Survival Pattern of Patients on Maintenance Haemodialysis for End Stage Renal Disease in a Nigerian Dialysis Centre
Article Information
Dada SA1*, Inubile AJ2, Thomas AA3, Dada OE4, Akinyemi RO4
1Nephrology Unit, Department of Medicine, Ekiti State University Teaching Hospital, Nigeria
2Department of Biostatistics, Ekiti State University Teaching Hospital, Ado-Ekiti, Nigeria
3Department of Radiodiagnosis, Ekiti State University Teaching Hospital, Ado-Ekiti, Nigeria
4Department of Health Information Management, Ekiti State University Teaching Hospital, Ado-Ekiti, Nigeria
*Corresponding Author: Samuel Ayokunle Dada, Nephrology Unit, Department of Medicine, Ekiti State University Teaching Hospital/Ekti State University Ado Ekiti, Nigeria
Received: 05 February 2019; Accepted: 11 February 2019; Published: 15 February 2019
Citation: Dada SA, Inubile AJ, Thomas AA, Dada OE, Akinyemi RO. Survival Pattern of Patients on Maintenance Haemodialysis for End Stage Renal Disease in a Nigerian Dialysis Centre. Archives of Nephrology and Urology 2 (2019): 001-012.
View / Download Pdf Share at FacebookAbstract
Background: The increasing incidence and prevalence of chronic kidney disease (CKD) is a serious health challenge globally. The need to examine the longevity of patients with CKD can never be over emphasized. We investigated the survival experience and factors that may contribute to the longevity of chronic kidney disease patient on haemodialysis.
Methods: Data for the study were extracted from the record of CKD patient on haemodialysis. Kaplan-Meier survival analysis was done to assess both short- and long-term survival. The impact of six covariates on survival chances were separately investigated using Log-rank test and collectively examined using both Cox and Weibull models. Akaike Information Criterion was then employed for determination of a better model between variables. P-value of <0.05 was considered as statistically significant.
Results: The overall median survival time was 182 days. Only 66.3% of all the patients survived their 90th days after starting dialysis and approximately 25% survived to 366 days. The hazard ratios for those patients with family history of chronic kidney disease was 0.45; 95% CI 0.23-0.90 and for those with urinary symptoms was 0.59; 95% CI 0.35-0.99. Model generated imply hi(t)= –5.1499 exp{–0.7850Family His. Of CKDi –0.5353Urinary symptomi}. The age of the patient was also found to be statistically significant when separately investigated.
Conclusion: Family history of chronic kidney disease and urinary symptoms were found to influence the survival of patients on haemodialysis. Early identification of at-risk family and prompt treatment of urinary symptoms is advocated.
Keywords
Chronic Kidney Diseases, Dialysis, Survival, Cox Proportional Hazard model, Weibull model
Chronic Kidney Diseases articles, Dialysis articles, Survival articles, Cox Proportional Hazard model articles, Weibull model articles
Chronic Kidney Diseases articles Chronic Kidney Diseases Research articles Chronic Kidney Diseases review articles Chronic Kidney Diseases PubMed articles Chronic Kidney Diseases PubMed Central articles Chronic Kidney Diseases 2023 articles Chronic Kidney Diseases 2024 articles Chronic Kidney Diseases Scopus articles Chronic Kidney Diseases impact factor journals Chronic Kidney Diseases Scopus journals Chronic Kidney Diseases PubMed journals Chronic Kidney Diseases medical journals Chronic Kidney Diseases free journals Chronic Kidney Diseases best journals Chronic Kidney Diseases top journals Chronic Kidney Diseases free medical journals Chronic Kidney Diseases famous journals Chronic Kidney Diseases Google Scholar indexed journals Dialysis articles Dialysis Research articles Dialysis review articles Dialysis PubMed articles Dialysis PubMed Central articles Dialysis 2023 articles Dialysis 2024 articles Dialysis Scopus articles Dialysis impact factor journals Dialysis Scopus journals Dialysis PubMed journals Dialysis medical journals Dialysis free journals Dialysis best journals Dialysis top journals Dialysis free medical journals Dialysis famous journals Dialysis Google Scholar indexed journals Survival articles Survival Research articles Survival review articles Survival PubMed articles Survival PubMed Central articles Survival 2023 articles Survival 2024 articles Survival Scopus articles Survival impact factor journals Survival Scopus journals Survival PubMed journals Survival medical journals Survival free journals Survival best journals Survival top journals Survival free medical journals Survival famous journals Survival Google Scholar indexed journals Cox Proportional Hazard model articles Cox Proportional Hazard model Research articles Cox Proportional Hazard model review articles Cox Proportional Hazard model PubMed articles Cox Proportional Hazard model PubMed Central articles Cox Proportional Hazard model 2023 articles Cox Proportional Hazard model 2024 articles Cox Proportional Hazard model Scopus articles Cox Proportional Hazard model impact factor journals Cox Proportional Hazard model Scopus journals Cox Proportional Hazard model PubMed journals Cox Proportional Hazard model medical journals Cox Proportional Hazard model free journals Cox Proportional Hazard model best journals Cox Proportional Hazard model top journals Cox Proportional Hazard model free medical journals Cox Proportional Hazard model famous journals Cox Proportional Hazard model Google Scholar indexed journals Weibull model articles Weibull model Research articles Weibull model review articles Weibull model PubMed articles Weibull model PubMed Central articles Weibull model 2023 articles Weibull model 2024 articles Weibull model Scopus articles Weibull model impact factor journals Weibull model Scopus journals Weibull model PubMed journals Weibull model medical journals Weibull model free journals Weibull model best journals Weibull model top journals Weibull model free medical journals Weibull model famous journals Weibull model Google Scholar indexed journals blood pressure articles blood pressure Research articles blood pressure review articles blood pressure PubMed articles blood pressure PubMed Central articles blood pressure 2023 articles blood pressure 2024 articles blood pressure Scopus articles blood pressure impact factor journals blood pressure Scopus journals blood pressure PubMed journals blood pressure medical journals blood pressure free journals blood pressure best journals blood pressure top journals blood pressure free medical journals blood pressure famous journals blood pressure Google Scholar indexed journals haemodialysis articles haemodialysis Research articles haemodialysis review articles haemodialysis PubMed articles haemodialysis PubMed Central articles haemodialysis 2023 articles haemodialysis 2024 articles haemodialysis Scopus articles haemodialysis impact factor journals haemodialysis Scopus journals haemodialysis PubMed journals haemodialysis medical journals haemodialysis free journals haemodialysis best journals haemodialysis top journals haemodialysis free medical journals haemodialysis famous journals haemodialysis Google Scholar indexed journals Mortality articles Mortality Research articles Mortality review articles Mortality PubMed articles Mortality PubMed Central articles Mortality 2023 articles Mortality 2024 articles Mortality Scopus articles Mortality impact factor journals Mortality Scopus journals Mortality PubMed journals Mortality medical journals Mortality free journals Mortality best journals Mortality top journals Mortality free medical journals Mortality famous journals Mortality Google Scholar indexed journals end stage renal disease articles end stage renal disease Research articles end stage renal disease review articles end stage renal disease PubMed articles end stage renal disease PubMed Central articles end stage renal disease 2023 articles end stage renal disease 2024 articles end stage renal disease Scopus articles end stage renal disease impact factor journals end stage renal disease Scopus journals end stage renal disease PubMed journals end stage renal disease medical journals end stage renal disease free journals end stage renal disease best journals end stage renal disease top journals end stage renal disease free medical journals end stage renal disease famous journals end stage renal disease Google Scholar indexed journals Urinary articles Urinary Research articles Urinary review articles Urinary PubMed articles Urinary PubMed Central articles Urinary 2023 articles Urinary 2024 articles Urinary Scopus articles Urinary impact factor journals Urinary Scopus journals Urinary PubMed journals Urinary medical journals Urinary free journals Urinary best journals Urinary top journals Urinary free medical journals Urinary famous journals Urinary Google Scholar indexed journals
Article Details
1. Introduction
The kidney is an important organ of the body which maintaining internal balance among other functions. When the kidney failed, the function is impaired with consequent retention of waste products of metabolism. Globally, the prevalence of renal disease is at a pandemic rate and has become a public concern. Report has shown that the prevalence of CKD is currently more than 10% in the general population and reaches 14% or more in high-risk subpopulations [1, 2]. There are currently over 1.4 million patients receiving renal replacement therapy (RRT) worldwide [3]. Haemodialysis, a form of RRT is beneficial to individuals with kidney failure. This treatment modality helps to remove waste and excess water from the blood. However, haemodialysis cannot completely reverse the function of the kidney. Mortality is substantially elevated in patients with chronic kidney disease (CKD) on dialysis [4, 5]. A Swedish population-based study compared the mortality in CKD stages 4 and 5 patients on renal replacement therapy. The authors found that during 6553 person-years, 766 patients with CKD stages 4 and 5 died (deaths/100 person-years 12, 95% CI 11 to 13) compared with 924 deaths during 3680 person-years in haemodialysis (25, 95% CI 23 to 27). In the same study, Neovius et al reported that against matched general population controls, the mortality HR was 3.6 (95% CI 3.2 to 4.0) for CKD and 12.6 (95% CI 10.8 to 14.6) for haemodialysis [6].
Survival of patient who cannot afford adequate treatment such as regular dialysis or renal transplant is unimaginable poor due to several complications that is associated with the disease [4, 5]. The financial burden of end stage renal disease (ESRD) is prohibitive. In our country, CKD patients are battling with poverty, inadequate health facilities, lack of subsidy for medical treatment among other challenges with a gloomy outlook. Survival analysis looks at how long it takes for an event to happen. The event outcome may either be positive or negative such as recovery or failure respectively. Analysis of survival data needs special consideration compared to other data because of the censored nature of the data as it does not follow a normal distribution. The survival analysis employed in this study is designed to handle time-to-event data, considering censored cases. These are cases where the event of interest has not occurred yet at the end of the study or before lost to follow-up or it has occurred due to some other causes. The degree of censoring can affect the reliability of the results. We investigated the survival experience and factors that may contribute to longevity of chronic kidney disease patient on haemodialysis in our dialysis centre. We determined the 4-week, 12-week, 26-week and 52-week survival probabilities and the best model while considering the effect of factors such as age, sex, blood pressure, family history of CKD, diabetes and urinary symptoms on the survival of the patient. Literature is scanty on the study of survival chance of dialysis patients in Nigeria. This study therefore, provided a scholarly impetus to improve the study of survival chance of dialysis patients in our practice.
2. Methods and Materials
2.1 Study design
This study was a five-year (2010-2014) retrospective review of patients with CKD who received treatment at the haemodialysis unit of Ekiti State University Teaching hospital, Ado Ekiti (EKSUTH), Nigeria. The analyzed data were extracted from the patient’s clinical and haemodialysis record. Patients with incomplete data were excluded from the study. The following information was extracted: date of first dialysis session, age of the patient, sex of the patient, blood pressure (BP) of the patient at commencement of dialysis, urinary symptoms, history of diabetes mellitus, family history of CKD, last date of dialysis session, status of patient after last dialysis and patient time. Ethical approval was obtained from the ethical committee of the institution.
2.2 Definition of terms
2.2.1 Date of first dialysis session: This is the date the patient commenced dialysis. This is the treatment starting point for the patient.
2.2.2 Age of the patient: This is the patient age at first dialysis. Age was categorized into two groups <60 and >/=60 using 60 years as cut off point. This was used in Cox proportional and Weibull analysis to determine whether age is a predictor of survival time.
2.2.3 Blood pressure (BP) of the patient at the commencement of dialysis: Blood pressure classification according to Eight Joint National Committee [7].
2.2.4 Last date of dialysis session: This is the date which the last dialysis was done for the patient. This is the treatment endpoint for the patient.
2.2.5 Status of the patient after last the dialysis: This is the treatment outcome of the patient after the last dialysis session. The status may be a failure (death) or censor.
2.2.6 Patient time: This is the time a patient spends in the study from the time of first to the last dialysis.
2.3 Data management and data analysis
Data analysis was performed with STATA version 12. Bivariate analysis was conducted using nonparametric technique; Kaplan-Meier Product Limit [8] Method was employed in estimation of the survival functions and hazard rates of the survival data. Also, both Log-Rank Test and Wilcoxon (Breslow) test was used for the comparison of survival functions. Cox’s proportional hazard regression (Multivariate analysis) was initially used to model the relationship between the survival time and explanatory variables such as age, sex, hypertension, diabetes, family history and urinary symptoms. The data used in this study follows Weibull distribution which enabled the fitting of the probability distribution of real-life time data of dialysis patients. To be able to choose the better model between Cox proportional and Weibull, Akaike Information Criterion (AIC) was employed. Model that has AIC estimate with the smallest value was considered to be more precise. Charts and tables were used to present the results.
3. Result
3.1 Sociodemographic distribution of the patients
One hundred and seven patients were the participants. Male constitute a higher proportion, 74 (69.2%). The mean age of the patients was 51 ± 15.4 years with a range of 21-89 years. The largest number of patients falls between age group 50-59 years (22.3%). Most of the patients (43.9%) were secondary school holders and petty trader (43.9%) constituted the highest proportion of their occupations (Table 1). The highest number of patient (46.7%) were treated in year 2013. Out of 107 patients, 40 (37.4%) were censored as alive up till or after the study time or were lost to follow up, 67 (62.6%) patients were reported to have died as a result of CKD.
|
Variables |
Count |
Percentage (%) |
|
Age Group (Years) |
||
|
20-29 |
8 |
7.8 |
|
30-39 |
22 |
20.4 |
|
40-49 |
20 |
18.7 |
|
50-59 |
24 |
22.3 |
|
60-69 |
20 |
18.7 |
|
70 and above |
13 |
12.1 |
|
All (21-89) |
107 |
100.0 |
|
Sex |
||
|
Male |
74 |
69.2 |
|
Female |
33 |
30.8 |
|
Occupation |
||
|
Civil Servant |
16 |
15.0 |
|
Petty Trader |
47 |
43.9 |
|
Farmer |
17 |
15.9 |
|
Artisan |
9 |
8.4 |
|
Others |
18 |
16.8 |
|
Education |
||
|
None |
10 |
9.3 |
|
Primary |
11 |
10.3 |
|
Secondary |
47 |
43.9 |
|
Tertiary |
39 |
36.4 |
Table 1: Social Demographic Characteristics of chronic kidney disease patients.
3.2 Overall survivorship experience
In the overall cumulative survival, 66.3% of all the patients survived their 90th days after starting dialysis and approximately 25% survived to 366 days (Table 2. Figure 1). The median survival time was 182 days.
|
Time (days) |
Start total |
[S(t)] |
SE[S(t)] |
95% CI |
|
|
5 |
107 |
0.9907 |
0.0093 |
0.9355 |
0.9987 |
|
15 |
99 |
0.9438 |
0.0223 |
0.8792 |
0.9744 |
|
21 |
90 |
0.8663 |
0.0333 |
0.7845 |
0.9186 |
|
31 |
79 |
0.826 |
0.0374 |
0.7379 |
0.8867 |
|
60 |
71 |
0.7518 |
0.0433 |
0.6547 |
0.8252 |
|
90 |
56 |
0.6632 |
0.0482 |
0.5593 |
0.748 |
|
183 |
34 |
0.4824 |
0.0544 |
0.3727 |
0.5836 |
|
206 |
25 |
0.4239 |
0.0551 |
0.3152 |
0.5284 |
|
216 |
20 |
0.3524 |
0.0562 |
0.2447 |
0.4617 |
|
222 |
19 |
0.3338 |
0.0562 |
0.2271 |
0.444 |
|
275 |
15 |
0.2712 |
0.0561 |
0.1686 |
0.3844 |
|
360 |
12 |
0.2486 |
0.0558 |
0.1482 |
0.3626 |
|
366 |
11 |
0.2486 |
0.0558 |
0.1482 |
0.3626 |
|
367 |
10 |
0.2238 |
0.0555 |
0.126 |
0.3389 |
|
397 |
9 |
0.1741 |
0.0531 |
0.0853 |
0.2889 |
|
399 |
7 |
0.1492 |
0.051 |
0.0668 |
0.2625 |
|
428 |
6 |
0.1492 |
0.051 |
0.0668 |
0.2625 |
|
457 |
5 |
0.1193 |
0.0488 |
0.0451 |
0.2326 |
|
463 |
4 |
0.0895 |
0.0448 |
0.0267 |
0.2004 |
|
491 |
3 |
0.0597 |
0.0385 |
0.0121 |
0.1654 |
|
517 |
2 |
0.0597 |
0.0385 |
0.0121 |
0.1654 |
|
611 |
1 |
0.0597 |
0.0385 |
0.0121 |
0.1654 |
Table 2: Kaplan Meier Estimate of survivorship for all patients since first dialysis.
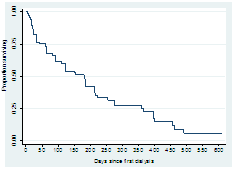
Figure 1: Survival curves of all patients since first dialysis.
3.3 Kaplan-Meier rate of survival according to selected characteristics and log rank or Wilcoxon test
3.3.1 Sex: There was no statistically significant association between sex and mortality. Log rank (Mantel-Cox): Chi-square=0.06; P-value=0.80. Median survival time was estimated for male and female to be 182 and 152 days respectively as shown in Figure 2.
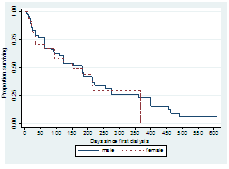
Figure 2: Survival Curves for male and female patients.
3.3.2 Age group: Figure 3 showed that survival (earlier survival difference) varies among the two age groups. Younger age group had a better survival experience. There was a statistically significant association between age group and mortality. Wilcoxon (Breslow): Chi-square=6.67; P-value=0.01.
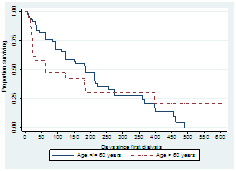
Figure 3: Survival curves for age group ≤ 60 years versus age group ?60 years.
3.3.3 Diabetes status: Median survival time for CKD patients with no diabetes is 183 days while patients with diabetes is 93 days (Table 4 and Figure 4). The relationship between the two diabetes status group was not statistically significant (Long-rank: Chi-square is 0.06, P-value=0.80) as shown in Table 4.
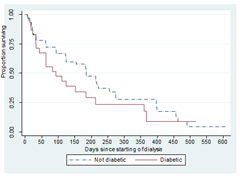
Figure 4: Survival curves by diabetes status.
3.3.4 Family history of chronic kidney diseases (CKD): The median survival time for patient with or without family history of CKD was 124 and 367 days respectively (Log rank statistic 6.78, p-value=0.009). Survival is better if there is no family history of CKD Figure 5.
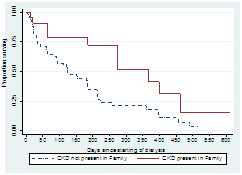
Figure 5: Survival curves by family history of chronic kidney disease patients.
3.3.5 Urinary symptoms: We found a statistically significant relationship among patient with and without urinary symptoms a median survival time of 83 and 213 days. (Long-rank: Chi-square is 5.79, P-value=0. 016) Figure 6.
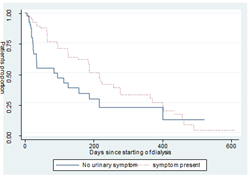
Figure 6: Survival curves for patient with or without urinary symptoms.
3.3.6 Blood pressure at commencement of dialysis: There was no statistically significant relationship in the median survival time among patient with normal BP, 1st stage hypertension and 2nd stage hypertension, Log rank (Mantel-Cox): Chi-square=0.836; P-value=0.80 Figure 7.
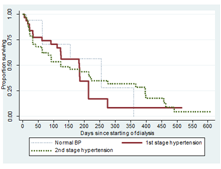
Figure 7: Survival curves of patients in different stages of blood pressure.
3.4 Justification that collected CKD data follows Weibull distribution
A straight (approximately) line in the plot of Log {-log S(t)} versus Log (t) indicates a Weibull models. In Figure 8 Gender and Age covariates showed linearity hence, Weibull model was considered appropriate for the data used for the study.
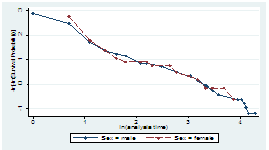
Figure 8: Justification that collected CKD data follows Weibull model.
3.5 Cox proportional hazard and weibull models
The consideration of only three variables here was as a result of their statistical significances among the initial six covariates. Under the Cox Proportional Hazard model (Table 3) therefore, only Family history of CKD is statistically significant among others (Age group and urinary symptoms). Computation from Weibull model (Table 4) on the other hands, confirmed family history of CKD and urinary symptom to be statistically significant.
|
Explanatory Variables |
Hazard Ratio |
S.E. |
Z |
P-values |
95% CI |
|
|
Age group |
1.146831 |
0.328604 |
0.48 |
0.633 |
0.654038 |
2.010926 |
|
F.history of CKD |
0.450776 |
0.157754 |
–2.28 |
0.023 |
0.227025 |
0.895051 |
|
Urinary symptoms |
0.62633 |
0.169884 |
–1.72 |
0.085 |
0.368066 |
1.065814 |
|
_cons |
- |
- |
- |
- |
- |
- |
Log likelihood= –246.18
Table 3: Cox proportional Hazard model.
|
Explanatory Variables |
Hazard Ratio |
S.E. |
Z |
P-values |
95% CI |
|
|
Age group |
1.181071 |
0.331647 |
0.59 |
0.553 |
0.681172 |
2.047836 |
|
Family history of CKD |
0.456093 |
0.158785 |
–2.25 |
0.024 |
0.230523 |
0.902385 |
|
Urinary symptoms |
0.585528 |
0.157515 |
–1.99 |
0.047 |
0.345592 |
0.992046 |
|
_cons |
0.005763 |
0.003747 |
–7.93 |
0 |
0.001612 |
0.02061 |
Log likelihood= –133.811
Table 4: Weibull model.
3.6 Comparison of the result form the Cox and the Weibull model
The comparison of both the Cox proportional hazard and the Weibull model as shown in table 5. gives different results. However, the Weibull model is supported by Akaike Information Criterion. Hence, computation of hazard ratio from Weibull model implies 0.45; 95% CI 0.23-0.90 for family history of CKD history, taking the patient with no family history of CKD as the reference category. Also, for urinary symptom, hazard ratio from Weibull model imply 0.59; 95% CI 0.35-0.99 for patients with urinary symptoms, taking patients with no urinary symptoms as the reference category.
|
Covariate |
Cox Model |
Weibull Model |
|
|
HR (95% Conf. Interval) |
HR (95% Conf. Interval) |
||
|
Age Class |
≤ 60 years (ref.) |
1.15 [0.65-2.01] |
1.18 [0.68-2.05] |
|
>60 years |
|||
|
Family history of CKD |
Not present (ref.) |
0.45 [0.23-0.90] |
0.46 [0.23-0.90] |
|
Present |
|||
|
Urinary symptoms |
No urinary symptoms (ref.) |
0.63 [0.37-1.07] |
0.59 [0.35-0.99] |
|
Akaike Information Criterion |
496.36 |
267.62 |
|
Table 5: Comparison of the result form the Cox and the Weibull model.
3.7 Model fitting
Family history of CKD and urinary symptoms are the only covariates compared to others that influence survival chance in this study. The hazard ratios of the two covariates are supportive. Therefore, better model using computations in table 6 imply; hi(t) = –5.1499 exp {– 0.7850Family His. Of CKDi – 0.5353Urinary symptomsi}
|
Covariate |
HR |
β |
|
Urinary status |
0.5855 |
–0.5352891 |
|
Family his. Of CKD |
0.4561 |
–0.7850432 |
|
Constant |
0.0058 |
–5.14989736 |
Table 6: Model fitting.
4. Discussion
Chronic kidney disease is common among young adults between 20 to 50 years in sub-Saharan Africa [9]. In this study, we found that the overall median survival time was 182 days. However, the overall cumulative survival was 66.3% for all the patients that survived their 90th days from starting of dialysis and approximately 25% survived to 366 days. This is higher than the 90th days reported in an Ethiopia study (61.5%) [10] and Ghana (45%) [11]. Van-Djik et al. in their analysis of renal data collected from the national renal registries of six European countries found that the 2-, 5- and 10-year patient survival was 67, 35 and 11% respectively among their dialysis patients. This figure is much higher than what we obtained in this study probably because Europe is a developed country with better healthcare system compared to Nigeria [12]. Individuals with a family history of kidney disease appear to be at higher risk of developing kidney disease. The presence of genes coding for susceptibility factors for the development or progression of kidney disease in general, as well as genes coding for specific kidney diseases such as nephropathic cystinosis, autosomal dominant polycystic kidney disease [2]. A family history of kidney disease is a risk factor for CKD. A high number of ESRD patients have an affected first-degree relative. This association has been found to be much stronger in African Americans than whites [13, 14]. Song et al. screened incident dialysis patients over 8 years period in the United States, nearly 23% of incident dialysis patients had close relatives with ESRD [15]. McClellan et al. [16] reported a significantly increased risk for ESRD associated with a positive family history of chronic kidney disease even after adjusting for confounders such as age, sex, race, diabetes, hypertension, and socio-economic status (adjusted hazard ratio of 1.93; 95% confidence interval, 1.22-3.07). In this study, patients with family history of CKD are 0.46 times as likely to be at the risk of death compared to those without family history. Similarly, Patients with urinary symptoms at starting dialysis are 0.59 times as likely to be at the risk of death compared to those without urinary symptoms similar to other reports [17, 18].
The presence of co-morbidity such as diabetes mellitus, high blood pressure and other covariates such as sex and age group which did not reach statistically significant were presumed not to have any influence on the survival chances of CKD patients on dialysis except age group when separately investigated. Reindl-Schwaighofer et al. [19] retrospectively analyzed the Austrian Dialysis and Transplant Registry and identified 8,622 patients who started maintenance hemodialysis after the age of 65 years between 2002 and 2009. The data set was compared with a cohort of 174 patients aged over 65 Years with CKD Stage 5 who progressed to an ESRD and were managed conservatively. Their result suggested that the benefit of dialysis treatment decreases with age when the data was further subjected to time to event analysis. The influences of family history of CKD and urinary symptoms in the patient may be supportive. Awareness of chronic kidney disease among members of family will enable other members to practice preventive measures as they are now better informed. Such an individual is expected to present early and comply with medical treatment. It is also likely that patient with urinary symptoms is likely to present or referred to the nephrologist early. The data used for this study followed Weibull regression model, being the one with the least AIC value when compared with the Cox model which indicates the best model for registered chronic disease patients in our institution. Cox regression model with highest AIC was considered to be comparatively less efficient. This result agreed with the results obtained by Hui et al. [20] where comparison of Cox and Weibull model was done using gastric cancer data.
5. Conclusion
This study provided information on the survival experience among a fairly large population of patients treated with dialysis at haemodialysis centre of Ekiti State University Teaching Hospital patient. Out of the entire explanatory variables investigated for their influence on survival chances during dialysis, only family history of chronic kidney disease and urinary symptoms were statistically significant among variables considered. Though the age-group of patients at the start of dialysis was also statistically significant when separately investigated. This study hence recommends appropriate attention to be paid to those with family history of CKD and urinary symptoms at the start of dialysis in addition to other treatment strategies.
References
- Eckardt KU, Coresh J, Devuyst O, et al. Evolving importance of kidney disease: From subspecialty to global health burden. The Lancet 382 (2013): 158-169.
- Levey AS, Coresh J, Bolton K, et al. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. American Journal of Kidney Diseases 39 (2002).
- Kazancioglu R. Risk factors for chronic kidney disease: an update. Kidney international supplements. 3 (2013): 368-371.
- Valdez-Ortiz R, Navarro-Reynoso F, Olvera-Soto MG, et al. Mortality in Patients with Chronic Renal Disease without Health Insurance in Mexico: Opportunities for a National Renal Health Policy. Kidney International Reports (2018).
- Tonelli M, Wiebe N, Culleton B, et al. Chronic Kidney Disease and Mortality Risk: A Systematic Review. Journal of the American Society of Nephrology 17 (2006): 2034-2047.
- Neovius M, Jacobson SH, Eriksson JK, et al. Mortality in chronic kidney disease and renal replacement therapy: A population-based cohort study. BMJ Open 4 (2014): 004251.
- James PA, Oparil S, Carter BL, et al. 2014 evidence-based guideline for the management of high blood pressure in adults: report from the panel members appointed to the Eighth Joint National Committee (JNC 8) Jama 311 (2014): 507-520.
- Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. Journal of the American statistical association 53 (1958): 457-481.
- Naicker S. End-stage renal disease in Sub-Saharan Africa. Kidney International Supplements 3 (2013): 161-163.
- Shibiru T, Gudina EK, Habte B, et al. Survival patterns of patients on maintenance hemodialysis for end stage renal disease in Ethiopia: summary of 91 cases. BMC Nephrology 14 (2013): 127.
- Eghan BA, Amoako-Atta K, Kankam CA, et al. Survival pattern of hemodialysis patients in Kumasi, Ghana: a summary of forty patients initiated on hemodialysis at a new hemodialysis unit. Hemodial Int 13 (2009): 467-471.
- Van Dijk PC, Jager KJ, de Charro F, et al. Renal replacement therapy in Europe: The results of a collaborative effort by the ERA-EDTA registry and six national or regional registries. Nephrol Dial Transplant 16 (2001): 1120-1129.
- Freedman BI, Parekh RS, Kao WL. Genetic basis of nondiabetic end-stage renal disease. Paper presented at: Seminars in nephrology (2010).
- Johnson DW, Atai E, Chan M, et al. KHA-CARI Guideline: early chronic kidney disease: Detection, prevention and management. Nephrology 18 (2013): 340-350.
- Song EY, McClellan WM, McClellan A, et al. Effect of community characteristics on familial clustering of end-stage renal disease. American journal of nephrology 30 (2009): 499-504.
- McClellan WM, Warnock DG, Judd S, et al. Association of family history of ESRD, prevalent albuminuria, and reduced GFR with incident ESRD. American Journal of Kidney Diseases 59 (2012): 25-31.
- Boulware LE, Troll MU, Jaar BG, et al. Identification and referral of patients with progressive CKD: A national study. American journal of kidney diseases 48 (2006): 192-204.
- DuBose TD, Behrens MT, Berns A, et al. The patient-centered medical home and nephrology. Journal of the American Society of Nephrology 20 (2009): 681-682.
- Reindl-Schwaighofer R, Kainz A, Kammer M, et al. Survival analysis of conservative vs. dialysis treatment of elderly patients with CKD stage 5. PloS one 12 (2017): 0181345.
- Zhu HP, Xia X, Chuan HY, et al. Application of Weibull model for survival of patients with gastric cancer. BMC gastroenterology 11 (2011): 1.


 Impact Factor: * 2.3
Impact Factor: * 2.3 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 