Endometrial Biopsy in the Luteal Phase Prior to IVF and ICSI: A Prospective Case Control Study
Article Information
Goeckenjan M1*, Glaß K1, Kramer M1, Hartig A2, Trinkaus I1, Wimberger P1
1Department for Gynecology and Obstetrics, Technical University Dresden, University Hospital, Dresden, Germany
2Institute for Pathology, Technical University Dresden, University Hospital, Dresden, Germany
*Corresponding Author: Goeckenjan M, Department for Gynecology and Obstetrics, Technical University Dresden, University Hospital, Fetscherstr. 74, 01307 Dresden, Germany
Received: 30 April 2020; Accepted: 11 May 2020; Published: 09 June 2020
Citation:
Goeckenjan M, Glaß K, Kramer M, Hartig A, Trinkaus I, Wimberger P. Endometrial Biopsy in the Luteal Phase Prior to IVF and ICSI: A Prospective Case Control Study. Obstetrics and Gynecology Research 3 (2020): 132-144.
View / Download Pdf Share at FacebookAbstract
Aims: The diagnostic and therapeutic impact of endometrial biopsy in the luteal phase of the preceding cycle before artificial reproductive technology (ART) was determined in a prospective case control study.
Methods: From 1/2017 until 8/2018 all women treated with ART were asked to participate in the study at a University Fertility Center. 115 endometrial biopsies were performed prior to controlled ovarian stimulation. The endometrial tissue was analyzed immunohistochemically for hCG and CD138 positive plasma cells (CD138+). The results of 84 ART cycles after endometrial biopsy were compared to 204 controls.
Results: The clinical pregnancy rates after or without endometrial biopsy did not differ significantly (35.7 vs. 39.2%, p=0.578). Lower live birth rates were seen after intervention but this difference was not significant (23.8% vs. 32.4%, p=0.150). In 19/84 samples of endometrial tissue CD138+-plasma cells were detected (22.6%). In this group the clinical pregnancy rate was lower than in the group without signs of inflammation (26.3 vs. 38.5%, p=0.337).
Conclusions: A therapeutic benefit of endometrial biopsy in the midluteal phase of the preceding menstrual cycle was not proven for the following ART cycle in this prospective study. Women with signs of chronic endometritis with CD138+-plasma cells showed a reduced clinical pregnancy rate. The detection of CD138+-plasma cells as a marker for impaired implantation should be investigated in larger studies as a routine diagnostic work-up for infertility prior to ART.
Keywords
ART, CD138, Chronic Endometritis, Endometrial Biopsy, hCG
ART articles ART Research articles ART review articles ART PubMed articles ART PubMed Central articles ART 2023 articles ART 2024 articles ART Scopus articles ART impact factor journals ART Scopus journals ART PubMed journals ART medical journals ART free journals ART best journals ART top journals ART free medical journals ART famous journals ART Google Scholar indexed journals CD138 articles CD138 Research articles CD138 review articles CD138 PubMed articles CD138 PubMed Central articles CD138 2023 articles CD138 2024 articles CD138 Scopus articles CD138 impact factor journals CD138 Scopus journals CD138 PubMed journals CD138 medical journals CD138 free journals CD138 best journals CD138 top journals CD138 free medical journals CD138 famous journals CD138 Google Scholar indexed journals Chronic Endometritis articles Chronic Endometritis Research articles Chronic Endometritis review articles Chronic Endometritis PubMed articles Chronic Endometritis PubMed Central articles Chronic Endometritis 2023 articles Chronic Endometritis 2024 articles Chronic Endometritis Scopus articles Chronic Endometritis impact factor journals Chronic Endometritis Scopus journals Chronic Endometritis PubMed journals Chronic Endometritis medical journals Chronic Endometritis free journals Chronic Endometritis best journals Chronic Endometritis top journals Chronic Endometritis free medical journals Chronic Endometritis famous journals Chronic Endometritis Google Scholar indexed journals Endometrial Biopsy articles Endometrial Biopsy Research articles Endometrial Biopsy review articles Endometrial Biopsy PubMed articles Endometrial Biopsy PubMed Central articles Endometrial Biopsy 2023 articles Endometrial Biopsy 2024 articles Endometrial Biopsy Scopus articles Endometrial Biopsy impact factor journals Endometrial Biopsy Scopus journals Endometrial Biopsy PubMed journals Endometrial Biopsy medical journals Endometrial Biopsy free journals Endometrial Biopsy best journals Endometrial Biopsy top journals Endometrial Biopsy free medical journals Endometrial Biopsy famous journals Endometrial Biopsy Google Scholar indexed journals hCG articles hCG Research articles hCG review articles hCG PubMed articles hCG PubMed Central articles hCG 2023 articles hCG 2024 articles hCG Scopus articles hCG impact factor journals hCG Scopus journals hCG PubMed journals hCG medical journals hCG free journals hCG best journals hCG top journals hCG free medical journals hCG famous journals hCG Google Scholar indexed journals menstrual cycle articles menstrual cycle Research articles menstrual cycle review articles menstrual cycle PubMed articles menstrual cycle PubMed Central articles menstrual cycle 2023 articles menstrual cycle 2024 articles menstrual cycle Scopus articles menstrual cycle impact factor journals menstrual cycle Scopus journals menstrual cycle PubMed journals menstrual cycle medical journals menstrual cycle free journals menstrual cycle best journals menstrual cycle top journals menstrual cycle free medical journals menstrual cycle famous journals menstrual cycle Google Scholar indexed journals reproductive technologies articles reproductive technologies Research articles reproductive technologies review articles reproductive technologies PubMed articles reproductive technologies PubMed Central articles reproductive technologies 2023 articles reproductive technologies 2024 articles reproductive technologies Scopus articles reproductive technologies impact factor journals reproductive technologies Scopus journals reproductive technologies PubMed journals reproductive technologies medical journals reproductive technologies free journals reproductive technologies best journals reproductive technologies top journals reproductive technologies free medical journals reproductive technologies famous journals reproductive technologies Google Scholar indexed journals embryo transfer articles embryo transfer Research articles embryo transfer review articles embryo transfer PubMed articles embryo transfer PubMed Central articles embryo transfer 2023 articles embryo transfer 2024 articles embryo transfer Scopus articles embryo transfer impact factor journals embryo transfer Scopus journals embryo transfer PubMed journals embryo transfer medical journals embryo transfer free journals embryo transfer best journals embryo transfer top journals embryo transfer free medical journals embryo transfer famous journals embryo transfer Google Scholar indexed journals endometrial articles endometrial Research articles endometrial review articles endometrial PubMed articles endometrial PubMed Central articles endometrial 2023 articles endometrial 2024 articles endometrial Scopus articles endometrial impact factor journals endometrial Scopus journals endometrial PubMed journals endometrial medical journals endometrial free journals endometrial best journals endometrial top journals endometrial free medical journals endometrial famous journals endometrial Google Scholar indexed journals fertility articles fertility Research articles fertility review articles fertility PubMed articles fertility PubMed Central articles fertility 2023 articles fertility 2024 articles fertility Scopus articles fertility impact factor journals fertility Scopus journals fertility PubMed journals fertility medical journals fertility free journals fertility best journals fertility top journals fertility free medical journals fertility famous journals fertility Google Scholar indexed journals
Article Details
1. Introduction
Almost 20 years ago the first studies were published suggesting a benefit of endometrial injury in the menstrual cycle prior to controlled ovarian stimulation for outcome parameters of artificial reproductive technologies (ART) in the following cycle [1]. Up to now the possible effect of the endometrial biopsy with injury of endometrium, “scratching”, prior to in vitro fertilization (IVF) or intracytoplasmatic sperm injection (ICSI) has been discussed extensively. Many authors stated that the endometrial biopsy in the preceding cycle or even the same cycle before embryo transfer could improve the receptivity of the endometrium in ART cycles [2]. In 2016 the procedure was used in several countries all over the world even without a proven benefit [3]. The majority of fertility centers performed the endometrial biopsy in the luteal phase of the preceding cycle before ART cycles with controlled ovarian stimulation (COS). In 2015 a Cochrane meta-analysis of 14 randomized controlled studies (RCT) in women with a history of implantation failure and in unselected women performing ART cycles showed that the procedure is safe and does not lead to impaired live birth rates [4].
The endometrial biopsy in the luteal menstrual phase can be used for the pathomorphological and immunohistochemical detection of a sufficient secretory phase. There is an ongoing debate which biochemical markers can predict an individual receptivity at the time of implantation. Individual patterns of the transcriptom influence the implantation [5]. It can be assumed that an ovulatory menstrual cycle which precedes the stimulation cycle with ART could improve the receptivity of the endometrium [6]. The detection of endometrial hCG was suggested as a differential marker for a sufficient luteal phase after ovulation and for the synchronous endocrine regulation of the menstrual physiology [7, 8]. The diagnostic approach of the study included the detection of a sufficient luteal phase with histological assessment and simultaneous measurement of endogenous endometrial hCG using standard immunohistochemical kits. The phase of the menstrual cycle can be clearly dated with histological criteria of the endometrial tissue after hematoxylin and eosin staining (H&E). Typical histological criteria for the luteal phase are [9].
- Basal vacuoles in the early secretory phase.
- Maximum of stromal edema in the mid luteal phase.
- Increasing vascularization in the late secretory phase.
- Increasing saw-blade-like curling of the glands.
- Glycogen in the lumen of the acini in the late secretory phase.
- Arteries surrounded by predecidual cells.
Chronic endometritis or a dysbalance in endometrial microbiome are discussed as important factors for infertility [10]. The effect of cervical and endometrial inflammation on implantation and pregnancy outcome was described almost 150 years ago: Under the title “On chronic cervical endometritis as a frequently overlooked cause of sterility and abortions” published 1878 in the British Medical Journal the author stated, that even without symptoms and clinical signs of infection, the inflammation of the endometrium can be a reason for infertility and miscarriages [11]. The typical histopathological signs of chronic endometritis in H&E staining are superficial mucosal stromal edema, increased stromal density with leukocytic infiltrate of glands and stroma, and plasma cells within the stroma. In addition to these diagnostic signs with low sensitivity and specificity the immunohistochemical staining with Syndecan-1 (CD138) increases the diagnostic concordance between pathologists [12, 13]. The systematic CD138+-plasma cell assessment as a diagnostic procedure for infertility was proposed by Liu et al. [14]. Because clinical studies have shown that endometrial biopsy in the preceding cycle prior to COS in unselected cycles as well as in cycles after repetitive implantation failure does not harm and may even have benefits, we planned this prospective case control study. The additional goal of the study was to examine the possible diagnostic value of endometrial biopsy with immunohistochemical detection in the clinical routine for hCG and CD138+-plasma cells in the endometrium. The treatment of a chronic endometritis was not part of the study protocol. At the time of the study, the diagnosis of chronic endometritis and the possible treatment have not been recommended for ART cycles routinely [15].
2. Methods
This study was conducted as a prospective case control study at a single-center in a University Fertility Center. The study protocol was approved by the local ethics committee (EK 68022015). The study protocol included all infertile women before ART cycles were performed. They were offered endometrial biopsy at day 20 of the menstrual cycle or 7 days after confirmed ovulation. Inclusion criteria were planned ART treatment with autologous oocytes, age of the treated women of 20-45 years, and written informed consent to the study protocol. All women regardless of previous cycles or diagnosis of repetitive implantation failure were asked, but each woman was included into the study only once. The results of the immunohistochemical testing for hCG and CD138 did not result in a change of treatment. The standard intervention was the endometrial biopsy with the single use catheter as previously described [16]. After disinfection of the vagina and external cervical os the endometrial biopsy catheter (Pipelle®) is inserted into the uterus. After insertion of the catheter, the inner cannula is pulled back to create a suction. With suction, the catheter is slowly turned and removed out of the uterine cavity. The procedure may be performed three times in maximum to obtain viable endometrial tissue. If the catheter cannot be inserted into the uterine cavity the uterus can be hooked and dragged into the vagina. The tissue samples were fixed in formaldehyde and embedded in paraffin. After cutting the blocked tissue into 4 µm serial tissue slices, they were prepared for H&E staining as a standard procedure in Benchmark XT and prepared for immunohistochemical staining. The sample size for the study was planned for 100 women with endometrial biopsy with 10% drop out rate and number of controls in an allocation of 1:2. Starting from January 2017 until August 2018, 328 cycles of IVF/ICSI were performed in the study period. 115 patients were included in the intervention group with endometrial biopsy. 84 ART cycles after biopsies with proven endometrial tissue in the preceding menstrual cycles with embryo transfer were analyzed in comparison to 204 cycles of IVF/ICSI without endometrial biopsy (Table 1). Figure 1 shows the flow chart of inclusion to the study.
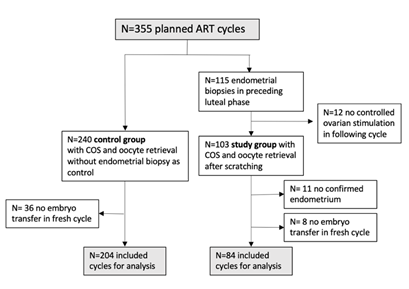
Figure 1: Flow chart of inclusion to the study and control group.
The hCG staining was performed as previously described by Zimmermann et al. [17]. The endometrial tissue specimens were stained with two different polyclonal antibodies for hCG as well as on slide controls of early placental tissue (DAKO A0231, Cell Marque, 234A-15). After heat preparation with CC1 mild for 32 minutes, the antibody staining with DAKO-antibodies in a dilution of 1:500 or Cell Marque 2234A-15 in a dilution of 1:400 were used for 28 minutes. After the primary peroxidase reaction, the ultraview DAB kit was applied for 8 min, H&E-staining followed for 4 min and bluing reagent for 4 min. All reagents were obtained from Ventana/Roche.
The immunohistochemical staining for CD138 was used in a similar protocol. As primary outcome parameters of the diagnostic part of the study, the detection of endogenous hCG and immunohistochemical detection of CD138+-plasma cells in the endometrial probe were defined. Primary endpoints of the safety and outcome parameter after endometrial biopsy were defined as clinical pregnancy rate, live birth rate, rate of ectopic pregnancy and miscarriage. The statistical analysis was performed using GraphPad Software version 7.0 for Windows, La Jolla California USA, www.graphpad.com. Categorical variables are presented as percentages compared via the chi-square test. Continuous variables are presented as mean standard deviation. The normality of the distribution of continuous variables was tested with the Shapiro-Wilk test. Levene´s test was used for equal variances. The unpaired t-test was performed. Group differences were considered to be significant at a P value of <0.05. The Ferring GmbH supported the study by a research funding grant.
3. Results
During the study period 355 planned ART cycles after COS were performed. According to the study protocol at day 20 of the preceding cycle or 7 days after confirmed ovulation, we included 115 women into the study and performed endometrial biopsy. The two subgroups for statistical analysis were defined as: controlled ovarian stimulation after confirmed endometrial biopsy in the preceding luteal phase and embryo transfer in the COS cycle. The data of the ART cycles in the study group compared to the control group without intervention are shown in Table 1.
The two groups did not differ significantly by mean age of treated women, number of treatment cycle, treatment either with IVF or ICSI and the number of transferred embryos. Controlled ovarian stimulation was performed in a standardized way. Due to the inclusion to the study at day 20 of the preceding menstrual cycle significantly more planned controlled ovarian stimulation cycles with the long protocol were performed in the study group than in the control group (75.0 vs. 54.4%, p=0.001). After transvaginal ultrasound-guided follicular aspiration the oocyte retrieval was performed with IVF (36.9%) or ICSI (63.1%). Only embryo transfers at day 2-3 were performed during the study period due to local legal regulations.
3.1 Safety parameters of endometrial biopsy in the preceding menstrual cycle before ART-treatment
In the group of women with or without endometrial biopsy and fresh embryo transfer in the subsequent cycle, we analyzed as parameters of safety and efficacy the clinical pregnancy rate, live birth rate, rate of ectopic pregnancy and miscarriage (Table 1). Even though the rate of pregnancy loss was 33.3% after endometrial biopsy vs. 17.5% in the control group, the possibly elevated risk of miscarriage in the study group was not statistically significant (p=0.070). The live birth rate in both groups did not differ significantly with 23.8% in the group after endometrial biopsy and 32.4% in the group without intervention (p=0.150). The risk of ectopic pregnancies was elevated in the study group (p=0.007).
3.2 Results of hCG-immunohistochemical staining
Of all performed endometrial biopsies (n=115), 103 samples showed histologically confirmed endometrial tissue (89.6%). 60/84 (71.4%) of the samples featured the typical pathomorphological criteria of secretory transformation in the luteal phase (Figure 2). All probes with confirmed endometrial tissue were prepared and examined for immunohistochemical staining with two different polyclonal antibodies for hCG (Cell Marque 234A-15, DAKO A0231). In contrast to previous studies (Zimmermann et al. 2009 and 2011), it was not possible to detect the endometrial hCG in the setting of this study. Even in the probes with histological signs of late secretory phase no endometrial endogenous hCG was found (Figure 3 a-d). At the day of endometrial biopsy serum progesterone was analyzed. 38 women had values higher than 31,8 nmol/l (=10 ng/ml), which can be interpreted as a sufficient luteal phase [18]. Endometrial tissue of these 38 women with confirmed luteal phase with pathomorphological timing of the cycle phase, as well as sufficient serum progesterone, also not show endometrial hCG.
3.3 Results of CD138 immunohistochemical staining
Of 84 samples of endometrial tissues the detection of CD138+-plasma cells was positive in 19 cases, with a mean section area of 36.57 mm2 (1 SD = 20.62 mm2). The Figure 4 depicts an example of the positive staining of CD138+-plasma cells. The detection of plasma cells in the H&E staining did not correlate in all cases with the immunohistochemical detection of CD138+-plasma cells. The Table 2 shows the results of plasma cell detection via H&E staining, CD138 immunohistochemistry, and clinical pregnancy rate in the study groups. It has to be mentioned, that no treatment was administered even after detection of positive plasma cells in H&E staining, as at the time of the study a treatment of chronic endometritis was not performed routinely in the study center. Therefore, clinical pregnancy rates after detection of plasma cells were analyzed without intervention.
|
Study group with endometrial biopsy before ART (n=84) and embryotransfer. Mean, SD (Min-Max) |
Control group without endometrial biopsy with embryotransfer after ART (n=204). Mean, SD (Min-Max) |
p-value |
|
|
Mean age (years) |
34.0 ± 3.8 (25-40) |
34.8 ± 3.8 (25-43) |
0.140 |
|
Treatment cycle |
1.6 ± 0.9 (1-5) |
1.8 ± 0.9 (1-5) |
0.810 |
|
Long agonist protocol |
63/84 (75.0%) |
11/204 (54.4%) |
0.001 |
|
ICSI |
53/84 (63.1%) |
109/204 (53.4%) |
0.133 |
|
Number of embryos per transfer at day 2 or 3 |
1.82 ± 0.52 (153/84) |
1.88 ± 0.45 (384/204) |
0.314 |
|
Clinical pregnancy rate/ embryo transfer |
30/84 (35.7%) |
80/204 (39.2%) |
0.578 |
|
Early pregnancy loss/ pregnancy |
10/30 (33.3%) of those ectopic pregnancy 4/30 |
14/80 (17.5%) of those ectopic pregnancy 1/80 |
0.070 0.007 |
|
Live birth/ embryo transfer |
20/84 (23.8%) |
66/204 (32.4%) |
0.150 |
Table 1: Characterization of study group (n=84) in comparison to control group (n=204, p-value after unpaired t-test, chi-square test).
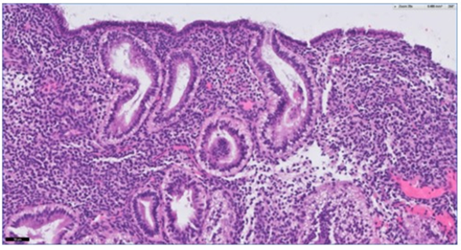
Figure 2: Endometrial biopsy after H&E staining, 20x zoom, markers of secretory transformation (elongated acini, glycogen in retronuclear vacuoles, increased vascularization).
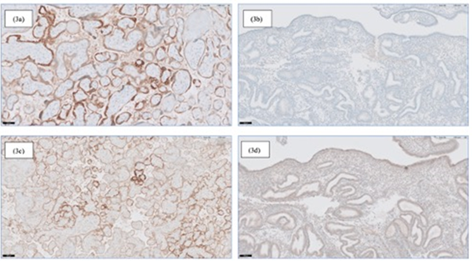
Figure 3 (a-d): Immunohistochemical staining with polyclonal antibody (Cell Marque 234A-15): on slide controls with placental tissue, 20x magnification (3a) and endometrial tissue (10x magnification) (3b). Immunohistochemical staining with polyclonal antibody (DAKO-A0231): on slide controls with placental tissue, 10x magnification (3c) and endometrial tissue (10x magnification) (3d).
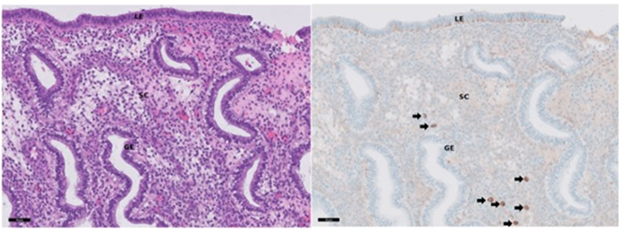
Figure 4: Immunohistochemical detection of CD138, endometrial biopsy with H&E-staining (4a), CD138 (4b): LE luminal epithelium, GE glandular epithelium, 138+-plasma cells.
|
CD138+ cells: >2/sample/probe |
CD138- cells |
||
|
Plasma cells after HE staining detected |
3 Clinical pregnancies: 1/3 (33.3%) |
7 Clinical pregnancies: 3/7 (42.8%) |
4/10 (40%) |
|
No plasma cells after HE staining detected |
16 Clinical pregnancies: 4/16 (25.0%) |
58 Clinical pregnancies: 22/58 (38.0%) |
26/74 (35.1%) |
|
5/19 (26.3%) |
25/65 (38.5%) |
p=0.331 p=0.763 |
Table 2: Detection of plasma cells either after H&E staining or after CD138 immunohistochemistry and correlation to pregnancy after ART cycles (n=84, p-value after chi-square test).
4. Discussion
This prospective interventional study was conducted to examine the effect of endometrial biopsy prior to IVF/ICSI for diagnostic purposes and possible therapeutic benefits. At our University Fertility Center, the endometrial biopsy as “endometrial scratching” in the midluteal phase was offered to 355 women before planned controlled ovarian stimulation and ART regardless of the number of preceding embryo transfers. The design of the study was based on randomized controlled studies which showed that endometrial biopsy might lead to improved pregnancy and birth rates after ART in the following menstrual cycle [4]. Newer studies show that endometrial biopsy does not lead to a benefit but also no harm prior to ART treatment cycles even in an unselected group [19]. This study could also not detect any therapeutic benefit after endometrial biopsy before ART cycles.
Clinical pregnancy rates of 35.7% in the intervention group vs. 39.2% in the control group (p=0.578) were statistically comparable. Nevertheless, early pregnancy loss rates (33.3% vs. 17.5%, p= 0.070) and birth rates (23.8 vs. 32.4%, p=0.150) showed a negative effect of endometrial biopsy compared to the control group, but this effect did not show statistical significance. The number of ectopic pregnancies was unexpectedly high in the interventional group (4/30 vs. 1/80, p=0.007). Ectopic pregnancies after ART are rare. In a retrospective analysis of more than 18 000 pregnancy following ART the incidence rate of ectopic pregnancy is calculated for cleavage stage embryo transfers with a risk of 3.5% [20]. In this study and another multicenter study with more than 160 000 pregnancies after ART [21], tubal infertility is the predominant risk factor. The endometrial biopsy in a preceding cycle has not been discussed as a risk factor yet. Therefore, the elevated risk of ectopic pregnancies after endometrial biopsy in this study is likely to be explained by the low number of included patients and the lack of randomization. The RCT of endometrial biopsy performed prior to first or repeated ART cycles published 2014 by Yeung [22] showed no significant differences in clinical outcome parameters: ongoing pregnancy rates were 26.7% (40/150) vs. 32% (48/150) in the group with prior endometrial biopsy compared to the control group without intervention (p=0.375) and miscarriage rates were 30.3% (17/56) vs. 18.6% (11/59). These results correspond to the results of our study with impaired but not statistically significant reduced clinical outcome parameters. Currently all current studies show that endometrial biopsy as scratching prior to ART has no benefit in unselected cycles, and even in cycles after repetitive implantation failure no benefit can be shown analyzing the pooled data of 14 RCTs [23]. The reduced birth rate in our study – even though not statistically significant – corresponds to a recently published RCT in which the birth rate is also lower after scratching [24]. A French RCT with endometrial scratching in women undergoing a first or second ART cycle was stopped prematurely due to impaired clinical pregnancy rates in the year 2014, the results of the study were published 2019 [25]. In contrast to many RCTs with endometrial biopsy as scratching prior to ART, the purpose of the endometrial biopsy in this study was the retrieval of endometrial tissue for diagnostic purposes. Only cycles with proven endometrial tissue were included in the analysis of pregnancy and birth rates after ART in this study. In 11 of 103 biopsies no endometrial tissue was detected (10.7%). In some RCTs on the endometrial biopsy prior to ART the intervention was not standardized by the histological proof of endometrium, this may impair the results [23].
The diagnostic approach of the study protocol using endometrial biopsy and performing pathomorphological and immunohistochemical tests was designed to answer the following questions:
- Is endometrial hCG a diagnostic parameter to detect a sufficient luteal phase in the preceding cycle?
- Can endometrial hCG be used as a prognostic marker for the success of an ART cycle?
- Is the diagnosis of chronic endometritis in the preceding luteal phase prior to IVF/ICSI useful to predict the success of an ART cycle in unselected infertile women?
It was not possible to detect hCG in the endometrial tissue retrieved at the 20th day of the preceding menstrual cycle prior to IVF/ICSI. Routine standard methods for immunohistochemical detection of hCG were used. Even in the subgroup of infertile women with proven ovulation and high serum progesterone at the time of endometrial biopsy no endometrial hCG was confirmed. It may be assumed that the biopsy at the midluteal cycle phase at day 20 of the menstrual cycle was too early to detect endometrial hCG. Zimmermann et al. 2012 showed that the intensity of immunohistochemical detection of hCG increases with the interval to ovulation [7]. In the late luteal phase of the menstrual cycle, corresponding to the late secretory phase of the endometrium, and especially in the predecidual late secretory phase, the expression of hCG was consolidated. Since the protocol for the study used the most frequently proposed time of endometrial biopsy for the purpose of endometrial “scratching” prior to ART at day 20 in the luteal phase, this may be a reason for the failure to detect endometrial hCG in the setting of this clinical study. This study could not prove that endometrial endogenous hCG may serve as a marker for sufficient luteal phase or implantation. The second diagnostic approach of the endometrium biopsy prior to controlled ovarian stimulation and IVF/ICSI was the detection or exclusion of chronic endometritis. This diagnostic procedure is proposed for infertile women with repetitive implantation failure and repetitive miscarriages [26, 27]. But recently the focus on microbiome and chronic endometritis is of growing interest for every infertile woman. As the most sensitive method to detect chronic endometritis the immunohistochemical staining for CD138 and counting of CD138+-plasma cells has been established [14].
In our study with 103 endometrial biopsies of infertile women without selection prior to ART, a prevalence of 22.6% of elevated CD138+-plasma cells > 2 per tissue or plasma cells in H&E was found. This corresponds to a review on the possible effect of endometrial infection or inflammation on implantation and pregnancy, in which incidence rates of 15-40% are described in infertile women [28]. Park et al. 2016 described a prevalence rate of 10% in infertile women [29]. Up to now the cut-off for CD138+ plasma cells regarding the diagnosis of chronic endometritis with impact on fertility are still not clearly defined. Liu et al. 2018 propose to use the cell density (cells/10mm2) in order to reduce the variability of results due to sample size [14]. We decided to define more than 2 plasma cells per tissue section as the diagnostic cut-off in this study, but for implementation in routine diagnosis the limits have to be defined in a larger study group including healthy and fertile women. The treatment of chronic endometritis is not standardized. Only a few retrospective and observational studies are published. The German groups which offer endometrial diagnostics with CD138, e.g. University Hospital Jena and Mannheim, have not published the treatment data yet. In women with repetitive implantation failure, it is proposed to treat the chronic endometritis prior to IVF/ICSI with antibiotics – as first-line treatment with doxycycline 200 mg for 14 days, as second-line metronidazole in combination with ciprofloxacin [30].
In retrospective studies, antibiotic treatment results in the resolution of inflammation in about 80-90% and leads to significantly higher pregnancy and live birth rates [30, 31]. As the antibiotic treatment of chronic endometritis has not been evaluated in prospective studies for unselected infertile women without repetitive implantation failure, the women in our study were not treated with antibiotics. The low pregnancy and birth rates of untreated women with confirmed chronic endometritis are in line with other retrospective studies. The strength of the study is the prospective design with a standardized protocol. The endometrial scratching with endometrial injury was confirmed by the histological examination of the tissue. The limitation of the study is the small number of included patients and the study concept as a single center study without randomization. A sample size calculation on the basis of 80% power for a 10% difference in pregnancy rates would result in 376 women in each group, but this was not realistic for a single center study. Chronic endometritis and intrauterine microbiome are promising approaches for treating infertile women. Current studies have resulted in the recommendation to avoid endometrial biopsy in the preceding menstrual cycle prior to ART cycles (Eskew et al. 2019) because of the lacking positive effect. Until now, the diagnostic procedure of endometrium biopsy to exclude chronic endometritis should be performed in every woman with repetitive implantation failure and probably in every woman with infertility.
The endometrial biopsy should be performed as a diagnostic workup for infertility, but not as an intervention to increase pregnancy and birth rates after IVF/ICSI. But it is important to consider that endometrial biopsy may itself increase the risk of inflammatory endometrial reactions and chronic endometritis. Every intrauterine intervention, either as a diagnostic procedure or a therapeutic measure with embryo transfer may itself cause inflammation.
5. Conclusion
A therapeutic benefit of endometrial biopsy in the midluteal phase of the preceding menstrual cycle was not proven for the following ART cycle in this prospective study. Endogenous endometrial hCG could not be detected in the midluteal phase by routine immunohistochemistry. Women with signs of chronic endometritis with CD138+-plasma cells showed a reduced clinical pregnancy rate. The detection of CD138+-plasma cells as a marker for impaired implantation should be investigated in larger studies as a routine diagnostic work-up for infertility prior to ART.
Acknowledgement
We thank Ferring GmbH for the financial grant. We thank Prof. Henry Alexander for the contribution to the study concept and his personal support.
Disclosure
The study was funded by a research grant of Ferring GmbH. All authors stated that they do not have further industrial links and affiliations with the topic of the study.
References
- Barash A, Dekel N, Fieldust S, et al. Local injury to the endometrium doubles the incidence of successful pregnancies in patients undergoing in vitro fertilization. Fertil Steril 89 (2003): 1317-1322.
- Li R, Hao G. Local injury to the endometrium: its effect on implantation. Curr Opin Obstet Gynecol 21 (2009): 236-239.
- Lensen S, Sadler L, Farquhar C. Endometrial scratching for subfertility: everyone’s doing it. Hum Reprod 31 (2016): 1241-1244.
- Nastri CO, Lensen SF, Gibreel A, et al. Endometrial injury in women undergoing assisted reproductive techniques. Cochrane Database Syst Rev (2015): CD009517.
- Diaz-Gimeno P, Ruiz-Alonso M, Sebastian-Leon P, et al. Window of implantation transcriptomic stratification reveals different endometrial subsignatures associated with live birth and biochemical pregnancy. Fertil Steril 108 (2017): 703.e3-710.e3.
- Strowitzki T, Germeyer A, Popovici R, et al. The human endometrium as a fertility-determining factor. Hum Reprod Update 12 (2006): 617-630.
- Zimmermann G, Ackermann W, Alexander H. Expression and production of human chorionic gonadotropin /hCG) in the normal secretory endometrium: evidence of CGB7 and/or CGB6 beta hCG subunit gene expression. Biol Reprod 88 (2012): 87.
- Schug S, Baunacke A, Goeckenjan M, et al. Endometrial human chorionic gonadotropin (hCG) expression is a marker for adequate secretory transformation of the endometrium. Arch Gynecol Obstet (2019).
- Dallenbach-Hellweg, Gisela: Endometrium. Pathologische Histologie in Diagnostik und Forschung, 2. Ausgabe (1981).
- Bracewell-Milnes T, Saso S, Nikolaou D, et al. Investigating the effect of an abnormal cervico-vaginal and endometrial microbiome on assisted reproductive technologies: A systematic review. Am J Reprod Immunol 80 (2018): e13037.
- Edis AW. On chronic cervical endometritis as a frequently overlooked cause of sterility and abortion. BMJ 2 (1878): 717-718.
- Bayer-Garner IB, Korourian S. Plasma cells in chronic endometritis are easily identified when stained with syndecan-1. Mod Pathol 14 (2001): 877-879.
- Kasius JC, Broekmans FJ, Sie-Go DM, et al. The reliability of the histological diagnosis of endometritis in asymptomatic IVF cases: a multicenter observer study. Hum Reprod 27 (2012): 153-158.
- Liu Y, Chen X, Huang J, et al. Comparison of the prevalence of chronic endometritis as determined by means of different diagnostic methods in women with and without reproductive failure. Fertil Steril 109 (2018): 832-839.
- Crosignani PG, Rubin BL. Optimal use of infertility diagnostic tests and treatments. The ESHRE Capri Workshop Group. Hum Reprod 15 (2000): 723-732.
- Lensen S, Martin W, Nastri C, et al. Pipelle for pregnancy (PIP): study protocols for three randomized controlled trials. Trials 17 (2016): 216.
- Zimmermann G, Ackermann W, Alexander. Epithelial human chorionic gonadotropin is expressed and produced in human secretory endometrium during the normal menstrual cycle. Biol Reprod 80 (2009): 1053-1065.
- Jordan J, Craig K, Clifton DK, et al. Luteal phase defect: the sensitivity and specificity of diagnostic methods in common clinical use. Fertil Steril 62 (1994): 54-62.
- Vitagliano A, Andrisani A, Alviggi C, et al. Endometrial scratching for infertile women undergoing a first embryo transfer: a systematic review and meta-analysis of published and unpublished data from randomized controlled trials. Fertil Steril 111 (2019): 734.e2-746.e2.
- Bu Z, Yiong Y, Wang K, et al. Risk factors for ectopic pregnancy in assisted reproductive technology: a 6-year, single-center study. Fetil Steril 106 (2016): 90-94.
- Santos-Riberio S, Tournaye H, Polyzos NP. Trends in ectopic pregnancy rates following assisted reproductive technologies in the UK: a 12-year nationwide analysis including 160 000 pregnancies. Hum Reprod 31 (2016): 393-402.
- Yeung TW, Chai J, Li RH, et al. The effect of endometrial injury on ongoing pregnancy rate in unselected subfertile women undergoing in vitro fertilization: a randomized controlled trial. Hum Reprod 29 (2014): 2474-2481.
- Van Hoogenhuijze NE, Kasius JC, Broekmans FJM, et al. Endometrial scratching prior to IVF; does it help and for whom? A systematic reviewa nd meta-analysis. Hum Reprod Open 2019 (2019): hoy025.
- Eskew AM, Reschke LD, Wollfolk C, et al. Effect of endometrial mechanical stimulation in an unselected population undergoing in vitro fertilization: futility analysis of a double-blind randomized controlled trial. J Assist Reprod Genet 36 (2019): 299-305.
- Frantz S, Parinaud J, Kret M, et al.. Decrease in pregnancy rate after endometrial scratch in women undergoing a first or second in vitro fertilization. A multicenter randomized controlled trial. Hum Repod 34 (2019): 92-99.
- Cicinelli E, Matteo M, Tinelli R, et al. Prevalence of chronic endometritis in repeated unexplained implantation failure and the IVF success rate after antibiotic therapy. Hum Reprod 30 (2015): 323-330.
- Mc Queen DB, Perfetto CO, Hazard FK, et al. Pregnancy outcomes in women with chronic endometritis and recurrent pregnancy loss. Fertil Steril 104 (2015): 927-931.
- Romero R, Espinoza J, Mazor M. Can endometrial infection/inflammation explain implantation failure, spontaneous abortion, and preterm birth after in vitro fertilization? Fertil Steril 82 (2004): 799-804.
- Park HJ, Kim YS, Yoon TK, et al. Chronic endometritis and infertility. Clin Exp Reprod Med 43 (2016): 185-192.
- Kitaya K, Matsubayashi H, Takaya Y, et al. Live birth rate following oral antibiotic treatment for chronic endometritis in infertile women with repeated implantation failure. Am J Reprod Immunol 78 (2017).
- Cicinelli E, Matteo M, Trojano G, et al. Chronic endometritis in patients with unexplained infertility: Prevalence and effects of antibiotic treatment on spontaneous conception. Am J Reprod Immunol 79 (2018).


 Impact Factor: * 3.2
Impact Factor: * 3.2 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 