The Effects of Synthesized Semicarbazone Copper Complex on Blood Pressure in Normotensive and L-NAME Induced Hypertensive Rats
Article Information
Keaton A Logan1*, Helen Asemota1,2#, Chukwuemeka Nwokocha3, Mark A Lawrence4, Rory Thompson5, Magdalene Nwokocha5, Mohammed Bakir4#
1Department of Basic Medical Sciences (Biochemistry Section), University of the West Indies, Mona Campus, Kingston, Jamaica
2Biotechnology Centre University of the West Indies, Mona Campus, Kingston, Jamaica
3Department of Basic Medical Sciences (Physiology Section), University of the West Indies, Mona Campus, Kingston, Jamaica
4Department of Chemistry University of the West Indies, Mona Campus, Kingston, Jamaica
5Department of Pathology University of the West Indies, Mona Campus, Kingston, Jamaica
#Principal Investigators
*Corresponding Author: Keaton Logan, Department of Basic Medical Sciences, The University of the West Indies, Mona Campus, Kingston, Jamaica
Received: 06 May 2020; Accepted: 16 May 2020; Published: 20 June 2020
Citation:
Keaton A Logan, Helen Asemota, Chukwuemeka Nwokocha, Mark A Lawrence, Rory Thompson, Magdalene Nwokocha, Mohammed Bakir. The Effects of Synthesized Semicarbazone Copper Complex on Blood Pressure in Normotensive and L-NAME Induced Hypertensive Rats. Journal of Biotechnology and Biomedicine 3 (2020): 67-77.
View / Download Pdf Share at FacebookAbstract
Hypertension has been for decades a major contributor to overall mortality and morbidity worldwide. This study was aimed towards evaluating changes in blood pressure in normotensive and L-NAME hypertensive induced rats subjected to treatment with a synthesized semicarbazone copper complex (CSCZ)**. CSCZ was synthesized from the reaction between di-2-pyridylketone semicarbazone (dpksc) and CuCl2.2H2O in refluxing ethanol and characterized from its spectroscopic and elemental analyses. The infrared and electronic absorption spectra are in accord with the proposed formulation of the complex. Animals were rendered hypertensive by oral administrations of Nω-nitro-L-arginine methyl ester (L-NAME) daily (dose 40 mg/kg body weight) for 5 weeks. Oral daily dosages (20 mg/kg body weight) of CSCZ was administered to the animals. A significant decrease in systolic blood pressure (SBP) at 132.91 ± 7.62 was observed in the L-NAME/CSCZ treated group (LCZ) when compared to the L-NAME group at 161.00 ± 5.34 while no significant changes were observed in heart rate (HR) and pulse pressure (PP). Treatment with CSCZ also seemingly normalised and prevented the development of L-NAME induced hypertension in the rats. A significant decrease in SBP and PP was also seen in the normotensive group treated with CSCZ at 91 ± 2.11 and 21.67 ± 1.43 respectively when compared to the control group at 130.63 ± 2.98 and 44.10 ± 5.15 respectively. These findings reveal the blood pressure lowering effects of CSCZ in normotensive rats and its ability to prevent L-NAME induced hypertension.
Keywords
L-NAME, Hypertensive, Hypotension, Copper, Dpksc, Normotensive, Semicarbazone, Complex
L-NAME articles L-NAME Research articles L-NAME review articles L-NAME PubMed articles L-NAME PubMed Central articles L-NAME 2023 articles L-NAME 2024 articles L-NAME Scopus articles L-NAME impact factor journals L-NAME Scopus journals L-NAME PubMed journals L-NAME medical journals L-NAME free journals L-NAME best journals L-NAME top journals L-NAME free medical journals L-NAME famous journals L-NAME Google Scholar indexed journals Hypertensive, Hypotension articles Hypertensive, Hypotension Research articles Hypertensive, Hypotension review articles Hypertensive, Hypotension PubMed articles Hypertensive, Hypotension PubMed Central articles Hypertensive, Hypotension 2023 articles Hypertensive, Hypotension 2024 articles Hypertensive, Hypotension Scopus articles Hypertensive, Hypotension impact factor journals Hypertensive, Hypotension Scopus journals Hypertensive, Hypotension PubMed journals Hypertensive, Hypotension medical journals Hypertensive, Hypotension free journals Hypertensive, Hypotension best journals Hypertensive, Hypotension top journals Hypertensive, Hypotension free medical journals Hypertensive, Hypotension famous journals Hypertensive, Hypotension Google Scholar indexed journals Copper articles Copper Research articles Copper review articles Copper PubMed articles Copper PubMed Central articles Copper 2023 articles Copper 2024 articles Copper Scopus articles Copper impact factor journals Copper Scopus journals Copper PubMed journals Copper medical journals Copper free journals Copper best journals Copper top journals Copper free medical journals Copper famous journals Copper Google Scholar indexed journals Dpksc articles Dpksc Research articles Dpksc review articles Dpksc PubMed articles Dpksc PubMed Central articles Dpksc 2023 articles Dpksc 2024 articles Dpksc Scopus articles Dpksc impact factor journals Dpksc Scopus journals Dpksc PubMed journals Dpksc medical journals Dpksc free journals Dpksc best journals Dpksc top journals Dpksc free medical journals Dpksc famous journals Dpksc Google Scholar indexed journals Normotensive articles Normotensive Research articles Normotensive review articles Normotensive PubMed articles Normotensive PubMed Central articles Normotensive 2023 articles Normotensive 2024 articles Normotensive Scopus articles Normotensive impact factor journals Normotensive Scopus journals Normotensive PubMed journals Normotensive medical journals Normotensive free journals Normotensive best journals Normotensive top journals Normotensive free medical journals Normotensive famous journals Normotensive Google Scholar indexed journals Semicarbazone articles Semicarbazone Research articles Semicarbazone review articles Semicarbazone PubMed articles Semicarbazone PubMed Central articles Semicarbazone 2023 articles Semicarbazone 2024 articles Semicarbazone Scopus articles Semicarbazone impact factor journals Semicarbazone Scopus journals Semicarbazone PubMed journals Semicarbazone medical journals Semicarbazone free journals Semicarbazone best journals Semicarbazone top journals Semicarbazone free medical journals Semicarbazone famous journals Semicarbazone Google Scholar indexed journals Complex articles Complex Research articles Complex review articles Complex PubMed articles Complex PubMed Central articles Complex 2023 articles Complex 2024 articles Complex Scopus articles Complex impact factor journals Complex Scopus journals Complex PubMed journals Complex medical journals Complex free journals Complex best journals Complex top journals Complex free medical journals Complex famous journals Complex Google Scholar indexed journals Blood Pressure articles Blood Pressure Research articles Blood Pressure review articles Blood Pressure PubMed articles Blood Pressure PubMed Central articles Blood Pressure 2023 articles Blood Pressure 2024 articles Blood Pressure Scopus articles Blood Pressure impact factor journals Blood Pressure Scopus journals Blood Pressure PubMed journals Blood Pressure medical journals Blood Pressure free journals Blood Pressure best journals Blood Pressure top journals Blood Pressure free medical journals Blood Pressure famous journals Blood Pressure Google Scholar indexed journals nervousness articles nervousness Research articles nervousness review articles nervousness PubMed articles nervousness PubMed Central articles nervousness 2023 articles nervousness 2024 articles nervousness Scopus articles nervousness impact factor journals nervousness Scopus journals nervousness PubMed journals nervousness medical journals nervousness free journals nervousness best journals nervousness top journals nervousness free medical journals nervousness famous journals nervousness Google Scholar indexed journals Animals articles Animals Research articles Animals review articles Animals PubMed articles Animals PubMed Central articles Animals 2023 articles Animals 2024 articles Animals Scopus articles Animals impact factor journals Animals Scopus journals Animals PubMed journals Animals medical journals Animals free journals Animals best journals Animals top journals Animals free medical journals Animals famous journals Animals Google Scholar indexed journals
Article Details
1. Introduction
Hypertension, one of the essential risk factors for coronary illness and stroke has become a great social and economic burden in both developing and non-developed countries [1]. Blood pressure can be more difficult to control in high risk patients as they usually require more frequent combination of antihypertensive drugs with other therapies, such as lipid lowering treatment (statins) [2]. While pharmacological therapies have been proven to reduce blood pressure in hypertensive patients, numerous destructive symptoms are related with them such as: coughs, diarrhoea or constipation, dizziness or light-headedness, erection problems, nervousness, lethargy, drowsiness or a lack of energy, headaches, nausea or vomiting.
Compounds used in medicine are often analogues of naturally occurring molecules; however, issues concerning drug potency and side effects still remains a concern. Several clinically active antihypertensive drugs share common structural features [3]: aromatic moieties with a core containing chelating regions of nitrogen and carboxylic groups as seen in ACE I for example. Metal complexes with ligands containing Nitrogen (N), Oxygen (O) or Sulphur (S) donors have shown to be effective as potential drugs [4]. Semicarbazones (SCs) are the condensation products of semicarbazide with aldehyde or ketone these having the formula R2C N–NH–(CO)–NH2. These compounds have versatile structural features and can exist in two tautomeric forms, keto/amido and enol/imido [5, 6]. SCs are known to possess effective O and N donor groups and have been shown to possess potent antihypertensive effects with less side effects compared to known antihypertensive drugs on the market. They also bare similarities to known antihypertensive drugs such as clonidine, guanabenz and guanoxabenz [7]. Therefore, these compounds could provide a strong basis for the synthesis of hypotensive drugs. There are however very few reports to date on the synthesis of potent hypotensive SC derivatives.
Literature records the numerous applications of SCs in light of their biological activities. Studies have shown that 4-substituted semicarbazone of mono and 2, 6-dichlorobenzaldehydes possess potent antihypertensive activity [7]. Literature also records its hypolipidemic activity [8, 9]. Other reported activities of SCs and their complexes includes: anticancer [10], analgesic [11] and as antimicrobial agents [12, 13]. The biological properties of SCs are related to metal ion coordination and importantly the metal complex can be more active than the free ligands [14, 15]. Some side effects may also decrease upon complexation and the complexes can exhibit bioactivities which are not shown by the free ligand [14]. The presence of the carboxyl group (C=O) on SCs also adds great lipophilicity to the compound and the SCs mode of action involve binding to a metal in vivo or its metal complexes can act as the activator for the ligand itself [15].
The present work explores a copper salt complex of the ligand di-2-pyridylketone semicarbazone (dpksc) due to its excellent chelating ability. Copper was chosen due to its importance in maintaining the integrity of blood vessels; deficiencies of copper in humans have shown to be a causative factor leading to increased blood pressure [16, 17]. Copper has also been implicated as a possible hypotensive agent [18]. While a considerable amount of work has been reported on metal complexes of dpksc no work has been done to assess the properties of copper complexes of dpksc on blood pressure. This study was therefore initiated to evaluate the effects of CSCZ in normotensive and L-NAME experimental hypertensive models in Sprague Dawley rats.
2. Materials and Methods
2.1 Chemicals
All chemicals and solvents used were obtained from commercial sources (di-2-pyridyl ketone from Aldrich Chemical, semicarbazide hydrochloride from Carlo Erba) and used as received. The di-2-pyridylketone semicarbazone (SC) was synthesized following a standard literature procedure [19]. All other chemicals and solvents used were obtained from commercial sources and required no further purification before use: methanol, hexane, ethyl ether, acetone, hydrochloric acid (HCL), sodium hydroxide (NaOH), dry acetonitrile, chloroform, dimethyl formamide (DMF), copper II chloride and Nω-nitro-L-arginine methyl ester (L-NAME).
2.2 Physical methods
Elemental Analysis was performed using the Perkin-Elmer Series II CHNS/O Analyzer, FT-IR spectra were recorded using a Burker-Tensor 37 FT-IR spectrometer with OPUS 6.5 software (Neat, ATR) and UV-VIS absorption spectra were recorded using the Hewlett Packard 8453 UV-VIS diode array spectrophotometer.
2.3 Preparation of [Cu(k3-N,N,O-dpksc)Cl2] (CSCZ)
A mixture of CuCl2.2H2O (256 mg, 1.50 mmol), dpksc (400 mg, 1.70 mmol) and ethanol was refluxed for 3 h. the resulting reaction mixture was allowed to cool to room temperature and the resulting precipitate was filtered off, washed with hexanes and diethyl ether and dried; yield 499 mg (89%), m.p. 238ºC. Anal. Calc. (%) for (C12H11Cl2CuN5O), C 38.36; H, 2.95; N 18.54. Found C 38.45; H 2.51; N 18.19. Selected I.R. (Neat ATR, n/cm-1): 3392 and 3247 n(NH2), 3185 n(NH), 1658 n(C O). UV-vis/nm (e ± 5% / M-1cm-1) DMF: 310 (13,240), 404 (900).
2.4 Animals
Adult male Sprague-Dawley rats (250-300 g) (n = 20) were procured from the University of the West Indies, Mona (UWI MONA) animal house and hosted in diurnal lighting condition (12 h/12 h) with a temperature of 25 ± 1°C. Animals had free access to food (Standard Chow Pellet) and water ad libitum. The experimental protocol was approved by the University of the West Indies, Mona Animal Ethics Committee and maintained in accordance to the rules and regulations for animal use in research (Ethical approval number: AN 10, 15/16).
2.5 Induction of hypertensionHypertension was induced in the animals by treatment with L-NAME orally (40 mg/kg body weight) for 5 weeks as seen in Nguelefack-Mbuyo et al. [20]. Group 1 (control) contained animals who received no CSCZ, only regular rat chow and drinking water. Group 2 (CSCZ) contained normotensive animals treated with CSCZ only (20 mg/kg body weight) administered as an aqueous solution. Group 3 (L-NAME) contained rats treated with L-NAME (40 mg/kg body weight) while Group 4 (LCZ) had rats treated with both L-NAME (40 mg/kg body weight) and CSCZ (20 mg/kg body weight) concomitantly. All animals were treated for a period of 5 weeks.
2.6 Blood pressure
The animals were acclimatized for 1 week prior to start of the experiment and the blood pressure readings were obtained using the tail cuff method (CODA) from Kent Scientific Corporation, USA). The mean arterial pressure (MAP) was calculated using the formula:
MAP = P diastole + 1/3 (P systole – P diastole).
2.7 Parameters
At the end of 5 weeks blood serum samples were analysed using the Beckman Coulter Counter to ascertain a complete blood count to assess the following haematological parameters: White blood cell tests (WBC), red blood cell tests (RBC), and platelets tests (PLT) along with their specific features.
2.8 Histopathological examinations
Histopathological analysis of the cardiac tissues of the Sprague Dawley rats were done using a Nikon Eclipse Ci research microscope (Nikon Instruments Inc., Americas). Micro-measurements were done via integrated mechanical stage with graduated locator margins and built in slide holder as well as X-Y translator knobs. The tissues were harvested and immediately submerged in 10% neutral buffered formalin for preservation. They were subsequently processed, embedded in wax, and serial sectioned to a thickness of four microns, then stained with haematoxylin and eosin (H&E) stain. Full-thickness transverse sections from the heart, immediately subjacent to the atrioventricular valves, were analysed and evaluated for oedema, mononuclear cell infiltration, fibrosis, interstitial haemorrhage and myocyte degeneration. The number of foci exhibiting degenerative features was recorded and the maximum diameter of the degenerated area was measured using the stage micrometre. Longitudinal and/or transverse sections of the large calibre abdominal blood vessels, i.e. aorta and caudal vena cava, were analysed and evaluated for evidence of endothelial injury, degeneration of the intima or elastic lamina, atherosclerotic change, or any features of adventitial cellular injury. The liver was analysed and evaluated for fibrosis, cholestasis, steatosis and inflammatory parameters. The Ishak systems were employed to ascribe a stage of fibrosis and to also grade the degree of inflammatory changes [21]. Cholestasis was scored using the scoring system proffered by Dixon & Crawford [22] and steatosis was graded using the Kleiner-Brunt scores [23]. In addition to these established parameters semi-objective parameters, subjective evaluation was performed for sinusoidal vascular congestion and the degree of nuclear chromatin density.
2.9 Statistical Analysis
Statistical analysis was performed using GraphPad Prism version 6 (Graph Pad Software, San Diego, CA, USA). A two way ANOVA was used followed by a Tukey’s multiple comparison test. Data obtained from the experiments are expressed as mean ± S.E.M. Values of p ≤ 0.05 were considered significant.
3. Results
3.1 Characterization of CSCZ
CSCZ used in this report was prepared from the reaction between CuCl2.2H2O with dpksc in refluxing ethanol. This reaction is similar to those reported previously for a variety of metal compounds containing k3-N,N,O-dpksc [24]. The isolated product was characterized from its infrared and electronic absorption spectra and the results of its elemental analysis. The infrared spectrum shows peaks in the n(N-H) and n(C=O) along with other peaks consistent with k3-N,N,O-dpksc coordination of the ligand, pointing to keto coordination of the ligand to Cu-ion [5]. The electronic absorption spectrum shows a highly intense peak at 310 nm similar to those reported for metal compounds containing k3-N,N,O-dpksc [24].
3.2 Effect of L-NAME on blood pressure
The animals were treated with L-NAME over a period of 5 weeks to ensure induction of hypertension. A significant increase in the blood pressure was observed over the aforementioned period when compared to the control group (Figure 1). This significant increase indicates the manifestation of hypertension in the animals.
3.3 Effect of CSCZ on L-NAME hypertension
The concomitant use of CSCZ and L-NAME in the LCZ treatment group showed a significant decrease in systolic blood pressure (SBP) and mean arterial pressure (MAP) when compared to the L-NAME group. Subsequently, this decrease in SBP and MAP of the LCZ group falls within a range comparative to that of the control group, signifying a potential normalisation of BP cause by CSCZ treatment. No significant changes in diastolic blood pressure (DBP) and pulse pressure (PP) were observed in the LCZ treatment groups (Table 1).
3.4 Effect of CSCZ on normal rats (normotensive group)
CSCZ treatment in normotensive animals produced a significant decrease in SBP and MAP when compared to the control group. No significant decrease was observed for DBP, however a significant decrease in PP was observed in the normotensive group treated with CSCZ when compared to the control group (Table 1).
3.5 Effect of L-NAME and CSCZ on blood parameters
A blood count was carried out to assess the effects of L-NAME and CSCZ treatment on various blood parameters (Table 2). No significant changes were observed among the hypertensive group and the treated group.
3.6 Effect of CSCZ and L-NAME on histopathology of organs
3.6.1 Myometrial Alternations: There were no observable changes in the normotensive control group (Figure 2A). There was also no significant myometrial hypertrophy, which is what is expected in long standing hypertension, instead there were areas of myocardial infarction identified in the L-NAME (Figure 2B) and CSCZ group (Figure 2C). In the L-NAME group the myocardial damage is multifocal, has a maximum dimension of 8.4 mm, and is accompanied by marked infiltrate of chronic inflammatory cells, in particular lymphocytes and macrophages. This is not present in the CSCZ group despite the focus of fibrosis. Chronicity of injury in the L-NAME group is further evidenced by cardiac myocyte atrophy coupled with hydropic cytoplasmic changes, which again is not appreciated in the CSCZ group.
3.6.2 Large calibre vessel changes: The large calibre vessel showed no abnormalities in the control normotensive groups (Figure 2D) The LCZ group demonstrated a lymphocytic infiltrate in the adventitia around the large calibre blood vessels (Figure 2E). The aorta showed no changes in the intima, media or external layer. The adventitia was composed primarily of brown fat. No inflammation was appreciated.
3.6.3 Hepatic alterations: Stained images of the liver for the control normotensive group did not show any signs of abnormalities (Figure 2F). The CSCZ group, however demonstrated mild sinusoidal congestion which may be indicative of poor cardiac function and early right-sided heart failure (Figure 2G). There was no fibrosis, or changes to the density of nuclear chromatin seen. Fat deposition within hepatocytes (steatosis) was not appreciated neither was there any cholestasis which would be indicative of bile duct obstruction and significant of ongoing hepatocyte damage.
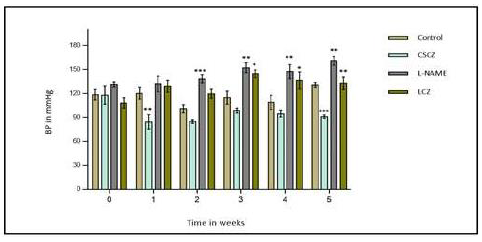
Figure 1: Changes in systolic blood pressure of rats treated with L-NAME (40 mg/kg) given alone or concomitantly with CSCZ (20 mg/kg) over a 5-week period. Each point represents the mean values of blood pressure ± S.E.M. Asterisks indicate significant difference from the normal control rats and also L-NAME in the case of the LCZ group at p<0.05 (*), p<0.01 (**) and p<0.001 (***) respectively.
|
mmHg |
Control |
CSCZ |
L-NAME |
LCZ |
|
SBP |
130.63 ± 2.98 |
91 ± 2.11*** |
161.00 ± 5.34** |
132.91 ± 7.62** |
|
DBP |
86.54 ± 4.72 |
69.33 ± 1.87* |
106.00 ± 6.69* |
83.00 ± 6.03* |
|
MAP |
101.24 ± 3.46 |
76.55 ± 1.84* |
124.92 ± 5.96* |
99.64 ± 5.98* |
|
Pulse Pressure |
44.10 ± 5.15 |
21.67 ± 1.43* |
54.12 ± 4.17 |
49.91 ± 5.94 |
|
Heart Rate |
246.22 ± 22.63 |
197.67 ± 10.18 |
218.13 ± 16.20 |
170.00 ± 13.48 |
The mean values of blood pressure ± S.E.M. Asterisks indicate significant difference from the normal control rats and also L-NAME in the case of the LCZ group at p<0.05 (*), p<0.01 (**) and p<0.001 (***) respectively.
Table 1: SBP, DBP, MAP, PP and HR expressed as mean ± SEM at the end of 5 weeks. *p<0.05, **p<0.01, n = 5 per group.
|
Parameters |
Control |
CSCZ |
L-NAME |
LCZ |
|
WBC |
9.45 ± 1.23 |
7.73 ± 1.28 |
10.30 ± 0.71 |
7.60 ± 0.17 |
|
LYM |
5.75 ± 0.62 |
5.33 ± 0.88 |
6.73 ± 0.69 |
5.47 ± 0.24 |
|
MID |
1.32 ± 0.27 |
1.17 ± 0.33 |
1.30 ± 0.14 |
1.20 ± 0.12 |
|
GRA |
2.40 ± 0.47 |
1.27 ± 0.26 |
2.28 ± 0.26 |
0.97 ± 0.07 |
|
RBC |
6.85 ± 0.16 |
6.40 ± 0.14 |
6.08 ± 0.20 |
6.40 ± 0.37 |
|
HGB |
14.80 ± 0.33 |
14.07 ± 0.23 |
13.35 ± 0.22 |
14.43 ± 0.91 |
|
HCT |
39.95 ± 0.79 |
36.93 ± 1.03 |
35.73 ± 0.60 |
38.73 ± 2.72 |
|
MCV |
58.38 ± 0.39 |
57.70 ± 0.59 |
58.88 ± 1.25 |
60.50 ± 0.71 |
|
MCH |
21.65 ± 0.22 |
21.97 ± 0.24 |
22.28 ± 0.56 |
22.53 ± 0.18 |
|
MCHC |
37.05 ± 0.38 |
38.10 ± 0.47 |
37.83 ± 0.20 |
37.30 ± 0.35 |
|
RDW |
15.03 ± 0.32 |
15.27 ± 0.35 |
16.90 ± 0.99 |
15.30 ± 0.35 |
|
PLT |
732.00 ± 56.11 |
855.00 ± 34.56 |
752.00 ± 50.47 |
874.00 ± 41.80 |
|
MPV |
6.60 ± 0.16 |
7.13 ± 0.03 |
6.93 ± 0.53 |
6.83 ± 0.15 |
Values are presented as mean ± SEM; p-value < 0.05 was considered significant and obtained using ANOVA. WBC-white blood cell count; LYM-lymphocytes; MID-combined value of other white blood cells; GRA-granulocyte; RBC-red blood cell count; HGB-haemoglobin; HCT-haematocrit; MCV-mean corpuscular volume; MCH-mean corpuscular haemoglobin; MCHC-mean corpuscular haemoglobin concentration; RDW-RBC distribution width; PLT-platelet; MPV-mean platelet volume
Table 2: Mean values of blood count parameters after 5 weeks of treatment with L-NAME and CSCZ.
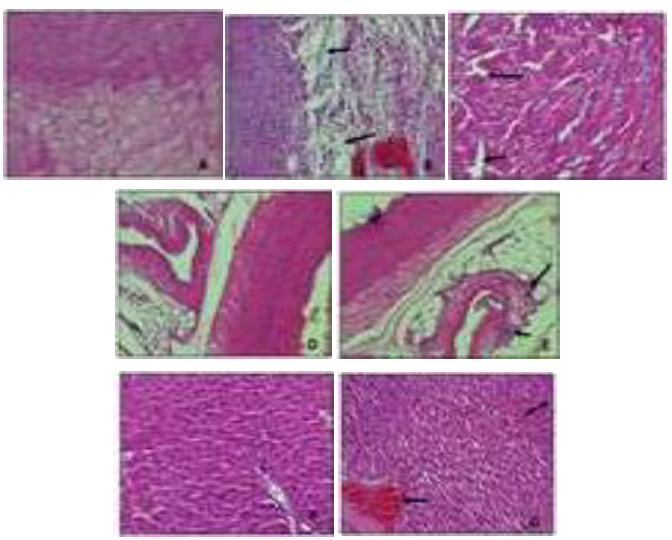
Figure 2: Sections of heart, large calibre vessel and liver were stained with H and E stain (20×), (A) Section of control normotensive heart showing normal structure and architecture; (B) L-NAME treated group showing signs of multifocal myocardial infarction with infiltration of inflammatory cells; (C) CSCZ treated group showed signs of mild myocardial infarction; (D) Section of control normotensive group showing normal structure and architecture of large calibre vessel; (E) LCZ group showing lymphocytic infiltrate of blood vessels; (F) Section of control normotensive group showing normal structure and architecture of the liver; (G) CSCZ group showing signs of mild sinusoidal congestion in the liver.
4. Discussion
In this study, an orally administered dose of 20 mg/kg body weight (CSCZ) resulted in a significant reduction of blood pressure in both L-NAME induced hypertensive and normotensive animals. Rats chronically treated with L-NAME showed a significant increase in blood pressure (SBP) over the treatment period in comparison to the control rats (Figure 1). Chronic L-NAME administration has been shown to cause persistent hypertension in rats [25]. This method of hypertension induction is due to the inhibitory effects of L-NAME on Nitric oxide synthase (NOS) which is responsible for the biosynthesis of NO - relaxes vascular smooth muscle [26]. Upon inhibition the concentration of NO decreases. This decrease subsequently results in excessive contraction of arteries hence increase in blood pressure due to increased pressure acting on the walls of these blood vessels. Studies have also shown that moderate inhibition of nitric oxide synthase by L-NAME does not seem to influence the cardiac sympathetic tone which is consistent with results obtained in this study as no significant changes in heart rate (HR) was noted for the L-NAME treated group [27]. However, the histopathological data indicates areas multifocal myocardial damage (myocardial infarction) (Figure 2). This is typical in chronic L-NAME administration and is also consistent with what may occur in clinical hypertensive heart disease [28, 29].
The concomitant administration of CSCZ to rats treated with L-NAME not only prevented the development of hypertension but seemingly normalised the blood pressure (Table 1). This result confirms the hypotensive effects of semicarbazones as explored in Warren et al. [7]. The mechanism of blood pressure reduction and normalisation is not known but may be as a result of several contributing factors. L-NAME hypertension has been known for its NOS inhibitory activity; however this method of hypertension induction has also been implicated in the production of free oxygen species [30, 31]. The production of these free oxygen species could have possibly been counteracted by CSCZ due to the potent antioxidant properties of semicarbazones [32], consequently preventing the development of hypertension. The presence of chelated copper ions in the CSCZ complex may have also contributed to the observed blood pressure reduction as explored in Loyke [18]. Chelated copper ions have also been reported to increase the hypotensive activity of complexes [33, 34].
Treatment of the normotensive group with CSCZ showed a significant decrease in SBP, MAP and PP (Table 1), indicating the blood pressure lowing effects of CSCZ in rats not induced with hypertension. A reduction in SBP in this study could suggest a low pressure in arteries during myocardial contractions which might be as a result of a reduction in sympathetic activity as no significant increases were observed in HR. Additionally, the reduction in arterial pressure might also be due to an increase in the pliability of arteries as a significant decrease in PP was observed. While no significant changes in blood parameters were observed (Table 2), there were signs of mild myocardial damage and mild sinusoidal congestion in the liver (Figure 2). This might suggest that while CSCZ is able to reduce blood pressure, the administered dosage may cause mild damage to both the sub-endocardial region of the myocardium and the hepatic tissue.
5. Conclusion
This study highlighted the ability of the synthesized semicarbazone complex (CSCZ) to significantly reduce blood pressure in normotensive animals. The ability of CSCZ to prevent hypertension in the L-NAME induced hypertensive rat model was also observed. Further comprehensive dose-dependent, biochemical and toxicity study could help in understanding the effects of CSCZ on blood pressure, its mechanism of action and its effect on the organs.
Acknowledgements
We would like to thank The Office of Graduate Studies and Research at the University of The West Indies, Mona Campus for providing funding through a grant.
Conflict of Interest
The authors declare that they have no conflicts of interest with the contents of this article.
References
- Chockalingam A, Campbell NR, Fodor JG. Worldwide epidemic of hypertension. Canadian Journal of Cardiology 22 (2006): 553-555.
- Council ES, Redon J, Narkiewicz K, et al. 2013 ESH/ESC Guidelines for the management of arterial hypertension. European Heart Journal 34 (2013): 2159-2219.
- Brown, Hall AS. Renin-angiotensin system modulation: the weight of evidence. American Journal of Hypertension 18 (2005): 127-133.
- Chandra S, Raizada S, Verma R. Synthesis, characterisation and biological activities of copper (II) complexes with semicarbazones and thiosemicarbazones. Journal of Chemical and Pharmaceutical Research 4 (2012): 1612-1618.
- Reena T, Kurup MP. Copper (II) complexes derived from di-2-pyridyl ketone-N 4-phenyl-3-semicarbazone: Synthesis and spectral studies. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy 76 (2010): 322-327.
- Reena T, Prathapachandra Kurup M. Spectral studies and crystal structures of some transition metal complexes of N4-substituted semicarbazones. Cochin University of Science & Technology (2011).
- Warren JD, Woodward DL, Hargreaves RT. 4-Substituted semicarbazones of mono- and dichlorobenzaldehydes as antihypertensive agents. J Med Chem 20 (1977): 1520-1521.
- Chapman JM, DeLucy P, Wong OT, et al. Structure activity relationships of imido N-alkyl semicarbazones, thiosemicarbazones and acethydrazones as hypolipidemic agents in rodents. Lipids 25 (1990): 391-397.
- Wong Hall IH, Chapman JM. The Hypolipidemic Activity of l-N-3-Methylphthalimido-butan-3-semicarbazone in Rodents. Pharmaceutical Research 6 (1989): 230-234.
- Jevtovic V, Ivkovic S, Kaisarevic S, et al. Anticancer activity of new copper (II) complexes incorporating a pyridoxal-semicarbazone ligand. Contemporary Materials (2010): 133-137.
- Singhal M, Paul A. Synthesis and analgesic activity of methylphenyl semicarbazone derivatives. Int J ChemTech Res 3 (2011): 1485-1489.
- Chandra S, Tyagi M. Ni (II), Pd (II) and Pt (II) complexes with ligand containing thiosemicarbazone and semicarbazone moiety: synthesis, characterization and biological investigation. Journal of the Serbian Chemical Society 73 (2008): 727-734.
- Sapna K, Kumar Sharma N, Kohli S. Synthesis, Characterization and Antimicrobial Studies of Copper (II) Complexes of Semicarbazone and Thiosemicarbazone of m-Hydroxy Benzaldehyde and p-Hydroxy Benzaldehyde. Oriental Journal of Chemistry 28 (2012): 969.
- Beraldo H, Gambinob D. The wide pharmacological versatility of semicarbazones, thiosemicarbazones and their metal complexes. Mini Reviews in Medicinal Chemistry 4 (2004): 31-39.
- Shirode P, Yeole P. Synthesis and Characterization of Mixed Ligand Complexes of Transition Metals with Schiff’s Bases. Chemical Science Transactions 3 (2014): 1186-1192.
- Klevay LM. Cardiovascular disease from copper deficiency-a history. The Journal of Nutrition 130 (2000): 489-492.
- Medeiros DM. Hypertension in the Wistar-Kyoto rat as a result of post-weaning copper restriction. Nutrition Research 7 (1987): 231-235.
- Loyke HF. Copper and zinc in experimental hypertension. Biological Trace Element Research 29 (1991): 45-49.
- Battaglia LP, Ferrari MB, Boggia R. Synthesis and characterization of lead (II)-semicarbazone complexes. Crystal structures of dinitrato di-2-pyridylketone semicarbazone lead (II) and dichloro cyclohexane-1, 2-dione bis (semicarbazone) dimethylformamide lead (II). Inorganica Chimica Acta 215 (1994): 85-90.
- Nguelefack-Mbuyo P, Nguelefack T, Dongmo A, et al. Anti-hypertensive effects of the methanol/methylene chloride stem bark extract of Mammea africana in L-NAME-induced hypertensive rats. Journal of Ethnopharmacology 117 (2008): 446-450.
- Goodman ZD. Grading and staging systems for inflammation and fibrosis in chronic liver diseases. Journal of Hepatology 47 (2007): 598-607.
- Dixon LR, Crawford JM. Early histologic changes in fibrosing cholestatic hepatitis C. Liver Transplantation 13 (2007): 219-226.
- Brunt EM, Tiniakos DG. Histopathology of nonalcoholic fatty liver disease. World Journal of Gastroenterology: WJG 16 (2010): 5286.
- Bakir M, McDermot C, Johnson T. Spectroscopic, and electrochemical studies of [MCl2 (η2-N, N-dpksc)](M= Zn, Cd, Hg and dpksc= di-2-pyridylketone semicarbazone). Journal of Molecular Structure 1040 (2013): 221-225.
- Gardiner SM, Kemp PA, Bennett T, et al. Nitric oxide synthase inhibitors cause sustained, but reversible, hypertension and hindquarters vasoconstriction in Brattleboro rats. European Journal of Pharmacology 213 (1992): 449-451.
- Sharma P, Vyawahare N, Ladhha A. Preclinical screening models for hypertension in rodents: a review. Pharmacologyonline 3 (2010): 458-472.
- Khayyal MT, El-Ghazaly MA, Abdallah DM, et al. Blood pressure lowering effect of an olive leaf extract (Olea europaea) in L-NAME induced hypertension in rats. Arzneimittel Forschung 52 (2002): 797-802.
- Ono Y, Ono H, Matsuoka H, et al. Apoptosis, coronary arterial remodeling, and myocardial infarction after nitric oxide inhibition in SHR. Hypertension 34 (1999): 609-616.
- Vaskonen T, Mervaala E, Krogerus L, et al. Cardiovascular effects of chronic inhibition of nitric oxide synthesis and dietary salt in spontaneously hypertensive rats. Hypertension Research 20 (1997): 183-192.
- Peterson D, Peterson D, Archer S, et al. The non specificity of specific nitric oxide synthase inhibitors. Biochemical and Biophysical Research Communications 187 (1992): 797-801.
- Sozer V, Uzun H, Gelisgen R, et al. The effects of atorvastatin on oxidative stress in L-NAME-treated rats. Scandinavian Journal of Clinical and Laboratory Investigation 73 (2013): 591-597.
- Safavi M, Foroumadi A, Nakhjiri M, et al. Complexes of 2-hydroxyacetophenone semicarbazones: a novel series of superoxide dismutase mimetics. Bioorganic & Medicinal Chemistry Letters 20 (2010): 3070-3073.
- Bontchev PR, Pantcheva IN, Bontchev RP, et al. Copper (II) complexes of the beta-blocker pindolol: properties, structure, biological activity. BioMetals 15 (2002): 79-85.
- Islas MS, Rojo T, Lezama L, et al. Improvement of the antihypertensive capacity of candesartan and trityl candesartan by their SOD mimetic copper (II) complexes. Journal of Inorganic Biochemistry 123 (2013): 23-33.


 Impact Factor: * 5.3
Impact Factor: * 5.3 CiteScore: 2.9
CiteScore: 2.9  Acceptance Rate: 11.01%
Acceptance Rate: 11.01%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 