Association of Socioeconomic Status with Glomerular Diseases at a Tertiary Care Hospital in Bangladesh
Md. Enamul Kabir1*, Afzalul Bashar1, Md. Obaidul Huq1, Muhammed Arshad Ul Azim1, A N M Ehsanul Karim2, Rana Mokarram Hossain3, Md. Omar Faroque3, Ferdous Jahan3, Md. Kabir Hossain3, A. K. M Shahidur Rahman3
1Department of Nephrology, Shaheed Sheikh Abu Naser Specialized Hospital, Khulna, Bangladesh
2Department of Nephrology, Shaheed Ziaur Rahman Medical College, Bogura, Bangladesh
3Department of Nephrology, Bangabandhu Sheikh Mujib Medical University (BSMMU), Dhaka, Bangladesh
*Corresponding Author: Dr. Md. Enamul Kabir, Assistant Professor and Head of the Department, Department of Nephrology, Shaheed Sheikh Abu Naser Specialized Hospital, Khulna, Bangladesh
Received: 12 July 2022; Accepted: 28 July 2022; Published: 28 August 2022
Article Information
Citation:
Kabir ME, Bashar A, Huq MO, Azim MAU, Karim ANME, Hossain RM, Faroque MO, Jahan F, Hossain MK, Rahman AKMS. Association of Socioeconomic Status with Glomerular Diseases at a Tertiary Care Hospital in Bangladesh. Archives of Nephrology and Urology 5 (2022): 57-63.
DOI: 10.26502/anu.2644-2833053
View / Download Pdf Share at FacebookAbstract
Background: Social deprivation is a well-known risk factor for chronic kidney disease (CKD), however its association with glomerular diseases is less well understood. This study was aimed to evaluate the association of Socioeconomic Status (SES) with glomerular diseases among patients attending at a tertiary care hospital in Bangladesh.
Methods: This cross sectional study was conducted at Department of Nephrology, Shaheed Sheikh Abu Naser Specialized Hospital, Khulna, Bangladesh from January 2016 to December 2020. A total of 184 diagnosed patients of glomerulonephritis (GN) were enrolled. Patient’s socioeconomic status (SES) was assessed using Modified BG Prasad socioeconomic classification. Morphological pattern of glomerular diseases were assessed according to the histopathology report. Relationship of glomerular diseases with particular socioeconomic status (SES) was identified.
Results: Among 184 study patients 99 were male and 85 were female, their mean age was 31.18 ± 13.55 years. It was observed that, in proliferative variety; Mesengial Proliferative Glomerulonephtis (Mes PGN) was the leading morphological variant (34.7%) followed by Membranoproliferative Glomerulonephtis [MPGN (16.8%)], Lupus Nephritis [LN (7.1%)], Focal Segmental Proliferative Glomerulonephritis [FSPGN (7.1%)], IgA Nephropathy (6.5%), IgM Nephropathy (4.3%) and Crescentic Glomerulonephtis [Cres GN (2.2%) serially. In the non-proliferative glomerulonephtis; Membranous Nephropathy (MN) was the commonest form (10.9%) followed by Focal Segmental Glomerulosclerosis [FSGS (7.6%)] and Minimal Change Disease [MCD (2.7%)]. All types of GN were significantly prevalent in the lower socioeconomic classes (Class- III, IV, V), while the frequency of Mes PGN and MPGN were significantly higher in Class- IV and Class- V socioeconomic classes.
Conclusion: Lower socioeconomic classes are associate
Keywords
<p>Chronic Kidney Disease (CKD); Glomerular Disease; Glomerulonephtis (GN); Socioeconomic Status (SES)</p>
Article Details
1. Introduction
Socioeconomic Status (SES) is an important contributory factor for different health problems including chronic kidney disease (CKD) and is a powerful predictor of mortality [1,2]. Socioeconomic status (SES) encompasses a number of susceptibilities that may contribute to an individual’s disease risk [3]. It was reported that lower socioeconomic status (SES) is constantly associated with CKD but its potential link to glomerular diseases is less clear [1,4]. Despite the improvement of public health measures, sanitization, and quality of life there is a paradoxical increase in allergic and autoimmune diseases, especially in industrialized countries in the last few years [5]. The hygiene hypothesis was originally proposed to explain the mechanism of marked increase in allergies in Westernized societies [6]. This hypothesis proposed that early and frequent exposure to bacterial and other antigens, which is common in developing nations, leads to a normal Th1 response; however, better public hygiene and fewer infections observed in industrialized nations may lead to persistence of the Th2 phenotype and thereby increase the risk for developing allergies [7]. There is a link between this hygiene hypothesis and geographical variation of morphological variants of glomerular diseases [8].
Glomerulonephritis (GN) literally means “inflammation of the glomeruli” although inflammation is not apparent in all varieties of the disease. Sometimes glomerulopathy is used to mention this entity [9]. Glomerular disease is the commonest cause of end-stage renal disease (ESRD) worldwide [10-12]. It comprises 25-45% cases of ESRD in developing nations, like Bangladesh [11]. While chronic ailments like diabetes and hypertension constitute the major bulk of chronic kidney disease (CKD) [10]. Idiopathic glomerulonephritis (GN) makes a major diagnostic challenge to both nephrologists and pathologists [11-12]. Glomerular disorders constitute one of the major causes of morbidity and mortality [10-12]. It has been suggested that apart from genetic profile, environmental factors and prevalence of infectious diseases, attribute to socioeconomic and demographic differences seen in patterns of glomerulopathy [13-14]. GN epidemiology follows racial and geographical distributions that are affected by a higher overall prevalence of infectious diseases and differences in social and economic statuses [13-14]. Little information exists on the factors leading these underlying changes, and most of the studies have conducted in Western countries [15]. Glomerulonephritis is one of the leading causes of chronic kidney disease (CKD) [10-12]. Timely intervention may limit this number or at least delay this progression which imposes a huge influence on resource limited national economy. In Bangladesh, most of the patients of glomerulonephritis present to the renal physician later and it is often impossible to alter the disease course with treatment. Moreover data is very limited addressing the influence of socioeconomic status (SES) on the pattern of glomerulonephritis. This study was aimed to evaluate the association of socioeconomic status with glomerular diseases among Bangladeshi population.
2. Methods
This cross sectional study was conducted at the Department of Nephrology, Shaheed Sheikh Abu Naser Specialized Hospital, Khulna, Bangladesh from January 2016 to June 2020. A total of 184 diagnosed patients of glomerulonephritis were selected purposively following selection criteria. Hospitalized patients age ≥12 years, having proteinuria >1 gm/24 hours and hemturia (>3 rbc/hpf) were included. Diabetic patients and patients with proteinuria who meet KDIGO [Kidney Disease Improving Global Outcomes] criteria for chronic kidney disease (CKD), patients having uncontrolled hypertension (HTN) and patients with bleeding diathesis were excluded from the study. Height, weight, blood pressure (BP) of the study patients was measured and body mass index (BMI) was calculated accordingly. Relevant investigations including- urine routine microscopic examination (R/M/E), complete blood count (CBC), serum creatinine, serum electrolytes, 24 hours urinary total protein (24h UTP), random blood sugar (RBS), serum albumin, fasting lipid profile [Total Cholesterol (TC), High Density Lipoprotein- Cholesterol (HDL- C), Low Density Lipoprotein- Cholesterol (LDL- C) and Triglyceride (TG)], serum calcium (S. Ca), serum phosphate (S. PO4), serological markers [like- antinuclear antibody (ANA), anti-ds DNA antibody, serum levels of complement components (C3 & C4) and antineutrophil cytoplasmic antibodies (ANCA)], HBsAg, Anti-HCV, Anti-HIV and ultrasonogram (USG) of kidneys of all study patients were done.
2.1 Study procedure: After admission all patients were evaluated by careful history taking, relevant clinical examination and routine investigations. Patient having suspected glomerulonephritis (GN) were further investigated to rule out primary from secondary GN. Those who had contraindication to kidney biopsy were excluded. After taking informed written consent kidney biopsy was done in each study patient following standard procedure. Then 2 core of renal tissue that obtained during renal biopsy were taken; one for light microscopy and another for direct immunofluorescence (DIF). Patient’s socioeconomic status (SES) was assessed using Modified BG Prasad Socioeconomic classification update 2019 [16]. Patient’s socioeconomic class was identified converting the Indian rupees into Bangladeshi taka (BDT). After getting biopsy report all the records were reviewed to correlate the variety of specific glomerular disease with different clinical and laboratory reports. Then relationship of glomerular diseases with particular socioeconomic status (SES) was identified. Thereafter each patient was treated accordingly and was advised for follow up as per schedule.
2.2 Socioeconomic Status (SES): Socioeconomic status (SES) refers to an individual’s position within a hierarchical social structure, which is one of the important determinants of health status. Composite scales are generally used to measure the SES, which has a combination of social and economic variables. The most commonly used scale for measuring socioeconomic status (SES) is modified BG Prasad scale [16]. B G Prasad's classification was based on per capita monthly income and the scale evolved in 1961. The updated values for the per capita monthly income (Rs/month converted to BDT*/month) for Jan 2019 are given here-
|
Socioeconomic classes |
Status |
Per capita income limit (Rupees) |
BDT* limit |
|
I |
Upper class |
7533 and Above |
8759 and Above |
|
II |
Upper-middle class |
3766-7532 |
4379-8758 |
|
III |
Middle class |
2260-3765 |
2628-4378 |
|
IV |
Lower middle class |
1130-2259 |
1313-2627 |
|
V |
Lower class |
1129 and Below |
1312 and Below |
*BDT= Bangladeshi taka
Table 1: Modified BG Prasad’s social classification- update 2019 [16].
2.2 Statistical Analysis: Data were recorded systematically and statistical analysis was performed using windows based software program Statistical Packages for Social Sciences 22 (SPSS- 22). All quantitative data were expressed as mean and standard deviation (SD), while frequency distribution and percentage (%) was used for qualitative data. Simple descriptive statistics was used to analyze the data. Comparison of continuous variables was done by unpaired ‘t’ test. Association between different socioeconomic classes with all varieties of glomerular disease was examined by chi-square test. A p value <0.05 was considered as significant.
3. Results and observation
A total of 184 patients were enrolled to evaluate the association of socioeconomic status (SES) with glomerular diseases among hospitalized patients at a sub-urban region of Bangladesh. Among 184 patients, majority (54%, n=99) were male and 46% (n=85) were female (Figure 1). Baseline demographic profile shows that, mean age of the patients was 31.18 ± 13.55 years. Youngest patient was 12 years old and the oldest patient was a 66 year old male patient. Their mean height was 154.13 ± 10.39 cm, mean weight was 55.32 ± 10.19 kg, mean body mass index (BMI) was 23.48 ± 4.45 kg/m2, mean body surface area (BSA) was 1.55 ± 0.16 m2, mean systolic blood pressure was 132.6 ± 24.98 mmHg, mean diastolic blood pressure was 81.84 ± 11.58 mmHg (Table- 2). It was observed that, the predominant socioeconomic class of study population was Class- IV which belongs to 45% of the study subjects (n=83). The next major class was Class- V including 64 study subjects (35%). While 13% patients was from Class- III group (n=24) and 5% from Class- II (n=10). The minor group was Class- I which includes only 3 of our study patients that covering 2% of the total study subjects (Figure 2).
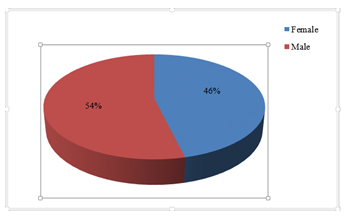
Figure 1: Distribution of the study patients according to gender (n=184).
|
Variables |
Mean ± SD |
Minmum – Maximum |
|
Age (year) |
31.18 ± 13.55 |
12 – 66 |
|
Height (cm) |
154.13 ± 10.39 |
112.5-177.5 |
|
Weight (kg) |
55.32 ± 10.19 |
26-81 |
|
BMI (kg/m2) |
23.48 ± 4.45 |
12.8 – 38.8 |
|
BSA (m2) |
1.55 ± 0.16 |
0.96-1.97 |
|
Systolic BP (mmHg) |
132.6 ± 24.98 |
90- 210 |
|
Diastolic BP (mmHg) |
81.84 ± 11.58 |
50 – 110 |
BMI= Body Mass Index, BSA= Body Surface Area, BP= Blood Pressure
Table 2: Demographic profile of the study patients (n=184).
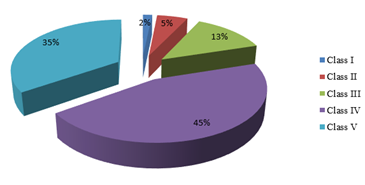
Figure 2: Distribution of the study patients based on socioeconomic status (n=184).
Among the study population baseline laboratory parameters showed that; mean serum creatinine was 1.68 ± 1.27 mg/dl, mean random blood sugar (RBS) was 6.78 ± 2.6 mmol/l, mean haemoglobin, (Hb) level was 11.59 ± 1.79 gm/dl, mean serum albumin was 3.2 ± 0.83 gm/dl, mean serum calcium was 8.84 ± 1.79 mg/dl, mean serum phosphate was 3.62 ± 0.96 mg/dl, mean serum triglyceride was 305.56 ± 226.7 mg/dl, mean serum cholesterol was 213.6 ± 116.1 mg/dl and mean 24 hour urinary total protein (24h UTP) was 4.6 ± 3.5 gm/24h (Table- 3).
|
Variables |
Mean ± SD |
Minmum – Maximum |
|
S.Creatinine (mg/dl) |
1.68 ± 1.27 |
0.6 - 8.3 |
|
RBS (mmol/l) |
6.78 ± 2.6 |
4.0 - 8.8 |
|
Hb (g/dl) |
11.59 ± 1.79 |
10.7 - 16.2 |
|
S. Albumin (g/dl) |
3.2 ± 0.83 |
1.4 - 5.7 |
|
S. Ca (mg/dl) |
8.84 ± 1.39 |
8.6– 10.0 |
|
S. PO4 (mg/dl) |
3.62 ± 0.96 |
3.2-4.3 |
|
S. TG (mg/dl) |
305.56 ± 126.7 |
205.0 - 1027.10 |
|
S. Cholesterol (mg/dl) |
213.6 ± 116.1 |
151.30 - 723.40 |
|
24h UTP (gm/24h) |
4.63 ± 2.52 |
1.38 - 7.20 |
RBS=Random Blood Sugar, Hb= Haemoglobin, S. Ca= Serum Calcium, S. PO4= Serum phosphate, S. TG= Serum Triglyceride, 24h UTP= 24 hours Urinary Total Protein
Table 3: Basic laboratory parameters of the study subjects (N=184).
Data analysis revealed that there was no statistically significant difference (p>0.05) between male and female in different clinical and laboratory parameters like- age, body mass index (BMI), body surface area (BSA), systolic blood pressure (SBP), serum creatinine, random blood sugar (RBS), haemoglobin, serum albumin, serum cholesterol and 24 hours urinary total protein (24h UTP) (Table- 4).
|
Variables |
Male (n=99) |
Female (n=85) |
p-value |
|
Age (years) |
31.68 ± 14.43 |
30.56 ± 12.52 |
0.581 |
|
BMI (kg/m2) |
22.95 ± 4.14 |
24.09 ± 4.74 |
0.084 |
|
BSA (m2) |
1.58 ± 0.168 |
1.48 ± 0.15 |
0.309 |
|
SBP (mmHg) |
132.93 ± 24.17 |
132.35 ± 26.03 |
0.877 |
|
S. Creatinine (mg/dl) |
1.39 ± 1.39 |
1.52 ± 1.09 |
0.093 |
|
RBS (mmol/l) |
6.97 ± 2.36 |
6.57 ± 2.97 |
0.314 |
|
Hb (gm/dl) |
11.83 ± 1.79 |
11.33 ± 1.75 |
0.060 |
|
S. Albumin (gm/dl) |
3.33 ± 0.86 |
3.18 ± 0.81 |
0.248 |
|
S. Cholesterol (mg/dl) |
219.3 ± 121.7 |
206.93 ± 109.6 |
0.469 |
|
24h UTP (gm/24h) |
4.54 ± 2.52 |
4.76 ± 2.56 |
0.668 |
BMI= Body Mass Index, BSA= Body Surface area, SBP= Systolic Blood pressure, RBS=Random Blood Sugar,
Hb= Haemoglobin, 24h UTP= 24 hours Urinary Total Protein
Table 4: Comparison of clinical and laboratory parameters of the study population among different gender (N=184).
Histopathological findings showed that, among the proliferative variety; Mesengial Proliferative Glomerulonephtis (Mes PGN) was the leading morphological variant (n=64, 34.7%). Membranoproliferative Glomerulonephtis (MPGN) came to the second (n=31, 16.8%). While 13(7.1%) patients were in the Lupus Nephritis (LN) group and another 7.1% was in the Focal Segmental Proliferative Glomerulonephritis (FSPGN) group. Thereafter, IgA Nephropathy (n=12, 6.5%), IgM Nephropathy (n=8, 4.3%) and Crescentic Glomerulonephtis (Cres GN) (n=4, 2.2%) serially. In the non-proliferative glomerulonephtis; Membranous Nephropathy (MN) was the commonest form (n=20, 10.9%) followed by Focal Segmental Glomerulosclerosis (FSGS) and Minimal Change Disease (MCD), accounts 7.6% and 2.7% respectively (Table- 5).
|
Variables |
Number (n) |
Percentage (%) |
|
Proliferative Glomerulonephritis (78.7%) |
||
|
Mes PGN |
64 |
34.7 |
|
MPGN |
31 |
16.8 |
|
FSPGN |
13 |
7.1 |
|
LN |
13 |
7.1 |
|
IgA Nephropathy |
12 |
6.5 |
|
IgM Nephropathy |
8 |
4.3 |
|
Cres GN |
4 |
2.2 |
|
Non Proliferative Glomerulonephritis (21.3%) |
||
|
MN |
20 |
10.9 |
|
FSGS |
14 |
7.6 |
|
MCD |
5 |
2.7 |
Mes PGN= Messangial proliferative Glomerulonephritis, MPGN= Membranoproliferative Glomerulonephritis, MN= Membranous Nephropathy, MCD= Minimal Change Disease, LN= Lupus Nephritis, IgM Nephropathy= Immunoglobulin M Nephropathy, IgA Nephropathy= Immunoglobulin A Nephropathy, FSPGN= Focal Segmental Proliferative Glomerulonephritis, FSGS= Focal and Segmental Glomerulosclerosis, Cres GN= Crescentic Glomerulonephritis
Table 5: Distribution of study population according to the morphological pattern of Glomerulonephritis (N=184).
Comparison of different morphological pattern of glomerulonephritis among the five socioeconomic classes showed that all varieties of GN were significantly prevalent in the lower socioeconomic classes (Class- III, IV, V), While the frequency of Mesengial Proliferative Glomerulonephtis (Mes PGN) and Membranoproliferative Glomerulonephtis (MPGN) were significantly higher in Class- IV and Class- V socioeconomic groups. The p value was significant (p<0.005) in all the instances (Table- 6).
|
Varieties of Glomerulonephritis |
Number (n) |
Class- I (n=3) |
Class- II (n=10) |
Class- III (n=24) |
Class- IV (n=83) |
Class- V (n=64) |
p-value |
|
Mes PGN |
64 |
1(1.6) |
1(1.6) |
4(6.3) |
29(45.3) |
29(45.3) |
<0.05 |
|
MPGN |
31 |
0(0.0) |
1(3.2) |
5(16.1) |
16(51.6) |
9(29.0) |
<0.05 |
|
FSPGN |
13 |
0(0.0) |
0(0.0) |
0(0.0) |
7(53.8) |
6(46.2) |
<0.05 |
|
IgA Nephropathy |
12 |
2(16.7) |
0(0.0) |
2(16.7) |
4(33.3) |
4(33.3) |
<0.05 |
|
LN |
13 |
0(0.0) |
0(0.0) |
1(7.7) |
8(30.8) |
4(30.8) |
<0.05 |
|
MN |
20 |
0(0.0) |
5(25.0) |
4(20.0) |
5(30.0) |
6(30.0) |
<0.05 |
|
IgM Nephropathy |
8 |
0(0.0) |
0(0.0) |
3(37.5) |
4(12.5) |
1(12.5) |
<0.05 |
|
FSGS |
14 |
0(0.0) |
2(14.3) |
2(14.3) |
6(28.6) |
4(28.6) |
<0.05 |
|
MCD |
5 |
0(0.0) |
1(20.0) |
2(40.0) |
2(40.0) |
0(0.0) |
<0.05 |
|
Cres GN |
4 |
0(0.0) |
0(0.0) |
1(25.0) |
2(25.0) |
1(25.0) |
<0.05 |
Values within the parenthesis denote corresponding percentage, Mes PGN= Messangial Proliferative Glomerulonephritis, MPGN= Membranoproliferative Glomerulonephritis, MN= Membranous Nephropathy, MCD= Minimal Change Disease, LN= Lupus Nephritis, IgM Nephropathy= Immunoglobulin M Nephropathy, IgA Nephropathy= Immunoglobulin A Nephropathy, FSPGN= Focal Segmental Proliferative Glomerulonephritis, FSGS= Focal and Segmental Glomerulosclerosis, Cres GN= Crescentic Glomerulonephritis
Table 6: Association of different varieties of Glomerulonephritis with all five socioeconomic classes (N=184).
4. Discussion
Socioeconomic status is a contributory factor to the different health problems and is an important indicator of mortality [5]. Moreover, individual’s socioeconomic position encompasses a number of susceptibilities that could contribute to different disease risk [8]. It was reported that lower socioeconomic status has been consistently associated with different chronic disease including chronic kidney disease (CKD) [1-4, 17]. In this background this study was aimed to describe the morphological pattern of primary glomerular diseases and to evaluate this pattern in the light of socioeconomic status of our study population. In this study, out of 184 patients; male sex was slightly predominant (54% versus 46%) which was almost similar to all of the studies depicting male predominance [18-19]. Although Habib et al. from Bangladesh found female predominance [18]. The mean age of the study subjects was 31.18 ± 13.55 years. A related study observed that the mean patient’s age was 31.9 ± 15.9 years which was also similar to this study [19]. Most of the participants of this study were hailing from lower socioeconomic status (80% belongs to Class IV and Class V- lower middle class and lower class respectively) reflecting the overall socioeconomic status in a sub-urban region of the Bangladeshi population. This finding was similar to a previous study where 80.1% of the studied patients were from lower socio-economic classes [20]. In both developing and developed countries, glomerular disease is a common cause of end stage renal disease (ESRD) [10-12]. The histological spectrum of glomerulonephritis (GN) varies widely between decades and nations, reflecting the possible effects of socio- economic, genetic and environmental factors [21]. IgA Nephropathy is the common primary glomerular disease from East Asia, as well as in white Europeans and Americans [5]. In contrast, Focal Segmental Glomerulosclerosis (FSGS) is the most common glomerular disease among African Americans, South Americans and in Middle East [5]. Proliferative glomerular disease was the most prominent renal disease in our biopsy cohort (78.7% versus 21.3%). This finding was supported by all recent studies [10, 18-20, 22]. In this current study Messangial proliferative Glomerulonephritis (Mes PGN) was the commonest primary GN seen. Mes PGN was found as the commonest primary and proliferative GN in several other studies from different area of Bangladesh [23]. Although it was the second most common GN reported by Ahmed et al. (11.50%) [19]. Tabssum et al. conducted a cross sectional study on 67 cases and found that Mes PGN was the second most common primary glomerulonephritis (30.95%) after Membranoproliferative Glomerulonephritis (MPGN) [12]. Another study also showed that Mes PGN is the predominant form of GN in the Southwestern region of Bangladesh (36.48%) [24]. A study done in Singapore on the spectrum and trends of glomerulonephritis for three decades showed that, in the 1st decade most Asians countries had MPGN and Mes PGN as the most common form of primary GN [25]. The similar trend was reported from some Asians countries like China, Japan and Thailand [26-28]. A recent study also found high prevalence of Mes PGN in India [29]. Hurtado et al. proposed that this variation in the morphological pattern of glomerulonephritis between regions can be explained by hygiene hypothesis [8]. Some types of human GN have Th1-predominant immune responses, including Crescentic Glomerulonephritis and MPGN. Other types of GN have predominant Th2 immune response, including Membranous Nephropathy (MN), Minimal Change Disease (MCD), and IgA nephropathy [7}. In developing countries, over stimulation of the immune system with numerous bacterial and other antigens may lead to a Th1 predominance and susceptibility to MPGN and other proliferative types of GN; in contrast, in Westernized nations fewer exposures to infections and better public hygiene may lead to a Th2-dominant phenotype and an increased risk for developing allergies with consequent IgA Nephropathy and MCD [8]. That may be possible explanation for higher prevalence of Mes PGN and MPGN in this study (34.7% and 16.8% respectively) as well as similar other studies from this region [25-28]. IgA Nephropathy is the most common form of primary GN as reported from different part of the globe, accounting for up to 30-40% of all biopsies from Asia, for 20% in Europe and for 10% in North America [5]. Although Asian studies showed variable result regarding the prevalence of IgA Nephropathy [25-29]. In comparison to the other Asian countries IgA Nephropathy is less common in Bangladesh (4.67%-7%) [20]. In this current study IgA Nephropathy constitute only 6.5% of the total GN. This finding was consistent with some other Asian studies reported from India and Pakistan [29-30]. The lower prevalence of IgA Nephropathy in our study may be explained by the benign presentation of this disease. As asymptomatic urinary abnormalities mostly go undetected and varying approaches for using renal biopsy. Another factor is that proteinuria less than 1 gm without having renal failure was not considered for renal biopsy in our biopsy protocol. Indeed, the cases of IgA Nephropathy detected in our patients probably represented the tip of the iceberg. It is likely that the vast majority of these cases go undetected as a result of the current indicative criteria for renal biopsy [12]. Membranous Nephropathy (MN) accounts for 10.9% and Focal Segmental Glomerulosclerosis (FSGS) 7.6% in our study. The finding slightly differs with similar previous studies as reported, MN (7.34%) and FSGS (2.8%) [19], while MN 7.37 % but FSGS was 11.58% [18] in two different studies. The incidence of IgM Nephropathy in our study was 4.3%. Most of the studies did not mention IgM nephropathy as a distinct category. We have only 5 cases of MCD (2.7%). Several studies have shown a decline in the relative frequency of MCD [27-30]. A study from China also reported a very low incidence of MCD [26]. In this study, we compared different morphological pattern of glomerulonephritis among the five socioeconomic classes according to the modified BG Prasad scale [16]. It was found that different types of glomerulonephritis were significantly (p<0.05) more frequent in the lower socioeconomic classes, particularly Mes PGN and MPGN were significantly higher in Class- IV and Class- V socioeconomic groups. This result was supported by a couple of previous study as reported that there is an inverse association between socioeconomic position and the incidence of glomerulonephritis [17, 31]. Glumerular diseases are complex conditions that require specialized care and expensive therapies, which may be more challenging for patients with a low socioeconomic status [17].
5. Conclusion
This study demonstrated that primary glomerulonephritis (GN) are more common in Bangladesh; Mesengial Proliferative Glomerulonephtis (Mes PGN) is the leading variety (accounts for 34.7%) of all morphological variant of glomerular disease followed by Membranoproliferative Glomerulonephtis [MPGN (16.8%)], other varieties are not very prevalent. Most of the study population belongs to Class- IV and Class- V socioeconomic groups (45% and 35% respectively). All varieties of GN are significantly prevalent in the lower socioeconomic classes particularly Mes PGN and MPGN follow relatively higher prevalence in the lower socioeconomic classes.
Limitation of the Study
It was a single center study with a relatively small sample size.
Recommendation
A large-scale multi-centered study should be conducted to see the reliability of the results.
Conflicts of Interest
The authors declare that they have no conflicts of interest regarding the publication of this article.
References
- Vart P, Gansevoort RT, Joosten MM, et al. Socioeconomic disparities in chronic kidney disease: a systematic review and meta-analysis. American journal of preventive medicine 48(2015):580-592.
- Stringhini S, Carmeli C, Jokela M, et al. Socioeconomic status and the 25× 25 risk factors as determinants of premature mortality: a multicohort study and meta-analysis of 1• 7 million men and women. The Lancet 389 (2017):1229-1237.
- Galobardes B, Shaw M, Lawlor DA, et al. Indicators of socioeconomic position (part 1). Journal of Epidemiology & Community Health 60(2006):7-12.
- Fored CM, Ejerblad E, Fryzek JP, et al. Socio-economic status and chronic renal failure: a population-based case-control study in Sweden. Nephrology Dialysis Transplantation 18(2003):82-88.
- Braden GL, Mulhern JG, O'Shea MH, et al. Changing incidence of glomerular diseases in adults. American journal of kidney diseases 35(2000):878-883.
- Maziak W. The hygiene hypothesis and the evolutionary perspective of health. Preventive medicine 35(2002):415-418.
- Holt PG, O'keeffe P, Holt BJ, et al. T-cell “priming” against environmental allergens in human neonates: sequential deletion of food antigen reactivity during infancy with concomitant expansion of responses to ubiquitous inhalant allergens. Pediatric Allergy and Immunology6(1995):85-90.
- Hurtado A, Johnson RJ. Hygiene hypothesis and prevalence of glomerulonephritis. Kidney international 68(2005):S62-7.
- Goddard J, Turner AN, Cumming AD, et al. Kidney and urinary tract disease. colledge NR, Brian RW, Ralston SH, eds. Davidson’s Principles and Practice of Medicine. 21st ed. Edinburgh. (2006):459-519.
- Gupta RD, Mamun AA, Morshed SM, et al. Mon-029 Pattern of Glomerulonephritis in Bangladesh-a Single Center Study at a Tertiary Care Hospital. Kidney International Reports 4(2019):S316.
- Rashid HU. Nephrotic syndrome-evidence based management. Bangladesh Renal Journal 22(2003):1-4.
- Tabassum S, Rahman KM, Mamun KZ, et al. Immunofluorescent microscopic findings in glomerulonephritis. Bangladesh Medical Research Council Bulletin. 23(1997):77-81.
- di Belgiojoso GB, Ferrario F, Landriani N. Virus-related glomerular diseases: histological and clinical aspects. Journal of nephrology 15(2002):469-479.
- Kong D, Wu D, Wang T, et al. Detection of viral antigens in renal tissue of glomerulonephritis patients without serological evidence of hepatitis B virus and hepatitis C virus infection. International journal of infectious diseases. 17(2013):e535-8.
- Chang JH, Kim DK, Kim HW, et al. Changing prevalence of glomerular diseases in Korean adults: a review of 20 years of experience. Nephrology Dialysis Transplantation 24(2009):2406-2410.
- Pandey VK, Aggarwal P, Kakkar R. Modified BG prasad socio-economic classification, update-2019. Indian journal of community health 31(2019).
- Canney M, Induruwage D, Sahota A, et al. Socioeconomic position and incidence of glomerular diseases. Clinical Journal of the American Society of Nephrology 15(2020):367-374.
- Habib MA, Badruddoza SM. Pattern of glomerular diseases among adults in Rajshahi, the Northern Region of Bangladesh. Saudi Journal of Kidney Diseases and Transplantation 23(2012):876.
- Ahmed PI, Zaman SU, Jahan F, et al. Pattern of primary glomerulonephritis in Dhaka medical college hospital, Bangladesh. Bangladesh Journal of Medicine 25(2014):42-46.
- Ahmed ZU, Fakir AH. Clinicopathological analysis of glomerulonephritis. Bangladesh Armed Force Medical Journal 17(1993):51-54.
- Di Belgiojoso GB, Baroni M, Pagliari B, et al. Is membranoproliferative glomerulonephritis really decreasing?. Nephron 40(1985):380-381.
- Das U, Dakshinamurty KV, Prayaga A. Pattern of biopsy-proven renal disease in a single center of south India: 19 years experience. Indian journal of nephrology 21(2011):250.
- Balakrishnan N, John GT, Korula A, et al. Spectrum of biopsy proven renal disease and changing trends at a tropical tertiary care centre 1990-2001. Indian Journal of Nephrology 13(2003):29.
- Azim MA, Rahaman MM, Zahir KM, et al. Changing Pattern of Post-infectious Glomerulonephritis: A Study in South-Western Region of Bangladesh. Medicine Today 32(2020):130-133.
- Woo KT, Chiang GS, Pall A, et al. The changing pattern of glomerulonephritis in Singapore over the past two decades. Clinical nephrology 52(1999):96-102.
- Hou JH, Zhu HX, Zhou ML, et al. Changes in the spectrum of kidney diseases: an analysis of 40,759 biopsy-proven cases from 2003 to 2014 in China. Kidney Diseases 4(2018):10-19.
- Sugiyama H, Yokoyama H, Sato H, et al. Japan Renal Biopsy Registry: the first nationwide, web-based, and prospective registry system of renal biopsies in Japan. Clinical and experimental nephrology 15(2011):493-503.
- Parichatikanond P, Chawanasuntorapoj R, Shayakul C, et al. An analysis of 3,555 cases of renal biopsy in Thailand. J Med Assoc Thai 89(2006):S106-11.
- Narasimhan B, Chacko B, John GT, et al. Characterization of kidney lesions in Indian adults: towards a renal biopsy registry. Journal of nephrology 19(2006):205-210.
- Hashmi AA, Hussain Z, Edhi MM, et al. Insight to changing morphologic patterns of glomerulopathy in adult Pakistani patients: an institutional perspective. BMC Research Notes 9(2016):1-6.
- McQuarrie EP, Mackinnon B, McNeice V, et al. The incidence of biopsy-proven IgA nephropathy is associated with multiple socioeconomic deprivation. Kidney international 85(2014):198-203.


 Impact Factor: * 3.3
Impact Factor: * 3.3 Acceptance Rate: 73.59%
Acceptance Rate: 73.59%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 