Spontaneous Regression of Congenital Cholesteatoma: A Report of Two Cases with Differing Ossicle Involvement
Article Information
Yoshimasa Tsuchiya, Taichi Kan*, Mayuko Kishimoto, Yasue Uchida, Tetsuya Ogawa, Hiromi Ueda
Department of Otorhinolaryngology Aichi Medical University School of Medicine, 1-1 Yazakokarimata, Nagakute, Aichi, Japan
*Corresponding Author: Dr. Taichi Kan, Department of Otorhinolaryngology Aichi Medical University School of Medicine, 1-1 Yazakokarimata, Nagakute, Aichi, 4801195, Japan
Received: 27 Sep 2019; Accepted: 07 October 2019; Published: 29 November 2019
Citation: Yoshimasa Tsuchiya, Taichi Kan, Mayuko Kishimoto, Yasue Uchida, Tetsuya Ogawa, Hiromi Ueda. Spontaneous Regression of Congenital Cholesteatoma: A Report of Two Cases with Differing Ossicle Involvement. Archives of Clinical and Medical Case Reports 3 (2019): 683-691.
View / Download Pdf Share at FacebookAbstract
The mechanism of congenital cholesteatoma has not been clarified yet. Usually, middle ear cholesteatoma (congenital and acquired) is a progressive and destructive disease that requires surgery for complete cure. In recent years, early detection of congenital cholesteatomas has become more frequent due to advances in endoscopy and microscopy. Along with these developments, there have been reports of congenital cholesteatoma regressing spontaneously, as seen by tympanic and computed tomography findings over time. Herein, we report two cases of spontaneous regression of cholesteatoma, both with ossicular chain malformation. One cholesteatoma was close to the ossicles. In the other case, the cholesteatoma and ossicle were separated; however, the ossicular morphology was inconsistent with that of congenital ossicular malformation. Further similarities and differences between these two cases are discussed herein.
Keywords
Congenital cholesteatoma; Spontaneous regression/ reduction; Conductive hearing loss; Ossicular chain malformation
Congenital cholesteatoma articles, Spontaneous regression/ reduction articles, Conductive hearing loss articles, Ossicular chain malformation articles
Article Details
Abbreviations:
CT- Computed tomography; OME- Otitis media with effusion
1. Introduction
In congenital cholesteatoma, it is thought that the cholesteatoma epithelium often pre-exists in the middle ear cavity from birth and is subsequently found in childhood. Epithelioma is derived from epidermis cells that have entered the embryonic period, and most epitheliomas are of a cystic form containing keratin (closed type), but there is also a membranous form of epithelioma that does not form a cyst (open type) [1-3]. Tos M. stated the incidence of congenital cholesteatoma is around 0.12 out of 100,000, and it is more common among boys [4]. In recent years, otolaryngologists and pediatricians alike have increased the awareness of this disease, and the number of cases diagnosed at a young age has increased due to the spread of improved screening and diagnostic techniques. In addition, there has been an increase in the number of cases diagnosed coincidentally from symptoms such as hearing loss and otitis media. Furthermore, there are reports that congenital cholesteatoma can spontaneously regress, identified by changes in tympanic and CT findings over time [5]. We examined if there is something with congenital cholesteatoma of spontaneous regression, and it can be used for follow-up. Herein we report two cases of spontaneously regressed cholesteatoma, each with different relationships between the cholesteatoma and ossicles.
2. Case Report
2.1 Case 1
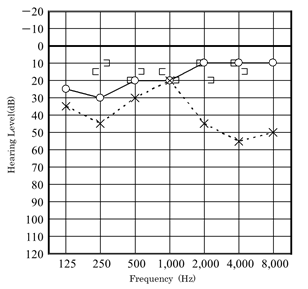
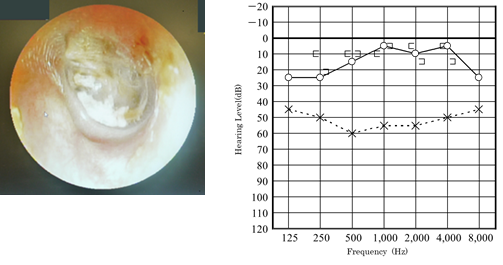
Our first patient was a boy who presented with a history of progressive hearing loss in the left ear. Left hearing loss was first noted during a school checkup at the age of 6 years in 2008, after which he visited a nearby clinic. Pure-tone audiometry showed a conductive hearing loss (Figure 1).
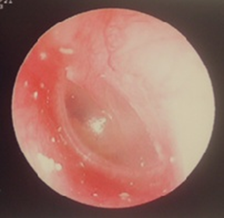
Magnetic resonance imaging showed no acoustic tumor. Since then, follow-up had been performed at the aforementioned nearby clinic. At the age of 7 years, in February 2010, the patient had no change in hearing loss and was referred to our hospital. Upon admission to our hospital, the eardrum findings were normal (Figure 2).
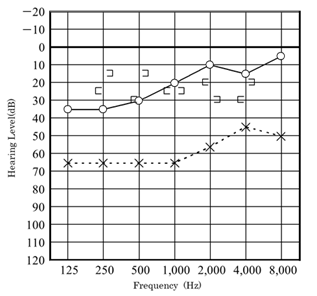
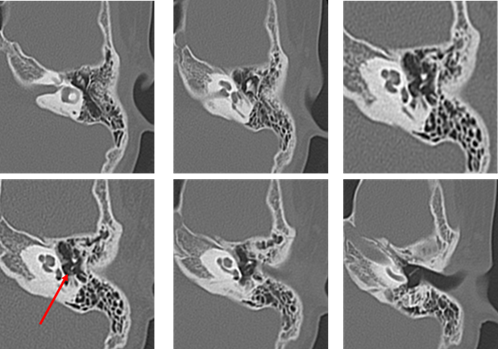
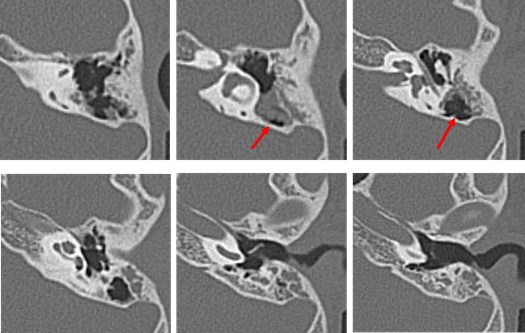
Audiometry again showed left conductive hearing loss and acoustic reflex tests were bilaterally positive. At this time, the patient was asked to continue with follow-up at the nearby clinic. In August 2011, computed tomography (CT) at the nearby clinic showed small, soft shadows around the tympanic segment of the facial nerve and the stapes (Figure 3). In December 2011, pure-tone audiometry at the clinic showed deterioration (Figure 4).
The patient visited our hospital again in January 2012 at the age of 9 years. Possible congenital cholesteatoma or ossicular chain malformation were considered, and the patient was scheduled for surgery. In July 2012, preoperative CT showed a reduction in the previously seen shadow (Figure 5); however, the hearing test showed no significant change. Left tympanoplasty was performed in August 2012.
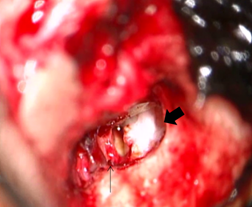
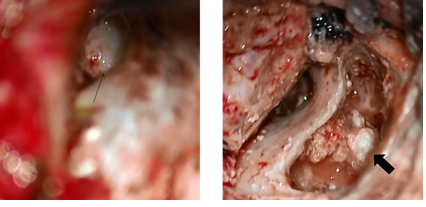
During the operation, the mucosa of the tympanic cavity was normal, but an open type cholesteatoma was found from the tympanic segment of the facial nerve and the long process of the incus to the stapes. The long process of the incus was eroded and the anterior and posterior crura of the stapes were missing, and the stapes superstructure was separated from the footplate. The cholesteatoma was removed; however, because the cholesteatoma had invaginated into the lower surface of the tympanic segment of the facial nerve, we considered the possibility of remaining tumor cells and scheduled a staged operation. The postoperative diagnosis was Potsic classification Stage II congenital cholesteatoma (Figure 6) [6]. In 2013, left tympanoplasty was performed, and there was no subsequent recurrence of cholesteatoma. In August 2018, 5 years after the second operation, no recurrence was found by CT, and follow-up at our hospital was ended.
2.2 Case 2
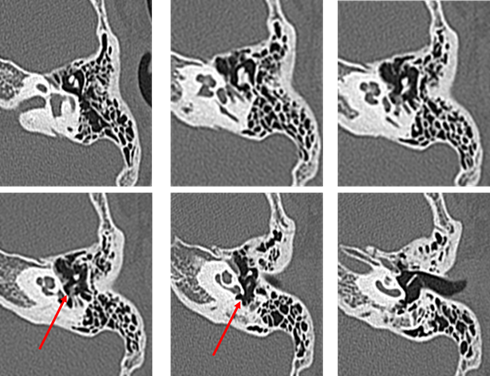
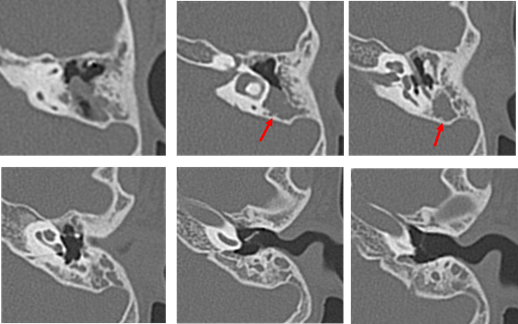
Our second was a boy diagnosed with hearing loss and left tympanic membrane abnormality at a medical checkup in April 2013. He visited a local doctor and was treated for left otitis media with effusion (OME). His left OME resolved, but there was no improvement in hearing, and the patient was thus referred to our hospital in August 2013. Examination revealed small perforations in the posterosuperior quadrant of the left eardrum (Figure 7). CT revealed a soft shadow from the epitympanum to the mastoid cavity, and the long process of the incus and the superstructure of the stapes appeared to be missing. However, CT showed discontinuity of these two lesions. Therefore, we suspected left pars tensa cholesteatoma or congenital cholesteatoma and ossicular chain malformation (Figure 8). In March 2014, preoperative CT showed a reduction in the previously seen soft shadow (Figure 9).
Figure 8: Computed tomography performed in August 2013 on the patient of Case 2 aged 7 years. The long process of the incus and stapes are missing. Suspected cholesteatoma from the soft shadow (indicated the red arrow) in the epitympanic attic and mastoid sinus. There are no suspicious findings around the ossicles.
Additionally, the small perforations in the eardrum had closed. Surgery was performed in April 2014, and open type cholesteatoma and cholesterol granuloma were found in the attic and mastoid sinus, and the lesions were removed. The malleus was intact, the long process of the incus was defective, and the stapes were displaced backward in the structure, but no cholesteatoma was found around the ossicle. The cholesteatoma was evaluated as Potsic classification Stage II. At the time of this writing, the patient continues to follow up at the outpatient clinic (Figure 10).
The thin arrow indicates that there is no cholesteatoma in tympanic cavity. The long process of the incus and the superstructure of the stapes were disfigured. The thick arrow indicates the cholesteatoma in the mastoid cavity.
3. Discussion
There have been reports on the spontaneous regression of congenital cholesteatomas [6]. There are several possibilities for the mechanism of spontaneous regression or reduction of congenital cholesteatoma. First, because closed type cholesteatoma may spontaneously morph to an open type, and the eustachian tube function of most congenital cholesteatoma patients is normal, debris excreted from the eustachian tube may move slowly, and the cholesteatoma may disappear. Second, ectodermal tissue improperly in the embryonic stage may resolve through apoptosis [7]. In early-childhood congenital cholesteatoma, the reasons for spontaneous regression are not clear, and the current standard of care for cholesteatoma remains surgical treatment. However, in infants and children, the eustachian tube function, the middle ear and the surrounding bone are still developing, and it may be difficult to predict changes in the external auditory canal and middle ear morphology several years after surgery. They may also have difficulty cooperating with postoperative procedures, which may be a reason for undesirable effects on surgical outcomes. Therefore, if the characteristics and cause of spontaneously regressed congenital cholesteatoma are clarified, it may be possible to select surgery after waiting for physical and mental growth.
In these two cases reported here, we confirmed that cholesteatoma spontaneously regressed over time. In Case 1, the patient’s hearing loss deteriorated gradually and there was a reduction in the soft shadow seen by CT; however, the possibility of congenital cholesteatoma was high, and thus surgery was performed. During the operation, open type cholesteatoma was found around the stapes, and both crura were missing. This ossicular malformation was clearly attributed to cholesteatoma. In Case 2, small perforations were observed in the eardrum at first and, after CT, we suspected pars tensa cholesteatoma that had progressed to the mastoid cavity. However, the mastoid cavity lesion appeared to be separated from the ossicular defect, which was thought to be the cause of hearing loss. During surgery, the separation between the cholesteatoma and the defect was confirmed. Interestingly, the ossicle abnormalities were different from the congenital ossicular malformations that we usually experience, and thus we suspected that there had initially been a cholesteatoma surrounding the ossicles that had spontaneously regressed, leaving behind the ossicular transection. The differences between the two cases are 1. the locations of the cholesteatomas, 2. the separation between the cholesteatoma and the ossicle defect in case 2, and 3. the sizes of the cholesteatomas. Accordingly, the common points of these two cases are as follows: 1) the cholesteatomas were both open type, 2) the long process of the incus was damaged and 3) the crura of the stapes were disconnected from the footplate in both patients. In the report of two cases by Kodama et al., one of them CT showed the soft shadow suspected congenital cholesteatoma around the incudostapedial joint. It was operated on, and the cholesteatoma had regressed completely, so the type was unknown, but the long process of the incus was damaged.
If preoperative exams demonstrate regression of cholesteatoma, and there is no infection, bone destruction, impairment to ventilation in the middle ear, or deteoration of hearing loss, watchful waiting may be indicated. There were similarities and differences between our two cases. Future retrospective analyses should be done on the spontaneous regression of congenital cholesteatoma to identify independent factors that indicate “watchful waiting” for these patients.
Acknowledgments
We would like to thank Editage (www.editage.com) for English language editing.
Conflicts of Interest
There are no conflicts of interest to declare.
References
- Aimi K. Role of the tympanic ring in the pathogenesis o congenital cholesteatoma. Laryngoscope 93 (1983): 1140-1146.
- Michaels L. An epidermoid formation in the developing middle ear; possible source of cholesteatoma. Journal of Otolaryngology-Head and Neck Surgery 15 (1986): 169-174.
- Michaels L. Origin of congenital cholesteatoma from a normally occurring epidermoid rest in the developing middle ear. International Journal of Pediatric Otorhinolaryngology 15 (1988): 51-
- Tos M. A new pathogenesis of mesotympanic (congenital) cholesteatoma. Laryngoscope 110 (2000): 1890-
- Kodama K, Hara M, Matsuzawa S, et al. Two cases of spontaneous regression of congenital cholesteatoma. International Journal of Pediatric Otorhinolaryngology 76 (2012): 142-
- Potsic WP, Samadi DS, Marsh RR, et al. A staging system for congenital cholesteatoma. Archives of Otolaryngology-Head & Neck Surgery128 (2002): 1009-
- Kojima H, Miyazaki H, Shiwa M, et al. Molecular Biological Diagnosis of Congenital and Acquired Cholesteatoma on the Basis of Differences in Telomere Length. Laryngoscope 111 (2001): 867-


 Impact Factor: * 3.1
Impact Factor: * 3.1 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
