Five-year survival reached 99% for early-stage disease and the population of cancer survivors in western countries is continuously growing [2]. Accordingly, unwanted effects of antineoplastic therapy are increasingly bringing patients to seek help outside the oncology clinical area. Of particular relevance is the impairment in cardiac function that may follow common adjuvant therapies, and which may produce in the long run deterioration of cardiac function, typically expressed by reduced Ejection Fraction (EF) and, eventually, heart failure. Ventricular dysfunction becomes evident after significant myocardial damage has already occurred. Thus, it may be considered a late marker, as underlined in a recent review [3]. From a clinical point of view it could be important to identify clinical tools capable of detecting early markers of cardiovascular impairment; in this regard, the study of autonomic nervous system could prove useful [3]. Cardiologists may thus be interested in the possible deleterious effects of adjuvant therapy on autonomic nervous system (ANS) [4-7] regulation of cardiac function because of the well-recognized link between autonomic alterations and cardiac prognosis suggesting to treat autonomic dysfunction as an antecedent of foreseeable cardiac conditions. For instance, ANS dysregulation, as suggested by reduced Heart Rate variability and/or impaired baroreflex gain, characterizes pre-hypertension [8, 9], overweight [9, 10], metabolic syndrome [11, 12], stress [13] and being associated to an elevated cardio-metabolic risk [3, 14], may predict mortality in cardiovascular patients [15, 16], particularly after myocardial infarction.
ANS performance is also linked to immunological and hormonal controls [17] and its dysfunction may be associated with chronic inflammation or endocrine dysregulation [18], which are both related to cardiometabolic diseases and cancer. Several studies show that breast cancer survivors present ANS dysfunction [19, 20], in particular HRV derived markers of reduced vagal activity [21, 22] that can be partly attributed to untoward effects of adjuvant therapy [3] and even more so to factors linked to unhealthy lifestyle, such as overweight, sedentariness, reduced cardiorespiratory fitness, stress, fatigue, etc. [3, 23, 24]. ANS dysfunction might also be traced to the effects of radiotherapy and chemotherapy [3, 22, 25-28]. There are however few data regarding the effects of long term systemic adjuvant therapy on ANS control. In this study we focus on the possible detrimental effects of endocrine adjuvant therapy on ANS performance on top of standard multifaceted adjuvant therapy, considering ANS dysregulation as a component of an emerging physiopathological ontology [29, 30]. To this end, we specifically explore the possibility to detect in a clinical setting difference in early markers of alteration in cardiovascular autonomic control in a population of breast cancer survivors with or without endocrine treatment. Given the major role of parasympathetic mechanisms both in clinical cardiology [16, 31, 32] and in oncology [21] we studied simple monovariate time and frequency domain classifiers from Heart Rate Variability (as assessed by ECG derived RR interval V), and a bivariate classifier such as the frequency domain index of cardiac baroreflex gain, at rest.
Furthermore, according to most distinguishable features extracted with factor analysis [30] from a large benchmark population, we computed a “unitary” Autonomic Nervous System Index (ANSI) [9] providing a rank of the dynamic of the paired antagonistic (sympathetic-parasympathetic), dual (positive and negative) feed-back cardiac neural regulation [33], usable as a simple clinical metaclassifier of autonomic dysfunction.
2. Methods
This retrospective proof of concept study involved anonymized data from 229 outpatients subdivided into three groups (see Table 1):
1) 94 breast cancer survivors who were treated with chemotherapy (CT) and/or radiotherapy (RT) and who were, at the moment of evaluation, under long term Adjuvant Endocrine Therapy (AET+), 11.7% were also treated with Trastuzumab.
2) 34 breast cancer survivors who were treated with CT and/or RT and were not on long term endocrine therapy (AET) at the moment of the evaluation. 11.8% were also treated with Trastuzumab.
3) 101 age matched healthy controls (Healthy Subjects).
Patients were treated at the Humanitas Research Hospital Cancer Center following best clinical practice and, together with healthy controls, were evaluated at the Exercise Medicine Unit, Humanitas Research Hospital. Eligibility criteria included age (30-75 years), no evidence of metastases and absence of acute conditions (within the past three months). In all controls the presence of disease or pharmacological treatment was excluded through history assessment and standard medical examination. General average characteristics are reported in Table 1. None of the patients underwent CT or RT treatments during the study period; they proceeded with endocrine therapy and in some cases, (similar percentage in both groups) biological (Trastuzumab) therapies. Endocrine therapy (inducing estrogen inhibition) considered the selective estrogen receptor modulator (SERM) Tamoxifen, LH-RH analogue, or Aromatase Inhibitors. The choice of the patient’s treatment protocol was based on patients’ and/or tumor characteristics (Table 2). Patients’ clinical assessment comprised history, standard medical examination, anthropometric and hemodynamic data, blood tests (see Table 1), and an ad hoc questionnaire [34, 35] to quantify lifestyle, with particular focus on physical activity [36], nutrition [37], perception of fatigue, stress and subjective stress related somatic symptoms (4SQ) (Stress Indices) [13]. Lastly, Fat (FM%), Free Fat Mass (FFM %) and % of dry free fat mass (Dry Lean) measurements were obtained by multi-frequency bioelectrical impedance analysis (Quadscan 4000 Multi-frequency, Bodystat LTD, Isle of Man, UK) (Table 2).
2.1 Autonomic evaluation
On the day of autonomic evaluation all subjects arrived at the clinic and had avoided caffeinated beverages since awakening as well as heavy physical exercise in the preceding 24 h. Recordings were performed between 09:00 am and 12:00 am in an air conditioned, quiet room. After a preliminary 10-min rest period in the supine position, ECG and respiratory activity (piezoelectric belt) were continuously recorded over a minimum 5-min period with a two-way radiotelemetry system (Marazza, Monza Italy). Subsequently subjects were asked to stand up unaided and remained in the upright position for 5 minutes, during which recordings were maintained. Data were acquired with a PC at 250 samples/second using a custom built software tool (HeartScope) [38] that automatically provided a series of indices describing Heart Rate variability (HRV) in the time domain: RR interval (in msec) and RR interval V (assessed as total power, i.e. variance, in msec2, taken as simple classifiers typical of vagal control [16, 32]); and in the frequency domain: autoregressive spectral components both in the low frequency (LF, center frequency ≈ 0.1 Hz) and in the high frequency (HF, centered with respiration, ≈ 0.25 Hz), assessed in msec2 as well as in normalized units (nu). To include an approximate evaluation of the effects of sympathetic activation, produced by active standing, the stand-rest difference (Δ) in LFnu was also computed [8, 39]. Arterial pressure was continuously monitored using a plethysmographic approach (Finometer Midi, FMS) calibrated against a sphygmomanometer and analyzed simultaneously with the same software tool [38]. The sensitivity of spontaneous cardiac baroreflex control of RR interval was assessed by a frequency-domain method (Alpha-Index=square root of the average of the ratio between RR interval and SAP Spectral powers in the LF and HF regions) [8, 40] usable as a bivariate metaclassifier. Recently to simplify clinical interpretation of multiple RR V variables, we described a unitary [41] autonomic index (ANSI) [9]. Computation of ANSI [9] depends on the combination of principal factor analysis and clinically optimized radar plot, considering information carried by RR, RR variance and Δ LFnu [9]. The computing procedure 1st corrects for age by percentile rank transformation, 2nd ranks the information (82.7% of Variance Accounted For) distributed across indices from the selected clusters of variability (considering amplitude and oscillatory code modalities [30]), 3rd using a radar plot [9] builds ANSI as a triangle area that is finally ranked against the benchmark population. ANSI is treated as a proxy of cardiac autonomic regulation.
2.2 Stress and somatic perception
Subjects completed a self-administered questionnaire [13, 34, 35] providing nominal self-rated scales (higher values indicate higher degrees of symptoms) that focused on: (i) the appraisal of overall stress and fatigue perception by Likert linear scales from 0 (‘no perception’) to 10 (‘highest perception’) for each measure; (ii) The Subjective Stress-related Somatic Symptoms Questionnaire (4S-Q), inquiring about 18 somatic symptoms accounting for the majority of somatic complaints. For scoring purpose, each response was coded from 0 (‘no feeling’) to 10 (‘a strong feeling’), thus the total score ranged from 0 to 180.
2.3 Statistics
Data are presented as mean ± SD. Significant differences were assessed by unpaired T Test, with Bonferroni correction, and by General Linear Model followed by post hoc comparisons. Trends were assessed with Jonckheere-Terpstra trend test. Group frequencies were assessed by the Chi-square test. Significance was set at p<0.05. Partial eta squared was also computed as a measure of effect size, defined as small, medium and large corresponding to values of 0.0099, 0.0588 and 0.1379, respectively [42]. Computations were performed with a commercial statistical package (SPSS version 24).
3. Results
Table 1 reports the general characteristics of the entire population. Heart rate, diastolic arterial pressure, Body Mass Index (BMI), and Waist circumference progressively (JT, p<0.001) increased from Controls to (AET-) Patients and to (AET+) Patients. In particular, BMI and Waist circumference were significantly greater in the latter group as compared to controls and to patients not receiving endocrine therapy. This latter finding was corroborated (Table 2) by greater %Fat Mass, (smaller %Free Fat Mass and %dry lean mass in patients on endocrine therapy compared with Patients who were not; no data available for controls). Note that no differences were observed between the two patient groups when considering nutrition score and the (self-reported) dose of aerobic physical activity. The percentage of subjects who were previously treated with CT was slightly lower in the group of patients on endocrine therapy, while the percentage of subjects who were previously treated with RT was slightly higher.
|
Variables |
Healthy subjects |
Endocrine T. (-) |
Endocrine T. (+) |
P |
|
Number |
101 |
34 |
94 |
|
|
Age [years] |
48.5 ± 6.4 |
48.6 ± 8.3 |
50.8 ± 9.1 |
0.107 |
|
SAP [mmHg] |
115.5 ± 12.6 |
114.2 ± 13.7 |
120.9 ± 17.2*† |
0.016 |
|
DAP [mmHg] |
72.6 ± 7.5 |
76.0 ± 8.7 |
78.5 ± 9.9* |
<0.001 |
|
HR [b/min] |
65.0 ± 8.9 |
68.2 ± 9.4 |
70.5 ± 11.0* |
0.001 |
|
BMI [Kg/m2] |
22.6 ± 2.8 |
26.0 ± 4.6 * |
28.6 ± 5.7*† |
<0.001 |
|
Waist C [cm] |
80.7 ± 7.8 |
88.5 ± 10.9 * |
95.3 ± 12.9*† |
<0.001 |
|
Smoke [%] |
11.7 |
17.6 |
7.5 |
0.252 |
Abbreviations: SAP-systolic arterial pressure; DAP-diastolic arterial pressure; HR-heart rate; BMI-body mass index; Waist C-waist circumference; JT Test-Jonckheere-Terpstra test; smoker positive if ≥ 1 cigarette/day; data are expressed as mean ± SD; GLM significant contrast for * vs Healthy subjects, † vs Endocrine T. (-), p<0.05.
Table 1: Study Population.
|
Variables |
AET- |
AET+ |
P |
|
Time Since Surgery [months] |
22.2 ± 29.6 |
23.1 ± 18.7 |
0.854 |
|
Physical Activity [MET*min/week] |
637.3 ± 538.0 |
729.6 ± 776.3 |
0.524 |
|
Nutrition Score [A. U.] |
2.50 ± 0.86 |
2.24 ± 0.90 |
0.143 |
|
FM [%] |
35.4 ± 6.3 |
38.6 ± 7.2 |
0.034 |
|
FFM [%] |
64.6 ± 6.3 |
61.6 ± 7.3 |
0.046 |
|
Dry Lean [%] |
16.0 ± 4.1 |
14.1 ± 3.5 |
0.018 |
|
Total Cholesterol [mg/dl] |
164.3 ± 96.6 |
196.2 ± 78.8 |
0.095 |
|
HDL Cholesterol [mg/dl] |
36.6 ± 33.6 |
46.3 ± 28.8 |
0.173 |
|
LDL Cholesterol [mg/dl] |
77.3 ± 69.9 |
99.2 ± 64.7 |
0.165 |
|
Triglycerides [mg/dl] |
67.6 ± 60 |
102.0 ± 97.6 |
0.101 |
|
FPG [mg/dl] |
67.6 ± 39.3 |
79.5 ± 37.8 |
0.186 |
|
Mastectomy/ Quadrantectomy [%] |
26.5/73.5 |
29.0/71.0 |
0.715 |
|
RT [%] |
61.7 |
80.1 |
0.735 |
|
CT [%] |
82.3 |
56.4 |
0.111 |
|
Trastuzumab [%] |
11.8 |
11.7 |
0.992 |
|
Cardiovascular Medication [%] |
5.9 |
13.8 |
0.217 |
Abbreviations: A.U.-arbitrary units; range 0-5 for nutrition score; MET-metabolic equivalent; FM%-% of fat mass; FFM%-% of free fat mass; HDL-high density lipoprotein; LDL-light density; Lipoprotein; FGP-fasting plasma glucose; RT-radiotherapy; CT-chemotherapy; cardiovascular medication comprise: ACEi, angiotensin receptor blockers, calcium channel blockers; data are expressed as mean ± SD; P- bold character if significance (p)<0.05.
Table 2: Life style and metabolic proxies for patients without Endocrine Therapy (AET-) and under Endocrine Therapy (AET+).
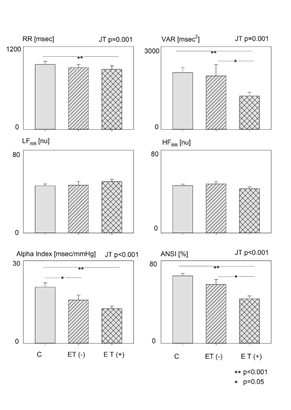
Indices of ANS regulation (see Table 3 and Figure 1) appeared progressively altered from Controls to (AET-) Patients and to (AET+) ones, as shown by a graded decline of RR interval, RR variance, LF and HF components in ms2, Δ LFRR nu, alpha Index and ANSI (all JT p ≤ 0.001, Δ LFRR p<0.007)). However post hoc contrasts suggested a selectively greater effect in (AET+) patients particularly regarding RR variance, LF and HF components in ms2 (i.e. amplitude coded variables) and ANSI. No difference was observed at rest regarding LF and HF in nu between controls and patients.
|
Variables |
Healthy subjects |
Endocrine T. (-) |
Endocrine T. (+) |
P |
?2 |
PW |
JT Test |
|
RR [ms] |
942.1 ± 145.7 |
896.4 ± 130.4 |
871.5 ± 135.4 * |
0.002 |
0.053 |
0.897 |
0.001 |
|
VARRR [ms2] |
2058 ± 1871 |
1940 ± 2396 |
1216 ± 1238 *† |
0.003 |
0.051 |
0.885 |
0.001 |
|
LFRRa [ms2] |
497 ± 541 |
520 ± 948 |
259 ± 310*† |
0.005 |
0.047 |
0.840 |
<0.001 |
|
LFRR [nu] |
45.7 ± 15.6 |
46.2 ± 20.8 |
49.7 ± 22.3 |
0.333 |
0.010 |
0.243 |
0.290 |
|
HFRRa [ms2] |
495 ±571 |
612 ±1307 |
285 ± 466*† |
0.030 |
0.031 |
0.659 |
<0.001 |
|
HFRR [nu] |
45.8 ± 16.7 |
47.4 ± 19.1 |
42.8 ± 21.3 |
0.373 |
0.009 |
0.221 |
0.304 |
|
LF/HF |
1.41 ± 1.47 |
1.56 ± 1.82 |
3.00 ± 7.25 |
0.057 |
0.025 |
0.563 |
0.319 |
|
ΔLFRR [nu] |
27.7 ± 16.0 |
26.2 ± 20.9 |
19.3 ± 25.8 * |
0.020 |
0.034 |
0.709 |
0.007 |
|
Alpha Index [ms/mmHg] |
20.4 ± 12.8 |
15.7 ± 10.3* |
12.6 ± 8.5 * |
<0.001 |
0.102 |
0.992 |
<0.001 |
|
SAPLFa [mmHg2] |
3.4 ± 3.7 |
2.9 ±2.5 |
5.3 ± 13.8 |
0.335 |
0.011 |
0.242 |
0.887 |
|
ANSI [%] |
65.2 ± 21.7 |
56.9 ± 27.7 |
43.1 ± 26.2 *† |
<0.001 |
0.149 |
1.000 |
<0.001 |
|
Stress[AU] |
4.3 ± 2.7 |
5.6 ± 3.4* |
5.8 ± 3.2* |
0.004 |
0.052 |
0.858 |
0.002 |
|
Fatigue [AU] |
4.8 ± 2.6 |
6.1 ± 2.7* |
6.0 ± 3.2* |
0.009 |
0.044 |
0.794 |
0.002 |
|
4SQ [AU] |
31.7 ± 23.1 |
44.4 ± 27.5* |
45.5 ± 30.2* |
0.002 |
0.057 |
0.896 |
0.003 |
Abbreviations: RR-RR mean interval; VARRR-RR interval variance; LFRRa-absolute values of low-frequency component of RR variability; HF-high frequency; Δ-stand-rest difference; nu-normalized units; SAPLFa-low-frequency component of systolic arterial pressure variability; ANSI-autonomic nervous system index; AU-arbitrary units; 4SQ-somatic stress symptom questionnaire; P-significance; ?2-partial eta squared; PW-observed power; JT Test- Jonckheere-Terpstra test; GLM significant contrast for * vs Healthy subjects, † vs Endocrine T. (-), p<0.05. Notice that only for Alpha Index and ANSI ?2 outnumbers 0.0588 and only for ANSI ?2outnumbers 0.1379 that are the limits, respectively, for medium and large effect size [42].
Table 3: Descriptive Statistics of Autonomic and Stress Variables in the Considered Groups.
Figure 1: Differences in indirect indices of cardiac Autonomic regulation in the examined groups: from controls (left bar) to Cancer survivors not on Endocrine therapy (-) (middle bar) and on Endocrine therapy (-) (right bar). Note the significant trend (assessed with JT test) in RR interval, RR Variance (var), the cardiac baroreflex (index α) as well as the unitary autonomic index (ANSI), but no effect on Low and High Frequency normalized powers (LFRR nu, and HFRR nu), suggesting selective autonomic impairment with Endocrine therapy. Significance of individual contrasts is indicated in the bottom right corner.
4. Discussion
In these preliminary, proof of concept, observations we report that, in breast cancer survivors, endocrine adjuvant therapy is associated with a more apparent impairment in autonomic regulation as suggested by: amplitude coded [30] proxies (RR variance and absolute power of LF and HF oscillations) and the unitary autonomic index ANSI, as well as cardiac baroreflex (index alpha, JT test only), hinting at an overall prevailing inhibitory effect on parasympathetic regulation. Autoregressive spectral analysis of heart rate variability may be an affordable and parsimonious tool that furnishes, in the clinical setting, early markers of cardiovascular dysregulation congruent with findings of cardiac baroreflex assessment. Anthropometric (BMI, waist circumference, FM%), but not glucolipid profile was also slightly worse in the AET+ group.
4.1 Autonomic dysregulation, cancer and adjuvant therapy
At more than 23000 hits in the Pubmed database HRV has gained the de facto role of non-invasive standard for autonomic evaluation. Time domain indices (such as RR variance or SD), as well as amplitude coded oscillations (particularly HF, in absolute units) reflect parasympathetic control, while stand induced changes in LF nu reflect the sympathetic excitatory effect of the upright posture. There is however still a debate as to the specific code underlying the link between nerve activity (usually obtained from leg muscle sympathetic activity) and coding of hidden information. We recently showed that multivariate statistics suggest that autonomic information is assembled into few clusters of spectral indices [30, 43], projected from hidden variables (specifically reflecting information about amplitude and oscillation). Accordingly we may surmise that the essential meaning could be reconstituted in a single unitary index, combining the information spread in multiple hidden variables [9]. This would largely simplify the assessment of autonomic performance, as expressed by a rank from 0 to100.
Clinically ANS dysregulation is observed in cancer patients [3, 19, 20, 22, 44, 45]. In breast cancer survivors ANS may be directly involved in the development and progression of cancer possibly through vagal modulation of the immune system [46], it may also mediate attendant, possibly late onset, cardiovascular alterations inducing an increase in cardiometabolic risk [5, 6]. Of clinical value is the possible ANS dysregulation associated with adjuvant therapy [3, 4, 22]. Both radiotherapy (RT) [28] and chemotherapy (CT) [25] with long term endocrine therapy [3, 22] seem to affect ANS control. Our data show that breast cancer survivors, who are on endocrine therapy (AET+) after RT or CT, present a more apparent ANS alteration than observed in patients who receive RT or CT and do not undergo long term endocrine treatment (AET-), despite no differences in nutrition score, physical activity and stress. The changes in indices of metabolic control observed in patients on endocrine therapy suggest metabolic impairment [47]. This in turn may foster a worsening in ANS dysfunction in breast cancer survivors. In this study, patients on endocrine therapy present an increase in BMI, waist circumference and percentage of fat mass, with reduced percentage of free fat mass despite no changes in lifestyle factors (smoke, diet endocrine treatment in the observed ANS alteration in our patient population [47]).
4.2 ANS impairment as initial cardiovascular alteration
ANS impairment may precede the development of some overt diseases, characterizing a status of increased risk [3]. It precedes the development of hypertension [8], diabetes, obesity [10] and is associated with functional diseases such as irritable bowel disease, chronic fatigue, fibromyalgia and chronic stress syndrome [48]. The possibility to detect an initial ANS impairment may be of potential interest also in cancer survivors in order to initiate secondary prevention programs to reduce cardio-metabolic risk before the onset of overt cardiovascular diseases. In these patients cancer and cardiovascular disease are inextricably intertwined [5, 6] and cardiovascular mortality is dramatically increasing especially in women older than 60 years of age [3, 49, 50]. The coexistence of cardiovascular disease and/or of risk factors already present before the onset of cancer worsens the prognosis [4-6]. In current oncology practice patients’ cardiovascular health and possible cardiovascular side effects of adjuvant therapy are primarily evaluated by simple medical assessment, changes in resting left ventricular ejection fraction (LV-EF%) and determination of lipid profile. Reduction of LV-EF% becomes evident after significant myocardial damage has already occurred [3] and thus should not be considered an early finding. Moreover it is associated, fortunately, with only some long term adjuvant treatments such as Trastuzumab [51]. Lipid profile may be differently affected by various adjuvant therapies. For instance, Tamoxifen seems to be associated with a reduction of total and LDL cholesterol (without a reduction in cardiovascular mortality) [5, 52-54], while the aromatase inhibitor, Letrozole, seems to increase serum lipids levels [5]. Notably, patient’s lifestyle (which is in general deteriorated in breast cancer survivors) as well as the presence of other risk factors, such as overweight and smoking, may further affect lipid profile [5-7, 44].
In view of the strong prognostic value of reduced HRV (and baroreflex gain) in cardiovascular conditions the observation that time domain indices of HRV are further reduced in AE+ patients might suggest autonomic evaluation as a tool to assess early changes in this component of cardiovascular health in breast cancer survivors. In this context the percent ranked modality of the reduced unitary index ANSI might represent a useful simplification.
4.3 Endocrine therapy and cardiometabolic risk
Several studies investigated the link between endocrine treatment and increased cardiovascular risk. Tamoxifen does not seem to increase cardiovascular mortality, however, despite improving lipid profile and decreasing the risk of death from myocardial infarction, it had no impact on the incidence of myocardial infarction [5, 52-54] and on the reduction of overall cardiovascular mortality [55]. Moreover, it is associated with a higher incidence of vascular events particularly thromboembolism and stroke [4]. Aromatase Inhibitors seem to worsen lipid profile and to increase cardiovascular death [51, 56, 57], and Anastrazole treatment is associated with an increase in hypertension and moderate angina [58]. On the other hand, other studies did not show a significant worsening of cardiovascular prognosis in patient treated with these drugs as compared to tamoxifen treatment [52, 58]. In our study it was not possible to derive significant information regarding the effects of Tamoxifen (or Tamoxifen+LHRH analogue) when compared to Aromatase Inhibitors due to the relative limited number of patients. When considering subgroups (patient who only received Tamoxifen treatment -23.08%, patient who received Tamoxifen+LHRH analogue treatments-51.64%, and patients who received alternatively one Aromatase Inhibitor among Anastrazole or Letrozole or Exemestane -25.28%), we did not observe significant differences in ANS variables; for instance, in the three subgroups ANSI was 42.41, 46.02, 38.68 %, respectively.
4.4 Fatigue, Stress and subjective stress related somatic symptoms perception
Stress represents an important cardiovascular risk factor acting directly on hormonal, immune and autonomic nervous controls [48] and indirectly favoring negative lifestyle habits such as poor nutrition, sedentariness and smoking [48]. Several studies report an ANS impairment (also detected using HRV) in chronic stress conditions [13, 59] also in breast cancer survivors [44]. This finding was confirmed by our study, as breast cancer survivors are characterized by higher levels of perceived stress, fatigue and stress related somatic symptoms compared to Controls. However no difference was observed between AET+ and AET-. Accordingly it is unlikely that increased levels of subjective stress might account for the greater ANS impairment observed in AET+ patients. Regarding stress indices, it should be noted that the effect size is in the range of small size. Overall it may be argued that autonomic deterioration is a somewhat selective effect of endocrine therapy, particularly evidenced by HRV analysis.
4.5 The study of ANS as a clinical tool
The study of ANS in clinical settings, although of recognized importance as a component of cardiac risk profile, is currently limited by several issues, particularly:
-large set of potentially redundant variables furnished by the analysis [9, 32]
-costs and specific competencies because of the need to continuously record both RR and SAP variabilities in order to obtain information regarding both cardiac and vascular ANS controls [60]
-influence of age and gender [9, 32].
Accordingly it may be useful to consider testing the usefulness of ANSI, a percent ranked multivariate unitary index of Autonomic control. ANSI in fact overcomes these issues, accounting by design [9] for age and gender bias and reducing into a single parameter (obtained solely from RR interval variability) all the main aspects needed to clinically describe individual ANS performance, defined by the percentile rank from the reference population, on a scale from 0 to 100. Simply, low and high ANSI values indicate impaired and functional autonomic performance [9]. A recent study showed that ANSI permitted to assess in a quantitative manner the different ANS impairment associated with different clinical conditions (hypertension, obesity, smoking) [9], and the greater impact of their combination.
In this investigation ANSI appears to complement findings with individual autonomic proxies from RRV, such as RR and RR variance (square of RR SD). Lower values in patients undergoing endocrine therapy suggest a potential increase of autonomic risk in this group of patients. Notably the effect size, assessed with partial eta square, appears greatest with ANSI not only as compared to other autonomic HRV proxies, but also with the index alpha. Although a word of caution must be said about effect size analysis as a measure of importance of effect, it can be concluded that ANSI compares favorably to the other HRV derived indices, and even to more complex indices such as the bivariate, frequency domain measure of baroreflex gain alpha, a recognized predictor of cardiovascular events) [8, 16]. ANSI has the additional advantage of reducing all autonomic information into a unitary value, which translates into easier interpretation of autonomic performance. The study of ANS may be further useful in revealing some beneficial effects of behavioral interventions, such as physical training [39, 40, 61, 62], weight reduction [10] and stress management [48, 63], which may represent convenient tools in breast cancer survivors in order to reduce cardiac risk, hence potentially cardiovascular events, and improving general wellbeing, particularly in those patients who require long term adjuvant therapy [50].
5. Limitations
Limitations should be considered: body composition and blood tests were not assessed in controls, whose dose of physical activity and nutrition score were not quantified, since lifestyle behavior was only assessed by history. The limited patient study population does not permit to assess the possible effect of Trastuzumab treatment on ANS control. Moreover, to differentiate between Tamoxifen and aromatase inhibitor effects larger clinical studies will be necessary to better define the individual effects of the various and different adjuvant treatments. Lastly, autonomic assessment, although performed with multiple indices, is still based on indirect measures [32] that are fraught by imprecisions and a still ongoing debate, particularly regarding sympathetic mediated information.
6. Conclusion
In conclusion, HRV evaluation might represent an additional tool to detect in breast cancer survivors early signs of cardiovascular autonomic involvement. This approach could foster the assessment of the important benefits following endocrine treatment. HRV, possibly complemented by ANSI, might provide data potentially useful in the cardiovascular management of cancer survivors. The value of this information needs larger longitudinal trials to be better defined.
Compliance with Ethical Standards
Funding
No funding was provided for this study.
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
The study was in accordance with the ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and Title 45, US Code of Federal Regulations, Part 46, Protection of Human Subjects, Revised November 13, 2001, effective December 13, 2001.
Informed Consent
All patients included in this study, signed an informed consent form.
Research data are confidential.
Acknowledgements
We thank native English speaker Dana Shiffer MD. for the English Language revision of the paper.
Credit Author Statement
Daniela Lucini: Conceptualization; Data curation; Funding acquisition; Investigation; Methodology; Project administration; Resources; Supervision; Validation; Visualization; Roles/Writing-original draft;
Mara Malacarne: Data curation; Investigation; Formal analysis; Software; Visualization ;
Gianluigi Oggionni: Data curation; Formal analysis; Visualization;
Wolfgang Gatzmeier: Investigation; Validation; Writing-Reviewing and Editing ;
Armando Santoro: Funding acquisition; Validation; Writing-Reviewing and Editing;
Massimo Pagani: Conceptualization; Data curation; Methodology; Supervision; Validation; Visualization; Roles/Writing-original draft;
Highlights
- Breast cancer survivors present altered ANS control independently from lifestyle
- Adjuvant endocrine therapy can further impair ANS control
- Spectral analysis of HRV may represent a convenient tool to assess ANS
- HRV may furnish in clinical setting early markers of cardiovascular dysregulation
- HRV may titrate autonomic effects of endocrine therapy on a personal basis.
References
- Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin 67 (2017): 7-30.
- Miller KD, Siegel RL, Lin CC, et al. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin 66 (2016): 271-289.
- Lakoski SG, Jones LW, Krone RJ, et al. Autonomic dysfunction in early breast cancer: Incidence, clinical importance, and underlying mechanisms. Am Heart J 170 (2015): 231-241.
- Khouri MG, Douglas PS, Mackey JR, et al. Cancer therapy-induced cardiac toxicity in early breast cancer: addressing the unresolved issues. Circulation 126 (2012): 2749-2763.
- Ewer MS, Gluck S. A woman’s heart: the impact of adjuvant endocrine therapy on cardiovascular health. Cancer 115 (2009): 1813-1826.
- Jones LW, Haykowsky MJ, Swartz JJ, et al. Early breast cancer therapy and cardiovascular injury. J Am Coll Cardiol 50 (2007): 1435-1441.
- Lucini D, Vigo C, Malacarne M, et al. Lifestyle changes as internal medicine. European Journal of Internal Medicine 43 (2017): 40-42.
- Lucini D, Mela GS, Malliani A, et al. Impairment in cardiac autonomic regulation preceding arterial hypertension in humans: insights from spectral analysis of beat-by-beat cardiovascular variability. Circulation 106 (2002): 2673-2679.
- Sala R, Malacarne M, Solaro N, et al. A composite autonomic index as unitary metric for heart rate variability: a proof of concept. Eur J Clin Invest 47 (2017): 241-249.
- Grassi G, Seravalle G, Colombo M, et al. Body weight reduction, sympathetic nerve traffic, and arterial baroreflex in obese normotensive humans. Circulation 97 (1998): 2037-2042.
- Lucini D, Cusumano G, Bellia A, et al. Is reduced baroreflex gain a component of the metabolic syndrome? Insights from the LINOSA study. J Hypertens 24 (2006): 361-370.
- Mancia G, Bousquet P, Elghozi JL, et al. The sympathetic nervous system and the metabolic syndrome. J Hypertens 25 (2007): 909-920.
- Lucini D, Di Fede G, Parati G, et al. Impact of chronic psychosocial stress on autonomic cardiovascular regulation in otherwise healthy subjects. Hypertension 46 (2005): 1201-1206.
- Karakas M, Koenig W. Sympathetic nervous system: a crucial player modulating residual cardiovascular risk. Circulation research 112 (2013): 13-16.
- Kleiger RE, Miller JP, Bigger JT, et al. Decreased heart rate variability and its association with increased mortality after acute myocardial infarction. Am J Cardiol 59 (1987): 256-262.
- La Rovere MT, Bigger JT, Marcus FI, et al. Baroreflex sensitivity and heart-rate variability in prediction of total cardiac mortality after myocardial infarction. ATRAMI (Autonomic Tone and Reflexes After Myocardial Infarction) Investigators. Lancet 351 (1998): 478-484.
- Pavlov VA, Tracey KJ. The vagus nerve and the inflammatory reflex-linking immunity and metabolism. Nature Reviews Endocrinology 8 (2012): 743.
- Wyss JM, Carlson SH. Effects of hormone replacement therapy on the sympathetic nervous system and blood pressure. Curr Hypertens Rep 5 (2003): 241-246.
- Giese-Davis J, Wilhelm FH, Tamagawa R, et al. Higher vagal activity as related to survival in patients with advanced breast cancer: an analysis of autonomic dysregulation. Psychosom Med 77 (2015): 346-355.
- Payne D, Mahmood S, Partridge AH, et al. Cardiac autonomic dysfunction in breast cancer survivors. American Society of Clinical Oncology (2017).
- De Couck M, Gidron Y. Norms of vagal nerve activity, indexed by Heart Rate Variability, in cancer patients. Cancer Epidemiol 37 (2013): 737-741.
- Caro-Moran E, Fernandez-Lao C, Galiano-Castillo N, et al. Heart Rate Variability in Breast Cancer Survivors After the First Year of Treatments: A Case-Controlled Study. Biol Res Nurs 18 (2016): 43-49.
- Dieli-Conwright CM, Mortimer JE, Schroeder ET, et al. Randomized controlled trial to evaluate the effects of combined progressive exercise on metabolic syndrome in breast cancer survivors: rationale, design, and methods. BMC Cancer 14 (2014): 238.
- Patnaik JL, Byers T, Diguiseppi C, et al. Cardiovascular disease competes with breast cancer as the leading cause of death for older females diagnosed with breast cancer: a retrospective cohort study. Breast Cancer Res 13 (2011): 64.
- Adams SC, Schondorf R, Benoit J, et al. Impact of cancer and chemotherapy on autonomic nervous system function and cardiovascular reactivity in young adults with cancer: a case-controlled feasibility study. BMC Cancer 15 (2015): 414.
- Zamorano JL, Lancellotti P, Munoz DR et al. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines. Eur Heart J 37 (2016): 2768-2801.
- Ekholm EM, Salminen EK, Huikuri HV, et al. Impairment of heart rate variability during paclitaxel therapy. Cancer 88 (2000): 2149-2153.
- Hoca A, Yildiz M, Ozyigit G. Evaluation of the effects of mediastinal radiation therapy on autonomic nervous system. Med Oncol 29 (2012): 3581-3586.
- Malliani A, Pagani M, Furlan R, et al. Individual recognition by heart rate variability of two different autonomic profiles related to posture. Circulation 96 (1997): 4143-4145.
- Lucini D, Solaro N, Pagani M. Autonomic differentiation map: a novel statistical tool for interpretation of heart rate variability. Frontiers in Physiology 9 (2018): 401.
- Eckberg DL. Sympathovagal balance: a critical appraisal. Circulation 96 (1997): 3224-3232.
- Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart-rate variability: standards of measurements, physiological interpretation and clinical use. Circulation 93 (1996): 1043-1065.
- Malliani A, Pagani M, Lombardi F, et al. Cardiovascular neural regulation explored in the frequency domain. Circulation 84 (1991): 482-492.
- Lucini D, Zanuso S, Blair S, et al. A simple healthy lifestyle index as a proxy of wellness: a proof of concept. Acta Diabetol 52 (2015): 81-89.
- Lucini D, Solaro N, Lesma A, et al. Health promotion in the workplace: assessing stress and lifestyle with an intranet tool. J Med Internet Res 13 (2011): 88.
- Physical Activity Guidelines for Americans. U.S. Department of Health and Human Services (2008).
- Lloyd-Jones DM, Hong Y, Labarthe D, et al. Defining and setting national goals for cardiovascular health promotion and disease reduction: the American Heart Association's strategic Impact Goal through 2020 and beyond. Circulation 121 (2010): 586-613.
- Badilini F, Pagani M, Porta A. Heartscope: a software tool adressing autonomic nervous system regulation. Computers in Cardiology 32 (2005): 259-262.
- Pagani M, Lombardi F, Guzzetti S, et al. Power spectral analysis of heart rate and arterial pressure variabilities as a marker of sympatho-vagal interaction in man and conscious dog. Circ Res 59 (1986): 178-193.
- Pagani M, Somers V, Furlan R, et al. Changes in autonomic regulation induced by physical training in mild hypertension. Hypertension 12 (1988): 600-610.
- Hess WR. Nobel Lecture: The Central Control of the Activity of Internal Organs. Nobelprize.org. Nobel Media AB (2014).
- Richardson JT. Eta squared and partial eta squared as measures of effect size in educational research. Educational Research Review 6 (2011): 135-147.
- Lucini D, Solaro N, Pagani M. May autonomic indices from cardiovascular variability help identify hypertension? J Hypertens 32 (2014): 363-373.
- Vigo C, Gatzemeier W, Sala R, et al. Evidence of altered autonomic cardiac regulation in breast cancer survivors. J Cancer Surviv 9 (2015): 699-706.
- Scott JM, Jones LW, Hornsby WE, et al. Cancer therapy-induced autonomic dysfunction in early breast cancer: implications for aerobic exercise training. Int J Cardiol 171 (2014): 50-51.
- Ondicova K, Mravec B. Role of nervous system in cancer aetiopathogenesis. Lancet Oncol 11 (2010): 596-601.
- Demark-Wahnefried W, Peterson BL, Winer EP, et al. Changes in weight, body composition, and factors influencing energy balance among premenopausal breast cancer patients receiving adjuvant chemotherapy. J Clin Oncol 19 (2001): 2381-2389.
- Lucini D, Pagani M. From stress to functional syndromes: an internist's point of view. European Journal of Internal Medicine 23 (2012): 295-301.
- Anthony FY, Jones LW. Breast cancer treatment-associated cardiovascular toxicity and effects of exercise countermeasures. Cardio-Oncology 2 (2016): 1.
- Park NJ, Chang Y, Bender C, et al. Cardiovascular disease and mortality after breast cancer in postmenopausal women: Results from the Women’s Health Initiative. PLoS One 12 (2017): 0184174.
- Valachis A, Nilsson C. Cardiac risk in the treatment of breast cancer: assessment and management. Breast Cancer (Dove Med Press) 7 (2015): 21-35.
- Haque R, Shi J, Schottinger JE, et al. Cardiovascular disease after aromatase inhibitor use. JAMA oncology 2 (2016): 1590-1597.
- Braithwaite RS, Chlebowski RT, Lau J, et al. Meta-analysis of Vascular and Neoplastic Events Associated with Tamoxifen. Journal of general internal medicine 18 (2003): 937-947.
- Early Breast Cancer Trialists' Collaborative Group. Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. The Lancet 365 (2005): 1687-1717.
- Buzdar A, Howell A, Cuzick J et al. Comprehensive side-effect profile of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: long-term safety analysis of the ATAC trial. Lancet Oncol 7 (2006): 633-643.
- Abdel-Qadir H, Amir E, Fischer HD, et al. The risk of myocardial infarction with aromatase inhibitors relative to tamoxifen in post-menopausal women with early stage breast cancer. Eur J Cancer 68 (2016): 11-21.
- Foglietta J, Inno A, de IF, et al. Cardiotoxicity of Aromatase Inhibitors in Breast Cancer Patients. Clin Breast Cancer 17 (2017): 11-17.
- Cuzick J, Sestak I, Baum M, et al. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 10-year analysis of the ATAC trial. Lancet Oncol 11 (2010): 1135-1141.
- Brotman DJ, Golden SH, Wittstein IS. The cardiovascular toll of stress. Lancet 370 (2007): 1089-1100.
- Lucini D, Marchetti I, Spataro A, et al. Heart rate variability to monitor performance in elite athletes: Criticalities and avoidable pitfalls. Int J Cardiol 240 (2017): 307-312.
- Lucini D, Zuccotti GV, Scaramuzza A, et al. Exercise might improve cardiovascular autonomic regulation in adolescents with type 1 diabetes. Acta Diabetol 50 (2013): 341-349.
- Lucini D, Milani RV, Costantino G, et al. Effects of cardiac rehabilitation and exercise training on autonomic regulation in patients with coronary artery disease. Am Heart J 143 (2002): 977-983.
- Lee MH, Kim DH, Yu HS. The effect of guided imagery on stress and fatigue in patients with thyroid cancer undergoing radioactive iodine therapy. Evidence-Based Complementary and Alternative Medicine (2013).


 Impact Factor: * 3.5
Impact Factor: * 3.5 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 14.80%
Acceptance Rate: 14.80%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 