Prognostic Analysis of Patients with Resectable T4 Colorectal Cancer
Article Information
Wei Chen1,2,3#, Xiao-ping Tan4#, Jun-Wen Ye1,2,3#, Yan Zhang2,3,5, Jing-Lin Liang1,2,3*, Mei-Jin Huang1,2,3*
#Authors contributed equally
1Department of Colorectal Surgery, the Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510655, China
2Guangdong Provincial Key Laboratory of Colorectal and Pelvic Floor Disease, The Sixth Affiliated Hospital of Sun Yat-sen University, 510655, Guangzhou, China
3Guangdong Research Institute of Gastroenterology, The Sixth Affiliated Hospital of Sun Yat-sen University, 510655, Guangzhou, China
4Department of Emergency, The Second Affiliated Hospital of Guangzhou medical university, Guangzhou, 510655, China
5Department of Medicine Oncology, the Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510655, China
*Corresponding Authors: Dr. Jing-Lin Liang, Department of Colorectal Surgery, the Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, 510655, P. R. China
Dr. Mei-jin Huang, Department of Colorectal Surgery, the Sixth Affiliated Hospital, Sun Yat-sen University, Guangzhou, Guangdong, 510655, P. R. China
Received: 29 July 2020; Accepted: 20 August 2020; Published: 29 August 2020
Citation: Wei Chen, Xiao-ping Tan, Jun-Wen Ye, Yan Zhang, Jing-Lin Liang, Mei-Jin Huang. Prognostic Analysis of Patients with Resectable T4 Colorectal Cancer. Journal of Cancer Science and Clinical Therapeutics 4 (2020): 325-332.
View / Download Pdf Share at FacebookAbstract
Background: To observe the factors related to the survival and prognosis of patients with resectable stage T4 colorectal cancer.
Methods: A total of 148 patients with resectable stage T4 colorectal cancer who underwent surgery at the First Affiliated Hospital of Sun Yat-sen University between August, 1994 and December, 2005 were retrospectively analysed. Univariate and multivariate analyses of the associations between clinicopathological variables and survival were analysed using the Cox regression model.
Results: The follow-up period ended in December 2010 or at patient death; the 5-year and 10-year overall survival (OS) rates were 49.0% and 32.2%, respectively, and the median OS duration was 25 months. The 5-year and 10-year disease-free survival (DFS) rates were 44.2% and 30. 3%, respectively. In univariate analysis, postoperative pathology indicating lymph node metastasis was associated with patient prognosis in terms of OS (all P< 0.01), and postoperative adjuvant therapy failed to improve OS or DFS (P>0.05). Postoperative pathology indicating lymph node metastasis was also associated with DFS (all P< 0.01). In multivariate analysis, postoperative pathology indicating lymph node metastasis was an independent factor affecting OS and DFS in colorectal cancer patients.
Conclusion: The postoperative prognosis of T4 colorectal cancer patients is poor, and postoperative pathology indicating lymph node positivity was an independent factor for OS and DFS.
Keywords
Colorectal cancer; Surgery; Prognosis; Resectable
Colorectal cancer articles, Surgery articles, Prognosis articles, Resectable articles
Colorectal cancer articles Colorectal cancer Research articles Colorectal cancer review articles Colorectal cancer PubMed articles Colorectal cancer PubMed Central articles Colorectal cancer 2023 articles Colorectal cancer 2024 articles Colorectal cancer Scopus articles Colorectal cancer impact factor journals Colorectal cancer Scopus journals Colorectal cancer PubMed journals Colorectal cancer medical journals Colorectal cancer free journals Colorectal cancer best journals Colorectal cancer top journals Colorectal cancer free medical journals Colorectal cancer famous journals Colorectal cancer Google Scholar indexed journals Surgery articles Surgery Research articles Surgery review articles Surgery PubMed articles Surgery PubMed Central articles Surgery 2023 articles Surgery 2024 articles Surgery Scopus articles Surgery impact factor journals Surgery Scopus journals Surgery PubMed journals Surgery medical journals Surgery free journals Surgery best journals Surgery top journals Surgery free medical journals Surgery famous journals Surgery Google Scholar indexed journals Prognosis articles Prognosis Research articles Prognosis review articles Prognosis PubMed articles Prognosis PubMed Central articles Prognosis 2023 articles Prognosis 2024 articles Prognosis Scopus articles Prognosis impact factor journals Prognosis Scopus journals Prognosis PubMed journals Prognosis medical journals Prognosis free journals Prognosis best journals Prognosis top journals Prognosis free medical journals Prognosis famous journals Prognosis Google Scholar indexed journals Resectable articles Resectable Research articles Resectable review articles Resectable PubMed articles Resectable PubMed Central articles Resectable 2023 articles Resectable 2024 articles Resectable Scopus articles Resectable impact factor journals Resectable Scopus journals Resectable PubMed journals Resectable medical journals Resectable free journals Resectable best journals Resectable top journals Resectable free medical journals Resectable famous journals Resectable Google Scholar indexed journals Peritoneal carcinomatosis articles Peritoneal carcinomatosis Research articles Peritoneal carcinomatosis review articles Peritoneal carcinomatosis PubMed articles Peritoneal carcinomatosis PubMed Central articles Peritoneal carcinomatosis 2023 articles Peritoneal carcinomatosis 2024 articles Peritoneal carcinomatosis Scopus articles Peritoneal carcinomatosis impact factor journals Peritoneal carcinomatosis Scopus journals Peritoneal carcinomatosis PubMed journals Peritoneal carcinomatosis medical journals Peritoneal carcinomatosis free journals Peritoneal carcinomatosis best journals Peritoneal carcinomatosis top journals Peritoneal carcinomatosis free medical journals Peritoneal carcinomatosis famous journals Peritoneal carcinomatosis Google Scholar indexed journals Complete cytoreduction articles Complete cytoreduction Research articles Complete cytoreduction review articles Complete cytoreduction PubMed articles Complete cytoreduction PubMed Central articles Complete cytoreduction 2023 articles Complete cytoreduction 2024 articles Complete cytoreduction Scopus articles Complete cytoreduction impact factor journals Complete cytoreduction Scopus journals Complete cytoreduction PubMed journals Complete cytoreduction medical journals Complete cytoreduction free journals Complete cytoreduction best journals Complete cytoreduction top journals Complete cytoreduction free medical journals Complete cytoreduction famous journals Complete cytoreduction Google Scholar indexed journals Hyperthermic intraperitoneal chemotherapy articles Hyperthermic intraperitoneal chemotherapy Research articles Hyperthermic intraperitoneal chemotherapy review articles Hyperthermic intraperitoneal chemotherapy PubMed articles Hyperthermic intraperitoneal chemotherapy PubMed Central articles Hyperthermic intraperitoneal chemotherapy 2023 articles Hyperthermic intraperitoneal chemotherapy 2024 articles Hyperthermic intraperitoneal chemotherapy Scopus articles Hyperthermic intraperitoneal chemotherapy impact factor journals Hyperthermic intraperitoneal chemotherapy Scopus journals Hyperthermic intraperitoneal chemotherapy PubMed journals Hyperthermic intraperitoneal chemotherapy medical journals Hyperthermic intraperitoneal chemotherapy free journals Hyperthermic intraperitoneal chemotherapy best journals Hyperthermic intraperitoneal chemotherapy top journals Hyperthermic intraperitoneal chemotherapy free medical journals Hyperthermic intraperitoneal chemotherapy famous journals Hyperthermic intraperitoneal chemotherapy Google Scholar indexed journals lymph node positivity articles lymph node positivity Research articles lymph node positivity review articles lymph node positivity PubMed articles lymph node positivity PubMed Central articles lymph node positivity 2023 articles lymph node positivity 2024 articles lymph node positivity Scopus articles lymph node positivity impact factor journals lymph node positivity Scopus journals lymph node positivity PubMed journals lymph node positivity medical journals lymph node positivity free journals lymph node positivity best journals lymph node positivity top journals lymph node positivity free medical journals lymph node positivity famous journals lymph node positivity Google Scholar indexed journals postoperative pathology articles postoperative pathology Research articles postoperative pathology review articles postoperative pathology PubMed articles postoperative pathology PubMed Central articles postoperative pathology 2023 articles postoperative pathology 2024 articles postoperative pathology Scopus articles postoperative pathology impact factor journals postoperative pathology Scopus journals postoperative pathology PubMed journals postoperative pathology medical journals postoperative pathology free journals postoperative pathology best journals postoperative pathology top journals postoperative pathology free medical journals postoperative pathology famous journals postoperative pathology Google Scholar indexed journals lymph node metastasis articles lymph node metastasis Research articles lymph node metastasis review articles lymph node metastasis PubMed articles lymph node metastasis PubMed Central articles lymph node metastasis 2023 articles lymph node metastasis 2024 articles lymph node metastasis Scopus articles lymph node metastasis impact factor journals lymph node metastasis Scopus journals lymph node metastasis PubMed journals lymph node metastasis medical journals lymph node metastasis free journals lymph node metastasis best journals lymph node metastasis top journals lymph node metastasis free medical journals lymph node metastasis famous journals lymph node metastasis Google Scholar indexed journals
Article Details
Abbreviations:
CRC- Colorectal cancer; PC- Peritoneal carcinomatosis; CCR- Complete cytoreduction; OS- Overall survival; HIPEC- Hyperthermic intraperitoneal chemotherapy
1. Introduction
Colorectal cancer is one of the most common malignant diseases in the world and has a high mortality. China is a high incidence area for colorectal cancer, with more than half of the world's morbidity and mortality [1]. Surgical resection is one of the main treatment methods for colorectal cancer patients. Early surgical treatment for colorectal cancer is effective, and the 5-year survival rate is as high as 60.5% [2]. Most patients have progressed to locally advanced disease before getting diagnosed and have lost the opportunity to undergo direct surgery. Even after surgery, it is difficult to achieve complete resection. Advances in surgical techniques and perioperative management have improved patient survival, but surgery alone does not improve the outcomes of patients with locally advanced colorectal cancer. To improve the resection rate, the guidelines [3] recommend that patients with locally advanced colorectal cancer can be treated with neoadjuvant chemoradiotherapy and/or chemotherapy combined with surgery, and radical chemoradiotherapy is also feasible. However, due to the limited accuracy of preoperative staging and patient willingness, some patients preferred to undergo surgical resection, and some doctors considered that neoadjuvant therapy may increase the risk and complications of surgery, so some doctors first chose surgery and then determined whether postoperative adjuvant therapy was feasible according to the postoperative pathology results. Patients with stage T4 colorectal cancer have poor prognosis and are prone to developing recurrence and distant metastasis. This study investigated the prognostic factors of patients with postoperative T4 colorectal cancer.
2. Materials and Methods
General information: A retrospective analysis of 148 patients with T4 stage colorectal cancer who underwent surgery at the First Affiliated Hospital of Sun Yat-sen University between August 1994 and December 2005 was performed. The inclusion criteria were as follows: postoperative pathological staging revealed neoplastic invasion out of the membrane (T4), and the survival time was longer than 2 months. The exclusion criteria were as follows: intolerance to surgery because of poor cardiopulmonary function; distant metastasis and so on. All patients and their families gave informed consent to participate in the study and signed informed consent forms. The study was approved by the Ethics Committee of Sun Yat-sen University.
Follow-up and review: The patients were followed up every 3 months after surgery for the first year, 6 months for the next 2 years and yearly thereafter. The first review was performed at the hospital one month after the operation. Chest and abdominal CT, routine blood, liver and kidney function tests, tumour marker tests, colonoscopy and other examinations were routinely performed; if necessary, a whole body bone scan and PET-CT were performed to determine whether there was systemic metastasis. Local recurrence was defined as an anastomotic stoma or regional lymph node recurrence. Distant recurrence was defined as positive, distant lymph nodes or distant organ metastasis.
Statistical method: The overall survival (OS) time was from the date of surgery to the date of death or end of follow-up. The disease-free survival (DFS) time was from the date of surgery to the date of tumour recurrence or metastasis. Using SPSS 23.0 software, the Kaplan-Meier method was used to calculate OS and DFS, and the log-rank method was used to test for differences in OS. The Cox model was used for single factor and multifactor analysis, and the χ2 test was used to analyse the effect of different treatment methods on the survival rate. P<0.05 was considered statistically significant.
3. Results
3. 1 Postoperative conditions
The whole group was followed up until December 2010. Among the 148 patients, 84 were male, and 64 were female. The patients were 17 to 86 years old, and the median age was 64 years. During the operation, the average number of lymph nodes removed was 13.5 (2~35). Postoperative pathology revealed 85 patients with lymph node metastasis and 63 patients with negative lymph nodes. Postoperative pathology showed that the tumour invaded the adventitia. Postoperative anastomotic leakage was found in 2 cases.
3. 2 Postoperative survival rate
The 5- and 10-year survival rates were 49.0% and 32.2%, respectively, in the whole group, and the median survival duration was 58.8 months (Figure 1). Cox univariate analysis showed that postoperative pathology indicating lymph node metastasis was associated with OS (P<0.05). Sex, age, obstruction, tumour site, liver metastasis, histological grade, family history, and postoperative adjuvant therapy were not associated with OS (P>0.05). Cox multivariate analysis showed that postoperative lymph node metastasis was an independent factor influencing patient survival. Compared with patients with negative lymph nodes postoperatively, those with positive lymph nodes had a 1.213 higher risk of death (95% Cl: 0.845-1. 564, P=0.045). See Table 1.
3. 3 Postoperative disease-free survival rate
The 5- and 10-year DFS rates after surgery of the whole group were 44.2% and 30.3%, respectively, and the median DFS duration was 45.2 months. Sex, age, obstruction, tumour site, liver metastasis, histological grade, family history, and postoperative adjuvant therapy were not associated with DFS (P>0.05). Cox multivariate analysis showed that postoperative pathological lymph node positivity was an independent factor affecting DFS in patients. Compared with patients with negative lymph nodes postoperatively, those with positive lymph nodes had a 1.425 times higher risk of tumour recurrence or metastasis (95% CI: 0.974-1.836, both P<0.01). See Table 2.
|
Variable |
Univariate analysis |
Multivariate analysis |
P |
||
|
10 year- OS |
P value |
HR |
CL (95%) |
||
|
Gender |
0.557 |
||||
|
Female |
87.6 |
||||
|
Male |
81.6 |
||||
|
Age (y) |
0.103 |
||||
|
≥65 |
82.4 |
||||
|
<65 |
83.8 |
||||
|
Obstruction |
0.27 |
||||
|
No |
81.5 |
||||
|
Yes |
64.3 |
||||
|
Tumor site |
0.117 |
||||
|
Rectal |
72.7 |
||||
|
Colon |
86.2 |
||||
|
N stage |
0 |
0.045 |
|||
|
N0 |
74.5 |
1 |
|||
|
N1-N2 |
60.7 |
1.213 |
0.845-1.564 |
||
|
Liver metastasis |
0.285 |
||||
|
No |
82.7 |
||||
|
Yes |
66.7 |
||||
|
Histological grade |
0.571 |
||||
|
Well |
90.9 |
||||
|
Moderately |
77.1 |
||||
|
Poorly |
65.2 |
||||
|
Family history |
0.415 |
||||
|
No |
84 |
||||
|
Yes |
50.2 |
0.531 |
|||
|
Postoperative adjuvant therapy |
|||||
|
No |
|||||
|
Yes |
60.1 |
||||
|
74.5 |
|||||
Table 1: Cox proportional hazards model univariate and multivariate analyses of individual parameters for correlations with overall survival (OS).
|
Variable |
Univariate analysis |
Multivariate analysis |
P |
||
|
10 year- DFS |
P value |
HR |
CL (95%) |
||
|
Gender |
0.436 |
||||
|
Female |
83.2 |
||||
|
Male |
75.3 |
||||
|
Age (y) |
0.201 |
||||
|
≥65 |
81.2 |
||||
|
<65 |
79.5 |
||||
|
Obstruction |
0.316 |
||||
|
No |
80.2 |
||||
|
Yes |
60.3 |
||||
|
Tumor site |
0.281 |
||||
|
Rectal |
71.2 |
||||
|
Colon |
83.1 |
||||
|
N stage |
0.014 |
0.032 |
|||
|
N0 |
73.1 |
1 |
|||
|
N1-N2 |
57.2 |
1.425 |
0.974-1.836 |
||
|
Liver metastasis |
0.132 |
||||
|
No |
80.2 |
||||
|
Yes |
60.1 |
||||
|
Histological grade |
0.231 |
||||
|
Well |
87.2 |
||||
|
Moderately |
73.1 |
||||
|
Poorly |
40.1 |
||||
|
Family history |
0.531 |
||||
|
No |
82.1 |
||||
|
Yes |
52.1 |
0.764 |
|||
|
Postoperative adjuvant therapy |
|||||
|
No |
|||||
|
Yes |
63.1 |
||||
|
75.2 |
|||||
Table 2: Cox proportional hazards model univariate and multivariate analyses of individual parameters for correlations with disease-free survival (DFS).
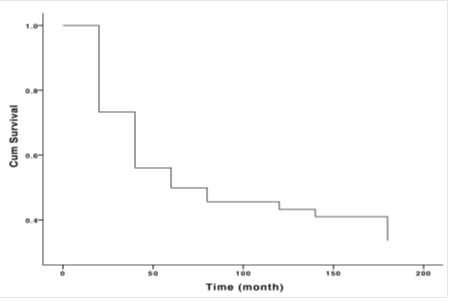
Figure 1: Survival curve for T4 CRC patients undergone surgery.
4. Discussion
In the 1970s, the surgical treatment of patients with T4 stage colorectal cancer mainly involved nutritional ostomy and palliative resection. With the continuous advancement of surgery and anaesthesia technology, patient management in the perioperative period has been gradually strengthened. The surgical resection rate of patients with T4 colorectal cancer is gradually increasing, but the survival rate of these patients is still not high. According to reports in the literature, the 5-year survival rate of patients with T4 stage colorectal cancer is 0% [4, 5]. Although patients with stage T4 colorectal cancer have a poor prognosis after surgery, surgical treatment is still feasible in patients with resectable colorectal cancer. The previous study showed that among 240 patients with T4 colorectal cancer, 77 patients received postoperative adjuvant chemotherapy [6]. The results showed that patients with T4 tumours who received postoperative chemotherapy had significantly better survival with respect to overall survival (p< 0.001) and recurrence-free survival (p=0.008). This suggests that surgical treatment is still important in patients with resectable T4 colorectal cancer.
This study selected patients who underwent surgery between August 1994 and December 2005. All patients had a 10-year survival rate of 32.2%, a median survival duration of 58.8 months, and poor overall survival. The main reasons for the poor prognosis in patients with colorectal cancer are roughly divided into two aspects. First, the accuracy of preoperative staging is not high, and patients do not receive standardized treatment. Second, the tumour is invasive, and local recurrence or distant metastasis can easily occur after surgery.
The 5-year and 10-year disease-free survival rates were 44.2% and 30.3%, respectively, and the median DFS duration was 45.2 months. More than one-third of the patients had tumour progression within 1 year after surgery, so R0 resection of the tumour was performed. Controlling postoperative recurrence or metastasis is the key to improving survival. Regarding T4 stage colorectal cancer, the probability of achieving complete R0 resection in a single operation is not high. Even if R0 resection is achieved, the 5-year survival rate is not satisfactory. Studies have shown that neoadjuvant chemoradiotherapy/chemotherapy can improve the resection rate of locally advanced resectable tumours and prolong patient survival. In terms of reducing the postoperative recurrence rate and improving postoperative survival, a previous study found that postoperative adjuvant therapy can improve OS in patients with advanced colorectal cancer and prolong DFS [7-13]. However, in our study, postoperative adjuvant chemotherapy did not lead to improved OS and DFS (both P>0.05) compared surgery alone. This may because the colorectal cancer and T stage were too advanced, and there were micro-metastases in the lymph nodes. Conventional pathological diagnostic techniques have a limited role in the diagnosis of micro-metastasis, resulting in inaccurate postoperative staging. There were only a few cases in this study, which may cause statistical errors.
This study found that after T4 stage colorectal cancer surgery, lymph node positive was an independent factor affecting OS and DFS (P<0.01). Compared with postoperative lymph node-negative patients, lymph node-positive patients had worse OS and DFS. During the follow-up period of this study, more than half of the patients had local recurrence or distant metastasis. Liver metastasis was also an independent factor affecting OS (both P<0.001). This study also has certain limitations. First: the number of patients selected was limited, and 148 patients were included in the study. Second, the study was retrospectively analyzed and still required multicenter prospective clinical trials and longer follow-up to confirm.
Acknowledgements
Funding for this trial was generously provided by Sun Yat-sen University.
Funding
This study was supported by the Guangdong Natural Science Foundation (2014A030310021).
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' Contributions
WC, JLL, JWY, XPT, YZ and MJH contributed to the study design, data collection, data analysis and interpretation, drafting of the manuscript, approval of the final manuscript, and supervision. All authors approved the final version of the manuscript.
Ethics Approval and Consent to Participate
The study was conducted in compliance with all national and international ethical standards for research with humans. All study procedures were approved by the Sun Yat-sen University and patients gave written informed consent before being enrolled.
Consent for Publication
Not applicable
Competing Interests
The authors declare that they have no competing interests.
References
- Arnold M, Mónica S Sierra, Laversanne M, et al. Global patterns and trends in colorectal cancer incidence and mortality. Gut 66 (2016): 683-691.
- Yang ZF, Wu DQ, Wang JJ, et al. Short- and long-term outcomes following laparoscopic vs open surgery for pathological T4 colorectal cancer: 10 years of experience in a single center. World Journal of Gastroenterology 24 (2018): 76-86.
- Benson RA, Venook AP, Alhawary MM, et al. NCCN Guidelines Insights: Colon Cancer, Version 2. 2018. Journal of the National Comprehensive Cancer Network Jnccn 16 (2018): 359-369.
- Laohavinij S, Maneechavakajorn J, Techatanol P. Prognostic factors for survival in colorectal cancer patients. Journal of the Medical Association of Thailand = Chotmaihet thangphaet 93 (2010): 1156-1166.
- Jeong WK, Shin J W, Baek SK. Oncologic outcomes of early adjuvant chemotherapy initiation in patients with stage III colon cancer 89 (2015): 124-130.
- Andreas T, Michael G, Janine H, et al. Benefit of adjuvant chemotherapy in patients with T4 UICC II colon cancer. Bmc Cancer 15 (2015): 1-9.
- Dehal A, Graffbaker AN, Vuong B, et al. Neoadjuvant Chemotherapy Improves Survival in Patients with Clinical T4b Colon Cancer. Journal of Gastrointestinal Surgery Official Journal of the Society for Surgery of the Alimentary Tract 152 (2017): 1-8.
- Capussotti L, Vigano’ L, Ferrero A, et al. Timing of Resection of Liver Metastases Synchronous to Colorectal Tumor: Proposal of Prognosis-Based Decisional Model. Annals of Surgical Oncology 14 (2007): 1143-1150.
- Veerasarn V, Phromratanapongse P, Lorvidhaya V, et al. Preoperative capecitabine with pelvic radiotherapy for locally advanced rectal cancer (phase I trial). Journal of the Medical Association of Thailand = Chotmaihet thangphaet 89 (2006): 1874-1884.
- Michael M, Zalcberg JR. Adjuvant Therapy for Colorectal Cancer. Cancer Forum 38 (2014): 44-52.
- Niloofar A, Mosalaei A, Shapour O, et al. Role of external irradiation in high-risk resected colon cancer. Indian Journal of Cancer 42 (2005): 133-137.
- Schrag, Deborah. Evolving Role of Neoadjuvant Therapy in Rectal Cancer. Curr Treat Options Oncol 14 (2013): 350-364.
- Denost Q, Kontovounisios C, Rasheed S, et al. Individualizing surgical treatment based on tumour response following neoadjuvant therapy in T4 primary rectal cancer. European Journal of Surgical Oncology the Journal of the European Society of Surgical Oncology & the British Association of Surgical Oncology 43 (2017): 92-99.


 Impact Factor: * 4.1
Impact Factor: * 4.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 