Hypokalemia: A Practical Approach to Diagnosis and Treatment
Article Information
Mohammad Tinawi*
Department of Internal Medicine and Nephrology, Nephrology Specialists, Munster, IN, USA
*Corresponding author: Mohammad Tinawi, Department of Internal Medicine and Nephrology, Nephrology
Specialists, P.C., 801 MacArthur Blvd., Ste. 400A, Munster, IN 46321, USA
Received: 09 March 2020; Accepted: 03 April 2020; Published: 13 April 2020
Citation: Mohammad Tinawi. Hypokalemia: A Practical Approach to Diagnosis and Treatment. Archives of Clinical and Biomedical Research 4 (2020): 048-066.
View / Download Pdf Share at FacebookAbstract
Potassium (K+) is the predominant intracellular cation. It is essential to the function of all living cells. Intracellular K+ concentration is over 30 times its extracellular concentration. Serum K+ is dependent on its intake, excretion, and transcellular distribution. Most of the body’s K+ is in the muscles. Aldosterone is the main regulator of K+ renal excretion. Hypokalemia (serum K+ < 3.5 mEq/l) has multiple manifestations affecting different organ systems. Careful history and basic laboratory tests are usually adequate to diagnose most cases of hypokalemia. K+ should be replaced orally whenever feasible. Intravenous K+ replacement is needed for emergency management of hypokalemia and in patients who cannot take oral potassium.
Keywords
Hypokalemia; Electrolyte disorder; Potassium disorders
Hypokalemia articles, Electrolyte disorder articles, Potassium disorders articles
Article Details
1. Potassium Homeostasis
Potassium is the most abundant intracellular cation. In humans, the intracellular concentration of K+ is around 150 mEq/l, while the extracellular concentration is 3.5-5 mEq/l.
Hypokalemia is defined as serum potassium concentration < 3.5 mEq/l [1].
The average intake of K+ on a western diet is 60-140 mEq/day. The kidneys excrete 90% of the daily intake while the remaining 10% is excreted in the stool. The amount excreted in the stool increases in advanced kidney disease as in patients on dialysis. The extracellular fluid (ECF) potassium content is only 60-80 mEq or about 2% of total body K+, while intracellular K+ content is 3000-4000 meq. The muscles contain 70% of total body K+, while the liver, the erythrocytes and the bone each contains about 7% [2]. K+ is exchanged between the ECF and the other compartments (muscle, liver, bone).
1.1 Potassium transport in the kidney
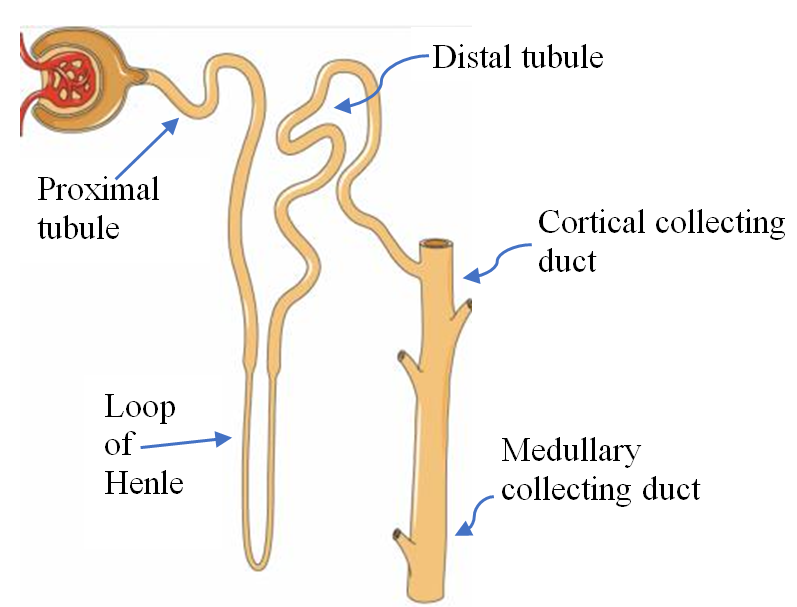
The major segments of the nephron are the proximal tubule, the loop of Henle with its thin and thick limbs, the distal tubule, and the collecting duct which consists of the connecting tubule, the cortical collecting duct and the medullary collecting duct, see Figure 1.
The collecting duct consists of two type of cells, the principal cells which reabsorb sodium (Na+) and secrete K+ under the effect of aldosterone, and the intercalated cells which maintain acid-base balance. The number of intercalated cells decline as the collecting duct slopes toward the medulla.
Potassium filtered through the glomerulus is almost completely absorbed before reaching the collecting duct. About 65% is absorbed in the proximal tubule and 25% in the loop of Henle. 10% of filtered K+ reaches the early distal tubule. It is important to note that almost all of the K+ in the urine is secreted by the collecting duct [3].
Figure 1: The Nephron. Courtesy of Servier Medical Art licensed under a Creative Commons Attribution 3.0 Unported License. https://smart.servier.com
There are several types of K+ channels in the kidney and other organs. Two types of K+ channels are found in the cortical collecting duct.
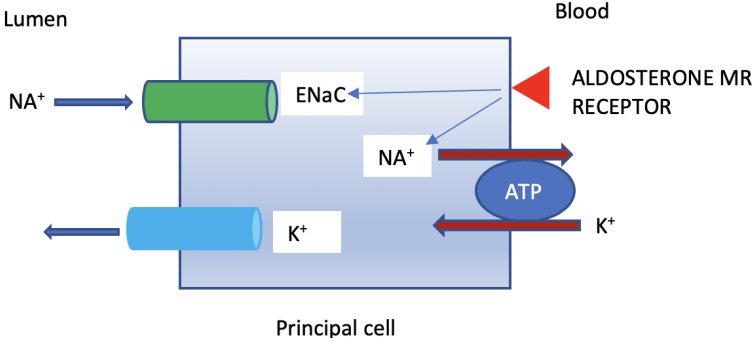
- The renal outer medullary potassium channel (ROMK) is the main K+ secretory channel and it is activated by aldosterone [4]. It is located in the principal cell of the collecting duct. Under physiological states it has a high likelihood of being open.
- The Maxi-K+ channel (BK channel) is activated by high flow rate through the collecting duct [5]. Maxi-K+ channels are located in both the principal and intercalated cells of the collecting duct.
Four major factors determine K+ secretion in the collecting duct [6]:
- Aldosterone: Aldosterone is secreted by the zona glomerulosa of the adrenal cortex. It is the major determinant of K+ secretion. It enhances Na+ absorption and K+ excretion via activation of the Na+-K+-ATPase pump and increasing the number of open K+ channels. The Na+-K+-ATPase pump is located on the basolateral membrane of the principal cell and exists in almost all living cells. Aldosterone also activates the epithelial sodium channel (ENaC) in the apical membrane of the principal cells of the collecting duct, Na+ absorption generates a negative charge which stimulates K+ secretion via ROMK channel, see Figure 2.
- Distal flow rate: A decrease in distal flow rate will decrease K+ secretion in the collecting duct. The reverse is true, an increase in distal flow rate (for example due to use of diuretics) will enhance K+ secretion in the collecting duct. As above, high flow rate activates the Maxi-K (BK channel). The kidneys have a great ability to preserve Na+ but there is a 10-15 meq/l obligatory loss of K+ even in case of hypokalemia.
- Serum potassium: An increase in serum K+ will directly increase aldosterone secretion from the zona glomerulosa, and the reverse is true.
- Delivery of anions to the collecting duct: Anions increase lumen negativity and K+ excretion. Examples are bicarbonate in metabolic alkalosis and non-absorbable anions such as nafcillin.
1.2 The Aldosterone paradox
As indicated above, aldosterone enhances Na+ absorption and K+ secretion in the collecting duct. In cases of low volume status, Na+ reabsorption is desirable, but a concomitant K+ excretion is not desirable because it will to hypokalemia. Applying the same logic to hyperkalemia, increasing K+ excretion is desirable but not Na+ reabsorption because it would lead to hypervolemia.
The aldosterone paradox [7] refers to the fact that in hypovolemia, aldosterone increases Na+ absorption without loss of K+. Additionally, in hyperkalemia aldosterone enhances K+ excretion in the collecting duct without increasing net Na+ absorption.
In case of low effective circulatory volume, the renin-angiotensin-aldosterone system (RAAS) is activated with subsequent increase in angiotensin II and aldosterone levels. RAAS activation leads to increased Na+ absorption in the proximal tubule due to the effect of angiotensin II, and in the collecting tubule due to the effect of aldosterone. Distal flow rate decreases and this in turn decreases K+ excretion minimizing the effect of aldosterone [8].
In case of hyperkalemia, aldosterone secretion is increased but angiotensin II is not activated. Distal delivery of Na+ is maintained which aids in K+ excretion without concomitant increase in net Na+ absorption [9]. Recently, the complex molecular mechanism of these phenomena has been elucidated [10].
1.3 Potassium balance
The kidneys maintain K+ homeostasis. In a steady state potassium intake equals potassium excretion. Extracellular K+ is maintained within a narrow range because K+ can move into or out of skeletal muscles. This prevents big shifts in extracellular K+ concentration. K+ movement is regulated by insulin and catecholamines [11].
Insulin shifts K+ intracellularly by activating the Na+-K+-ATPase pump. The same effect is achieved by catecholamines by activating β2 receptors. After a meal, insulin secretion shift K+ into the cell until it is excreted by the kidney thereby preventing hyperkalemia.
Normal anion gap hyperchloremic metabolic acidosis (mineral acidosis) results in K+ exit from the cells and a rise in extracellular K+. This is due to the effect of mineral acidosis on the Na+/H+ exchanger in the skeletal muscle [12]. High anion gap metabolic acidosis (organic acidosis) and respiratory acidosis have a minimal effect on K+ distribution.
An increase in serum osmolality as in hyperglycemia will result in water movement out of the cell, and subsequent K+ efflux. This will raise K+ in the extracellular fluids [13]. See Table 1.
|
Causes of intracellular K+ shift |
Causes of extracellular K+ shift |
|
Insulin |
Increase in serum osmolality |
|
Catecholamines (β2 receptors) |
Mineral acidosis (normal anion-gap metabolic hyperchloremic acidosis) |
|
Alkalemia |
Table 1: Causes of potassium shift
K+ execration in the kidneys follows a circadian rhythm [14]. K+ excretion is lower during the night and the early hours of the morning, and it increases as the day progresses concurring with increased intake of K+ rich food.
2. Hypokalemia
2.1 Prevalence
Hypokalemia is common in hospitalized and community dwelling subjects. A study in about 5000 community subjects aged 55 years or older (the Rotterdam Study) found hypokalemia in about 2.5% [15]. The prevalence in women was twice as in men. Hypokalemia was most prevalent in patients on thiazide diuretics, odds ratio (OR): 7.68 (4.92-11.98), P < 0.001.
A study in about 8000 patients admitted to the emergency department (ED) found hypokalemia in 39% [16]. Hypokalemia is seen in about 20% of hospitalized patients [17].
2.2 Etiology
Hypokalemia results from renal or non-renal loss of K+. Intracellular K+ shift will lead to transient hypokalemia, while inadequate dietary intake is a rare cause of hypokalemia. Inadequate intake is seen in starvation, dementia, and anorexia. See Table 2.
2.2.1 Pseudohypokalemia
Pseudohypokalemia is seen when blood samples containing very high number of white blood cells (>75 x 109/L) are stored at room temperature. Hypokalemia is the result of K+ uptake by white blood cells. This is seen in acute myelogenous leukemia (AML). Measuring K+ after quick plasma separation prevents this error [18].
2.2.2 Intracellular potassium shift or redistribution
Insulin and β2 receptors agonists (such as epinephrine, albuterol, and ephedrine) are the major causes of intracellular K+ shift. Redistribution is seen in hypokalemic periodic paralysis. This is a rare disorder that is seen more commonly in Asians in association with thyrotoxicosis [19]. Other rare causes are intoxications with verapamil, cesium salts, chloroquine or barium [20]. The antipsychotic medications risperidone and quetiapine can rarely cause intracellular K+ shift [21].
2.2.3 Non-renal potassium loss
The most common causes in this category are gastrointestinal such as diarrhea, vomiting, nasogastric (NG) suctioning, and laxatives. Hypokalemia due to K+ loss via excessive perspiration is uncommon. It is important to note that the above conditions are associated with dehydration with subsequent secondary hyperaldosteronism and renal K+ wasting.
In vomiting and NG suctioning, hypokalemia results from secondary hyperaldosteronism (due to dehydration) and metabolic alkalosis (due to loss of chloride). K+ concentration in gastric juice is small (around 8 mEq/l). Direct K+ loss is important in diarrhea because K+ concentration in stool is 80-90 mEq/l. Metabolic alkalosis is associated with hypokalemia due to intracellular K+ shift, renal K+ excretion due to bicarbonaturia and secondary hyperaldosteronism (when volume depletion is present) [22]. Hemodialysis and peritoneal dialysis are common causes of hypokalemia in patients on renal replacement therapy.
2.2.4 Renal potassium loss
Renal loss of K+ is the most common etiology of hypokalemia. This category includes medications, hormones, hypomagnesemia, and renal tubular acidosis [23].
Diuretics such as thiazides and loop diuretics are a common cause of hypokalemia due to increased distal flow, and secondary hyperaldosteronism resulting from volume depletion. Combining two diuretics such as metolazone and a loop diuretic, or acetazolamide and a loop diuretic may lead to severe hypokalemia. Several antibiotics can cause hypokalemia due to a variety of mechanisms. High dose penicillin G and penicillin analogues can cause hypokalemia due to distal tubule delivery of non-reabsorbable anions which increases K+ excretion.
Aldosterone is the main K+ regulating hormone and excess aldosterone as in primary aldosteronism leads to hypokalemia [24].
Magnesium deficiency may result in refractory hypokalemia. Magnesium inhibits ROMK channels, subsequently hypomagnesemia increases potassium secretion in the collecting duct [25].
Both proximal and distal renal tubular acidosis cause hypokalemia due to renal K+ loss [26]. Renal K+ loss is also seen in certain rare disorders such as Bartter’s syndrome, Gitelman’s syndrome, and Liddle’s syndrome [27].
Sodium reabsorption due to overactivity of ENaC (gain of function mutation) increases the negative charge in the lumen of the collecting duct which enhances K+ excretion. This is the mechanism of hypokalemia in Liddle syndrome. Liddle syndrome is an autosomal dominant disorder and a rare cause of hypertension. It is characterized by early onset of HTN, suppressed renin and aldosterone, hypokalemia and metabolic alkalosis. It is treated with amiloride or triamterene which block ENaC. Thiazides aggravates K+ loss [28].
|
1.Pseudohypokalemia: as in AML. Hypokalemia is the result of K+ uptake by white blood cells when the sample is stored at room temperature. WBC (>75 x 109/L). 2.Intracellular K+ shift: insulin, β2 receptors agonists such as albuterol, theophylline, alkalemia, hypothermia, risperidone, quetiapine, intoxications (chloroquine, verapamil, barium or cesium), thyrotoxicosis and hypokalemic periodic paralysis 3.Non-renal K+ loss: diarrhea, laxatives, repeated enemas, vomiting, NG suctioning, enteric fistula, vipoma, Zollinger-Ellison syndrome, clay ingestion, and skin loss (uncommon) 4.Renal loss:
5.Inadequate intake: patients on total parenteral or enteral nutrition, anorexia, and starvation |
Table 2. Causes of Hypokalemia
2.3 Symptoms and complications
Mild hypokalemia can be asymptomatic. Most symptomatic patients have a serum K+ < 3 mEq/l. The severity of the symptoms is also related to the rate of K+ decline.
Muscle weakness and fatigue are the most common symptoms upon presentation. Both hypokalemia and hyperkalemia can result in muscle weakness starting in the lower extremities and ascending to the trunk and upper extremities [32].
In severe hypokalemia muscle weakness can progress to flaccid paralysis, but this is rare. Some patients develop muscle cramps. Severe hypokalemia can lead to rhabdomyolysis. Gastrointestinal muscle involvement can lead to ileus, nausea, vomiting and constipation.
ECG changes in hypokalemia include flat T waves, ST segment depression, and prominent U waves. Hypokalemia can result in palpitations in addition to ventricular and supraventricular tachyarrhythmias. Digitalis increases the likelihood of arrhythmias [33].
Hypokalemia can result in a variety of renal manifestations including polyuria, polydipsia, and nephrogenic DI [34]. Chronic hypokalemia can rarely result in chronic tubulointerstitial nephritis (CIN).
Hypokalemia is associated with glucose intolerance due to a decrease in insulin secretion [35].
Hypokalemia has been associated with psychological manifestations including, psychosis, delirium, hallucinations and depression [36].
2.4 Diagnosis
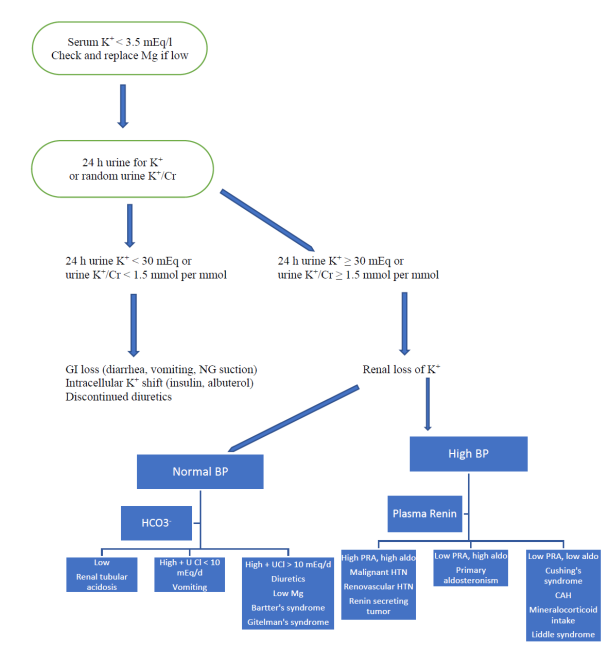
When approaching a patient with hypokalemia, remember the following principles [37]: See Figure 3.
- Obtaining a good history is essential. Most patients have either GI or renal loss of K+. Intracellular shift causes transient hypokalemia. Inadequate dietary intake and pseudohyokalemia are rare causes. Vomiting and diarrhea are the most common GI causes, while diuretic use is the most common renal etiology. The focus of physical exam is on blood pressure, volume status and musculoskeletal exam.
- Hypokalemia is diagnosed after ordering an electrolyte panel. Hypokalemia is serum K+ < 3.5 mEq/l or < 3.5 mmol/l in SI units. HCO3- level may help in assessing acid-base status. Serum glucose, blood urea nitrogen and creatinine are usually obtained. Serum magnesium should be checked especially in recalcitrant hypokalemia [38]. Most patients do not require extensive testing. K+ replacement and addressing the etiology (such as diarrhea, or diuretic use) is usually sufficient.
- If the etiology of hypokalemia is unclear, a 24 h urine collection for K+ is helpful. If hypokalemia is due to GI loss, the kidneys will preserve K+ and 24 h urine K+ is < 30 meq. In patients with renal loss of K+, 24 h urine K+ is ≥ 30 meq. If a 24 h urine collection is not feasible, the ratio of urine K+ to urine creatinine is obtained in a random specimen. If hypokalemia is due to renal loss of potassium the ratio of K+ in mmol to creatinine in mmol is > 1.5. If non-SI unit are used, renal loss is diagnosed if the ratio of K+ in mEq to creatinine in g is >13. Note that in case of K+, the value is the same in mEq/l or mmol/l [39].
- Some patients require further testing such as urine electrolytes (including urine Na+, K+, Cl-, Ca2+, and Mg2+), thyroid function tests, plasma renin activity, and plasma aldosterone level [40]. Creatine kinase is obtained if rhabdomyolysis is suspected. ECG is ordered in the appropriate setting to diagnose cardiac arrhythmias.
- In case of GI loss, patients with diarrhea or laxative abuse usually have low HCO3-, while patients with vomiting usually have high HCO3-. Patients with vomiting have characteristically low urine Cl- < 10 mEq/24 h [41].
- If diuretic abuse is suspected, urine diuretic screen is ordered [42]. A clue to diuretic abuse is inconsistent values of random urine K+/Cr (high while taking a diuretic, and low hours after the last diuretic dose).
2.5 Treatment
- Patients with serum K+ in the range of 3.0-3.5 mEq/l are usually treated with oral K+ salts as long as they can take oral medications. Patients with serum K+ < 3 mEq/l may require IV K+ especially in emergencies such as arrhythmias, rhabdomyolysis and respiratory failure. In many situations both PO and IV K+ salts are used [43].
- IV replacement of K+ is appropriate for patients with ECG changes, and in hypokalemia associated with diabetic ketoacidosis (DKA) or the use of digitalis. K+ deficit is about 200-400 mEq for every 1 mEq/l drop in K+, but the actual amount varies among individuals [44].
- Most patient are treated with potassium chloride (KCl). See table 3. KCl is widely available in multiple forms (extended release (ER) tablets, capsules, liquid, and IV). KCl works quickly and is the preferred agent especially in patients with concomitant metabolic alkalosis. In those patients, replenishment of Cl- is paramount [45]. Cl- stays mostly in the extracellular compartment. If K bicarbonate is given, HCO3- largely enters the cell and K+ follows, this make K bicarbonate (and citrate/acetate, which are precursor of bicarbonate) less effective.
|
Drug |
K chloride |
K bicarbonate |
K citrate |
K acetate |
K phosphate |
K gluconate |
|
Forms |
PO (tablets, capsules, liquid), IV |
PO (effervescent tablets) |
PO |
IV |
IV |
PO |
|
Indication |
Almost all causes of hypokalemia, especially with metabolic alkalosis |
Hypokalemia due to renal tubular acidosis or diarrhea |
Hypokalemia due to renal tubular acidosis or diarrhea |
Mainly in TPN |
Used only when both K and phos are low |
Available with no prescription |
|
Precautions |
ER forms can cause GI ulcerations |
Can worsen metabolic alkalosis |
Infuse slowly 7.5 mM/h |
|||
|
Amount needed to provide 40 mEq of K |
3.0 g |
4.0 g |
4.3 g |
3.9 g |
Each ml has 3 mM phos and 4.4 mEq K |
9.4 g |
|
Remarks |
Do not crush ER tablets |
K phos tablets are only used for low phos |
Table 3: Comparison of different potassium salts [46]
- Intravenous KCl should be given at a rate that does not exceed 10 mEq/h. A higher rate up to 20 mEq/h is a consideration in emergency situations such as cardiac arrhythmias, telemetry monitoring is required [47].
- Administration of IV KCl should be done through a central venous catheter if available. IV KCl can cause phlebitis, and many patients find the infusion painful. It is preferable to give intravenous KCl in 0.9 NS (usually 20 mEq KCl in 100 ml of 0.9 NaCl). Giving IV KCl in a dextrose solution may stimulate insulin release and potentially aggravate hypokalemia.
- Potassium chloride salt substitutes are a good source of oral K+. They contain about 13.6 mEq/g [48]. K+ containing foods are appropriate for chronic management of mild hypokalemia. There are not effective for emergency treatment because the amount needed for correction is large, and potassium in diet is K+ citrate or phosphate which is less effective compared to KCl as explained above. Bananas are a good source of K+. They contain about 1 mEq/cm [49]. Therefore, one needs to eat 2 large bananas to get 40 mEq of K+. Examples of foods that are high in K+ include: dried fruits (dates, figs, prunes), spinach, broccoli, kiwis, mangos, oranges, tomatoes, avocados, bananas, milk, raisins, and lima beans [20].
- Potassium sparing diuretics [50] may be appropriate for the chronic management of hypokalemia especially in patients who are already on a thiazide or a loop diuretic. Aldosterone receptor antagonists (spironolactone and eplerenone) may help in the management of patients with advanced heart failure and in patients with resistant hypertension. Amiloride is well tolerated as well, it blocks the epithelial sodium channel ENaC in the collecting duct. Triamterene is rarely associated with kidney stones, and the use of amiloride, eplerenone or spironolactone is preferable.
- Laxatives and diuretics should be stopped if hypokalemia is due to their abuse. Symptomatic treatment of diarrhea and vomiting is helpful [51].
- If the patient needs administration of both bicarbonate and potassium intravenously, potassium should be given first because bicarbonate results in intracellular K+
2.6 Clinical Vignettes
- 50-year-old man with a history of chronic systolic congestive heart failure (CHF) presents to the emergency department (ED) with weakness and palpitations. His medications include furosemide, digoxin, carvedilol, quinapril and atorvastatin. ECG shows paroxysmal atrial tachycardia with 2:1 block, serum K+ is 2.9 mEq/l, digoxin level is 3.1 ng/ml. How would you manage his hypokalemia?
Answer: The patient has digoxin toxicity associated with hypokalemia; he should be monitored on telemetry. KCl should be given intravenously. Oral KCl can be started concomitantly. Digoxin should be held.
- 76-year-old woman with chronic systolic CHF, her ejection fraction is 20%. She is on furosemide, bisoprolol and enalapril. On a routine chemistry profile her serum K+ is 3.4 mEq/l. BP is 144/93. What is the best approach to her hypokalemia?
Answer: The patient has chronic mild hypokalemia; her BP control is suboptimal. Spironolactone is appropriate for this patient with chronic systolic CHF, uncontrolled hypertension and mild hypokalemia. In the RALES trial aldosterone reduced morbidity and mortality in patients with severe heart failure [52]. Patients should be monitored for hyperkalemia.
- A 71 year-old-man who weighs 60 kg is started on 25 mg of hydrochlorothiazide (HCTZ) for hypertension. Initial chemistry panel is unremarkable. He presents to the ED 2 weeks later with weakness. Laboratory studies: Na+ 127 mEq/l, K+ 2.7 mEq/l. How would you manage his electrolyte disorder?
Answer: The patient has hyponatremia and hypokalemia due to HCTZ. He needs intravenous replacement of Na+ and K+. Since both Na+ and K+ are active osmoles, K+ replacement should be taken into account when replacing Na+, otherwise overcorrection of hyponatremia will ensue [53]. HCTZ was discontinued and the patient was given 4 doses of 20 mEq KCl over 8 h (each in 100 ml of 0.9 NaCl, total volume is 400 ml) and started on 0.9 NaCl infusion at 75 ml/h (600 ml in 8 h). The change in Na+ after 8 h is calculated using the formula:
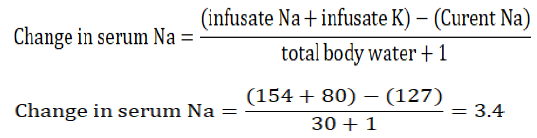
The infusate is the infused solution, and in case of 0.9 NS, it contains 154 mEq of Na+ per liter. The sodium will rise to approximately 130 mEq/l which is an appropriate rate of correction.
- A 20-year-old woman presents with weakness and nausea; her BP is 105/52. Electrolytes: Na+ 139, K+ 2.7, Cl- 109, HCO3- 21 (mEq/l), anion gap (AG = Na – Cl+bicarb) = 9, Urine electrolytes (in mEq/l): Na+ 50, K+ 11, Cl- 70, urine anion gap (U AG = Na+K-Cl) = -9.
The renal consultant insists that her hypokalemia is not due to Bartter’s syndrome or type I distal RTA (renal tubular acidosis), why?
Answer: This patient presents with severe symptomatic hypokalemia. Her electrolyte panel is suggestive of metabolic acidosis due to low HCO3-; she has normal serum anion gap and a negative urine anion gap. All of this is consistent with diarrhea or laxative abuse.
Patients with Bartter’s syndrome have normal blood pressure and renal wasting of K+, in this patient urine K+ is 9 consistent with GI loss of potassium and renal preservation of K+. Bartter’s syndrome usually presents with metabolic alkalosis and not acidosis. The clinical picture of Bartter’s syndrome is similar to intake of loop diuretics (renal wasting of K+ and metabolic alkalosis). Patients with type I distal RTA also have renal wasting of K+, and a positive urine anion gap.
Finally, in any patient with hypokalemia, one needs to rule out vomiting, bulimia, diarrhea, laxative abuse, use of diuretics, and diuretic abuse. These causes are far more common than RTA, Bartter’s syndrome, Gitelman’s syndrome or Liddle’s syndrome.
- A 40-year-old man with a known diagnosis of AML was found to have a serum K+ of 1.9 mEq/l on a routine lab. Leukocytes 290 x 109/l. How would you replace his K+?
Answer: The patient was asymptomatic despite severe hypokalemia. Pseudohypokalemia due to AML was suspected. This is the result of delayed analysis of a blood sample left at room temperature. A second sample was drawn and placed on ice, then it was immediately analyzed in the lab. K+ in the second sample was 3.7 mEq/l. No action is required.
- A 26-year-old man presents with a BP of 161/101mmHg. Initial electrolyte panel: Na+ 144, K+ 3.5, Cl- 109, HCO3- 29 (mEq/l). The patient was started on chlorthalidone 25 mg po daily for HTN. One week later his electrolyte: Na+ 139, K+ 2.2, Cl- 101, HCO3- 32. How would you manage his hypokalemia?
Answer: Work up for secondary hypertension should be considered in patients who present with hypertension at an early age. This patient developed severe hypokalemia in response to a thiazide diuretic. This is suggestive of primary aldosteronism. Note that his initial K+ was at the lower range of normal. It is recommended to follow the Endocrine Society guidelines for work up of primary aldosteronism [54].
- A 19-year-old woman was brought to the ED by her family due to nausea, proximal muscle weakness and fatigue. Her family is concerned because she has been ingesting a large amount of a clay powder she bought on the internet. She is using it for "detoxification". Labs: Na+ 135, K+ 2.1, Cl- 105, HCO3- 23 (mEq/l), CK was elevated at 1200 U/l, urine electrolytes: Na+ 85, K+ 18, Cl- 60 (mEq/l). How would you manage her hypokalemia?
Answer: Her hypokalemia is due to ingestion of Bentonite clay which binds K+ in the GI tract. She has low urine K+ consistent with a non-renal cause of hypokalemia. Bentonite clay powder is advertised as a toxin cleanser and as a homeopathic treatment for constipation and nausea. Some use it externally as a face mask for oily skin.
This patient required oral and intravenous replacement of K+ with instructions to avoid the use of Bentonite.
There is a report of a 3-year-old girl who presented with a K+ of 0.9 mEq/l due to oral and rectal use of Bentonite for constipation [55]. She improved with saline hydration and multiple doses of IV KCl.
- A 72- year-old man was admitted to the ICU due to osteomyelitis. He was started on nafcillin 2 g IV q 4h. He has underlying sever COPD and was started on BiPAP non-invasive ventilation and an intensive nebulizer regimen. He has stage 4 chronic kidney disease. Enteral feeding via NG tube using a renal formula was initiated at 55 ml/h. Due to increased lower extremities edema, he was placed on furosemide 80 mg IV q 12 h in addition to D5W at 70 ml/h due to a serum Na+ of 146 mEq/l.
His serum K+ dropped from 4 to 2.4 mEq/l on his 3rd hospital day. What is the etiology of his hypokalemia?
Answer: The above is a common scenario. Hypokalemia in complicated patients is multifactorial. This patient is on renal formula which is low in K+. He is getting D5W which is free of K+ and will stimulate insulin secretion and drive K+ intracellularly. Albuterol in his nebulizer therapy will also shift K+ intracellularly. Nafcillin acts as non-absorbable anion stimulating K+ excretion in the collecting duct [56]. Finally, furosemide will cause renal loss of K+.
- A 50-year-old woman was referred to the renal clinic for persistent hypokalemia. She is on extended release KCl 20 mEq twice daily. She is complaining of arms and legs cramping, fatigue, urinary frequency and nocturia. BP is 105/54.
Labs: Na+ 135, K+ 2.7, HCO3- 29(mEq/l), Mg2+ 1, Ca2+ 9 (mg/dl), 24 h urine shows: Na+ 130, Cl-140, K+ 45 (mEq/24 h), Ca2+ 30 mg/24 h.
Answer: The patient has high urine K+ due to renal loss of K+. Her HCO3- is high consistent with metabolic alkalosis. She has concomitant severe hypomagnesemia. Urine studies show high Na+ and Cl- and low urine Ca2+.
This presentation is not due to vomiting, because she has high urine K+ and high urine Cl- (both should be low in vomiting). It is not due to diarrhea or laxative use due to renal loss of K+ and the presence of metabolic alkalosis (metabolic acidosis is expected in diarrhea).
Diuretic abuse is high on the differential diagnosis list. The patient has renal loss of K+, hypomagnesemia, metabolic alkalosis, low urine Ca++, high urine Na+ and Cl-. All these manifestations can be seen in a patient abusing a thiazide diuretic. This patient denied using any diuretics and her urine screen for diuretics came back negative. The diagnosis is consistent with Gitelman's syndrome [57] and it was confirmed with genetic testing.
Gitelman's syndrome is an autosomal recessive disorder and is a salt wasting tubulopathy. This explains the normal or low normal BP. It is usually due to mutations in the SLC12A3gene, resulting in dysfunction of the thiazide sensitive Na-Cl cotransporter (NCC) channel in distal tubule. The late onset and low urine Ca2+ distinguish Gitelman’s from Bartter’s syndrome; however, genetic testing is the only way to ascertain the diagnosis.
- A 63-year-old man takes furosemide 40 mg po bid for management of his chronic systolic CHF. On routine labs his K+ was 3.1 mEq/l and he was started on extended release KCl 20 meq bid. Repeat K+ one week later was 3.8 mEq/l. A month later his K is 3.3 mEq/l after he decided to switch to non-prescription potassium gluconate 99 mg tablets twice daily to save money. How would you advise this patient?
Answer: Potassium gluconate 99 mg tablets contain only 2.5 mEq of KCl per tablet which is far less than his previous dose of KCl. Patients on furosemide may develop metabolic alkalosis, and KCl is the preferred potassium salt. If cost is an issue, the patient should switch to KCl salt substitute which contains 13.6 mEq/g. 3 g (about ½ teaspoon) will provide him with the required 40 mEq of KCl daily.
2.6 Conclusion
- Hypokalemia is common in the inpatient and outpatient settings.
- Aldosterone is the main potassium regulating hormone.
- Hypokalemia should be treated orally. Intravenous treatment is reserved for severe hypokalemia (K+ < 3 mEq/l) or emergencies (e.g. arrhythmias).
- Potassium chloride is the preferred potassium salt for the treatment of hypokalemia.
- The cause of hypokalemia is usually ascertained by obtaining a careful history, checking BP, and ordering a few laboratory tests. Distinguishing renal loss from GI loss of potassium is essential.
- A specialist consultation is required if an endocrine cause of hypokalemia is suspected.
Conflict of interest
The author declares no conflict of interest.
References
- B Palmer and D Clegg. Physiology and Pathophysiology of Potassium Homeostasis: Core Curriculum. Am J Kid Dis 74 (2019): 682-695.
- FV Osorio and SL Linas. Disorders of Potassium Metabolism. Atlas of Diseases of The Kidney 1 (2002).
- Palmer. Regulation of Potassium Homeostasis. Clin J Am Soc Nephrol 10 (2015): 1050-1060.
- Welling, Paul A and Kevin Ho. A comprehensive guide to the ROMK potassium channel: form and function in health and disease. American Journal of Physiology-Renal Physiology 297 (2009): F849-F863.
- Palmer, Lawrence G and Gustavo Frindt. High-conductance K channels in intercalated cells of the rat distal nephron. American Journal of Physiology-Renal Physiology 292 (2007): F966-F973.
- Stanton BA. Renal potassium transport: morphological and functional adaptations. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 257 (1989): R989-R997.
- Arroyo, Juan Pablo, et al. Aldosterone paradox: differential regulation of ion transport in distal nephron. Physiology 26 (2011): 115-123.
- Giebisch G. Renal potassium transport: mechanisms and regulation. American Journal of Physiology-Renal Physiology 274 (1998): F817-33.
- Palmer BF. A physiologic-based approach to the evaluation of a patient with hypokalemia. American journal of kidney diseases 56 (2010): 1184-90.
- Kamel KS, Schreiber M, Halperin ML. Renal potassium physiology: integration of the renal response to dietary potassium depletion. Kidney international 93 (2018): 41-53.
- Ho K. A critically swift response: insulin-stimulated potassium and glucose transport in skeletal muscle. CJASN 6 (2011): 1513-1516
- Palmer BF, Clegg DJ. Physiology and pathophysiology of potassium homeostasis. Advances in physiology education 40 (2016): 480-90.
- Rastegar. Serum Potassium in Clinical Methods: The History, Physical, and Laboratory Examinations. 3rd edition., Boston, MA: Butterworths, 1990.
- Gumz ML, Rabinowitz L, Wingo CS. An integrated view of potassium homeostasis. New England Journal of Medicine 373 (2015): 60-72.
- Liamis G, Rodenburg EM, Hofman A, Zietse R, Stricker BH, Hoorn EJ. Electrolyte disorders in community subjects: prevalence and risk factors. The American journal of medicine 126 (2013): 256-63.
- Giordano M, Ciarambino T, Castellino P, Malatino L, Di Somma S, Biolo G, Paolisso G, Adinolfi LE. Diseases associated with electrolyte imbalance in the ED: age-related differences. The American journal of emergency medicine 34 (2016): 1923-6.
- Lippi G, Favaloro EJ, Montagnana M, Guidi GC. Prevalence of hypokalaemia: the experience of a large academic hospital. Internal medicine journal 40 (2010): 315-6.
- Weiner ID, Wingo CS. Hypokalemia--consequences, causes, and correction. Journal of the American Society of Nephrology 8 (1997): 1179-88.
- Correia M, Darocki M, Hirashima ET. Changing Management Guidelines in Thyrotoxic Hypokalemic Periodic Paralysis. The Journal of emergency medicine 55 (2018): 252-6.
- Bhoelan BS, Stevering CH, Van Der Boog AT, Van der Heyden MA. Barium toxicity and the role of the potassium inward rectifier current. Clinical Toxicology 52 (2014): 584-93.
- Lim S. Approach to hypokalemia. Acta Med Indones 39 (2007): 56-64.
- Gennari FJ. Hypokalemia. New England Journal of Medicine 339 (1998): 451-8.
- Viera AJ, Wouk N. Potassium disorders: hypokalemia and hyperkalemia. American family physician 92 (2015): 487-95.
- Gennari FJ. Disorders of potassium homeostasis: hypokalemia and hyperkalemia. Critical care clinics 18 (2002): 273-88.
- Huang CL, Kuo E. Mechanism of hypokalemia in magnesium deficiency. Journal of the American Society of Nephrology 18 (2007): 2649-52.
- Soriano JR. Renal tubular acidosis: the clinical entity. Journal of the American Society of Nephrology 13 (2002): 2160-70.
- Subramanya AR, Ellison DH. Distal convoluted tubule. Clinical Journal of the American Society of Nephrology 9 (2014): 2147-63.
- Tetti M, Monticone S, Burrello J, Matarazzo P, Veglio F, Pasini B, Jeunemaitre X, Mulatero P. Liddle syndrome: review of the literature and description of a new case. International journal of molecular sciences 19(2018): 812.
- Kardalas E, Paschou SA, Anagnostis P, Muscogiuri G, Siasos G, Vryonidou A. Hypokalemia: a clinical update. Endocrine connections 7 (2018): R135-46.
- McMahon GT, Dluhy RG. Glucocorticoid-remediable aldosteronism. Arquivos Brasileiros de Endocrinologia & Metabologia 48 (2004): 682-6.
- Yau M, Haider S, Khattab A, Ling C, Mathew M, Zaidi S, Bloch M, Patel M, Ewert S, Abdullah W, Toygar A. Clinical, genetic, and structural basis of apparent mineralocorticoid excess due to 11β-hydroxysteroid dehydrogenase type 2 deficiency. Proceedings of the National Academy of Sciences 114 (2017): E11248-56.
- Knochel JP. Neuromuscular manifestations of electrolyte disorders. The American journal of medicine 72 (1982): 521-35.
- Skogestad J, Aronsen JM. Hypokalemia-induced arrhythmias and heart failure: new insights and implications for therapy. Frontiers in physiology 9 (2018): 1500.
- Yalamanchili HB, Calp-Inal S, Zhou XJ, Choudhury D. Hypokalemic Nephropathy. Kidney international reports 3 (2018): 1482-8.
- Grunfeld C, Chappell DA. Hypokalemia and diabetes mellitus. The American journal of medicine 75 (1983): 553-4.
- Hong E. Hypokalemia and Psychosis: A forgotten association. American Journal of Psychiatry Residents' Journal 11 (2016): 6-7.
- Groeneveld JH, Sijpkens YW, Lin SH, Davids MR, Halperin ML. An approach to the patient with severe hypokalaemia: the potassium quiz. Qjm 98 (2005): 305-16.
- Whang R, Flink EB, Dyckner T, Wester PO, Aikawa JK, Ryan MP. Magnesium depletion as a cause of refractory potassium repletion. Archives of internal medicine 145 (1985): 1686-9.
- Kamel KS, Ethier JH, Richardson RM, Bear RA, Halperin ML. Urine electrolytes and osmolality: when and how to use them. American journal of nephrology 10 (1990): 89-102.
- Wu KL, Cheng CJ, Sung CC, Tseng MH, Hsu YJ, Yang SS, Chau T, Lin SH. Identification of the causes for chronic hypokalemia: Importance of urinary sodium and chloride excretion. The American journal of medicine 130 (2017): 846-55.
- Woywodt A, Herrmann A, Eisenberger U, Schwarz A, Haller H. The tell?tale urinary chloride. Nephrology Dialysis Transplantation 16 (2001): 1066-8.
- Reimann D, Gross P. Chronic, diagnosis-resistant hypokalaemia. Nephrology Dialysis Transplantation 14 (1999): 2957-61.
- Cohn JN, Kowey PR, Whelton PK, Prisant LM. New guidelines for potassium replacement in clinical practice: a contemporary review by the National Council on Potassium in Clinical Practice. Archives of internal medicine 160 (2000): 2429-36.
- Sterns RH, Cox MA, Feig PU, Singer IR. Internal potassium balance and the control of the plasma potassium concentration. Medicine 60 (1981): 339-54.
- Kassirer JP, Berkman PM, Lawrenz DR, Schwartz WB. The critical role of chloride in the correction of hypokalemic alkalosis in man. The American journal of medicine 38 (1965): 172-89.
- com. Drugs.com, 1 3 2020. [Online]. Available: https://www.drugs.com/monograph/potassium-supplements.html. [Accessed 1 3 2020].
- Kruse JA, Carlson RW. Rapid correction of hypokalemia using concentrated intravenous potassium chloride infusions. Archives of internal medicine 150 (1990): 613-7.
- Hueston WJ. Use of salt substitutes in the treatment of diuretic-induced hypokalemia. J Fam Pract 29 (1989): 623-6.
- Kopyt N, Dalal F, Narins RG. Renal retention of potassium in fruit. The New England journal of medicine 313 (1985): 582.
- Horisberger JD, Giebisch G. Potassium-sparing diuretics. Kidney and Blood Pressure Research 10 (1987): 198-220.
- Unwin RJ, Luft FC, Shirley DG. Pathophysiology and management of hypokalemia: a clinical perspective. Nature Reviews Nephrology 7 (2011): 75.
- Pitt B, Zannad F, Remme WJ, Cody R, Castaigne A, Perez A, Palensky J, Wittes J. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. New England Journal of Medicine 341 (1999): 709-17.
- Tinawi M. Hyponatremia and Hypernatremia: A Practical Guide to Disorders of Water Balance. Archives of Internal Medicine Research 3 (2020): 74-95.
- Funder JW, Carey RM, Mantero F, Murad MH, Reincke M, Shibata H, Stowasser M, Young Jr WF. The management of primary aldosteronism: case detection, diagnosis, and treatment: an endocrine society clinical practice guideline. The Journal of Clinical Endocrinology & Metabolism 101 (2016): 1889-916.
- Bennett A, Stryjewski G. Severe hypokalemia caused by oral and rectal administration of bentonite in a pediatric patient. Pediatric emergency care 22 (2006): 500-2.
- Mohr JA, Clark RM, Waack TC, Whang R. Nafcillin-associated hypokalemia. JAMA 242 (1979): 544.
- Blanchard A, Bockenhauer D, Bolignano D, Calo LA, Cosyns E, Devuyst O, Ellison DH, Frankl FE, Knoers NV, Konrad M, Lin SH. Gitelman syndrome: consensus and guidance from a kidney disease: improving global outcomes (KDIGO) controversies conference. Kidney international 91 (2017): 24-33.


 Impact Factor: * 3.1
Impact Factor: * 3.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 