Role of Pathology in the Decision Making of SFA Chronic Total Occlusion: A Narrative Review Article
Article Information
Ahmed MT Ghanem1*, Amira Roshdy Eltabbakh2
1Nasser Institute for research and Treatment, Cairo, Egypt
2Tanta University, Radiology department and medical imaging, Tanta, Egypt
*Corresponding Author: Ahmed Mohamed Taher Ghanem, Department of Vascular Surgery, Nasser Institute for Resarch and Treatment, 1351 Kournish El-Nile, Aghakhan, Cairo 11614, Egypt
Received: 10 June 2019; Accepted: 24 June 2019; Published: 05 July 2019
Citation: Ahmed MT Ghanem, Amira Roshdy Eltabbakh. Role of Pathology in the Decision Making of SFA Chronic Total Occlusion: A Narrative Review Article. Journal of Surgery and Research 2 (2019): 34-46.
View / Download Pdf Share at FacebookAbstract
Long segment femoral chronic total occlusion (CTO) intervention is considered to be among the most challenging procedures in patients with peripheral artery disease. The anatomical TransAtlantic InterSociety Consensus (TASC) classification in SFA total occlusion may be not enough for the selection between endovascular or surgical intervention. This article reveals the importance of clear understanding and visualization of the detailed pathological nature of the lesion and how it will create a more accurate and safe decision.
Keywords
CTO (chronic total occlusion), SFA (superficial femoral artery), Pathological nature
chronic total occlusion articles chronic total occlusion Research articles chronic total occlusion review articles chronic total occlusion PubMed articles chronic total occlusion PubMed Central articles chronic total occlusion 2023 articles chronic total occlusion 2024 articles chronic total occlusion Scopus articles chronic total occlusion impact factor journals chronic total occlusion Scopus journals chronic total occlusion PubMed journals chronic total occlusion medical journals chronic total occlusion free journals chronic total occlusion best journals chronic total occlusion top journals chronic total occlusion free medical journals chronic total occlusion famous journals chronic total occlusion Google Scholar indexed journals superficial femoral artery articles superficial femoral artery Research articles superficial femoral artery review articles superficial femoral artery PubMed articles superficial femoral artery PubMed Central articles superficial femoral artery 2023 articles superficial femoral artery 2024 articles superficial femoral artery Scopus articles superficial femoral artery impact factor journals superficial femoral artery Scopus journals superficial femoral artery PubMed journals superficial femoral artery medical journals superficial femoral artery free journals superficial femoral artery best journals superficial femoral artery top journals superficial femoral artery free medical journals superficial femoral artery famous journals superficial femoral artery Google Scholar indexed journals Pathological nature articles Pathological nature Research articles Pathological nature review articles Pathological nature PubMed articles Pathological nature PubMed Central articles Pathological nature 2023 articles Pathological nature 2024 articles Pathological nature Scopus articles Pathological nature impact factor journals Pathological nature Scopus journals Pathological nature PubMed journals Pathological nature medical journals Pathological nature free journals Pathological nature best journals Pathological nature top journals Pathological nature free medical journals Pathological nature famous journals Pathological nature Google Scholar indexed journals peripheral artery disease articles peripheral artery disease Research articles peripheral artery disease review articles peripheral artery disease PubMed articles peripheral artery disease PubMed Central articles peripheral artery disease 2023 articles peripheral artery disease 2024 articles peripheral artery disease Scopus articles peripheral artery disease impact factor journals peripheral artery disease Scopus journals peripheral artery disease PubMed journals peripheral artery disease medical journals peripheral artery disease free journals peripheral artery disease best journals peripheral artery disease top journals peripheral artery disease free medical journals peripheral artery disease famous journals peripheral artery disease Google Scholar indexed journals surgical intervention articles surgical intervention Research articles surgical intervention review articles surgical intervention PubMed articles surgical intervention PubMed Central articles surgical intervention 2023 articles surgical intervention 2024 articles surgical intervention Scopus articles surgical intervention impact factor journals surgical intervention Scopus journals surgical intervention PubMed journals surgical intervention medical journals surgical intervention free journals surgical intervention best journals surgical intervention top journals surgical intervention free medical journals surgical intervention famous journals surgical intervention Google Scholar indexed journals proximal tibial lesions articles proximal tibial lesions Research articles proximal tibial lesions review articles proximal tibial lesions PubMed articles proximal tibial lesions PubMed Central articles proximal tibial lesions 2023 articles proximal tibial lesions 2024 articles proximal tibial lesions Scopus articles proximal tibial lesions impact factor journals proximal tibial lesions Scopus journals proximal tibial lesions PubMed journals proximal tibial lesions medical journals proximal tibial lesions free journals proximal tibial lesions best journals proximal tibial lesions top journals proximal tibial lesions free medical journals proximal tibial lesions famous journals proximal tibial lesions Google Scholar indexed journals peripheral vascular disease articles peripheral vascular disease Research articles peripheral vascular disease review articles peripheral vascular disease PubMed articles peripheral vascular disease PubMed Central articles peripheral vascular disease 2023 articles peripheral vascular disease 2024 articles peripheral vascular disease Scopus articles peripheral vascular disease impact factor journals peripheral vascular disease Scopus journals peripheral vascular disease PubMed journals peripheral vascular disease medical journals peripheral vascular disease free journals peripheral vascular disease best journals peripheral vascular disease top journals peripheral vascular disease free medical journals peripheral vascular disease famous journals peripheral vascular disease Google Scholar indexed journals Repetitive knee flexions articles Repetitive knee flexions Research articles Repetitive knee flexions review articles Repetitive knee flexions PubMed articles Repetitive knee flexions PubMed Central articles Repetitive knee flexions 2023 articles Repetitive knee flexions 2024 articles Repetitive knee flexions Scopus articles Repetitive knee flexions impact factor journals Repetitive knee flexions Scopus journals Repetitive knee flexions PubMed journals Repetitive knee flexions medical journals Repetitive knee flexions free journals Repetitive knee flexions best journals Repetitive knee flexions top journals Repetitive knee flexions free medical journals Repetitive knee flexions famous journals Repetitive knee flexions Google Scholar indexed journals duplex ultrasonography articles duplex ultrasonography Research articles duplex ultrasonography review articles duplex ultrasonography PubMed articles duplex ultrasonography PubMed Central articles duplex ultrasonography 2023 articles duplex ultrasonography 2024 articles duplex ultrasonography Scopus articles duplex ultrasonography impact factor journals duplex ultrasonography Scopus journals duplex ultrasonography PubMed journals duplex ultrasonography medical journals duplex ultrasonography free journals duplex ultrasonography best journals duplex ultrasonography top journals duplex ultrasonography free medical journals duplex ultrasonography famous journals duplex ultrasonography Google Scholar indexed journals
Article Details
1. Introduction
Peripheral vascular disease (PVD) is devastating health problem all over the world [1]. Endovascular first strategy revascularization is one of the preferred strategies in many vascular centers. Surgical options should always a ready option for revascularization especially with long CTO 14 lesions. So, Chronic total occlusions (CTOs) of arteries are major challenges for revascularization. The caps of CTO lesions, distal popliteal and proximal tibial lesions and long CTO lesions (more than 50 mm length) are between the most important characteristics. The best decision making (whether endovascular or surgical therapy) in CTO lesions of the superficial femoral arteries (SFA) is the main focus of this article [2].
1.1 Aim of the work
The authors here discuss how the decision of the best method of an intervention in SFA total occlusion is difficult.
The anatomical TASC classification in SFA total occlusion may be not enough for the selection between endovascular or surgical intervention. We try to prove in this article the importance of clear understanding and visualization of the detailed pathological nature of the lesion and how it will create more accurate and safe decision.
2. Multiple Dynamic Forces affecting SFA and its Peculiar Nature
2.1 Shortening and length excess
Femoral and popliteal arteries elongate with age 26 and in peripheral vascular disease (PVD) [3, 4]. Also, during flexion, the artery bends smoothly (instead of acutely) and becomes longer [5]. The SFA shortens about 25% during (90 degrees) knee flexion [6] in comparison to (14%) shortening in the popliteal artery [7]. Also, the unstented portion of the artery tends to flex and bend more than the stented segment of the artery during the flexion, while overlapped stent portion cannot shorten as non overlapped one [7, 8].
2.2 Arterial tortuosity and axial twisting
This factor could be explained by the fact that the natural gliding of the femoral vessels 36 in the fascia of the adductor canal is impaired in the old age due to perivascular fibrosis. The adductor canal plays an important role to prevent traction to the vessel wall [9]. Also, the superficial femoral artery deformations in different fetal position have been proved [10].
2.3 Blood flow, wall shear stress, and early atherosclerotic lesions
Atherosclerotic Lesions and diminished arterial elasticity could result in disturbances of blood flow [5]. Wall shear stress (WSS) is directly correlated with blood flow and viscosity of the blood and inversely with the arterial radius [11, 12]. There is also a strong relation between both increased tortuosity and disturbed hemodynamic patterns in the SFA. Regions of low wall shear stress and disturbed flow were investigated, especially in the adductor canal. Repetitive knee flexions, lead to tortuous SFA with disturbed hemodynamic patterns and could cause atherosclerosis to progress [13]. On the opposite side, frequent movements of the leg lead to changes in the shape of the artery and redistribution of WSS may delay the progression of atherosclerosis [14].
3. Clinical Presentation of CTO in Superficial Femoral Artery
Asymptomatic patients with a reduced ankle brachial index (ABI) but no symptoms may have significant impairment of leg function when tested objectively.
3.1 Claudication
The patient suffered from intermittent claudications experiences leg pain (in the 50 calf, thigh or buttock) while walking and alleviated by rest [15].
3.2 Chronic limb threatening ischemia
Chronic Limb Threatening Ischemia (CLTI) or known also as Critical limb ischemia (CLI) is the most aggressive
form of PAD in approximately 1% of all patients with PAD [16]. It is very important to notice that the natural history of the critical limb ischemia is markedly aggressive in comparison to claudication. CLI is associated with a very high risk of limb loss if not promptly treated [17]. Rest pain and ischemic ulceration or gangrene of the forefoot or toes, representing different manifestations of CLI [18].
4. Options for Treatment
Femoral artery (CFA) or superficial femoral artery (SFA) occlusions (TASC D lesion), bypass surgery is preferable to endovascular treatment [19]. The main etiology for intervention in claudicants is to improve lifestyle, given that the risk of severe clinical deterioration (20%) or major limb amputation (5%) during a 3 to 5 year period is low [20]. Most studies demonstrate that infrainguinal endovascular therapy can be carried out with limited periprocedural risk [21]. Nevertheless, the decision to proceed with infrainguinal endovascular therapy rather than an open surgical approach requires a thorough risk benefit analysis based on the information acquired from the history, physical examination, noninvasive imaging, and diagnostic angiography. Salient features of the history and physical examination, when planning an intervention, include (1) indication (claudication 112 versus critical limb ischemia [CLI]); (2) disease location, extent, and severity; (3) degree of disability and lifestyle limitations; (4) medical comorbidities and anesthetic risk; (5) prior lower extremity reconstructions or interventions; and (6) prospects for long term functional status and survival. Also, detailed discussions of duplex ultrasonography, computed tomographic angiography (CTA), magnetic resonance angiography (MRA), and digital subtraction angiography are crucial [19]. So, We need to weigh both surgical and endovascular modalities to select the best method for intervention.
5. Surgical Bypass… is it the Best Option?
5.1 Bypass versus angioplasty in severe ischaemia of the Leg (BASIL) trial (BASIL–1)
That for patients who lived more than 2 years after randomization, a bypass-first revascularization strategy was associated with a significant increase in survival as well as better amputation free survival. The BASIL trial clearly demonstrated that patients who used prosthetic grafts for the bypass surgery (constituting 25% of the surgical group) experienced decreased amputation-free survival compared with those who received autogenous vein grafts. The BASIL authors concluded that patients who underwent bypass surgery after failed first endovascular therapy experienced significantly worse amputation free survival than did those who underwent bypass first as the initial therapy [22]. Despite Basil study-1 recommends surgical bypass over endovascular management, but we still have major complications related to surgical bypass.
5.2 Lower extremity vein graft failure
Although intimal hyperplasia is the main cause of vein graft failure, but geometric remodeling of the healing vein graft is another important factor [23]. Other factors include diabetes, race and perioperative management could be adjusted with total understanding of the risk of early graft failure. So, bypass surgery is not the best strategy for the claudicant patients. Also, bypass to the tibial arteries must be reserved to limb salvage situations and every effort should be done to use the autogenous vein [24].
6. Endovascular First Strategy
Because many patients with TASC C/D lesions have medical comorbidities that place them at high risk for surgical bypass and therefore endovascular first strategy still an important alternative. Many centers are now using endovascular first therapy as the main treatment line for TASC-C/D lesions and reserving surgical therapy for failed endovascular therapy. We have acceptable short and mid-term outcomes following endovascular intervention of femoropopliteal artery, but with mixed results on longer term outcomes [25].
6.1 Complications of endovascular treatment
Most of them are minor and can be treated with endovascular therapies. A frequent complication of percutaneous access including bleeding, hematoma and arteriovenous fistula.The angle of approach is believed to be the cause of arteriovenous fistula. A pseudoaneurysm (PSA) also may develop after endovascular techniques if the arteriotomy has not sealed adequately. One of the more common problems in the percutaneous procedures is intimal-medial dissection. Balloon inflation causes stretching and disruptions of of the plaque and overlying intima and occasionally the media that results in a tear or dissection. Embolization is a devastating complication of endovascular procedures. Arterial perforation can result from wire manipulation, implantation and removal of endovascular devices or balloon angioplasty [26, 27]. We need a step forward after TASC classification for decision making in the treatment of total SFA occlusion lesion. We need a clear correlation between pathological features, clinical presentation and anatomical distribution of the arterial lesion.
7. Pathological Nature of Chronic Total Occlusion
In coronaries, a chronic total occlusion is defined as total interruption of the coronary antegrade blood flow of greater than 3 months whether documented angiographically or suspected clinically [28]. The CTO has a body, proximal and distal cap. The proximal cap is often calcified or fibrotic and either tapered or not. The body of the occlusion consists histologically from fibrous tissue, whether loose or dense, calcification, atheroma, lymphocytes aggregates and is mostly has also a neovascularization [29]. Negative remodeling was frequent and particularly with long standing CTOs; while shorter duration CTOs characterized by abundant organized thrombus and large necrotic core. Collateral circulation opacifies the distal cap in most cases and appears to be more tapered than the proximal cap, facilitating wire entry with the retrograde techniques [30, 31].
8. Pathological Stages of Chronic Total Occlusion
Three stages of CTO development have been identified: first two weeks is the early stage, (6-12 weeks) is the intermediate stage and (18-24 weeks) is the advanced one. The early stage was characterized by the formation of the immature extracellular matrix with more proteoglycan and low collagen content as an acute response to the vessel injury. In the early intermediate stage (6 weeks), there is a high negative arterial remodeling, disruption of the internal elastic lamina, and intraluminal neovascularization. In the later intermediate stage (12 weeks), there is reduction in the blood volume, low proteoglycan content and increase in the collagen with microvessels formation that remains till the advanced stage. There is also reduction in the perfusion with advanced CTO lesion.Bidirectional thrombus formation is considered an essential component of the initiating event in the pathogenesis of human CTO development [32]. The physical properties Four pathological features represent a real obstacle preventing the success of the endovascular technique in the intervention of the long standing CTO lesion (Figure 1). These pathological changes include negative remodeling with the resultant reduction in the vessel size up to 80% by 6 weeks, the tough fibrous proximal cap that studded with collagen, progressive decrease in the proteoglycan content with increase in the rigid collagen content and discontinuity of the microvascular network with reduction in the perfusion of CTO [33, 34].
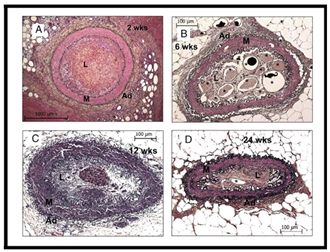
Figure 1: Movat Stained Sections Showing Temporal Changes in Vessel Size and Intraluminal Microvessels. Representative histological sections of occlusions at 2 (A), 6 (B), 12 (C), and 24 weeks (D). There was marked reduction in vessel size at 6 weeks (note the differences in calibration). Microvessels (indicated by *) were maximal at 6 weeks with a decrease at the later time period. Ad-adventitia; L-lumen; M-media [34].
9. Lesion Calcification Classification
Curved multiplanar reconstructions (MPR) as well as axial slices were used for calcification assessment. Depending upon the global amount of calcification on curved MPR, lesions were classified as (1) grade 1, with absent or mild calcification; or (2) grade 2, with severe calcification. On the basis of axial location, lesions were classified as (1) grade 1, with high-density plaque involving less than 50% of the vessel’s cross sectional area; or (2) grade 2, with high density plaque involving at least 50% of the vessel’s cross sectional area. Only the highest grade was taken in to account in case of multiple types of calcifications [34, 35].
10. Role of Plaque Cap Morphology in Determining CTO Crossing Approach
The CTOP study is the first to categorize peripheral CTOs and identify factors. CTO mapping using the CTOP classification actually increases the rate of crossing without increase the rate of complications.
10.1 Proposed crossing approach based on CTOP classification
CTOP classification represents one of the most detailed and practical algorithm for the selection of the best approach, whether antegrade or retrograde (Table 1) (Figure 2). The retrograde tibiopedal access is mostly used for long (>10 cm) and heavily calcified CTO lesions. We have four types of CTOs depending on the proximal and distal cap appearance. Type I CTO, it could be treated easily with the antegrade endovascular route. Type II CTO, in this type the proximal cap could be crossed with antegrade technique while the distal cap because of the antegrade convexity- needs the retrograde technique or the subintimal route via with antegrade technique. Type III CTO, in this type both antegrade and retrograde techniques have been recommended to be used. Type IV CTO, the retrograde fashion is the best because both the proximal and distal caps have retrograde concavity [36]. (Table 1) (Figure 2).
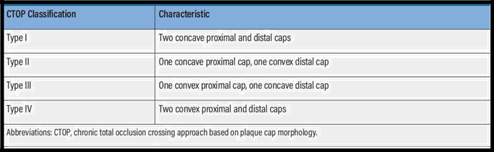
Table 1: Characteristics of caps in the CTOP study when descend from antegrade approach [36].
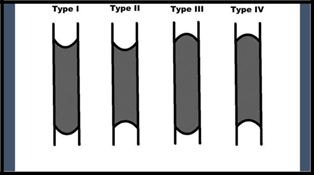
Figure 2: The four types of CTOs based on the proximal and distal cap appearance [36].
11. How the Pathological Nature of Chronic Total Occlusion of SFA appears by Angioscopy
Angioscopy has been used for the imaging of the thrombus, and the vessel wall from inside and the proximal and distal cap of the CTO lesion of the superficial femoral artery. It is generally considered that CTO starts with occlusion of the artery with thrombus, followed by replacement with collagen formation and deposition of calcium. Angioscopy enables direct visualization of the lumen, which makes it useful for the diagnosis of thrombus, but it cannot reach the distal side of CTO lesions in coronary arteries. Therefore, the femoral artery of patients with arteriosclerosis obliterans was examined, and may be, this is the first reported angioscopic finding of the distal side of CTO lesions. In both of our cases, multiple thrombi of variable nature were identified with patchy distribution on the distal side, but not on the proximal side, of the CTO lesions. Those thrombi could not be identified with angiography (Figure 3, 4). There was no coagulation disorder in these patients. Our observations support an idea that one mechanism of formation of a long CTO lesion can be the result of a distal extension of the thrombus that started from the initial site of occlusion. The presence of red and mixed thrombi on the distal side of our CTO lesions suggests that the combined use of anticoagulation with antiplatelet agents may be more effective to minimize the extending growth of the CTO lesion [37].
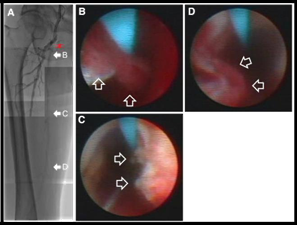
Figure 3: Images of peripheral arterial angiogram and angioscope of preendovascular treatment in case 1. Control angiography (A) showed that the superficial femoral artery (SFA) was occluded immediately after branching of the deep femoral artery (DFA). Collateral circulation from the deep femoral artery irrigated the SFA distal to the occlusion, and its filling with contrast media was observed. The red 2 way arrow indicates the range of chronic total occlusion of the SFA. Angioscopic images (B through D) respectively, correlates with the sites marked by solid arrows in A, and thrombi are indicated by hollow arrows. Different types of thrombi (B, a red thrombus partly covered by white thrombi; C, a white thrombus; D, mixed thrombus) could be identified over the light yellow plaques [37].
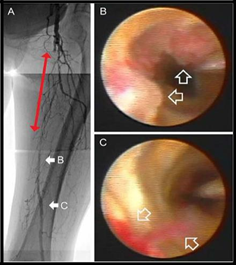
Figure 4: Images of peripheral arterial angiogram and angioscope of preendovascular treatment in case 2. Diagnostic angiography (A) showed that the superficial femoral artery (SFA) was occluded immediately after the branching of deep femoral artery (DFA), and the distal side of the occlusion was contrast filled by collateral flow from the DFA. The red 2 way arrow indicates the range of chronic total occlusion of the SFA. An Angioscopic image (B and C) respectively, correlates with the sites indicated by solid arrows in A, and thrombi are indicated by hollow arrows. Different types of thrombi (B, white thrombi; C, red thrombus attached with a flapping white thrombus) could be identified over the dark yellow plaques [37].
We need not only to draw the anatomical distribution of the arterial lesion but also the detailed pathological nature of the chronic total occlusion. More calcified hyperechoic lesion will prone to endovascular treatment failure specially at proximal and distal cap. Hypoechoic lesion is a soft lesion that usually will respond successfully to endointervention.
12. Role of Duplex Ultrasound in Describing the Pathology of Chronic Total Occlusion in SFA
Doppler US is a safe and effective imaging modality for functional and morphological assessment of SFA. It can be performed by obtaining gray scale images and the color Doppler study. On a gray scale image, the presence and the size of a plaque could be described, as well as whether the plaque is calcified or not. Three dimensional US has been recently used for measuring plaque volume. On color Doppler ultrasound, total SFA occlusion is seen in the form of absent color signal within the lumen. SFA stenosis shows aliasing artifacts on color doppler Us due to turbulence in blood flow caused by high grade velocities. On spectral flow Doppler, intra-arterial peak systolic velocity (PSV) and ratio of PSV between the site of stenosis and adjacent normal vessel are used as a primary criteria to estimate the degree of stenosis. Stenotic segments will show increase in the peak systolic velocity, which is proportionate to the degree of stenosis. Total absence in spectral flow is seen at totally occluded segments. An area of flow disturbance is seen within 2 cm beyond the area of stenosis due to loss of the laminar flow pattern, it show spectral broadening. Monophasic wave pattern is also described distal to the site of occlusion. It is characterized by a “damped” pattern, which means that systolic flow acceleration is slowed, peak systolic velocity is reduced, and diastolic flow is increased [38]. The use of US is essential in obtaining access, Adequate visualization of vessels, evaluation of proximal and distal CTO cap (morphology, architecture and shape), Length of the CTO, Collaterals at the proximal CTO cap and Calcium content [39].
13. Role of Optical Coherence Tomography in describing the pathology of chronic total occlusion in SFA
Optical Coherence Tomography (OCT) imaging can differentiate between the occluded lumen and the underlying arterial wall in peripheral CTOs. OCT correctly identified tissue composition within the CTO, such as the presence of collagen and calcium and was also able to identify intraluminal micro channels. CTOs could be classified by OCT into different types according to the histological appearance (Figure 5):
- Extensively calcified wall, dense collagen occupying the lumen.
- Microcalcifications within the lumen, embedded in collagen.
- Extensive smooth muscle cell infiltration with collagen in lumen.
- High lipid content in lumen.
- Dense collagen within lumen.
In the OCT images, dense fibrotic tissue appeared bright, while highly cellular areas and looser connective tissue appeared darker. Microcalcifications were seen as highly reflective spots under OCT within the CTO that greatly attenuate the OCT signal with depth. Regions of lipids were observed as signal spaces within poor the CTO and confirmed using Oil Red stain. Intraluminal microchannels within the occlusion were identified in most OCT images as small crevices on the longitudinal slices and holes on the cross sectional slices; that were confirmed by histology. The appearance of the different potential components of CTOs under OCT is summarized in Table 2.
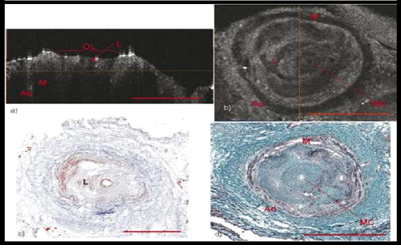
Figure 5: OCT images of an occluded anterior tibial artery demonstrating high lipid content are shown in (a) and (b). A small central microchannel (MC) is seen in both reconstructed cross sectional OCT slices and histology. Lipid deposition, labeled L, is seen both within the collagen matrix of the CTO as well as accumulation around the central microchannel on the Oil Red O histology shown in (c). These regions are seen as weakly scattering regions in the OCT images. Lipid deposition around the central microchannel appears as small segmental deposits seen in the longitudinal OCT image shown in (a). Areas of the lumen containing a high collagen content onceagain appear as bright under the OCT images. Histology is Oil Red O in (c) and Elastin Trichrome (d) [40].
|
CTO component |
OCT appearance |
|
Perivascular tissue (loose connective |
Dark border surrounding the artery |
|
Adventitia |
Signal rich peripheral of the vessel |
|
Media |
Signal rich in significant fibrosis or signal poor |
|
Collagen within the lumen |
Uniformly backscattering region. Denser collagen has a higher back scattering signal. |
|
Smooth muscle cells within the lumen |
Dark region within collagen matrix of the |
|
Intraluminal microchannels |
Fine cracks within the CTO. Residual blood shows as a bright reflective lining. |
|
Lipid |
Lightly scattering in large pools. |
|
Microcalcifications within the CTO |
Highly reflective dots. When abundant they create shadows. |
|
Intraluminal calcium |
Highly reflective in the surface, otherwise signal poor. |
Table 2: OCT Signal characteristics of CTO constituents. Quoted from (Nigel R. Munce et al, 2007)
It is clear now that we need a step forward beyond the TASC anatomical classification as a main classification for selection of the best therapeutic approach for the chronic total occlusion of SFA. We need a classification correlates between clinical presentation, anatomical distribution, morphology and pathological nature of the lesion for better therapeutic method selection and better prediction of intervention complications. We have till now a mangel in the available tools to perform this mission, but maybe the duplex scan, optical coherence Tomography and angioscopy are valuable options.
Acknowledgements
To Amira Roshdy Eltabbakh.
Conflict of Interest
No conflict of interest.
References
- Fowkes FG, Rudan D, Rudan I, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: A systematic review and analysis. Lancet 382 (2013): 1329-1340.
- Mustapha JA and Fadi S. Long Peripheral CTOs: Femoropopliteal to Mid and Distal Tibial Arteries. Proper patient assessment and current access and revascularization techniques to treat challenging CLI anatomy. Endovascular Today 15 (2016).
- Chilvers AS, Thomas ML, Browse NL. The progression of arteriosclerosis, a radiological study. Circulation 50 (1974): 402-408.
- Smedby O, Hogman N, Nilsson S, et al. Two-dimensional tortuosity of the superficial femoral artery in early atherosclerosis. J Vasc Res 30 (1993): 181-191.
- Wensing PJ, Scholten FG, Buijs PC, et al. Arterial tortuosity in the femoropopliteal region during knee flexion: a magnetic resonance angiographic study. J Anat 187 (1995): 133-139.
- Vernon P, Delattre JF, Johnson EJ, et al. Dynamic modifications of the popliteal arterial axis in the sagittal plane during flexion of the knee. Surg Radiol Anat 9 (1987): 37-41.
- Smouse HB, Nikanorov A, LaFlash D. Changes in major peripheral arteries during joint movement before and after stent placement in the cadaver model. In: Program and Abstracts, Transcatheter Cardiovascular Therapeutics; Washington, DC (2004).
- Zocholl G, Zapf S, Schild H, et al. Functional angiography of the arteries near the knee joint: consequences for stent implantation?. Rofo 153 (1990): 658-662.
- De Souza RR, Ferraz de Carvalho CA, Merluzzi Filho TJ, et al. Functional anatomy of the perivascular tissue in the adductor canal. Gegenbaurs Morphol Jahrb 130 (1984): 733-738.
- Cheng CP, Wilson NM, Hallett RL, et al. In vivo MR angiographic quantification of axial and twisting deformations of the superficial femoral artery resulting from maximum hip and knee flexion. J Vasc Interv Radiol 17 (2006): 979-987.
- Wood NB, Zhao SZ, Zambanini A, et al. Curvature and tortuosity of the superficial femoral artery: a possible risk factor for peripheral arterial disease. J Appl Physiol 101 (2006): 1412-1418.
- Stonebridge PA, Brophy CM. Spiral laminar flow in arteries? Lancet 338 (1991): 1360-1361.
- Lindbom A. Arteriosclerosis and arterial thrombosis in the lower limb; a roentgenological study. Acta Radiol Suppl 80 (1950): 1-80.
- Frederik HWJ, Felix JVS, Frans LM, et al. Dynamic Forces in the SFA and Popliteal Artery During Knee Flexion Consequences of stress to consider for stent durability and design. Endovascular today (2008).
- Sluka KA. Pain mechanisms involved in musculoskeletal disorders. J Orthop Sports Phys Ther 24 (1996): 240-254.
- Dormandy JA, Rutherford RB. The TransAtlantic Inter-Society Consensus on the management of peripheral arterial disease. J Vasc Surg 31 (2000): 1-296.
- Tang GL, Chang DS, Sarkar R, et al: The effect of gradual or acute arterial occlusion on skeletal muscle blood flow, arteriogenesis, and inflammation in rat hindlimb ischemia. J Vasc Surg 41 (2005): 312-320.
- Hasan HD. Lower Extremity Arterial Disease: General Considerations. In Eds.: Cronenwett JL, Johnston KW. Rutherford’s vascular surgery (8th) Philadelphia: Saunders (2014): 1660-1674.
- Joseph L and Mills SR. Infrainguinal Disease: Surgical Treatment. In Eds.: Cronenwett JL, Johnston KW. Rutherford’s vascular surgery (8th) Philadelphia: Saunders (2014): 1758-1881.
- Norgren L, Hiatt WR, Dormandy JA, et Inter Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J Vasc Surg 45 (2007): 5-6.
- Wolfe JH, Wyatt MG. Critical and subcritical ischaemia. Eur J Vasc Endovasc Surg 13 (1997): 578-582.
- Andrew WB, Donald JA, Jocelyn B, et al. Bypass versus Angioplasty in Severe Ischaemia of the Leg (BASIL) trial: Analysis of amputation free and overall survival by treatment received. Journal Of Vascular surgery 51 (2010).
- Christopher DO, Karen JH and Michael SC. Lower extremity vein graft failure: a translational approach. Vascular Medicine 13 (2008): 63-74.
- Niten S, Anton NS, Kent JD, et al. Factors associated with early failure of infrainguinal lower extremity arterial bypass Washington, DC, Journal of vascular surgery 47 (2008).
- Jeffrey LG, Kong TT, Hadas M, et al. Endovascular first strategy for denovo TransAtlantic InterSociety Consensus C and D femoro-popliteal disease: Midterm outcomes from a single tertiary referral center.Vascular 23 (2015): 31-40.
- Sherev DA, Shaw RE, Brent BN. Angiographic predictors of femoral access site complications: implication for planned percutaneous coronary intervention. Catheter Cardiovasc Interv 65 (2005): 196-202.
- Elliot BL and James FM. Local Complications: Endovascular. In Eds.: Cronenwett JL, Johnston KW. Rutherford’s vascular surgery (8th) Philadelphia: Saunders (2014): 704-722.
- Sianos G, Werner GS, Galassi AR, et al. Recanalisation of Chronic Total coronary Occlusions: 2012, consensus document from the Euro CTO club. Euro Intervention 8 (2012): 139-145.
- Srivatsa SS, Edwards WD, Boos CM, et al. Histologic correlates of angiographic chronic total coronary artery occlusions: influence of occlusion duration on neovascular channel patterns and intimal plaque composition. J Am Coll Cardiol 29 (1997): 955-963.
- Sakakura K, Nakano M, Otsuka F, et al. Comparison of pathology of chronic total occlusion with and without coronary artery bypass graft. Eur Heart J 35 (2014): 1683-1693.
- Georgios S, Nikolaos VK, Carlo DM, et al. Theory and practical based approach to chronic total occlusions, BMC Cardiovascular Disorders 16 (2016): 33.
- Wakefield TW, Linn MJ, Henke PK, et al. Neovascularization during venous thrombosis organization: a preliminary study. J Vasc Surg 30 (1999): 885-892.
- Strauss BH, Segev A, Wright GA, et al. Microvessels in chronic total occlusions: pathways for successful guidewire crossing. J Interv Cardiol 18 (2005): 425-436.
- Ronen J, General L, Nigel RM, et al. Natural History of Experimental Arterial Chronic Total Occlusions. J Am Coll Cardiol 53 (2009): 1148-1158.
- Li MW, Yue QZ, Pei LZ, et al. Morphological characteristics of chronic total occlusion:predictors of different strategies for long segment femoral arterial occlusions. Vascular Interventional, Eur Radiol 28 (2018): 897-909.
- Fadi S and Jihad AM. Using Plaque Cap Morphology to Determine CTO Crossing Approach. Initial findings from the CTOP study and how they could apply to clinical practice. Endovascular Today 17 (2018).
- Kenji Y, Masafumi U, Hiroyuki Y, et al. Images in Cardiovascular Medicine, Backyards of Chronic Total Occlusion Scenery Revealed Through Angioscope. Circulation 129 (2014): 2715-2716.
- Ji YH. Doppler ultrasonography of the lower extremity arteries: anatomy and scanning guidelines. Ultrasonography 36 (2017): 111-119.
- Fadi S. How Can Ultrasound help me Lets talk beyond the access? (2015).
- Nigel RM, Victor XDY, Beau AS, et al. Ex Vivo Imaging of Chronic Total Occlusions Using Forward Looking Optical Coherence Tomography. Lasers in Surgery and Medicine 39 (2007): 28-35.


 Impact Factor: * 4.2
Impact Factor: * 4.2 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 