Hyponatremia and Hypernatremia: A Practical Guide to Disorders of Water Balance
Article Information
Mohammad Tinawi*
Department of Internal Medicine and Nephrology, Nephrology Specialists, Munster, IN, USA
*Corresponding author: Mohammad Tinawi, Department of Internal Medicine and Nephrology, Nephrology Specialists, P.C., 801 MacArthur Blvd., Ste. 400A, Munster, IN 46321, USA
Received: 10 February 2020; Accepted: 18 February 2020; Published: 24 February 2020
Citation:
Mohammad Tinawi. Hyponatremia and Hypernatremia: A Practical Guide to Disorders of Water Balance. Archives of Internal Medicine Research 3 (2020): 074-095.
View / Download Pdf Share at FacebookAbstract
Hyponatremia and hypernatremia are disorders of water balance and are very common especially in hospitalized patients. Hyponatremia is defined as serum sodium < 135 mEq/l (mmol/l). Hypernatremia is defined as serum sodium > 145 mEq/l (mmol/l). Most of hyponatremia and hypernatremia cases are mild but they are clinically significant. Even mild hyponatremia is associated with many non-specific symptoms and may quickly evolve into severe hyponatremia. Quick and uncontrolled correction of chronic hyponatremia may lead to severe clinical consequences. Hypernatremia is associated with high mortality due to associated co-morbid conditions even after successful correction. The following review will cover the most salient aspects of hyponatremia and hypernatremia and provide the clinician with a practical guide to the diagnosis and treatment. Complex tables, flow charts and algorithms will be avoided. The review will conclude with clinical cases that apply the discussed principles in diagnosis and treatment.
Keywords
Hyponatremia, Hypernatremia, Water balance, Electrolyte disorder
Hyponatremia articles Hyponatremia Research articles Hyponatremia review articles Hyponatremia PubMed articles Hyponatremia PubMed Central articles Hyponatremia 2023 articles Hyponatremia 2024 articles Hyponatremia Scopus articles Hyponatremia impact factor journals Hyponatremia Scopus journals Hyponatremia PubMed journals Hyponatremia medical journals Hyponatremia free journals Hyponatremia best journals Hyponatremia top journals Hyponatremia free medical journals Hyponatremia famous journals Hyponatremia Google Scholar indexed journals Hypernatremia articles Hypernatremia Research articles Hypernatremia review articles Hypernatremia PubMed articles Hypernatremia PubMed Central articles Hypernatremia 2023 articles Hypernatremia 2024 articles Hypernatremia Scopus articles Hypernatremia impact factor journals Hypernatremia Scopus journals Hypernatremia PubMed journals Hypernatremia medical journals Hypernatremia free journals Hypernatremia best journals Hypernatremia top journals Hypernatremia free medical journals Hypernatremia famous journals Hypernatremia Google Scholar indexed journals Water balance articles Water balance Research articles Water balance review articles Water balance PubMed articles Water balance PubMed Central articles Water balance 2023 articles Water balance 2024 articles Water balance Scopus articles Water balance impact factor journals Water balance Scopus journals Water balance PubMed journals Water balance medical journals Water balance free journals Water balance best journals Water balance top journals Water balance free medical journals Water balance famous journals Water balance Google Scholar indexed journals Electrolyte disorder articles Electrolyte disorder Research articles Electrolyte disorder review articles Electrolyte disorder PubMed articles Electrolyte disorder PubMed Central articles Electrolyte disorder 2023 articles Electrolyte disorder 2024 articles Electrolyte disorder Scopus articles Electrolyte disorder impact factor journals Electrolyte disorder Scopus journals Electrolyte disorder PubMed journals Electrolyte disorder medical journals Electrolyte disorder free journals Electrolyte disorder best journals Electrolyte disorder top journals Electrolyte disorder free medical journals Electrolyte disorder famous journals Electrolyte disorder Google Scholar indexed journals Internal Medicine articles Internal Medicine Research articles Internal Medicine review articles Internal Medicine PubMed articles Internal Medicine PubMed Central articles Internal Medicine 2023 articles Internal Medicine 2024 articles Internal Medicine Scopus articles Internal Medicine impact factor journals Internal Medicine Scopus journals Internal Medicine PubMed journals Internal Medicine medical journals Internal Medicine free journals Internal Medicine best journals Internal Medicine top journals Internal Medicine free medical journals Internal Medicine famous journals Internal Medicine Google Scholar indexed journals urological articles urological Research articles urological review articles urological PubMed articles urological PubMed Central articles urological 2023 articles urological 2024 articles urological Scopus articles urological impact factor journals urological Scopus journals urological PubMed journals urological medical journals urological free journals urological best journals urological top journals urological free medical journals urological famous journals urological Google Scholar indexed journals gynecological articles gynecological Research articles gynecological review articles gynecological PubMed articles gynecological PubMed Central articles gynecological 2023 articles gynecological 2024 articles gynecological Scopus articles gynecological impact factor journals gynecological Scopus journals gynecological PubMed journals gynecological medical journals gynecological free journals gynecological best journals gynecological top journals gynecological free medical journals gynecological famous journals gynecological Google Scholar indexed journals serum osmolality articles serum osmolality Research articles serum osmolality review articles serum osmolality PubMed articles serum osmolality PubMed Central articles serum osmolality 2023 articles serum osmolality 2024 articles serum osmolality Scopus articles serum osmolality impact factor journals serum osmolality Scopus journals serum osmolality PubMed journals serum osmolality medical journals serum osmolality free journals serum osmolality best journals serum osmolality top journals serum osmolality free medical journals serum osmolality famous journals serum osmolality Google Scholar indexed journals jaundice articles jaundice Research articles jaundice review articles jaundice PubMed articles jaundice PubMed Central articles jaundice 2023 articles jaundice 2024 articles jaundice Scopus articles jaundice impact factor journals jaundice Scopus journals jaundice PubMed journals jaundice medical journals jaundice free journals jaundice best journals jaundice top journals jaundice free medical journals jaundice famous journals jaundice Google Scholar indexed journals Total body water (TBW) articles Total body water (TBW) Research articles Total body water (TBW) review articles Total body water (TBW) PubMed articles Total body water (TBW) PubMed Central articles Total body water (TBW) 2023 articles Total body water (TBW) 2024 articles Total body water (TBW) Scopus articles Total body water (TBW) impact factor journals Total body water (TBW) Scopus journals Total body water (TBW) PubMed journals Total body water (TBW) medical journals Total body water (TBW) free journals Total body water (TBW) best journals Total body water (TBW) top journals Total body water (TBW) free medical journals Total body water (TBW) famous journals Total body water (TBW) Google Scholar indexed journals
Article Details
1. Hyponatremia
1.1 Hyponatremia and serum osmolality
Hyponatremia can be hypotonic, hypertonic or isotonic [1, 2]. See table 1.
|
1. Hypotonic hyponatremia (plasma osmolality < 280 mOsm/kg H2O): serum osmolality and sodium are both low due to excess water that was not excreted. This is by far the most encountered type of hyponatremia. 2. Hypertonic hyponatremia (plasma osmolality >295 mOsm/kg H2O): serum osmolality is high, and serum sodium is low as in hyperglycemia and administration of intravenous immune globulin (IVIG suspended in disaccharide such as sucrose). Water shifts from the intracellular to the extracellular compartment. 3. Isotonic hyponatremia (plasma osmolality is normal 280-295 mOsm/kg H2O): is encountered after some urological and gynecological surgeries due to absorption of sodium-free irrigation solutions such as mannitol, sorbitol or glycine which expand extracellular fluid space. No shift of water from the intracellular space occurs. 4. Pseudohyponatremia: is rare and is avoided by measuring sodium by direct ion-selective electrode. It is seen in certain clinical scenarios such as: a. Hypertriglyceridemia b. Paraproteinemia c. Obstructive jaundice due to high level of lipoprotein X (LpX) |
Table 1: Types of hyponatremia.
For the purpose of this discussion, hyponatremia refers to hyponatremia with hypoosmolality (hypotonic hyponatremia). Water is increased in the extracellular compartment relative to sodium, and the body’s ability to excrete excess water is impaired [1, 2]. Sodium is the main cation in the extracellular fluid and the main contributor (with its accompanying anion: chloride or bicarbonate)to serum osmolality [1]. Based on Edelman’s equation [3] serum sodium is approximately the sum of exchangeable sodium Nae+ and potassium Ke+ divided by total body water (TBW). Exchangeable sodium and potassium are osmotically active. Not all sodium and potassium in the body is osmotically active. An example of osmotically inactive sodium is the bound sodium in bone, cartilage and skin [4]. It is important to know that both Nae+ and Ke+ contribute to serum sodium, but the contribution of Nae+ is significantly larger because of its higher concentration. This issue becomes relevant in case of hyponatremia and severe hypokalemia, because potassium replacement will lead to a rise in serum sodium in a similar fashion to sodium replacement.

In hypokalemia sodium shifts from the extracellular space intracellularly to maintain cellular volume and osmolality [5]. Once potassium is replaced, sodium exits the cells, resulting in a rise in serum sodium. For example, giving 1 mEq/kg of IV potassium chloride to correct hypokalemia will raise serum sodium by 2 mEq/l assuming that TBW is 50% of body weight. Serum osmolality is a measurement of different solutes that exist in the serum [6]. The normal range in adults is280-295 mOsm/kgH2Oor280-295 mmol/kg (in SI units). Significant hypoosmolality is <285 mOsm/kg H2O, while significant hyperosmolality is >310 mOsm/kg H2O. Normal urine osmolality is 50-1200 mOsm/kg H2O.
Serum osmolality is estimated based on the following equation [6]:
Serum osmolality (mOsm/kg H2O) = 2 x Na (mEq/l) + glucose/18 (mg/dl) + BUN/2.8 (mg/dl)
If SI units are used the equation becomes:
Serum osmolality (mmol/kg) = 2 x Na (mmol/l) + glucose (mmol/l) + BUN (mmol/l)
In case of sodium the value is the same in mEq/l or mmol/l. If serum sodium is found to be low (< 135 mEq/l) on a laboratory test, the next step is to prove hypoosmolality. Clinical laboratories measure osmolality by anosmometerusing either a freezing point depression osmometer or vapor pressure osmometer. The above equation cannot be used to confirm hypoosmolality, rather it is used to calculate osmolar gap.
Osmolar gap = measured osmolality - calculated osmolality
See table 2 for important definitions [7].
1.2 Pseudohyponatremia
In hypotonic or “true” hyponatremia, sodium level in plasma water is decreased. Plasma consists of 7% lipids and proteins and 93% water. Pseudohyponatremia is associated with severe hyperlipidemia or hyperproteinemia (such as paraproteinemia) which decrease the water portion of plasma resulting in artificial hyponatremia, while sodium concentration in plasma water remains unchanged. It does not cause hypoosmolality. A triglyceride level of 1000 mg/dl artificially reduces serum sodium by about 2 mEq/l [8]. Measurement of serum sodium via ion-specific electrode (ISE) potentiometry makes pseudohyponatremia rare. Hyponatremia associated with severehyperglycemia is not considered pseudohyponatremia. It is the result of sodium dilution due to water shift from the intracellular compartment [9, 10]. This is hyponatremia with hyperosmolality (hypertonic hyponatremia) resulting from hyperglycemia. Hyponatremia quickly resolves with resolution of hyperglycemia; therefore, aggressive measures to correct serum sodium are not indicated is this case. In case of hyperglycemia, serum sodium decreases by about 1.6 mEq/l for every 100 mg/dl of glucose above the normal range which is around 100 mg/dl. The 1.6 correcting factor was originally reported by Katz in 1973 [9], others have disputed its accuracy [10].
Corrected sodium = measured sodium + [1.6 x (measured glucose -100/100)]
For example, if blood sugar is788 mg/dl and serum sodium is 122 mEq/l, corrected serum sodium is: 122 + [1.6 x(788-100/100)] =11, therefore true serum sodium is estimated to be: 122+ 11=133 mEq/l.
1.3 Pathogenesis of hyponatremia
Total body water (TBW) is approximately 60% of lean body weight in men and 50% of lean body weight in women. Extracellular fluid (ECF) is one-third of TBW, and intracellular fluid (ICF) is two-thirds of TBW. Extracellular fluid has two components: interstitial fluid (26% of TBW) and Plasma (7% of TBW). See Table 3.
It is critical to know that hyponatremia is usually due to an increase in extracellular water rather than a decrease in extracellular sodium [1, 2]. In other words, hyponatremia is a water balance disorder. Total body water may be increased (as in congestive heart failure, CHF), decreased (as in vomiting and diarrhea) or unchanged (as in the syndrome of inappropriate ADH secretion SIADH). Irrespective of total body sodium, hyponatremia results from an increase in extracellular water relative to ions (sodium with accompanying chloride are the main extracellular ions).
Water moves freely between the intracellular and extracellular space. As a result, the osmolality of the ECF and the ICF are equal. Serum sodium is determined by balance between water intake and excretion. In hyponatremia serum sodium declines because consumed water is not properly excreted, while in hypernatremia water intake is inadequate [1, 2, 5]. A patient with decompensated CHF and a serum sodium of 120 mEq/l has an increase in total body sodium. Hyponatremia is due to an increase in total body water relative to sodium; therefore, such a patient is treated with a loop diuretic to correct this imbalance [11]. Increasing sodium in the patient’s diet will worsen his CHF symptoms. Antidiuretic hormone (ADH)or vasopressin is the main hormone responsible for maintaining water and sodium balance in the body [1]. It is secreted from the posterior pituitary. An increase in serum osmolality even by 1% will increase ADH secretion and increase water absorption from the collecting tubules in the kidney resulting in the return of serum osmolality to the normal range of 280-295 mOsm/kg H2O [2]. ADH secretion is stimulated either by an increase in serum osmolality (osmotic stimulation) or a decrease in the effective circulatory blood volume (non-osmotic stimulation) as in cases of hemorrhage, dehydration and heart failure [11]. Broadly speaking ADH secretion is either appropriate (compensatory) as in patients with heart failure, liver cirrhosis and dehydration or inappropriate as in patients with the syndrome of inappropriate secretion of antidiuretic hormone (SIADH) [11].
1.4 Etiology of hypotonic hyponatremia
There are three general categories of hypotonic hyponatremia according to the volume of ECF [12]. See table 4.
1.4.1 Hypovolemic hyponatremia (hyponatremia associated with a decrease in ECF volume): Examples include loss of fluids via the GI track (vomiting, diarrhea), via the skin (excessive sweating, burns) or due to bleeding [13]. Some patients lose sodium due to renal causes such as use of diuretics or mineralocorticoids deficiency. Hypovolemiawill lead to increased water retention due to an increase in ADH secretion. If feasible, BP and pulse should be measured in the supine and upright positions. Orthostatic drop in BP associated with tachycardia may be indicative of volume loss. High BUN may indicate dehydration or GI bleeding. As indicated above, hyponatremia is water excess relative to sodium. If replacement fluids were hypotonic (for example D5W or 0.45 NaCl), hyponatremia may ensue irrespective of the tonicity of the fluids lost. Urine sodium is elevated if loss of fluid is due to a renal mechanism and is low (< 20 mEq/l) if the cause of fluidloss is extra-renal [14]. Urine sodium may be low if the last dose of a diuretic was taken several hours prior to the measurement. Thiazide diuretics are a common cause of hyponatremia, while loop diuretic tend to cause hypernatremia. Both types of diuretics lead to loss of sodium and water in urine; however, thiazide diuretics lead to greater loss of sodium relative to water, the reverse is true in loop diuretics [15-17]. A rare but an intriguing cause of hyponatremia with ECF decrease is cerebral salt wasting (CSW) [18]. It is critical to know that patients with CSW are dehydrated which is a distinguishing feature from patients with SIADH who are euvolemic. CSW is seen in patients with CNS disease especiallyseveral days after subarachnoid hemorrhage [19]. CSW is characterized by hyponatremia, serum hypoosmolality, urine osmolality>100-300 mOsm/kg H2O, urine Na > 40 mEq/l. Some patients with chronic kidney disease especially chronic interstitial nephritis have salt wasting nephropathy which is characterized by hypovolemia, hyponatremia and high urine Na > 40 mEq/l [20].
1.4.2 Hypervolemic hyponatremia (Hyponatremia associated with an increase in ECF volume): Patients in this category have signs of hypervolemia such as peripheral edema and ascites. Heart failure and liver cirrhosis are important causes. Due to a decrease in effective circulatory blood volume the kidneys receive a lesser amount of the cardiac output which results in an increase in ADH secretion and water retention [21]. Again, both water and sodium are retained but there is higher water retention relative to sodium and hence hyponatremia. Some refer to hyponatremia is these cases as dilutional. Urine sodium is low, unless the patient is taking diuretics. Other causes include patients with the nephrotic syndrome; and acute and chronic kidney disease. A common scenario is hospitalized patients when given excessive amount of hypotonic IVF such as D5W and 0.45 NaCl. Thishas led to serious complications and even death in some patients (especially young females)postoperatively when ADH is elevated due to stress [22]. Isotonic IVF such as 0.9 NaCl should be given to hospitalized patients especially postoperatively unless they have hypernatremia. Hyponatremia in beer potomania (beer is hypotonic) [23] and primary polydipsia [24] is the result of excess intake of water that overwhelms the kidneys' excretory ability. This happens when the amount of water ingested exceeds 10-15 L. In these patients ADH is maximally suppressed, water osmolality is low but since it cannot get lower than 50-100 mOsm/kg H2O, water retention ensues. Primary polydipsia is seen in patients with psychiatric history such as schizophrenia. Another important cause of hyponatremia is high intake of fluids accompanied by low intake of solutes [25]. This is encountered frequently in hospitalized patients especially elderly patients on low solute diet (tea and toast diet). Hyponatremia improves if intake of solutes increases. In these patients the ability to maximally dilute the urine (reduce urine osmolality) is impaired which leads to water retention.
1.4.3 Euvolemic hyponatremia: This category includes SIADH and also hypothyroidism [26] and adrenal insufficiency [27] (in both cases ADH secretion is increased). To ascertain the diagnosis of SIADH one should exclude hypothyroidism (by measuring TSH and free T4) and adrenal insufficiency (by performingACTH stimulation test). In some cases, the diagnosis isobvious and evaluating thyroid and adrenal functions may not be needed. In most cases, hypothyroidism is not the sole cause of hyponatremia unless the patient has myxedema or a significantly elevated TSH level > 50 mIU/ml [26].
Performing the ACTH stimulation test requires measurement of serum cortisolin early am, then the patient is given 250 mcg of cosyntropin IV. Serum cortisol is measured 30 minutes and 60 minutes later. A serum cortisol level > 18-20 mcg/dl is a normal response. A lower value would necessitate an endocrinology consultation [28]. Since SIADH is hyponatremia with euvolemia by definition, the patient is not expected to have peripheral edema, pulmonary edema or ascites [29, 30]. The normal response to hyponatremia is the excretion of a dilute urine (i.e. a decrease in urine osmolality), the reverse occurs in SIADH. SIADH is characterized by hyponatremia, low serum osmolality (<280 mOsm/kg H2O), high urine osmolality relative to serum osmolality(>100 mOsm/kg H2O) and high urine sodium (>30 mEq/l). Serum uric acid is usually below 4 mg/dl [2, 31]. Measurement of ADH is not readily available in most laboratories and is not indicated to confirm the diagnosis. See table 5 for causes of SIADH.
|
· Plasma osmolality · Plasma osmotic pressure is created by every ion and molecule dissolved in plasma; therefore, it depends on the total number of particles in plasma. · Tonicity = effective osmolality · Plasma tonicity is determined by effective osmoles, i.e. Solutes that do not cross cellular membrane (mainly Na and glucose), and not urea and alcohol (if present). · Plasma tonicity (mmol/kg) = 2 x Na (mmol/l) + glucose (mmol/l) |
Table 2: Definitions.
|
Examples |
TBW (L) |
ECF (L) |
ICF (L) |
|
60 kg woman |
30 |
10 (plasma = 2.1 L) |
20 |
|
70 kg man |
42 |
28 (plasma = 2.9 L) |
14 |
Table 3: Examples of TBW in a woman and a man.
|
1. Hyponatremia with hypovolemia Renal causes Diuretics especially thiazide diuretics Mineralocorticoid deficiency Cerebral salt wasting Salt wasting nephropathy Extrarenal causes GI (vomiting, diarrhea, bowel obstruction) Skin (excessive sweating, burns) Bleeding |
|
2. Hyponatremia with euvolemia SIADH Hypothyroidism Adrenal insufficiency |
|
3. Hyponatremia with hypervolemia Heart failure Liver cirrhosis Nephrotic syndrome Acute kidney injuryand chronic kidney disease Beer potomania Primary polydipsia Low intake of solutes |
Table 4: Causes of hypotonic hyponatremia.
|
1. CNS disease: tumors, abscesses, subdural hemorrhage, subarachnoid hemorrhage, encephalitis, meningitis, CVA and Guillain-Barrésyndrome. 2. Malignancies: especially small cell lung cancer (due to ectopic ADH secretion), other lung tumors, mediastinal tumors, pancreatic cancer, prostate cancer, uterine cancer, and leukemia. 3. Pulmonary disease: such as tuberculosis, pneumonia and empyema. 4. Medications: such as selective serotonin reuptake inhibitors (SSRIs), nicotine, tricyclic antidepressants, chlorpropamide, carbamazepine, phenothiazine, and intravenous cyclophosphamide. 5. AIDS. 6. Hereditary SIADH (rare): due to gain-of-function mutationsin the renal V2 receptor gene. 7. Idiopathic. |
Table 5: Causes of SIADH [2, 29, 32].
1.5 Symptoms of hyponatremia
Hyponatremia is seen in 15%-30% of hospitalized children and adults and it is mostly due to administration of hypotonic intravenous fluids [33, 34]. It is the most common electrolyte disturbance in hospitalized patients. Even mild hyponatremia is associated with falls, fractures and neurological impairment [35, 36]. In a Dutch study involving about 5000 community dwelling elderly residents (The Rotterdam Study), mild hyponatremia (133.4 ± 2.0 mEq/l) was seen in 7.7%, and was associated with an increase in all-cause mortality by 21%, and an increased risk of vertebral fractures and falls [35]. The symptoms are nonspecific and variable [1, 2]. Common symptoms include headaches, nausea and gait problems. Falls are common even in patients with mild hyponatremia. Severe symptoms include disorientation, delirium, seizures and coma. Severe manifestations are referred to as hyponatremic encephalopathy and are attributed to cerebral edema resulting from water shift into the brain due to serum hypoosmolality. The rapidity of hyponatremia determines the severity of symptoms. Slow and gradual decline in sodium gives the brain adequate time for adaptation. Brain cells get rid of sodium and potassium salts in addition to osmolytes. Major osmolytes are the amino acids taurine, glutamine and glutamate. This lowers the osmolality inside brain cells and protect from cerebral edema. Acute drop in sodium does not give the brain cells enough time to adapt and in severe cases will lead to cerebral edema, herniation and then death [37, 38]. See table 6.
1.6 Approach to the diagnosis of hyponatremia
In approaching the diagnosis of hyponatremia, the clinician focuses on history, physical examination and laboratory testing [1, 5, 13].
1.6.1 History: The cause of hyponatremia may be readily apparent from the history. Examples include history of GI losses (diarrhea, vomiting), medications (such as SSRIs, carbamazepine, thiazides), pulmonary or neurological conditions and high intake of water or alcohol. A prior history of hyponatremia is helpful as well.
1.6.2 Physical exam: The main goal is to determine volume status (hypovolemia vs euvolemia vs hypervolemia) which narrows the differential diagnosis considerably. Therefore, skin and mucous membranes examination; andBP and pulse measurements are paramount.
1.6.3 Laboratory testing: Extensive testing is not indicated if the diagnosis is obvious from the history and physical examination, and in cases of chronic and recurrent hyponatremia where a full work up had been done previously. Laboratory testing includes checking serum electrolytes, BUN, creatinine, serum osmolality, urine osmolality, urine sodium and potassium in a random specimen. In cases of SIADH it may be necessary to check TSH and free T4 to rule out hypothyroidism, and to do ACTH stimulation test to rule out adrenal insufficiency.
1.7 Management of hyponatremia
Hyponatremia can be mild, moderate or severe. Depending on duration, hyponatremia can be acute or chronic [1, 2, 5, 13]. See table 7. Management of hyponatremia requires considerable care and experience. Severe hyponatremia (Na <125 mEq/l) usually requires a specialist consultation. Most patients present with chronic hyponatremia (³ 48 h in duration). Acute hyponatremia that evolves over less than 48 is encountered less often. Examples include hyponatremia in users of 3, 4-Methyl?enedioxy?methamphetamine (ecstasy) [39], marathon runners [40] and psychotic patients with self-inflicted water intoxication [24]. The following are the general principles in the management of hyponatremia:
- Prevention of hyponatremia is paramount. Many cases of hyponatremia in hospitalized patients may be prevented by avoidance of hypotonic solutions such as D5W and 0.45 NaCl [22]. Other cases may be prevented by avoiding thiazide diuretics in patients with tendency for hyponatremia.When thiazide diuretics are initiated, electrolytes should be measured after one week, 4 weeks, and then every 3 months [15, 17]. If hyponatremia develops, thiazides should be discontinued, and the patient should not be rechallenged.
- If the cause of hyponatremia is a certain medication, effort should be made to stop it (for example: thiazides, SSRIs) [41]. If this is not feasible, measures should be taken to lessen the effect of hyponatremia in addition to frequent monitoring (such as institution of fluid restriction and increase in solutes intake).
- Correcting serum sodium up to 125 mEq/l is usually adequate to prevent major complications. Further correction should be done slowly over several days [42].
- Slow correction of sodium is paramount. There is no indication to increase serum sodium by more than 6-8 mEq/24 h [1, 2, 43, 44]. Quick correction especially >12 mEq/ 24 h might lead to osmotic demyelination syndrome (ODS) [37, 38, 45]. This disorder is encountered more often after rapid correction of chronic hyponatremia rather than acute hyponatremia. It is a rare but seriouscondition that occur 2-6 days after rapid correction of sodium. It manifests with dysarthria, dysphagia, behavioral disturbances, paraplegia or quadriplegia, coma and seizures. Risk factors for ODS include sodium level <120 mEq/l, alcoholism, liver disease and malnutrition. The diagnosis is made with MRI. A retrospective Swedish study in patients with ODS showed that at 3 months, 7.2% had died and 60.2% were functionally independent [45]. 70% ofthestudy patients were alcoholic andonly 7%had their sodium corrected at a rate ≤8mmol/L/24 h.
- If hyponatremia is associated with hypovolemia as in cases of dehydration or thiazide diuretics, the treatment should start with administration of 0.9 NaCl. Potassium should be replaced as well [1, 2, 13].
- If sodium is below 125 mEq/l and the patient is symptomatic, a specialist consult should be obtained to administer 3% hypertonic saline [46].
- In cases of hypervolemia as in heart failure, loop diuretics and water and saltrestrictions are utilized. Administration of sodium containing IV solutions may worsen the symptoms. Spironolactone may worsen hyponatremia [47].
- SIADH patients who aresymptomatic and have a sodium below 125 mEq/l, are treated with 3% saline [46]. Water restriction is helpful. If appropriate, a high solute diet (a diet high in salts and protein) may be helpful. In some cases, salt tablets and loop diuretics may be of benefit. 0.9 NaCl infusion is unlikely to be effective in patients with SIADH. Many have high urine osmolality. Hyponatremia may worsen with 0.9 NaCl if the sum of urine (Na+K) exceeds serum sodium. Infusion of 0.9 NaCl will decrease serum sodium because water is retained, and sodium is excreted in urine [1]. Aquaretics such as tolvaptan (vasopressin V2 receptor blocker) are particularly helpful in SIADH [48]. They may be utilized in heart failure patients with hypervolemic hyponatremia as well. Tolvaptan is initiated only in ahospitalized setting with sodium monitoring every 6 hours. While on tolvaptan, patients should have access to water and should not be water-restricted. Tolvaptan and other aquaretics should not be used in patients with impaired sensorium because they cannot drink to thirst. It is given orally with a starting dose of 15 mg daily. The dose may be increased if needed. It canbe continued after the patient is dischargedfora total of30 days with monitoring of liver enzymes [49]. Examples of other aquaretics include conivaptan, satavaptan and lixivaptan [1, 29]. Demeclocycline should no longer be used due to its nephrotoxicity and hepatotoxicity and the availability of tolvaptan [50]. It is useful to note that oral urea may be useful in the chronic management of hyponatremia. It is available for purchase without a prescription. It comes in 15 g packets, and it is mixed with water or juice. Patients use 1-4 packets per day [1, 51]. Palatability may be an issue for some patients.
- Serum sodium should be measured every 4-6 hours in hospitalized patientsundergoing acutetreatment for hyponatremia [1].
- When correcting hyponatremia, one should remember that both potassium and sodium contribute to the tonicity of the serum and therefore correcting hypokalemia will lead to a rise in serum sodium. Based
on Edelman Equation, serum sodium is the ratio of exchangeable sodium and potassium to total body water. This issue becomes critical in patients with hyponatremia and severe hypokalemia. Correcting hypokalemia with intravenous potassium chloride should be taken into account, otherwise excess correction of hyponatremia will ensue [52, 53].
- Follow these steps to determine the volume of IV solution that will be administered to the patient [29]:
a. Calculate TBW as above.
b. Determine the desired serum sodium level.
c. Determine the sodium content of the IV solution (154 mEq/l of 0.9 NaCl and 513 mEq/l of 3% NaCl). See table 8.
d. Use this simple formula to determine the volume:

For example, a 60 kg woman has TBW of 30 L. If her serum Na is 120 mEq/l and the desired Na is 125 mEq/l, the volume of 3% saline needed is:
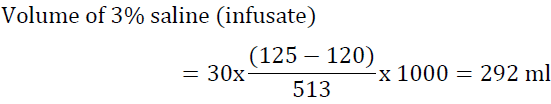
If the situation is not urgent it would be reasonable to infuse this amount over 15 h at a rate of 20 ml/h. Another way to look at the problems is to determine the increase in serum sodium after infusing one liter of the proposed solution. To accomplish this task, utilize the formula of Adrogue ?–Madias [54]:
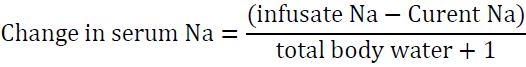
The “1” in the denominator is added because we are adding 1 liter of the infusate to total body water. For example, if the patient’s sodium is 123 mEq/l and total body water is 30 L, infusing 1 L of 0.9 NaCl which contains 154 mEq of Na will raise serum Na by 1 meq/l only.
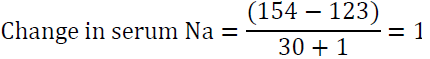
Modification of this formula is very useful in case of hyponatremia associated with severe potassium deficiency [54]. As detailed above both Na and K are active osmoles and correcting K will lead to a rise in serum sodium. The modified formula is:
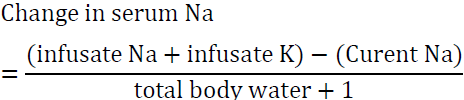
In the above example if the patient was also very hypokalemic and 60 meq of KCL was added to 1 L of 0.9 NS, the rise in Na after infusing one liter is approximately 3 meq as opposed to 1 meq in the first example. This situation illustrates the importance of factoring in potassium correction to avoid overcorrecting serum Na.
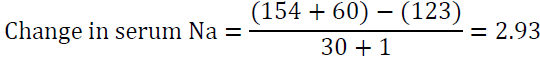
- A quick way to determine the approximate volume of 3% saline needed for infusion relies on the fact that 3% saline has 513 mEq/l or approximately 0.5 mEq/ml. If we assume that TBW is 0.5 L per Kg of lean body weight, then 1 ml/kg of 3% saline increases serum sodium by1 mEq/l. If we use the above example, we need 60 ml of 3% saline to raise serum sodium in a 60 kg woman by 1 mEq/l, to reach a 5 mEq/l increase, we need 5 x 60 = 300 ml which is a good approximation.
- All of the above calculations do not take into account urine output, insensible water losses or free water intake; therefore, it is paramount to check serum sodium every 4-6 hours during correction and to adjust the IVF rate accordingly [1, 2].
- Free water restriction is useful in patients with euvolemic and hypervolemic hyponatremia [55]. Severe water restriction (less than 1200 ml per 24 h) is not well tolerated and is poorly accepted by patients. A reasonable water restriction target is 500 ml below current urine output. Water restriction is unlikely to be effective in patients with high urine osmolality (> 500 mOsm/kg H2O) or if the sum of urine Na and K exceeds serum Na. It is common to see patients with hyponatremia placed on water restriction in the hospital for days without achieving any significant rise in Na. Water restriction is inappropriate as the sole management for hypervolemic and euvolemic hyponatremia if emergent treatment is needed, if the hyponatremia is severe (< 125 mEq/l), urine osmolality is > 500 mOsm/kg H2O or if urine Na + K > serum Na [1, 56]. A large registry of over 3000 hospitalized patients with hyponatremia (Na < 130 mEq/l) revealed that fluid restriction was the most common treatment followed by administration of 0.9 NaCl [57]. Hypertonic saline and tolvaptan were used in about 7% of patients. Fluid restriction without other measures was usually ineffective and most patients were discharged from the hospital with hyponatremia.
- Exercise hyponatremia is increasingly recognized in athletes due to excessive intake of hypotonic fluids (water overload) during strenuous physical activities such as marathon running [40]. Ingesting as much water as possible during such activities is unwarranted. Hyponatremia is the result of excess water drinking, and water retention due to non-osmotic release of ADH. Sodium loss through sweat contributes to a lesser degree. Symptoms can be severe including collapse, delirium and seizures. This necessitates quick infusion of 100 ml of 3% saline over 15-30 minutes to achieve a 4-6 mEq/l rise in serum sodium [42, 58, 59]. This quick infusion may be repeated up to two times if needed; a 10-minute interval between infusions is advisable. The recommended correction of serum sodium by no more than 8 mEq/24 h still applies. The total increase in sodium over 24 h is what matters rather than the hourly rate of increase. For example, if one raises serum sodium by 5 mEq/l over 15 minutes in a medical emergency, correction should not exceed another 3 mEq/l for the remainder of the 24 h. The following measures should be taken in case of overcorrection of serum sodium by more than 8 mEq/l in the first 24 h (these measures are more pressing if initial sodium is <120 mEq/l or if Na has risen by more than 12 mEq/l) [1, 60]:
- Discontinue NaCl solutions and tolvaptan.
- Measure sodium frequently (every 2-4 hours) until goal is achieved (current sodium is £ initial sodium + 8 mEq/l).
- Give 5% dextrose in water intravenously at 3 ml/kg/h.
- Consider desmopressin at 1-2 mcg intravenously every 8 h until goal is achieved.
- Two situations warrant special precautions regarding the development of ODS:
- The first is in patients with hyponatremia due to thiazide diuretics [61]. Upon discontinuation of the thiazide diuretic and initiation of treatment with a NaCl solution, a severe water diuresis can ensue leading to overcorrection of hyponatremia and the development of ODS. As alluded to earlier, these patients may have severe potassium deficit and potassium replacement should be taken into account to avoid sodium overcorrection.
- The second is in patient with primary polydipsia who maintain high water intake and high urine output. Upon institution of water restriction, these patients may continue to have significant water diuresis with subsequent overcorrection of hyponatremia. Clozapine (atypical antipsychotic) may help in managing hyponatremia in patients with primary polydipsia [62].
|
Manifestation |
Sequala |
|
Rapid increase in serum Na due to rapid correction of chronic hyponatremia |
Osmotic demyelination |
|
Rapid increase in serum Na due to acute onset of hypernatremia |
Osmotic demyelination |
|
Rapid decrease in serum Na due to acute hyponatremia |
Cerebral edema |
|
Rapid decrease in serum Na due to rapid correction of chronic hypernatremia |
Cerebral edema |
Table 6: Clinical sequalae of abrupt changes in serum sodium.
|
Mild hyponatremia: serum Na 130-134 mEq/l Moderate hyponatremia: serum Na 125-129 mEq/l Severe or profound hyponatremia: serum Na <125 mEq/l |
|
Acute hyponatremia < 48 h in duration Chronic hyponatremia ≥ 48 h in duration |
Table 7: Classification of hyponatremia.
|
IV solution (infusate) |
3% NaCl |
0.9 NaCl |
0.45 NaCl |
Ringer’s lactate |
0.2%NaCl in 5% dextrose |
5% dextrose |
|
Na content (mEq/l) |
513 |
154 |
77 |
130 |
34 |
0 |
Table 8: Sodium content of commonly used IV solutions.
2. Hypernatremia
Hypernatremia is seen in patient who do not have ready access to water. This includes infants, incapacitated patients (with hypodipsia) and patients in the intensive care unit (ICU). It is usually indicative of severe illnesses and multiple co-morbidities [63]. Thirst resulting from hypernatremia or hyperosmolality in general, will lead to a strong drive to seek and drink water which prevents hypernatremia in community dwelling people with water access.
Most hypernatremic patients are volume depleted. Some ICU patients have hypernatremia with hypervolemia due to administration of sodium bicarbonate or hypertonic IV solutions [64]. See table 9. Diabetes insipidus (DI) can be central (partial or complete absence of ADH) or nephrogenic (impaired response to ADH by renal collecting tubules) and is an important cause of hypernatremia [65]. Central DI can be complete or partial [66]. It can be the result of head injury, brain tumors (both malignant and benign), or brain surgery, while some cases remain idiopathic. Post-operative DI can be transient. Hereditary central DI is uncommon (10% of cases), both autosomal dominant and recessive forms have been described. Nephrogenic DI can be seen in hypokalemia, hypercalcemia, sickle cell renal disease and renal amyloidosis. Medications can cause nephrogenic DI, see Table 9. Hereditary nephrogenic DI is rare [67]. Hypernatremia can result in malaise, fatigue, weakness, and agitations. Symptoms can progress in severe cases to seizures, metabolic encephalopathy and coma. Hypernatremia is associated with high mortality when serum sodium exceeds 180 meq/l even if corrected properly [63, 64, 65]. Severe hypernatremia is associated with significant co-morbidities.
|
Dehydration (water loss) · Osmotic diarrhea: loss of water exceeds loss of solutes · Loop diuretics such as furosemide, bumetanide and torsemide · Patients with central and nephrogenic diabetes insipidus (DI) with no access to water. This becomes apparent upon hospitalization or loss of consciousness. Under normal circumstances their sodium is normal or slightly elevated. · Drug-induced nephrogenic diabetes insipidus: most commonly: lithium, amphotericin B, ofloxacin, ifosfamide, foscarnet, clozapine, cidofovir, orlistat and demeclocycline · Osmotic diuresis due to hyperglycemia or mannitol · Hypothalamic lesions affecting thirst (rare) · Enteral feeding if free water administration is deficient |
|
Sodium gain · Administration of hypertonic intravenous solutions (sodium bicarbonate or sodium chloride) to infants or patients in the intensive care unit · Acute salt poisoning: accidental or due to attempted suicide · Hypertonic feeding solutions |
Table 9: Causes of hypernatremia [63, 64, 65, 66, 68].
2.1 Water loss formula
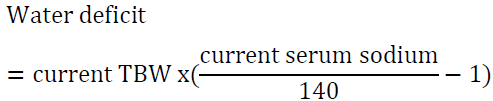
For example, a man who weighs 70 kg has TBW of 42 L. If his serum sodium is 160 meq/l, then his water deficit is 6 L. Another way to look at the problem is to set the desired sodium level then to calculate the amount of water needed to reach that goal. For example, a woman who weighs 60 kg has a TBW of 30 L. If she presents with a serum sodium of 176 mEq/l, the total water deficit based on the above formula is 7.7 L. Let us say than one would like to correct the sodium to 166 mEq/l in the first 24 h, we can calculate the amount of water needed by algebraic rearrangement of the above formula:
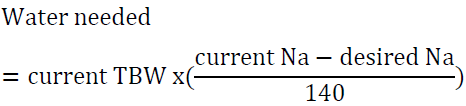
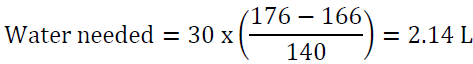
This amount can be given as D5W at 90 ml/h for the first 24 h. Insensible and ongoing losses will have to be taken into account as well.
2.2 Diagnosis of hypernatremia
Water deprivation test is useful in the differential diagnosis of hypernatremia when diabetes insipidus is suspected [69]. It is done when the diagnosis is not obvious because it is labor-intensive and time consuming. It requires a specialist consultation. It is usually done in a hospitalized setting, especially in patients suspected of having complete central DI who tend to have very low urine osmolality < 100 mOsm/kg/ H2O associated with severe polyuria. The patient cannot consume anything by mouth (NPO). Urine volume and body weight are recorded hourly. Urine osmolality, serum sodium and serum osmolality are checked on admission, then urine osmolality is check hourly, while serum sodium and serum osmolality are checked every 2 hours. Once serum sodium is >145 mEq/l and serum osmolality is > 300 mOsm/kg H2O, the level of vasopressin (ADH) is measured, followed by subcutaneously (SQ) injection of 5 units of aqueous vasopressin. Some use desmopressin (2-4 mcg IV or SQ) instead of aqueous vasopressin. Some experts use 3% NaCl infusion to achieve the desired rise in serum sodium and serum osmolality. If the patient presents with serum Na > 145 mEq/l and urine osmolality > 300 mOsm/kg H2O proceed immediately with vasopressin measurement and administration. In normal subjects, urine osmolality starts to rise quickly and reaches a plateau after about 4 hours. In patients with complete central DI or with nephrogenic DI, urine osmolality does not rise significantly. After administration of vasopressin, urine osmolality rises significantly in complete central DI while if fails to change in nephrogenic DI. Patients with partial central DI and primary polydipsia are usually differentiated based on history. In both conditions, urine osmolality rises with water deprivation but not to a maximum level. No change in urine osmolality is noted post vasopressin injection in patients with primary polydipsia while some rise is noted in patients with partial central DI.
2.3 Management of hypernatremia
Correction of hypernatremia should be done slowly and at a rate that does not exceed 12 meq/24 h [63]. Hypotonic solutions such as D5W and 0.45 NaCl are used, and serum sodium is checked every 6 hours. If the patient is hypotensive, 0.9 NaCl is used first to stabilized BP followed by hypotonic IV solutions. If we use the above example (Na 160 mEq/l) one can utilized D5W at 125 ml/h to provide 3 L per 24 h and to correct the deficit over 48 h. The rate may need to be increased depending on insensible losses. Checking sodium every 6 hours remains critical to avoid over or undercorrection. Patients with hypervolemic hyponatremia should be treated with a hypotonic IV solution and a diuretic to achieve a negative balance of sodium that exceeds the negative balance of water [70]. A combination of D5W and intravenous furosemide may be utilized with frequent electrolytes monitoring. Patients with central DI are treated with desmopressin [66]. Thiazide diuretics may be useful in nephrogenic DI in addition to correcting the underlying cause if possible [71].
3. Clinical Vignettes
1. A 73-year-old man underwent left hip open reduction and internal fixation after a fall at home. Postoperatively he was started on IVF: D5W at 100 ml/h. He became lethargic on the third post-operative day. A chemistry panel revealed a creatinine 1.4 mg/dl, Na 124 mEq/l, K 3.3 mEq/l. What is the etiology of his hyponatremia?
Answer: This is a very common scenario. Administration of hypotonic IVF (in this case D5W) is the most common cause of hyponatremia in hospitalized patients. In this case D5W was stopped, the patient was given 0.9 NaCl and K was replaced. Serum sodium was corrected to 134 mEq/l over 72 h. Use 0.9 NaCl or Ringer’s lactate in postoperative patients. Use hypotonic IV solutions only in patients with hypernatremia.
2. A 40-year-old woman with liver cirrhosis due to chronic hepatitis C infection presents with the following labs: Na 126 mEq/l, K 4.4 mEq/l, Cr 1.2 mg/dl, serum Osmolality 285 mOsm/kg H2O, Urine Osmolality 500 mOsm/kg H2O, urine Na 33 mEq/l, total protein 12 gm/dl, albumin 2.6 g/dl. What is the etiology of her hyponatremia?
Answer: Hyponatremia in this case is not due to SIADH, note that the patient is not hypoosmolar (serum osmolality is 285 mOsm/kg H2O). This is an example of pseudohyponatremia due to hypergammaglobulinemia due to hepatitis C.
3. A 50-year-old man presents with hyponatremia following subarachnoid hemorrhage. Na 120 mEq/l, serum osmolality 255 mOsm/kg H2O, urine Na 82 mEq/l, Urine osmolality 445 mOsm/kg H2O. Supine BP 105/62 mmHg and sitting BP 90/51 mmHg. What is the etiology of his hyponatremia?
Answer: Both SIADH and cerebral salt wasting (CSW) are associated with hypotonic hyponatremia, high urine Na and high urine osmolality. Patients with SIADH are euvolemic; however, patients with CSW are volume depleted. This patient has orthostatic hypotension consistent with volume depletion and his hyponatremia is due to CSW following subarachnoid hemorrhage.
4. An 80-year-old woman who weighs 56 kg is started on 25 mg chlorthalidone daily for management of hypertension. Her initial electrolytes panel was normal. One month later she presented to the emergency department with nausea and vomiting due to a viral gastroenteritis. Her laboratory studies showed: Na 118 mEq/l, K 2.3 mEq/l, serum osmolality 253 mOsm/kg H2O, urine Na 20 mEq/l, urine K, 22 mEq/l, urine osmolality 254 mOsm/kg H2O. How would you manage her electrolyte disorder?
Answer: The patient has severe hyponatremia and hypokalemia due to her viral gastroenteritis and chlorthalidone. Administration of potassium chloride to correct her potassium will also result in raising her sodium and should be taken into account. In this case the patient was given 4 doses of 20 mEq KCL in 100 ml of NaCl over 8 hours. She was also started on 0.9 NS at 75 ml/h.
Over 8 h, the total volume infused is (4 x 100) + (75 x 8) = 1000 ml
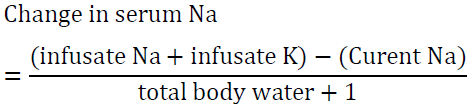
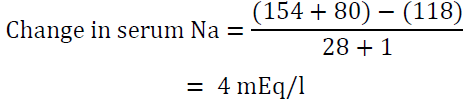
Therefore, the above regimen will raise serum sodium by 4 mEq/l in the first 8 hours which is a desirable rate of correction. If potassium was not taken into account, we reach an erroneous conclusion that the rise would be only 1.2 mEq/l. This may lead to the use of higher volume of 0.9 NaCl or the utilization of 3% NaCL and overcorrection of hyponatremia.
5. A 60-year-old man who weighs 80 kg presents with a seizure. Serum sodium is 105 mEq/l. He has been on sertraline for major depression and was started on hydrochlorothiazide for hypertension one week ago. What is the best approach to management?
Answer: This patient requires emergency treatment for hyponatremia with 3% NaCl. The use of 0.9 NaCl or tolvaptan is inappropriate for emergency management. Raising serum Na by 5 mEq/l is reasonable.
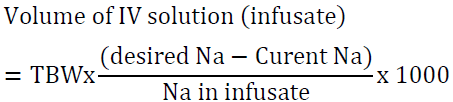
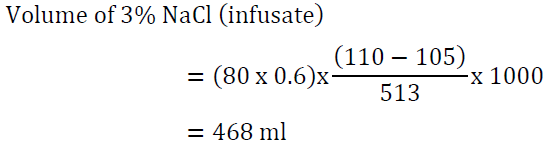
It would be reasonable to give the first 100 ml of the 3% NaCl solution over 15 minutes given the patient’s presentation with seizures. The other 365 ml can be given slowly over 10 hours. Serum sodium measurement every 2-4 hours is paramount.
6. A 55-year-old woman with chronic hyponatremia due to carbamazepine presents with a fall. Serum sodium is 120 mEq/l. Urine Na 45 mEq/l, urine osmolality 526 mOsm/kg H2O. Weight is 60 kg. What are the options for her management?
Answer: The most likely diagnosis is SIADH due to carbamazepine. 0.9 NS and/or water restriction are unlikely to be of significant benefit due to high urine osmolality. This is not an emergency. The patient has chronic hyponatremia that appears to have worsened. Tolvaptan is an appropriate choice starting at 15 mg daily. While on tolvaptan the patient should not be fluid restricted and serum sodium should be measured every 6 h. Alternatively, 3% NaCl may be used. If we utilized the formula above, we need 292 ml to raise serum sodium by 5 mEq/l. This should be infused at a rate of 20-30 ml/h. There is no need to infuse at a faster rate since this is a chronic condition and 3% NaCl can be damaging to the peripheral veins.
7. A 40-year-man with paranoid schizophrenia admits to excessive water drinking. He presents with gait disturbance and a serum sodium of 122 mEq/l. He was started on fluid restriction at 1200 ml/day and serum Na was ordered every 4 hours. You notice that his urine output is 1400 ml/h and his serum Na has risen to 132 mEq/l after 8 hours. How would you manage this patient?
Answer: This patient has primary polydipsia. He was placed on water restriction. His high urine output is due to water diuresis which leads to overcorrection of hyponatremia. The patient should be started on D5W at 3 ml/kg/h, in addition to intravenous desmopressin 1-2 mcg every 8 hours until the goal of correcting serum Na by no more than 6-8 mEq/l is achieved (serum Na ~ 128 mEq/l).
8. A 72-year-old man was admitted to the intensive care unit with pneumococcal pneumonia. He was intubated and started on mechanical ventilation. Three days later he was started on enteral nutrition with 50 ml of free water flushes down NG tube every 6 h. His sodium rose gradually from 142 mEq/l to 155 mEq/l over 3 days. How would you approach his hypernatremia?
Answer: The patient is critically ill with no free access to water. Hypernatremia is commonly encountered in patients on enteral nutrition. The first action is to increase free water flushes down NG tube. If this is ineffective an infusion of D5W should be started.
9. An 85-year-old woman with advanced dementia was sent to the emergency department from a skilled nursing facility due to obtundation. Her serum sodium was 181 mEq/l. She weighs 56 kg. How should her hypernatremia be corrected?
Answer: first, we utilize the water loss formula:
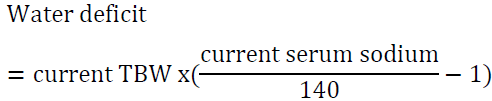
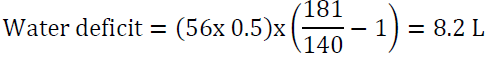
It would be reasonable to start D5W at 150 ml/h and to check serum sodium every 6 h. Avoid lowering Na by more than 12 mEq/24 h. The above water deficit does not take into account ongoing water loss (for example due to urination, vomiting or diarrhea) or insensible water loss. The rate may need to be adjusted depending on serum sodium measurements. Correcting serum sodium in this case will take about 4 days using the above guidelines: her sodium is 181-140 = 41 mEq/l above the normal range, if we correct by10 mEq/l per 24 h period, it will take 4 days to achieve the desired target.
10. A 44-year-old man with manic-depressive disorder has been stable for 3 years on lithium. He is now complaining of frequent urination. A 24 h urine collection revealed a urine volume of 3.2 Liters. Urine Na 35 mEq/l, urine K 33 mEq/l, urine protein is 13 mg/24 h, urine glucose is 0, urine osmolality 180 mOsm/kg H2O. Serum sodium is 144 mEq/l. How would you manage his condition?
Answer: The patient has nephrogenic diabetes insipidus due to lithium. His serum sodium is at the upper range of normal because he is able to drink, and significant hypernatremia is not expected. Urine osmolality is low, and he is polyuric (urine volume is above 3 L per 24 h). There is no evidence of solute (osmotic) diuresis. It would be reasonable to try amiloride in this case [68, 72]. Amiloride is a weak diuretic that blocks the sodium epithelial channel (the main entry site for lithium in the principal cells of the collecting duct) and may be particularly helpful in lithium-induced diabetes insipidus. Changing to a different agent is another option after consultation with the patient’s psychiatrist.
11. A 75-year-old woman presented with a serum Na of 128 mEq/l three weeks after initiation of hydrochlorothiazide 25 mg po daily for management of hypertension and lower extremities edema. Hydrochlorothiazide was stopped and she was started on an ACE inhibitor for her hypertension. One month later her serum Na is 134 mEq/l, she is asking if she can be restarted on hydrochlorothiazide for management of lower extremities edema.
Answer: This patient should not be started on any thiazide type diuretic because of her tendency for hyponatremia. A loop diuretic can be utilized at a low dose for edema management, for example, torsemide 10 mg po daily. Loop diuretics increase free water excretion in urine and are unlikely to cause hyponatremia. They are associated with hypernatremia. Electrolytes should be checked to monitor the patient’s sodium and potassium.
12. An 82-year-old man presents to the emergency department with a fall. Serum Na is 129 mEq/l. His appetite is poor. His only medication is candesartan for the management of hypertension. He is euvolemic on physical exam. Serum osmolality is 275 mOsm/kg H2O, urine volume is 1.5 L, sodium is 22 mEq/l, and urine osmolality is 155 mOsm/kg H2O. What is the etiology of his hyponatremia?
Answer: his hyponatremia is not due to SIADH. His urine osmolality and urine sodium are low. His hyponatremia is due to low intake of solutes. This is seen on a regular basis in hospitalized older adults with high fluid intake relative to their solute intake (tea and toast diet). The treatment is by implementing fluid restriction and increasing intake of solids. Salt tablets and oral urea packets may be considered as well.
Conclusion
- Hyponatremia and hypernatremia are encountered commonly in hospitalized patients.
- Hyponatremia and hypernatremia are disorders of water balance. Hyponatremia is usually due to failure to excrete excess water rather than excess sodium loss. Hypernatremia is usually due to inadequate water intake or water loss rather than excess sodium intake.
- Use of hypotonic intravenous fluids is the most common cause for hyponatremia in hospitalized patients.
- Both hyponatremia and hypernatremia should be corrected slowly.
- 3% NaCl is indicated for emergency treatment of hyponatremia.
- Patients on thiazide diuretics should be monitored for hyponatremia. Thiazides should not be restarted after correction of hyponatremia.
- Potassium replacement should be taken into account when correcting hyponatremia.
- Aquaretics such as tolvaptan are effective for treatment of euvolemic and hypervolemic hyponatremia.
- Water restriction is ineffective as the sole treatment for severe hyponatremia especially if urine osmolality is significantly elevated.
References
- Verbalis JG, Goldsmith SR, Greenberg A, et al. Diagnosis, Evaluation, and Treatment of Hyponatremia: Expert Panel Recommendations. The American Journal of Medicine 126 (2013): S1-S42.
- Spasovski G, Vanholder R, Allolio B, et al. Clinical practice guideline on diagnosis and treatment of hyponatremia. European Journal of Endocrinology 170 (2014): G1-G47.
- Edelman IS, Leibman J, O’Meara MP, et al. Interrelations between serum sodium concentration, serum osmolarity and total exchangeable sodium, total exchangeable potassium and total body water. J Clin Invest 37 (1958): 1236-1256.
- Selvarajah V, Connolly K, McEniery C, et al. Skin Sodium and Hypertension: a Paradigm Shift?. Curr Hypertens Rep 20 (2018): 94.
- Androgué HJ, Madias NE. The Challenge of Hyponatremia. Journal of The American Society of Nephrology 23 (2012):1140-1148.
- Faria DK, Mendes ME, Sumita NM. The measurement of serum osmolality and its application to clinical practice and laboratory: literature review. J Bras Patol Med Lab 53 (2017): 38-45.
- Vujovic P, Chirillo M, Silverthorn U. Learning (by) osmosis: an approach to teaching osmolality and tonicity. Adv Physiol Educ 42 (2018): 626-635.
- Weisberg LS. Pseudohyponatremia: a reappraisal [Review]. Am J Med 86 (1989): 315-318.
- Katz MA. Hyperglycemia-induced hyponatremia-calculation of expected serum sodium depression. N Engl J Med 289 (1973): 843-844.
- Hillier TA, Abbott RD, Barrett EJ. Hyponatremia: evaluating the correction factor for hyperglycemia. American Journal of Medicine 106 (1999): 399-403.
- Schrier RW. Pathogenesis of sodium and water retention in high- output and low-output cardiac failure, nephrotic syndrome, cirrhosis, and pregnancy. N Engl J Med 319 (1988): 1127-1134.
- Chung HM, Kluge R, Schrier RW, et al. Clinical assessment of extracellular fluid volume in hyponatremia. Am J Med 83 (1987): 905-908.
- Sahay M, Sahay R. Hyponatremia: A practical approach. Indian J Endocr metab, 18 (2014): 760-771.
- Milionis HJ, Liamis GL, Elisaf MS. The hyponatremic patient: a systematic approach to laboratory diagnosis. CMAJ 166 (2002): 1056-1062,
- Chow KM, Kwan BC, Szeto CC. Clinical studies of thiazide-induced hyponatremia. J Natl Med Assoc 96 (2004): 1305-1308.
- Sonnenblick M, Friedlander Y, Rosin AJ. Diuretic-induced severe hyponatremia. Review and analysis of 129 reported patients [Review]. Chest 103 (1993): 601-606.
- Hix JK, Silver S, Sterns RH. Diuretic-associated hyponatremia. Semin Nephrol 31 (2011): 553-566.
- Hannon MJ, Finucane FM, Sherlock M, et al. Clinical review: disorders of water homeostasis in neurosurgical patients. J Clin Endocrinol Metab 97 (2012): 1423-1433.
- Sherlock M, O’Sullivan E, Agha A, et al. The incidence and pathophysiology of hyponatraemia after subarachnoid haemorrhage. Clinical Endocrinology 64 (2006): 250-254.
- Uribarri J, Oh M S, Carroll H J, et al. Clinical presentation and mechanisms. American Journal of Nephrology 39 (1983): 193-198.
- Schrier RW. An Odyssey into the milieu intérieur: pondering the enigmas. J Am Soc Nephrol 2 (1992): 1549-1559.
- Steele A, Gowrishankar M, Abrahamson S, et al. Postoperative Hyponatremia despite Near-Isotonic Saline Infusion: A Phenomenon of Desalination. Ann Intern Med 126 (1997): 20-25.
- Demanet JC, Bonnyns M, Bleiberg H, et al. Coma due to water intoxication in beer drinkers. Lancet 2 (1971): 1115-1117.
- de Leon J, Verghese C, Tracy JI, et al. Polydipsia and water intoxication in psychiatric patients: a review of the epidemiological literature. Bio Psychiatry 35 (1994): 408-419.
- Thaler SM, Teitelbaum I, Berl T. "Beer potomania" in non-beer drinkers: effect of low dietary solute intake. Am J Kidney Dis 31 (1998): 1028-1031.
- Derubertis FR Jr, Michelis MF, Bloom ME, et al. Impaired water excretion in myxedema. Am J Med 51 (1971): 41-53.
- Ishikawa S, Schrier RW. Effect of arginine vasopressin antagonist on renal water excretion in glucocorticoid and mineralocorticoid deficient rats. Kidney Int 22 (1982): 587-593.
- Ospina NS, Nofal A, Bancos I, et al. ACTH Stimulation Tests for the Diagnosis of Adrenal Insufficiency: Systematic Review and Meta-Analysis. The Journal of Clinical Endocrinology & Metabolism, 101 (2016): 427-434.
- Ellison D, Berl T. The Syndrome of Inappropriate Antidiuresis. N Engl J Med 356 (2007): 2064-2072.
- Robertson GL. Regulation of arginine vasopressin in the syndrome of inappropriate antidiuresis. Am J Med 119 (2006): :S36-S42.
- Beck LH. Hypouricemia in the syndrome of inappropriate secretion of antidiuretic hormone. N Engl J Med, 301 (1979): 528-530.
- Chonchol M, Berl M. Hyponatremia. In: DuBose TD Jr, Hamm LL, eds. Acid- base and electrolyte disorders: a companion to Brenner & Rector’s The Kidney. Philadelphia: Saunders (2002): 229-239.
- Upadhyay A, Jaber BL, Madias NE. Incidence and prevalence of hyponatremia. Am J Med 119 (2006): S30-S35.
- Carandang F, Anglemyer A, Longhurst CA, et al. Association between maintenance fluid tonicity and hospital-acquired hyponatremia. J Pediatr 163 (2013): 1646-1651.
- Hoorn EJ, Rivadeneira F, van Meurs JB, et al Mild hyponatremia as a risk factor for fractures: The Rotterdam Study. J Bone Miner Res 26 (2011): 1822-1828.
- Sajadieh A, Binici Z, Mouridsen MR, et al. Mild hyponatremia carries a poor prognosis in community subjects. Am J Med 122 (2009): 679-686.
- Sterns RH, Riggs JE, Schochet SS Jr. Osmotic demyelination syndrome following correction of hyponatremia. N Engl J Med 314 (1986): 1535-1542.
- Sterns RH, Cappuccio JD, Silver SM, et al. Neurologic sequelae after treatment of severe hyponatremia: a multicenter perspective. J Am Soc Nephrol 4 (1994): 1522-1530.
- Campbell GA, Rosner MH. The Agony of Ecstasy: MDMA (3, 4-Methylenedioxymethamphetamine) and the Kidney. Clin J Am Soc Nephrol 3 (2008): 1852-1860.
- Almond CS, Shin AY, Fortescue EB, et al. Hyponatremia among runners in the Boston Marathon. N Engl J Med 352 (2005): 1550-1556.
- Jacob S, Spinler SA. Hyponatremia associated with selective serotonin-reuptake inhibitors in older adults. Ann Pharmacother 40 (2006):1618-1622.
- Sterns R, Nigwekar S, Hix J. The treatment of hyponatremia. Semin Nephrol 29 (2009): 282-299.
- Hsu Y-J, Chiu J-S, Lu K-C, et al. Biochemical and etiological characteristics of acute hyponatremia in the emergency department. Journal of Emergency Medicine 29 (2005): 369-374.
- Sterns RH. Treating hyponatremia: why haste makes waste. Southern Medical Journal 87 (1994): 1283-1287.
- Aegisdottir H, Cooray C, Wirdefeldt K, et al. Incidence of osmotic demyelination syndrome in Sweden: A nationwide study. Acta Neurol Scand 140 (2019): 342.
- Sterns RH. Severe symptomatic hyponatremia: treatment and outcome. A study of 64 cases. Ann Intern Med 107 (1987): 656-664.
- Goland S, Naugolny V, Korbut Z, et al. Appropriateness and complications of the use of spironolactone in patients treated in a heart failure clinic. European Journal of Internal Medicine 22 (2011): 424-427.
- Schrier RW, Gross P, Gheorghiade M, et al. SALT Investigators: Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med 355 (2006): 2099-2112.
- Berl T. Vasopressin Antagonists. N Engl J Med 372 (2015): 2207-2216.
- Miller PD, Linas SL, Schrier RW. Plasma demeclocycline levels and nephrotoxicity. Correlation in hyponatremic cirrhotic patients. JAMA 243 (1980): 2513-2515.
- Coussement J, Danguy C, Zouaoui-Boudjeltia K, et al. Treatment of the syndrome of inappropriate secretion of antidiuretic hormone with urea in critically ill patients. Am J Nephrol 35 (2012): 265-270.
- Fichman MP, Vorherr H, Kleeman CR, et al. Diuretic-induced hyponatremia. Ann Intern Med 75 (1971): 853-863.
- Berl T, Rastegar A. A patient with severe hyponatremia and hypokalemia: osmotic demyelination following potassium repletion. Am J Kidney Dis 55 (2010): 742-748.
- Adrogué HJ, Madias NE. Hyponatremia. N Engl J Med 342 (2000): 1581-1589.
- Goh KP. Management of Hyponatremia. Am Fam Physician 69 (2004): 2387-2394.
- Cuesta M, Ortolá A, Garrahy A, et al. Predictors of failure to respond to fluid restriction in SIAD in clinical practice; time to re-evaluate clinical guidelines? QJM: An International Journal of Medicine 110 (2017): 489-492.
- Greenberg A, Verbalis JG, Amin AN, et al. Current treatment practice and outcomes. Report of the hyponatremia registry. Kidney Int 88 (2015): 167-177.
- Francony G, Fauvage B, Falcon D, et al. Equimolar doses of mannitol and hypertonic saline in the treatment of increased intracranial pressure. Crit Care Med 36 (2008): 795-800.
- Hew-Butler T, Rosner MH, Fowkes-Godek S, et al. Statement of the Third International Exercise-Associated Hyponatremia Consensus Development Conference, Carlsbad, California. Clin J Sport Med 25 (2015): 303-320.
- Sood L, Sterns RH, Hix JK, et al. Hypertonic saline and desmopressin: a simple strategy for safe correction of severe hyponatremia. Am J Kidney Dis 61 (2013): 571-578.
- George J, Zafar W, Bucaloiu ID, et al. Risk Factors and Outcomes of Rapid Correction of Severe Hyponatremia. Clin J Am Soc Nephrol 13 (2018): 984-992.
- Canuso CM, Goldman MB. Clozapine restores water balance in schizophrenic patients with polydipsia-hyponatremia syndrome. J Neuropsychiatry Clin Neurosci 11 (1999): 86-90.
- Adrogué HJ, Madias NE. Hypernatremia. N Engl J Med 342 (2000): 1493-1499.
- Palevsky PM, Bhagrath R, Greenberg A. Hypernatremia in hospitalized patients. Ann Intern Med 124 (1996): 197-203.
- Sterns RH: Disorders of plasma sodium–causes, consequences, and correction. N Engl J Med 372 (2015): 55-65.
- Arima H, Azuma Y, Morishita Y, et al. Central diabetes insipidus. Nagoya J Med Sci 78 (2016): 349-358.
- Milano S, Carmosino M, Gerbino A, et al. Hereditary Nephrogenic Diabetes Insipidus: Pathophysiology and Possible Treatment. An Update. Int J Mol Sci 18 (2017): 2385.
- Bedford J, Weggery S, Ellis G, et al. Lithium-induced Nephrogenic Diabetes Insipidus: Renal Effects of Amiloride. Clin J Am Soc Nephrol 3 (2008): 1324-1331.
- Fenske W, Allolio B. Clinical review: Current state and future perspectives in the diagnosis of diabetes insipidus: a clinical review. J Clin Endocrinol Metab 97 (2012): 3426-3437.
- Nguyen MK, Kurtz I. Correction of hypervolaemic hypernatraemia by inducing negative Na+ and K+ balance in excess of negative water balance: a new quantitative approach. Nephrol Dial Transplant 23 (2008): 2223-2227.
- Singer I, Oster JR, Fishman LM. The management of diabetes insipidus in adults. Arch Intern Med 157 (1997): 1293-1301.
- Lodin M, Dwyer J. The role of amiloride in managing patients with lithium-induced nephrogenic diabetes insipidus. Journal of Pharmacy Practice and Research 47 (2017): 389-392.


 Impact Factor: * 3.6
Impact Factor: * 3.6 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 