Our Challenges in an Extraordinary Case of AL Amyloidosis
Article Information
Gomercic Palcic Marija1*, Vrbanic Luka1, Mandic Peric Marija1, Ulamec Monika2, Šokcevic Marijka2, Galešic Ljubanovic Danica3
1University hospital center Sestre Milosrdnice, Department for internal diseases, Division for clinical immunology, pulmonology and rheumatology, Zagreb, Croatia
2University hospital center Sestre Milosrdnice, Ljudevit Jurak Department of Pathology, Zagreb, Croatia
3University hospital Dubrava, Department of pathology, Zagreb, Croatia
*Corresponding Author: Dr. Marija Gomercic Palcic, University Hospital Center Sestre Milosrdnice, Departmet for Internal Diseases, Vinogradska cesta 29, 10000 Zagreb, Croatia
Received: 15 July 2019; Accepted: 29 July 2019; Published: 30 September 2019
Citation: Gomerčić Palčić Marija, Vrbanić Luka, Mandić Perić Marija, Ulamec Monika, Šokčević Marijka, Galešić Ljubanović Danica. Our Challenges in an Extraordinary Case of AL Amyloidosis. Archives of Clinical and Medical Case Reports 3 (2019): 382-389.
View / Download Pdf Share at FacebookAbstract
Background: Light-chain (AL) amyloidosis is a condition in which extracellular tissue deposition of protein derived from light chain immunoglobulin fragments can affect any organ in the body, and therefore has a variety of clinical manifestations, mimics variety of diseases and can easily be misdiagnosed.
Case Presentation: This is a case of 79-year-old man with 9-year history of IgM monoclonal gammopathy of undetermined significance. Patient presented with right-sided effusion with pulmonary infiltrate. After antibiotic treatment the infiltrate resolved, but effusion progressed and appeared also on the left side. Extensive workup revealed generalized lymphadenopathy and no signs of any organ failure. Lymph node biopsy was analyzed applying Congo red, immunohistochemistry, immunofluorescence and electron microscope, and diagnosis of AL amyloidosis with monoclonal kappa chains was established.
Conclusions: Since prognosis and natural history of the disease is dependent on time of discovery, it is important to raise suspicion for AL amyloidosis and use all available diagnostic tools and collaborate with appropriate institutions.
Keywords
Chylous pleural effusion; Amyloidosis; Electron microscopy; Monoclonal gammopathy; Generalized lymphadenopathy
Article Details
Abbreviations:
AL amyloidosis - light-chain amyloidosis; MGUS - monoclonal gammopathy of undetermined significance; TSH - thyroid stimulating hormone; NT-proBNP - N-terminal serum brain natriuretic peptide; CT – computed tomography; TBNA - transbronchial needle aspiration; HCT - hematopoietic cell transplantation
1. Introduction
AL amyloidosis is a condition in which extracellular tissue deposition of protein derived from light chain immunoglobulin fragments can affect any organ in the body, and therefore has a variety of clinical manifestations, mimics variety of diseases and can easily be misdiagnosed. Monoclonal gammopathy rarely progresses into systemic amyloidosis (incidence around 1% yearly) [1] and IgM monoclonal gammopathy of undetermined significance (MGUS) more often than other types [2]. Diagnostic procedure requires a biopsy of an affected organ. On light microscopy it shows typical Congo red-positivity with birefringence under polarized light. Additionally, electric microscopy can be used to confirm the diagnosis. Presence of a monoclonal protein alone is not sufficient to diagnose AL amyloidosis in a patient with documented amyloidosis, unless immunofluorescence has demonstrated monoclonal light chains in the amyloid deposits. There are several possible therapeutic regimens, and the choice of treatment should be individualized. Persistent pleural effusions develop in 1 to 2 percent of patients with systemic amyloidosis and appear to be caused by pleural infiltration with amyloid deposits [3-5] although, mediastinal lymph nodes, if infiltrated, can additionally obstruct lymph drainage. Here we will present a case of a 79-year-old man with a 9-year history of IgM MGUS and insignificant systemic lymphadenopathy, who presented with right sided pleuropneumonia and later on progressive bilateral exudative pleural effusions, one of which was chylous. Light microscopy showed a negative Congo red birefringence, so the biopsy specimen of lymphoid tissue was reexamined by electronic microscope, immunohistochemistry, immunofluorescence, and diagnosis of AL amyloidosis with monoclonal kappa chains was established. Patient was treated with bortezomib and dexamethasone, with a good response on short follow up. Since there was a satisfying regression of pleural effusions, pleurodesis wasn’t performed. We present this case report based on our experience in AL amyloidosis, including challenges in diagnostics and treatment concerns regarding this rare disease.
2. Case Presentation
A 79-year old patient with 9-year history of IgM MGUS and insignificant systemic lymphadenopathy was admitted to our hospital in April 2018 because of fever, productive cough and dyspnea. So far there was no need for IgM MGUS treatment because patient had no B symptoms, sings of affected organs or hyperviscosity syndrome. At admission chest X-ray revealed moderate right side effusion and pneumonic infiltrate. Arterial blood gas analysis revealed type I respiratory insufficiency with partial pressure of oxygen 7.42 kPa, partial pressure of carbon dioxide 5.9 kPa, and oxygen saturation (SaO2) 89.6%. Pneumonic infiltrate resolved after a course of antibiotics, but right side pleural effusion progressed and left side effusion appeared. During the treatment non-specific systemic symptoms such as fatigue and unintentional weight loss were noted. After two right-side thoracentesis without an adequate clinical improvement, pleural drainage system (Pleuracan) was inserted on the right and 7 L of thick and pale-yellow fluid was evacuated. The analyzed fluid contained 30 g/L of protein, glucose was 6.5 mmol/L, lactate dehydrogenase 105, triglycerides 7.5 mmol/L.
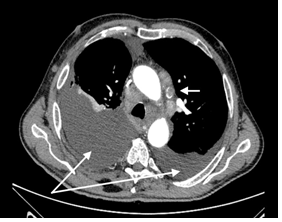
Cytological examination of the pleural effusion revealed predominantly lymphocytic type of effusion without malignant cells. Cultures of the effusion showed no bacterial, mycobacterial nor fungal except a normocytic anemia. Bone marrow aspirate showed only signs of anemia of chronic disease without elevated plasma-cell count. Electrophoresis of the serum proteins revealed an M protein (14.6%), and serum immunofixation revealed a monoclonal augmentation of immunoglobulin M (18.53 g/L) and kappa chains (29.2 mg/L). Immunofixation of urine proteins demonstrated kappa chains. 24-hour urinary protein, creatinine clearance, troponin T, and thyroid stimulating hormone (TSH) were all in referent ranges. Levels of N-terminal serum brain natriuretic peptide (NT-proBNP) were slightly elevated (502 pg/mL) in the absence of known primary heart disease. Electrocardiogram and echocardiogram were normal; without any signs of restrictive cardiomyopathy. No tumor markers were elevated. A computed tomography (CT) scan of thorax, abdomen and pelvis showed a systemic lymphadenopathy (partly calcified), much larger in size compared to previous examinations (Figure 1). During work-up, axillar, inguinal and cervical lymphadenopathy was also established. On bronchoscopy no intraluminal infiltrate was seen, only extramural compression due to bilateral effusions. Again there were no microorganisms isolated in specimens taken during bronchoscopy. We performed transbronchial needle aspiration (TBNA) of mediastinal lymph nodes but no malignant cells were found. Although the right-side effusion was completely resolved after undertaken therapeutical measures, patient stayed depended on oxygen therapy (2 L/min). During hospitalization pleural drainage system had obstructed due to thick effusion and was extracted. After only few days patient complained with dyspnea during minimal exertion, there was a progression of pleural effusions so we performed thoracentesis on both sides. Right-side was still chylothorax (Figure 2a) and left- side effusion was yellow, turbid and exudate like (Figure 2b). Since right-side effusion progressed rapidly chest drain was reinserted (Rocket, 12F).
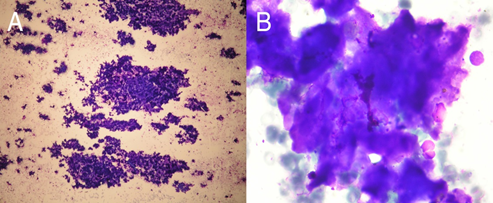
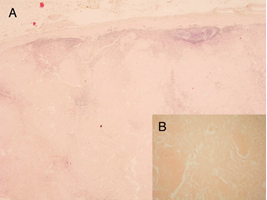
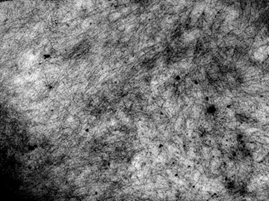
Cytological analysis of peripheral lymph nodes, in different regions, revealed extracellular amorphous hyaline material without malignant cells (Figure 3), so we decided to surgically remove the largest accessible lymph node that was in right inguinal region. On light microscopy lymph node was completely permeated with amorphous hyaline material, but after binding Congo red green birefringence under polarized light wasn’t found, even after repeated staining (Figure 4). Electron microscopy found straight and unbranching fibrils and amyloidosis was confirmed. Immunohistochemical and immunofluorescent analysis was positive for monoclonal kappa chains and negative for lambda chains, so AL amyloidosis, was identified (Figure 5). With additional testing autonomic neuropathy and other neurological complications were excluded. There was no sign of gastroparesis, and findings of upper endoscopy made few months before were normal. After diagnosis was established two cycles of bortezomid with dexamethasone were applied. Pleural drain was removed and ever since there was no need for repeating thoracentesis. Patient was still depended on oxygen therapy but pleural effusions were in regression by each applied cycle.
3. Discussion
In “primary” amyloidosis precursor proteins are monoclonal immunoglobulin light chains produced by an underlying clonal plasma cell proliferative disorder or, rarely, lymphoplasmacytic neoplasm. However, new nomenclature is AL amyloidosis, in which AL implies that the amyloid protein is immunoglobulin light chain-derived. The fibrils in AL light chains in approximately 75 percent of cases, and kappa representing the remaining cases [6]. Patient in our case had monoclonal kappa chain AL amyloidosis and an unusual clinical presentations, and we encountered obstacles in the diagnostic process and in the choice of appropriate therapy. Studies showed that MGUS precedes AL amyloidosis, with monoclonal gammopathy being present in 80 percent of cases at least four years prior and in 42 percent more than 11 years prior to the diagnosis of amyloidosis [7].
AL amyloidosis with IgM MGUS history is a disease of older adults with male predominance (65 to 70 percent of patients) [8] with median age at diagnosis of 64 years. Demographic characteristics of our patient correspond to these observations. The patient was a 79-years-old man with a 13 year history of IgM MGUS. He had systemic lymphadenopathy for several years and the first presentation of the disease were pleural effusions and pneumonic infiltrate. Thoracentesis excluded a possible parapneumonic effusion since it was a chylous exudate with high levels of triglycerides. Chylous effusions occur in patients with extensive amyloid burden due to direct infiltration of mediastinal nodes by amyloid rather than by mechanical compression by enlarged lymphonodes [9]. Partial leak from ductus thoracicus is also a possible mechanism of chylous effusion genesis. Pleural effusion on the right was exudative in nature, probably due to primary disruption of the pleural surface and its function by amyloid (surrounding pleural inflammation and the disturbed lymphatic drainage caused by the amyloid deposition) [10]. The most common cause of the respiratory amyloid disease is secondary to systemic AL amyloidosis counting for up to 80% of pulmonary amyloidosis [11]. Besides, 88% of patients with systemic amyloid have pulmonary disease [12].
In our patient CT scan of thorax, abdomen and pelvis, as well as ultrasound of peripheral lymph nodes confirmed minor systemic lymphadenopathy nine years ago. Now a notable progression in size and partial calcification was found, compared to previous scans. Although patients presenting with amyloid lymphadenopathy usually have AL amyloidosis, they have to go thorough evaluation for other organ involvement at diagnosis [13]. Respiratory amyloid disease can present with nodular or diffuse submucosal involvement, interstitial lung disease, single or multiple parencyhmal nodules, mediastinal lymphadenopathy with egg shell or popcorn calcification, and unilateral or bilateral effusions. In our case there were pleural effusions and intrathoracic lymphadenopathy with no pathological signs on lung parenchyma. Bronchoscopy revealed normal bronchial mucosa without any visible infiltrate. Mediastinal or hilar lymphadenopathy is less common than extrathoracic lymphadenopathy in patients with systemic AL disease. It can cause luminal narrowing of the airways and thus may lead to postobstructive atelectasis and/or pneumonia, which was the case in our patient. Specimen obtained by TBNA of mediastinal lymph nodes, showed no malignant cells nor lymphocytes. Although it was suggested that pulmonary involvement is accompanied by myocardial involvement, in this patient electrocardiogram, as well as echocardiography, were normal and therefore no other invasive tests were performed. Elevations of NT-proBNP in patients with AL amyloidosis are seen before the onset of clinical heart failure and are a marker of cardiac involvement [14, 15].
Since all lymph nodes (peripheral, intrathoracic and intraabdominal) had similar characteristics on imagining methods, and cytological analysis of multiple peripheral lymph nodes revealed amorphous hyaline material, we decided to surgically remove the largest lymph node (in the right inguinal region). For establishing diagnosis of amyloidosis, it is important to identify the amyloid fibril protein in tissue deposits, which exhibits typical histochemical staining on Congo red and green birefringence under polarization microscope [16]. However, in our case it showed typical Congo red staining, but without green microscopy for amyloid are 79 and 80%, respectively [17]. The sensitivity for kappa (74%) is lower than for lambda (84%). As there was a negative Congo red birefringence electronic microscopy, immunohistochemistry and immunofluorescence were indicated. The diagnosis of AL amyloidosis with monoclonal kappa chains was finally established. Examination of the ultrastructure of amyloid deposits on electronic microscopy generally demonstrates straight and unbranching fibrils [18]. Immunofluorescent microscopy can be used to identify the type of amyloid fibril unit.
There are several therapeutic options to treat pleural effusions in AL amyloidosis. One of the options is pleurodesis, which we didn’t perform in this case, since after initiating the treatment there was no more need for thoracentesis. To determinate the best treatment for the patients with AL amyloidosis, initial evaluation must confirm the diagnosis, establish the extent and sites of disease, and evaluate for comorbidities that are likely to have an impact on treatment options. Virtually all patients with systemic AL amyloidosis require treatment at the time of diagnosis. All patients with newly diagnosed AL amyloidosis need to be assessed to determine eligibility for autologous hematopoietic cell transplantation (HCT). Our patient was not a candidate for HTC regarding his age, effusions and dependence on oxygen therapy. There is controversy regarding the preferred management of patients who are not eligible for HCT, and participation in clinical trials should be encouraged. Outside of a clinical trials, it is suggest to apply a bortezomib-based triple regime such as bortezomib, cyclophosphamide and dexamethasone or bortezomib, melphalan and dexamethasone. We decided to start with bortezomib and dexamethasone (two cycles so far) and additionally introduce cyclophosphamide when we see how the patient will tolerate this treatment. So far there was no need for thoracentesis because pleural effusions regressed and patient is clinically improving. He is still depended of oxygen therapy.
4. Conclusion
In conclusion, if there is a high suspicion of AL amyloidosis with a history of IgM MGUS, a whole spectrum of diagnostic procedures should be done even though Congo red staining and polarization microscopy are negative. Since prognosis in patients with AL amyloidosis depends on the time of diagnosis, it is important to shorten the overall diagnostic time. In our case collaboration with institution owning electron microscopy and methods was crucial for our patient.
Declarations
This case report was approved by “Sestre milosrdnice” Ethic committee.
Competing Interests
Not applicable
Declaration of Interest Statement
The authors report no conflict of interest.
References
- Kyle RA, Therneau TM, Rajkumar SV, et al. A long- term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med 346 (2002): 564-569.
- Weiss BM, Hebreo J, Cordaro DV, et al. Increased serum free light chains precede the presentation of immunoglobulin light chain amyloidosis. J 12 Clin Oncol 32 (2014): 2699-2704.
- Schade L, Carmes ER, de Barros JA. Mediastinal lymph node amyloidosis in a patient with sarcoidosis. J Bras Pneumol 33 (2007): 222-225.
- Kavuru MS, Adamo JP, Ahmad M, et al. Amyloidosis and pleural disease. Chest 98 (1990): 20-23.
- George S, Ravindran M, Anandan PT, et al. Primary systemic amyloidosis: A rare cause for pleural effusion. Respir Med Case Rep 13 (2014): 39-42.
- Perfetti V, Casarini S, Palladini G, et al. Analysis of V(lambda)-J(lambda) expression in plasma cells from primary (AL) amyloidosis and normal bone marrow identifies 3r (lambdaIII) as a new amyloid-associated germline gene segment. Blood 100 (2002): 948-953.
- Weiss BM, Hebreo J, Cordaro DV, et al. Increased serum free light chains precede the presentation of immunoglobulin light chain amyloidosis. J Clin Oncol 32 (2014): 2699-2704.
- Kyle RA, Linos A, Beard CM, et al. Incidence and natural history of primary systemic amyloidosis in Olmsted County, Minnesota, 1950 through 1989. Blood 79 (1992): 1817-1822.
- Berk JL, Keane J, Seldin DC, et al. Persistent pleural effusions in primary systemic amyloidosis: etiology and prognosis. Chest 124 (2003): 969-977.
- Berk JL. Pleural effusions in systemic amyloidosis. Curr Opin Pulm Med 11 (2005): 324-328.
- Pitz MW, Gibson IW, Johnston JB. Isolated pulmonary amyloidosis: case report and review of the literature. Am J Hematol 81 (2006): 212-213.
- Smith RR, Hutchins GM, Moore GW, et al. Type and distribution of pulmonary parenchymal and vascular amyloid: correlation with cardiac amyloid. Am J Med 66 (1979): 96-104.
- Fu J, Seldin DC, Berk JL, et al. Lymphadenopathy as a manifestation of amyloidosis: a case series. Amyloid 21 (2014): 256-260.
- Merlini G, Wechalekar AD, Palladini G. Systemic light chain amyloidosis: an update for treating physicians. Blood 121 (2013): 5124-5130.
- Merlini G, Palladini G. Differential diagnosis of monoclonal gammopathy of undetermined significance. Hematology Am Soc Hematol Educ Program (2012): 595-603.
- Glenner GG. Amyloid deposits and amyloidosis. The beta-fibrilloses (first of two parts). N Engl J Med 302 (1980): 1283-1292.
- Fernández de Larrea C, Verga L, Morbini P, et al. A practical approach to the diagnosis of systemic amyloidoses. Blood 125 (2015): 2239-2244.
- Jahn TR, Makin OS, Morris KL, et al. The common architecture of cross-beta amyloid. J Mol Biol 395 (2010): 717-727.



 Impact Factor: * 3.1
Impact Factor: * 3.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
