Generation of Bio-Electricity from Sewage Sludge Using Single Chamber Microbial Fuel Cell
Article Information
Kumar Sonu*
Department of Mechanical Engineering, Anand International College of Engineering, Jaipur, Rajasthan, India
*Corresponding Author: Kumar Sonu, Department of Mechanical Engineering, Anand International College of Engineering, Near Kanota, Agra Road, Jaipur - 303012, Rajasthan, India, Tel: 01429 234 994;
Received: 03 July 2017; Accepted: 20 July 2017; Published: 02 August 2017
View / Download Pdf Share at FacebookAbstract
A Single Chamber Microbial Fuel Cell (MFC) has been fabricated to generate electricity from the sludge of the sewage treatment plant at Anand International College of Engineering, Jaipur, at two different ambient temperature range of 25 ± 4°C and 32 ± 4°C under aerobic condition. The maximum voltage obtained was about 2890 mV after 80 (hrs.) at temperature range of25 ± 4°C, with the surface power density of 1108.29 mW/m2. When the ambient temperature was 32 ± 4°C, the maximum voltage obtained was 1652 mV after 40 (hrs.) surface power density reduced to 865.57 mW/m2.While changing the amount of substrate for certain area of the electrode at 25 ± 4°C range it showed that the electricity generation decreased with the available substrate and it also shortens the time to peak voltage. On the other way when the ambient temperature increased to 32 ± 4°C, the maximum potential energy generated is less than the previous temperature for the same substrate per unit area of the electrode and also the time to peak voltage decreases to 40 hrs. At the end of the 152 (hrs.), the maximum COD reduction for the sewage sludge was 30% for 32 ± 4°C. When comparing with other single chambers MFC, the present model is generating more electricity that any MFC using sewage sludge as substrate except platinum electrode, which is much costlier that electrode used in the present study.
Keywords
Graphite electrode, Microbial fuel cell, Sewage sludge, Used dry cell
Graphite electrode articles Graphite electrode Research articles Graphite electrode review articles Graphite electrode PubMed articles Graphite electrode PubMed Central articles Graphite electrode 2023 articles Graphite electrode 2024 articles Graphite electrode Scopus articles Graphite electrode impact factor journals Graphite electrode Scopus journals Graphite electrode PubMed journals Graphite electrode medical journals Graphite electrode free journals Graphite electrode best journals Graphite electrode top journals Graphite electrode free medical journals Graphite electrode famous journals Graphite electrode Google Scholar indexed journals Microbial fuel cell articles Microbial fuel cell Research articles Microbial fuel cell review articles Microbial fuel cell PubMed articles Microbial fuel cell PubMed Central articles Microbial fuel cell 2023 articles Microbial fuel cell 2024 articles Microbial fuel cell Scopus articles Microbial fuel cell impact factor journals Microbial fuel cell Scopus journals Microbial fuel cell PubMed journals Microbial fuel cell medical journals Microbial fuel cell free journals Microbial fuel cell best journals Microbial fuel cell top journals Microbial fuel cell free medical journals Microbial fuel cell famous journals Microbial fuel cell Google Scholar indexed journals Sewage sludge articles Sewage sludge Research articles Sewage sludge review articles Sewage sludge PubMed articles Sewage sludge PubMed Central articles Sewage sludge 2023 articles Sewage sludge 2024 articles Sewage sludge Scopus articles Sewage sludge impact factor journals Sewage sludge Scopus journals Sewage sludge PubMed journals Sewage sludge medical journals Sewage sludge free journals Sewage sludge best journals Sewage sludge top journals Sewage sludge free medical journals Sewage sludge famous journals Sewage sludge Google Scholar indexed journals Used dry cell articles Used dry cell Research articles Used dry cell review articles Used dry cell PubMed articles Used dry cell PubMed Central articles Used dry cell 2023 articles Used dry cell 2024 articles Used dry cell Scopus articles Used dry cell impact factor journals Used dry cell Scopus journals Used dry cell PubMed journals Used dry cell medical journals Used dry cell free journals Used dry cell best journals Used dry cell top journals Used dry cell free medical journals Used dry cell famous journals Used dry cell Google Scholar indexed journals sewage articles sewage Research articles sewage review articles sewage PubMed articles sewage PubMed Central articles sewage 2023 articles sewage 2024 articles sewage Scopus articles sewage impact factor journals sewage Scopus journals sewage PubMed journals sewage medical journals sewage free journals sewage best journals sewage top journals sewage free medical journals sewage famous journals sewage Google Scholar indexed journals temperature articles temperature Research articles temperature review articles temperature PubMed articles temperature PubMed Central articles temperature 2023 articles temperature 2024 articles temperature Scopus articles temperature impact factor journals temperature Scopus journals temperature PubMed journals temperature medical journals temperature free journals temperature best journals temperature top journals temperature free medical journals temperature famous journals temperature Google Scholar indexed journals electrode articles electrode Research articles electrode review articles electrode PubMed articles electrode PubMed Central articles electrode 2023 articles electrode 2024 articles electrode Scopus articles electrode impact factor journals electrode Scopus journals electrode PubMed journals electrode medical journals electrode free journals electrode best journals electrode top journals electrode free medical journals electrode famous journals electrode Google Scholar indexed journals Sewage sludge articles Sewage sludge Research articles Sewage sludge review articles Sewage sludge PubMed articles Sewage sludge PubMed Central articles Sewage sludge 2023 articles Sewage sludge 2024 articles Sewage sludge Scopus articles Sewage sludge impact factor journals Sewage sludge Scopus journals Sewage sludge PubMed journals Sewage sludge medical journals Sewage sludge free journals Sewage sludge best journals Sewage sludge top journals Sewage sludge free medical journals Sewage sludge famous journals Sewage sludge Google Scholar indexed journals wastewater articles wastewater Research articles wastewater review articles wastewater PubMed articles wastewater PubMed Central articles wastewater 2023 articles wastewater 2024 articles wastewater Scopus articles wastewater impact factor journals wastewater Scopus journals wastewater PubMed journals wastewater medical journals wastewater free journals wastewater best journals wastewater top journals wastewater free medical journals wastewater famous journals wastewater Google Scholar indexed journals
Article Details
- Introduction
The microbial fuel cell is a device which generates electricity by the metabolic activities of the microbes. Microorganisms transfer the electrons obtained from the metabolism of organic matters in the anode and thereby to the cathode through an external circuit, hence generate electricity [1, 2]. Though a century old technique, which was initially recognized for treatment of wastewater, MFC is gaining its interest for the generation of electricity, bio-hydrogen and also used as biosensor [3, 4]. Any biodegradable organic ranging from pure compounds such as glycerol, acetate, starch, glucose, cysteine, and ethanol [3, 5] to complex mixtures of organic matter such as wastewater [6], cow dung [2], and kitchen waste [6] are the ideal candidates of sustainable energy source for MFC. The performance of microbial fuel cell depends upon the types of electrode materials, microbe & substrate and its concentration, pH, temperature and ionic strength.
The various types of the electrode that can be used in the construction of the microbial fuel cell are graphite; graphite felt, Platinum (Pt.), Pt. black, carbon paper, reticulated vitreous carbon (RVS). The design of MFC may be 1. Two compartment MFC systems consisting of an anodic chamber and catholic chamber separated by proton exchange membrane (PEM) or sometime salt bridge to allowing the proton to move across the blocking the diffusion of oxygen into the anode [3]. 2. Single ?compartment MFC systems consisting of the single anodic chamber without the any aerated catholic chamber [3]. The anode chamber is coupled to a porous air-cathode exposing directly to the air.
The Single?compartment MFC systems is cheaper than that of two compartments MFC. The traditional dual chamber which is bulky due to two separate anode and cathode chambers, the single chamber containing single anode chamber omitting the cathode chamber where the cathode is placed as membrane and exposed to air is much more efficient and cost effective than dual chamber MFC.
Table 1 shows the performance of single chamber microbial fuel cell for the different substrate on the basis of maximum power density. The maximum surface power density of 6000 mW/m2 using platinum electrode modified polyanilineco and sewage sludge was found from the literature available [7]. When the sewage sludge used with graphite electrode 152 mW/m2 maximum surface power density could be achieved [7].
Sl. No. |
Industry/Substrate |
System configuration |
Maximum surface power density |
Refs. |
|
1 |
Sewage sludge |
Electrode: Single chamber (graphite electrode) |
152 mW/m2 |
[7] |
|
Single chamber (Platinum and |
6000 mW/m2 |
[7] |
||
|
2 |
Glycerol -waste water |
Single-chamber |
600 mW/m2 |
[8] |
|
3 |
Meat packing wastewater |
One-chamber, Carbon paper loaded with0. 35 mg platinum/cm2 |
80 ± 1 mW/m2 |
[9] |
|
4 |
Starch processing wastewater |
One-chamber air-cathode MFC with carbon paper anode (25 cm2) |
239.4 mW/m2 |
[10] |
|
5 |
Swine wastewater |
Single-chambered air cathode |
261 mW/m2 |
[11] |
|
6 |
Brewery wastewater |
One-chamber air-cathode MFC with non-wet proofed carbon cloth as anode (7 cm2) and wet proofed carbon cloth containing Pt as cathode |
205 mW/m2 |
[12] |
|
7 |
Paper recycling wastewater |
One-chamber MFC with graphite fiber-brush anode (5418 m2/m3 brush volume) |
501 ± 20 mW/m2 |
[13] |
Table 1: Performance of single chamber MFC under different types of industrial waste.
In conventional single chamber MFC the anode and cathode are separated by a membrane or sometime cathode is directly exposed to air. In both the cases, anode and cathode are completely different material in design and composition. In our present studies, we are using single chamber MFC where the carbon rod extracted from the used dry cell is used as anode and copper as a cathode. The substrate used here is domestic sewage sludge.
- Materials and Methodology
2.1 Collection and preservation of sample
The sewage sludge is collected from the waste water treatment plant at Anand International College of Engineering, Jaipur. The sludge was preserved at 4°C after collection and before using in MFC [12]. The physiochemical characteristics of the sludge used in MFC are shown in the Table 2.
|
Sl.No |
Parameters |
Values |
|
1 |
COD |
80000 mg/l |
|
2 |
% of Total Solid |
17.2 |
|
3 |
% of Volatile Solid |
11.6 |
|
5 |
Colour |
Grey |
|
6 |
p? |
5.3 |
Table 2: Physiochemical properties of sewage sludge used in MFC.
2.2 Design
Six numbers of single chamber MFCs having carbon electrode as anode and copper as a cathode connected externally in series with copper wire have been used in this present study. Each electrode is submerged into the sludge and copper cathode is exposed to the atmosphere. Figure 1 shows the experimental setup in details.
The various components in microbial fuel cell are as follow;
2.2.1 Electrode: The carbon electrodes were taken from the used dry cell to absorb electrons from the substrate, before using the electrodes were cleaned with 0.1 M HCl and stored in deionized water for 12 hr. The dimension of the electrode is 10 mm diameter and 57 mm, respectively having 40 mm (70% length) as the anode and 17 mm as cathode over which the copper wire was bounded acting as the cathode. Hence the surface area of an anode is 1256 mm2.
2.2.2 Copper wires: The copper wires of 0.7 ohms resistance were connected with the electrodes. The external circuit was in series connections.
2.2.3 Calibrated multimeter: To measure the voltage output (Model No.MAS83OL).
2.2.4 pH meter:To measure the pH of the sewage sludge ( Make: Systronics)
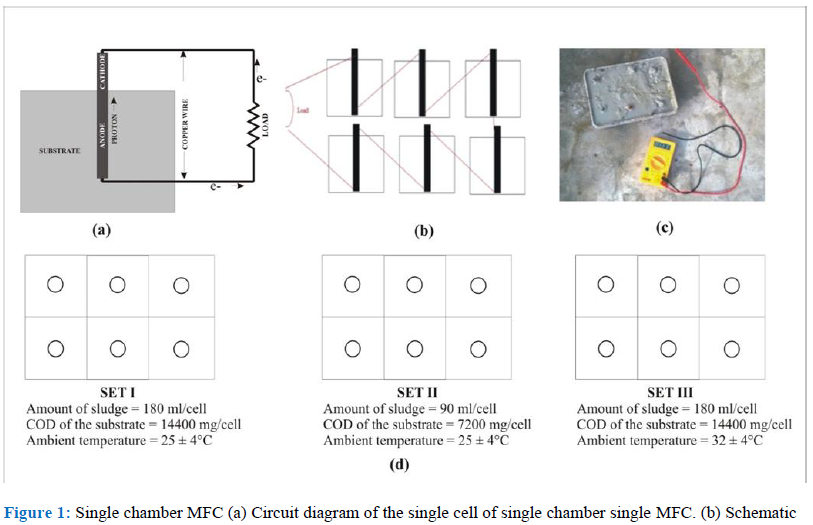
The carbon electrodes were fixed in the containers with the help of the copper wires and are connected in series externally. The collected sewage sludge was mixed uniformly and was poured into the container and allowed the bacteria present in it to grow and start the metabolic activities in aerobic condition (Figure 1).
Figure 1: Single chamber MFC (a) Circuit diagram of the single cell of single chamber single MFC. (b) Schematic representation of the circuit diagram of single chamber MFC in series (c) Photograph of the experimental setup of the combination of the single chamber. (d) Schematic representation of the three different setups of MFC studied.
2.3 Performance evaluation criteria
For all the three sets the voltage output was recorded by the multimeter across the resistor of 1000 ohm at the regular interval of 8 hrs and continuously operated until the energy generation is diminished to zero (least count of the multimeter is 0.01 mV). The first two sets were operated at the ambient temperature of 25 ± 4°Cwhile the third set is operated at the ambient temperature of 32 ± 4°C, which was within 10 days.
The surface power density P (mW/m2) is calculated as , where V (mV) is the measured fuel cell voltage, R(ohm) external resistant and A ( m2) projected surface area of the anode, 1000 is needed for maintaining the units.
- Result and Discussion
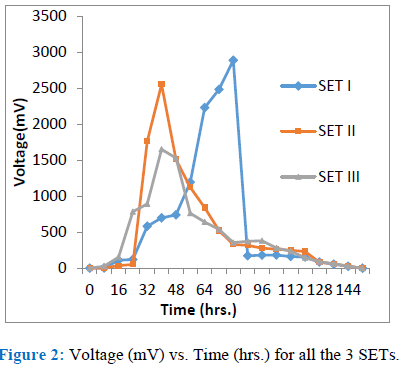
The maximum voltage obtained from the study was about 2890 mV after 80 (hrs.) for the SET Iat ambient temperature of 25 ± 4°C. At the same average ambient temperature in SET II , we could able to achieve 2554 mV after 40 (hrs.) where the quantity (hence the amount of available COD) of the sewage sludge is half that of SET I. In SET III though the quality of sewage sludge is same as that of SET I but we could able to achieve only 1652 mV after 40 (hrs.) at an average ambient temperature of 32 ± 4°C.
|
SET I |
SET II |
SET III |
||||
|
Initial |
Final |
Initial |
Final |
Initial |
Final |
|
|
pH |
5.3 |
4.5 |
5.3 |
4.6 |
5.3 |
6.1 |
|
COD (mg/L) [% decrease] |
80000 |
58000 [27.5%] |
80000 |
72000 [10%] |
80000 |
56000 [30%] |
Table 3: Change of pH and COD in three different sets of Microbial Fuel Cell.
The pH of the sewage sludge found to decrease from 5.3 to 4.5 in SET I (Table 3). For SET II the pH reduced from 5.3 to 4.6 But in SET III the pH increased from 5.3 to 6.1. At the end of the152 (hrs.), the COD of the sewage sludge for the SET I was 58000 mg/l (reduction of 27.5%), In case of SET II the COD was 72000 mg/l (reduction of 10%), finally for the SET III it is 56000 mg/l (reduction of 30%).
Results obtained from the SET III clearly indicate that at elevated temperature the metabolic activities of the microbes reduce and even the pH of the sewage sludge is increased, thereby reducing the surface power density. But at the same time there is the slight improvement in the COD reduction.
- Conclusion
The present study is a novel and economical (cost of each cell Rs.250/-only) single chamber microbial fuel cell for the generation of electricity. Result found is very encouraging when compared to other single chamber MFC. The highest surface power density found to be 1108.29 mW/m2 which is more than any other single chamber MFC excluding platinum electrode (Table 1). Both SET I and SET II are experimented at the same ambient temperature and substrate, where SET II is giving less potential energy. This is because the amount of substrate (hence COD) available for anode at SET II is less than SET I. SET I and SET II are designed in such a way that the same amount of surface area of the electrode is exposed to the substrate in both cases. Thus, in SET II reducing the volume of the substrate in half will also decrease in substrate availability per unit area of the electrode to half. The reduced available substrate per unit area of the electrode not only reduced the peak potential energy, but it also shortens the time to peak voltage. SET III which is operated at the elevated ambient temperature (32 ± 4°C) gives less peak potential energy than SET I and SET II thus indicating that among the two different range of ambient temperature 25 ± 4°C and 32 ± 4°C, the previous one is more promising.
As per the very preliminary study conducted on the presented single chamber, MFC is can be undoubtedly said that the current model is capable of generation of electricity from the domestic sewage sludge economically and effectively.
- Acknowledgement
We are very grateful to Anand International College of Engineering, Jaipur for the financial support to carry out the above project work.
References
- Potter MC. Electrical effects accompanying the decomposition of organic compounds. Proc R Soc London Ser B 84 (1911): 260-276.
- Guang Zhao, Fang Ma, Li Wei, et al. Electricity generation from cattle dung using microbial fuel cell technology during anaerobic acidogenesis and the development of microbial populations. Waste Management 32 (2012): 1651-1658.
- Logan BE, Hamelers B, Rozendal R, et al. Microbial Fuel Cells: Methodology and Technology. Environ Sci Technol 40 (2006): 5181-5192.
- Wang H, Park JD, Ren ZJ. Practical Energy Harvesting for Microbial Fuel Cells: A Review. Environ Sci Technol 49 (2015): 3267-3277.
- Kim JR, Jung SH, Regan JM et al. Electricity generation and microbial community analysis of alcohol powered microbial fuel cells. Biores Technol 98 (2007): 2568-2577.
- Das S, Mangwani N. Recent development in microbial fuel cell: a review. Journal of Scientific and Industrial Research. 69 (2010): 727-731.
- Rahimnejad M, Adhami A, Darvari S, et al. Microbial fuel cell as new technology for bioelectricity generation: A review. Alexandria Engineering Journal 54 (2015): 745-756.
- Nimji VR, Chen CJ, Chen CC, et al. Glycerol degradation in single-chamber microbial fuel cells. Bioresource Technolog 102 (2010): 2629-2634.
- Heilmann J, Logan BE. Production of electricity from proteins using a microbial fuel cell. Water Environ Res 78 (2006): 531-537.
- Liu H, Cheng S A, Logan BE. Production of electricity from acetate or butyrate using a single-chamber microbial fuel cell. Environ Sci Technol 39 (2005): 658-662.
- Min B, Kim JR, Oh S, et al. Electricity generation from swine wastewater using microbial fuel cells. Water Res 39(2005): 4961-4968.
- Feng Y, Wang X, Logan BE, et al. Brewery wastewater treatment using air-cathode microbial fuel cells. Appl Microbiol Biotechnol 78 (2008): 873-880.
- Huang LP, Logan BE. Electricity generation and treatment of paper recycling wastewater using a microbial fuel cell. Appl Microbiol Biotechnol 80 (2008): 349-355.


 Impact Factor: * 3.6
Impact Factor: * 3.6 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 