Intestinal Polyparasitism in a Mexican Family and Morphological Diversity of the Ascaris Lumbricoides Eggs
Article Information
Ana Luisa Madriz-Elisondo1, María de la Luz Galván-Ramírez2*, Marco Antonio Cardona-López1
1Departamento de Ciencias Médicas y de la Vida, Centro Universitario de la Ciénega, Universidad de Guadalajara, Mexico
2Departamento de Fisiología, Centro Universitario de Ciencias de la Salud, Universidad de Guadalajara, Mexico
*Corresponding Author: María de la Luz Galván-Ramírez, Departamento de Fisiología, Centro Universitario de Ciencias de la Salud, Universidad de Guadalajara, Av. Sierra Mojada No. 950, 44320 Guadalajara, Jalisco, México
Received: 03 August 2020; Accepted: 18 August 2020; Published: 27 August 2020
Citation:
Madriz-Elisondo AL, Galván-Ramírez ML, Cardona-Lopez MA. Intestinal Polyparasitism in a Mexican Family and Morphological Diversity of the Ascaris Lumbricoides Eggs. Journal of Pediatrics, Perinatology and Child Health 4 (2020): 093-098.
View / Download Pdf Share at FacebookAbstract
Intestinal polyparasitism represents a public health problem. It is more prevalent in developing countries like Mexico and children are the most affected. It has been associated with several factors, including the number of family members. However, intestinal polyparasitism in the family is underestimated. After explanation of the study and informed consent, the family was invited to provide three stool samples which were analyzed by direct and concentration methods as well as methods such as Graham and Kato Katz. The prevalence of intestinal polyparasitism in the family was 100%. The protozoa and helminths identified were: Entamoeba histolytica/Entamoeba dispar /Entamoeba moshkovskii, Blastocystis spp, Endolimax nana, Entamoeba coli, Ascaris lumbricoides y Enterobius vermicularis. The father and one of the children presented 6 parasites. The 5 year-old children presented severe infection by Ascaris lumbricoides, with 68,436 eggs per gram of faeces. Most of the participants reported more than two gastrointestinal symptoms. A morphological diversity of the Ascaris lumbricoides eggs were found in the family. The differentiation between infertile eggs of this helminth and detritus transcends for a reliable diagnosis. This case of familial intestinal polyparasitism shows the need for the timely diagnosis of intestinal parasitosis for specific treatment and the prevention of complications as well as for implementation of health education.
Keywords
Polyparasitism, Helminths, Protozoa, Ascaris lumbricoides, Enterobius vermicularis, Entamoeba spp, Risk factors, Mexico
Polyparasitism articles, Helminths articles, Protozoa articles, Ascaris lumbricoides articles, Enterobius vermicularis articles, Entamoeba spp articles, Risk factors articles, Mexico articles
Article Details
1. Introduction
Parasitic intestinal infections represents a public health problem worldwide because cause significant morbidity in children. These types of infections are widely prevalent in developing countries such as Mexico, although the national deworming campaign is administered twice a year [1, 2]. Polyparasitism (infection with two or more parasites and can be to protozoans, helminths or both) [2, 3] has been investigated in children and adults [4]. On the other hand, intestinal polyparasitism has been associated with drinking non-potable water, to walk barefoot, the presence of other family members with enteroparasites, consumption of unwashed vegetables, absence of a toilet in the house, poor hygiene habits [5] and the number of family members [6]. Intestinal polyparasitism in children is important because it could affect their physical and mental development, especially when they have severe helminth infections [7]. Intestinal polyparasitism is significantly more common than single infections [3, 7] and its prevalence has been reported up to 71.4% [5]. In Mexico, intestinal polyparasitism is over 50% [8]. However, intestinal polyparasitism in families has been under-studied [9]. Hence the importance of the present clinical case.
2. Case Report
We present the case of a family with intestinal polyparasitism, including a 30-year-old father working as a mason, a 29-year-old housewife mother and their four children. The family lived in a home consisting of one room and one bathroom with no flushing toilet. The home had a sheet metal roof, dirt floors and adobe walls. It had electricity and running water. With prior written informed consent, stool samples were requested from the family. They were analysed using direct coproparasitoscopic methods with Lugol’s solution, concentration methods with zinc sulfate and formalin-ethyl acetate, the Kato-Katz technique to estimate the severity of infection by A. lumbricoides and the Graham method for E. vermicularis for diagnosis in children [10] Figure 1.
The family members, their age, symptoms and the parasites identified in each are shown in (Table 1). After diagnosis, the adults were treated with nitazoxanide (500 mg) + mebendazole (167 mg) + quinfamide (767 mg) in a single dose per day. The children were referred to the health centre of their community for clinical evaluation and treatment in case necessary.
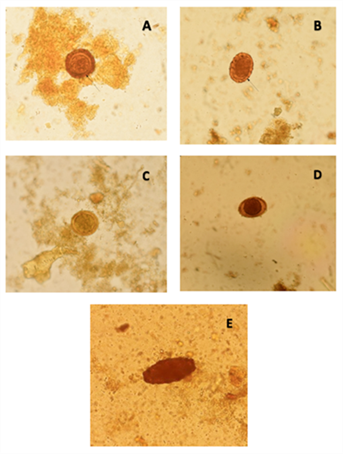
Figure 1: Morphological diversity of A. lumbricoides eggs observed by the direct Lugol’s solution method (40 X). (A) Round egg with an outer cover; (B) Oval egg with an outer cover. The arrows in A and B indicate the presence of an albuminoid outer layer with a wavy and irregular appearance; (C) Round decorticated egg; D) Oval decorticated egg. The lack of an outer layer is due to prolonged exposure to pancreatic secretions; (E) Infertile egg with an external cover.
Table 1: Polyparasitism in the members of the family studied.
|
Relationship |
Age (years) |
Symptoms referred by patients |
Parasites identified |
|
Mother |
29 |
Abdominal pain, constipation, tenesmus, abdominal distention, headache and decreased appetite. |
Endolimax nana Blastocystis spp. A. lumbricoides |
|
Father |
30 |
Abdominal and epigastric pain; liquid, mucoid, bloody diarrhoea, abdominal cramps, tenesmus, anal pruritus and abdominal distension. |
Entamoeba spp* Entamoeba coli Endolimax nana Blastocystis spp. A. lumbricoides E. vermicularis** |
|
Son |
9 |
Epigastric pain and anal pruritus. |
Blastocystis spp. E. vermicularis ** A. lumbricoides |
|
Son |
7 |
Anal pruritus. |
Entamoeba spp* Blastocystis spp. E. vermicularis** A. lumbricoides |
|
Son |
5 |
Abdominal pain, tenesmus, anal pruritus, decreased appetite, bloating and cramping. |
Entamoeba spp* Entamoeba coli Endolimax nana Blastocystis spp. E. vermicularis** A. lumbricoides |
|
Son |
1.4 |
Constipation, anal pruritus and abdominal distension. |
Blastocystis spp. E. vermicularis** |
*Entamoeba spp (Entamoeba histolytica/Entamoeba dispar/ Entamoeba moshkovskii).
**E. vermicularis was found by direct examination with Lugol’s solution in the father and by the Graham method in the children.
3. Discussion
Epidemiological studies that report intestinal polyparasitism have been performed [3, 5]. However, we present the case where all the members of the family were parasitized. Number of family members have been associated with intestinal parasites in children [6]. In our case, the father and son presented up to six parasites y the family members reported having several gastrointestinal symptoms at the time of the study. Protozoa and helminths were diagnosed.
The father was who reported signs and symptoms suggestive of amebiasis. Particularly, in endemic countries like Mexico, this criterion is useful for making a clinical diagnosis. Furthermore, microscopy represents a valuable tool because it is considered the gold standard. However, to differentiate between the species of the genus Entamoeba the Multiplex real-time PCR can be used [11]. This method was not applied in the present case, representing a limitation because the clinical manifestations of amoebic infection by E. histolytica vary from asymptomatic to severe symptoms, such as dysentery and extraintestinal abscesses, whereas E. dispar, E. moshkovskii and are unable to cause invasive disease; therefore, they do not require treatment [12, 13]. In the present case of familial polyparasitism, Blastocystis spp. were diagnosed in all family members. Other authors have reported high prevalence of this microorganism. The most common symptoms associated with this microorganism are nausea, loss of appetite, abdominal distention and pain, flatulence, acute or chronic diarrhoea and irritable colon [14].
Furthermore, two helminths Enterobius vermicularis and Ascaris lumbricoides coexisted in four members of the family. The first is easily transmitted by direct person-to-person. Anal and vaginal pruritus are the most common symptom associated with this parasite in symptomatic patients. The anal pruritus was presented by five family members which were parasitized with this helminth. Scratching of the perianal region directly contributes to the spread of the parasite eggs in bed sheets, pyjamas and mattresses; therefore, infections occur in families, kindergartens and primary schools. The prevalence of this parasite is mainly related to public health and personal hygiene [15].
Adults of Ascaris lumbricoides in the small intestine causes nausea, vomiting, discomfort, pain epigastric. Of the six family members, five were diagnosed with A. lumbricoides. The parents and the 9-year-old child presented mild infection with counts lower than 5,000 eggs per gram of faeces (epg). Severe infections were found in the other two parasitised children aged 7 and 5 years, with 50,376 and 68,436 epg, respectively. Knowing the epg value is important because moderate to severe infections may result in impaired digestion and malabsorption of protein, lactose, some fat-soluble vitamins and therefore malnutrition in children [16]. On the other hand, the probability of intestinal obstruction is related to the intensity of the infection [17]. A. lumbricoides eggs proliferate more intensively in children ranging in age from 5 to 10 years, decreasing dramatically without disappearing in adults [18]. This coincides with what was presented in this case, where the highest counts per gram of stool were in children aged 5 and 7 with respect to their parents who had mild to moderate infections. A morphological diversity of the A.lumbricoides eggs was observed. It is important to distinguish between different types of eggs so as not to confuse an infertile egg with an artefact as well as to apply the appropriate methods for correct diagnosis. For example, infertile eggs do not float with the zinc sulfate solutions used to concentrate the parasitic stages [10]. This information is important because when egg counting is done, both types of eggs should be considered [Figure 1].
4. Conclusion
This case of familial polyparasitism shows the various socioeconomic and hygienic factors that combined to favour conditions conducive to acquisition of up to six parasites. It is necessary to implement health education programmes that emphasise hygiene habits, especially hand washing, as well as timely and proper diagnosis and treatment of the entire family to prevent spread not only among the family but also in schools.
References
- Panti-May JA, Zonta ML, Cociancic P, et al. Occurrence of intestinal parasites in Mayan children from Yucatán, Mexico. Acta Tropica 195 (2019): 58-61.
- Quihui-Cota L, Morales-Figueroa GG. Persistence of intestinal parasitic infections during the national de-worming campaign in schoolchildren of northwestern Mexico: a cross-sectional study. Ann Gastroenterol 25 (2012): 57-60.
- Donohue RE, Cross ZK, Michael E. The extent, nature, and pathogenic consequences of helminth polyparasitism in humans: A meta-analysis. PLoS Negl Trop Dis 13 (2019): e0007455.
- Galván-Ramírez ML, Madriz-Elisondo AL, Ramírez CGT, et al. Enteroparasitism and Risk Factors Associated with Clinical Manifestations in Children and Adults of Jalisco State in Western Mexico. Osong Public Health Res Perspect 10 (2019): 39-48.
- Al-Delaimy AK, Al-Mekhlafi HM, Nasr NA, et al. Epidemiology of Intestinal Polyparasitism among Orang Asli School Children in Rural Malaysia. PLOS Neglected Tropical Diseases 8 (2014): 3074.
- Forson AO, Arthur I, Ayeh-Kumi PF. The role of family size, employment and education of parents in the prevalence of intestinal parasitic infections in school children in Accra. PloS one 13 (2018): e0192303.
- Keiser J, N'Goran EK, Traoré M, et al. Polyparasitism with Schistosoma Mansoni, Geohelminths, and Intestinal Protozoa in Rural Côte D'Ivoire. The Journal of Parasitology 88 (2002): 461-466.
- Quihui-Cota L, Valencia ME, Crompton DW, et al. Prevalence and intensity of intestinal parasitic infections in relation to nutritional status in Mexican school children. Trans R Soc Trop Med Hyg 98 (2004): 653-659.
- Pullan R, Brooker S. The health impact of polyparasitism in humans: are we under-estimating the burden of parasitic diseases?. Parasitology 135 (2008): 783-794.
- Clinical and Laboratories Standards Institute. Procedures for the recovery and identification of parasites from the intestinal tract; approved guideline, 2nd CLSI document M28-A2. Clinical and Laboratories Standards Institute (2005).
- Saidin S, Othman N, Noordin R. Update on laboratory diagnosis of amoebiasis. Eur J Clin Microbiol Infect Dis 38 (2019): 15-38.
- Parija SC, Mandal J, Ponnambath DK. Laboratory methods of identification of Entamoeba histolytica and its differentiation from look-alike Entamoeba spp. Trop Parasitol 4 (2014): 90-95.
- Sharbatkhori M, Nazemalhosseini-Mojarad E, Cheraghali F, et al. Discrimination of Entamoeba Spp. in children with dysentery. Gastroenterol Hepatol Bed Bench 7 (2014): 164-167.
- Tan KS. New insights on classification, identification, and clinical relevance of Blastocystis spp. Clinical Microbiology Reviews 21 (2008): 639-665.
- Taylor A, Saichua P, Rhongbutsri P, et al. A preliminary epidemiological study of pinworm infection in Thaklong Municipal Early Childhood Development Center and Rangsit Babies' Home, Pathum Thani, Thailand. BMC Research Notes 11 (2018): 603.
- Ojha SC, Jaide C, Jinawath N, et al. Geohelminths: public health significance. Journal of Infection in Developing Countries 8 (2014): 5-16.
- World Health Organization. Prevention and control of intestinal parasitic infections. Report of a WHO Expert Committee. Geneva: Technical Report WHO. (Technical Report Series No. 749) (1987).
- Carrera E, Nesheim MC, Crompton DW. Lactose maldigestion in Ascaris-infected preschool children. Am J Clin Nutr 39 (1984): 255-264.


 Impact Factor: * 3.8
Impact Factor: * 3.8 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 