Recurrent Nodular Glomerulosclerosis in a Second Kidney Allograft
Nasreen H Mohamed1, Ali Alshaqaq2*, Khaled Hamawi2, Meteb M AlBugami2, Fahad Otaibi2, AbdulNasser AlAbadi2, Khalid Bel’eed-Akkari2
1Department of Pathology and Laboratory Medicine, King Fahad Specialist Hospital, Dammam, Saudi Arabia
2Multiorgan Transplant center, King Fahad Specialist Hospital, Dammam, Saudi Arabia
*Corresponding Author: Dr. Ali Alshaqaq, Multiorgan Transplant Center, King Fahad specialist hospital, building 14, Dammam, Saudi Arabia
Received: 07 July 2021; Accepted: 19 July 2021; Published: 27 July 2021
Article Information
Citation: Nasreen H Mohamed, Ali Alshaqaq, Khaled Hamawi, Meteb M AlBugami, Fahad Otaibi, AbdulNasser AlAbadi, Khalid Bel’eed-Akkari. Recurrent Nodular Glomerulosclerosis in a Second Kidney Allograft. Archives of Nephrology and Urology 4 (2021): 093-100.
DOI: 10.26502/anu.2644-2833040
View / Download Pdf Share at FacebookAbstract
Kidney transplantation is considered the best choice for patients with end stage renal disease. However, recurrence of primary disease, especially glomerular disease represents a major problem. Nodular glomerulosclerosis with its wide differential diagnosis can recur post kidney transplantation and might results in graft failure. Here, we report a unique case of 35 years old male with idiopathic nodular glomerulosclerosis that recurred twice after two successful living donor kidney transplants.
Keywords
<p>Nodular glomerulosclerosis, kidney transplantation, diabetes</p>
Article Details
1. Introduction
Recurrence of glomerular disease after kidney transplantation is the third most common cause of graft loss at 10 years after chronic rejection and death with functioning graft [1, 2]. Many secondary forms of glomerulonephritis (GN) are no longer considered a potential contraindication to renal transplantation; however, the risk of a post-transplant recurrence of GN remains real, but variable. The histologic recurrence is most frequent in lupus nephritis, Henoch-Schonlein nephritis, light-chain amyloidosis, and diabetic nephropathy which often recur after many years [2].
Nodular glomerulosclerosis is a pathological entity first described by Kimmelstiel and Wilson in 1936 [3]. Although initially thought to be pathognomonic for diabetic nephropathy, it has a wide differential diagnosis that includes the primary and secondary forms of chronic membranoproliferative glomerulonephritis, chronic thrombotic microangiopathy, amyloidosis, monoclonal immunoglobulin deposition disease, fibrillary glomerulonephritis, immunotactoid glomerulopathy, fibronectin glomerulopathy, and collagen III glomerulopathy [4, 5]. In 1989, Alpers and Biava described the renal histopathological findings in 5 non-diabetic patients showing nodular mesangial matrix expansion devoid of immune deposits [6], however, the term idiopathic nodular glomerulosclerosis (ING) was first coined by Herzenberg et al. in 1999 [7].
ING is a rare clinico-pathologic entity that is by definition a diagnosis of exclusion. Cases usually presents with nephrotic-range proteinuria, renal insufficiency, and a clinical history of hypertension and obesity. Nevertheless, careful exclusion of subclinical evidence of glucose intolerance or early DM is essential. Histologically, it is characterized by expansion of mesangial matrix with nodule formation, accentuation of glomerular lobularity, and thickening of glomerular basement membranes.
While there have been several reports of patients with post- transplant diabetes mellitus whose graft biopsies showed nodular glomerulosclerosis [8-11], there have been few reports of kidney transplant recipients developing nodular glomerulosclerosis with variable levels of proteinuria in the absence of glucose intolerance or other recognized causes of nodular glomerulosclerosis [12-14]. To the best of our knowledge this is the first case of ING to have recurred twice after two successful living donor kidney transplants, in a recipient who is not overweight, non-diabetic and who has never smoked.
2. Case Presentation
We report the case of a 35 years old hypertensive male whose cause of end stage renal disease (ESRD) was uncertain. He had never smoked. At the age of 22 years he had his first living unrelated kidney transplant in 2006, and was followed up at another facility. Three years later and due to slowly progressive graft dysfunction (serum creatinine 341umol/l and ACR 73 mg/mmol), he had an allograft biopsy. The biopsy revealed 10 viable glomeruli, some with mild expansion of mesangial matrix and nodule formation (Figure 1). The diagnosis was nodular glomerulosclerosis, and the patient was investigated for diabetes which showed average fasting blood glucose if 5.1mmol/l and HbA1c 4.9%.
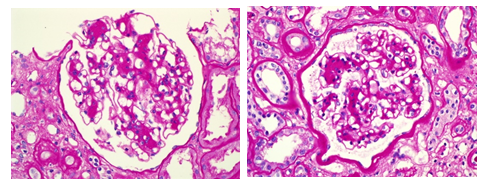
Figure 1: Allograft biopsy showing mesangial matrix expansion and nodule formation.
His graft failed few months later and was he started on haemodialysis before receiving a second, zero HLA mismatched, living related kidney transplant in June 2010. He had an uneventful post-operative course. His serum creatinine remained around 85umol/l on regular maintenance immunosuppression of prednisone, tacrolimus and mycophenolate mofetil.
Two years later, he had acute allograft dysfunction and underwent a renal biopsy. This showed mild tubular injury with 42 viable and within normal limits glomeruli. There was no evidence of nodular glomerulosclerosis. His graft function stayed fairly stable (mean serum creatinine 140umol/l, mean trough tacrolimus level 5.4 mg/dl, and no proteinuria) until three years later when his serum creatinine rose to 206 umol/l. Clinical evaluation at the time revealed BMI of 19.6 Kg/m2 and blood pressure 137/81 mmHg. Urinalysis was negative for blood and protein, and tacrolimus trough level was 6.9 mg/dl. Peripheral white cell count was 2.37x109/l, haemoglobin 8.4g/dl and platelets 93x109/l. CMV and BK virus PCR were negative and ultrasound examination of the graft was unremarkable. He had a renal allograft biopsy which showed 56 viable glomeruli, some with mild to moderate expansion of the mesangial matrix and nodule formation. There was no increase in mesangial cellularity and the glomerular capillaries were patent with no glomerulitis. The glomerular capillary walls were within normal limits with no spikes or double contouring, and interstitial fibrosis was estimated around 50- 60%. Immunofluorescence studies were negative for immunoglobulins, light chains and complements. Staining for C4d and SV40 was negative. The ultrastructural examination showed expansion of mesangial matrix, with mild increase in glomerular basement membrane thickness. Furthermore, there were no immune complex or paraprotein type deposits, and podocyte foot processes were mainly intact. The dose of MMF was reduced to 500 mg twice daily and target tacrolimus trough level kept at 4 mg/dl.
Retrospectively, immunofluorescences were performed on formalin fixed paraffin embedded tissue from the first transplant biopsies, and this was negative for IgG, IgA, kappa, and lambda light chains. There was no tissue reserved for ultrastructural examination. Since his second transplant the patient’s fasting blood glucose ranged between 4.9- 5.3 mmol/l (HbA1c 4.4- 5.2%), and a two hour glucose tolerance test showed glucose of 4.9 mmol/l. ANA speckled titer was 1:160, with C3 of 1.01 (0.91- 2.41 g/l), and C4 0.3 (0.078- 0.52 g/l). Serum and urine protein electrophoresis and immunofixation were negative. Serum free light chains were elevated: kappa 771 mg/l (3.3- 19.4 mg/l), lambda 55 mg/l (5.71- 26.3 mg/l), with kappa/ lambda ratio of 14 (0.26- 1.65). Urine free light chains were elevated (kappa 139 mg/l (1.35- 24.19 mg/l), lambda 16.6 mg/l (range 0.24- 6.66 mg/l), and kappa/ lambda ratio 8.37 (2.04- 10.37). Skeletal survey for myeloma was negative, and bone marrow biopsy showed hypocellular marrow with no evidence of myeloma. Serum and urine electrophoresis, free light chains, and a bone marrow biopsy repeated 4 months later only confirmed the previous findings. Trace proteinuria ensued and his graft function progressively deteriorated until he commenced dialysis a year later.
3. Discussion
We are reporting a case of ING that seemed to have recurred twice after two successful living donor kidney transplants in a hypertensive, non-obese, non-diabetic male who never smoked. Our patient did not have proteinuria until late in the course, and his graft dysfunction seemed to progress to ESRD over a period of 18 months.
Nodular glomerulosclerosis is a histologic pattern with a wide range of differentials, most importantly diabetic nephropathy, monoclonal light chain deposition disease, amyloidosis and chronic thrombotic microangiopathy. ING, therefore is a diagnosis of exclusion. In our case, the histologic features were subtle with mild to moderate expansion of mesangial matrix and occasional nodule formation within glomeruli, suggesting an early development of nodular glomerulosclerosis.
In a recent review, Wu et al found only 72 reported cases after excluding those with impaired glucose tolerance [15]. A biopsy incidence of ING of 0.45% was reported by Markowitz et.al. in a retrospective review of 23 cases of ING, where the mean age was 68 years in patients who were mostly males (78%); smokers (91%); hypertensive (96%); had renal insufficiency (83%) and with proteinuria > 3 g/d in 70% [4]. The literature contains several reported cases of native kidneys with nodular glomerulosclerosis in the absence of known glucose intolerance [16-19], but the definition of glucose intolerance in some of these reports may now be questionable in view of the changing diagnostic criteria of diabetes mellitus [20, 21].
Nodular glomerulosclerosis has been reported in post-kidney transplant patient as a sequel to post-kidney transplant diabetes [22]. Wojciechowski et al. Reported three cases of biopsy proven diabetic nephropathy, developing within 2 years of the diagnosis of post-transplant diabetes mellitus (NODAT), despite excellent glycaemic control (HbA1c ≤ 7%) [23]. In a retrospective study by Prasad et al. the period from developing NODAT to allograft diabetic nephropathy varied between 27-188 months (mean 110 months) [24].
Nodular glomerulosclerosis has previously been reported in non-diabetic deceased donor kidney transplant recipients [12, 13]. Rivera et al. reported a case of nodular glomerulosclerosis in allograft biopsy in 1997, prior to the recognition of ING in 1999 by Herzenberg et al. Although the patient was normotensive with no diabetes or glucose intolerance, the authors did not comment on history of chronic smoking. In contrast to the case by Rivera et.al., where nodular glomerulosclerosis was documented 8 years post-transplant and the patient had nephrotic range proteinuria, our patient did not have proteinuria upon presentation and developed ING within 3 years (first transplant) and 5 years (second transplant). Others reported cases of nodular glomerulosclerosis 21 years post kidney transplantation.
Several other studies also highlighted the prevalence of smoking among patients with ING [4, 14, 25, 26]. Nasr et al. have argued that idiopathic nodular glomerulosclerosis is probably not idiopathic, and pointed to emerging evidence suggesting a distinct clinico-pathological entity of smoking-associated nodular glomerulosclerosis [5]. Proteinuria has been a prominent feature of ING in several case series. In the retrospective report by Li and Verani on the clinic-pathologic features of 15 patients with ING between 1998 and 2007, all 15 had proteinuria (mean 24 hour urinary protein excretion, 5.6 g/24 h), with 11 (73%) having nephrotic-range proteinuria [27]. Similar case series by Wu and colleagues from Shanghai, all 20 patients had proteinuria (mean 2.85 ± 0.33 g/day, and 7 had nephrotic-range proteinuria [15].
Several case reports highlighted the diagnostic difficulty presented by light chains in post-kidney transplant patients [28-30]. Monoclonal light chains may be present in the serum and/or urine only intermittently and at low concentrations, due to rapid degredation and variable rates of production, and some 40% of patients with LCDD do not have a demonstrable lymphoplasmacytic disorder even when followed up for periods of 2–17 years [31, 32]. In our patient, examination for light chain deposits by immunofluorescence and electron microscopy was negative.
Despite the several reported factors associated with ING like obesity, smoking and hypertension, a significant degree of heterogeneity remains between the reported cases. This was highlighted in a recent review by López-Revuelta et al. Where they hypothesized that Diabetic Nephropathy without Diabetes is not an independent histopathological entity per se, but a stage in a rather “special form of atherosclerotic or metabolic glomerulopathy” that can occur with or without diabetes [26]. In addition to recurring twice, several other distinctive features differentiate our case from the characteristics of previous ING patients reported in the literature. Our patient is a post kidney transplant patient who is not overweight, had never smoked, and had no evidence of glucose intolerance and minimal late onset proteinuria.
Conflicts of Interest
The authors declared no potential conflicts with respect to the research, authorship, and /or publication.
Ethical Approval
The research was approved by the institutional review boards of king Fahad specialist hospital.
Authors Contribution
All authors participated in collecting the data, writing and reviewing the manuscript.
Funding
The authors received no financial support for research, authorship, and /or publication.
References
- Briganti EM, Russ GR, McNeil JJ, et al. Risk of Renal Allograft Loss from Recurrent Glomerulonephritis. N Engl J Med 347 (2002): 103-109.
- Ponticelli C, Moroni G, Glassock R. J. Recurrence of Secondary Glomerular Disease after Renal Transplantation. CJASN May 6 (2011): 1214-1221.
- Kimmelstiel P, Wilson C. Intercapillary lesions in the glomeruli of the kidney. Am J Pathol 12 (1936): 83-98.
- Markowitz GS, Lin J, Valeri AM, et al. Idiopathic nodular glomerulosclerosis is a distinct clinicopathologic entity linked to hypertension and smoking. Hum Pathol 33 (2002): 826-835.
- Nasr SH, D’Agati VD. Nodular Glomerulosclerosis in the Nondiabetic Smoker. J Am Soc Nephrol 18 (2007): 2032-2036.
- Alpers CE, Biava CG. Idiopathic lobular glomerulonephritis (nodular mesangial sclerosis): a distinct diagnostic entity. Clin Nephrol 32 (1989): 68-74.
- Herzenberg AM, Holden JK, Singh S, et al. Idiopathic nodular glomerulosclerosis. Am J Kidney Dis 34 (1999): 560-564.
- Gimenez LF, Watson AJ, Burrow CR, et al. De novo diabetic nephropathy with functional impairment in a renal allograft. Am J Nephrol 6 (1986): 378-381.
- Schwarz A, Krause PH, Offremann G, et al. Recurrent and de novo renal disease after kidney transplantation with or without cyclosporine. Am J Kidney Dis 17 (1991): 524-531.
- Sharma UK, Jha V, Gupta KL, et al. De novo diabetic glomerulosclerosis in a renal allograft recipient. Am J Kidney Dis 23 (1994): 597.
- Owda AK, Abdallah AH, Haleem A, et al. De novo diabetes mellitus in kidney allografts: nodular sclerosis and diffuse glomerulosclerosis leading to graft failure. Nephrol. Dial. Transplant 14 (1999): 2004-2007.
- Rivera M, Marce´n R, Mampaso F, et al. Nodular glomerulosclerosis after renal transplantation without diabetes mellitus. Nephrol Dial Transplant 12 (1997): 219-220.
- Abdi R, Chavin K, Nadasdy T. Nodular glomerulosclerosis in a renal allograft of a non-diabetic recipient. Nephrol Dial Transplant 14 (1999): 493-496.
- Suneja M, Khan A, Katz DA, et al. Nodular Glomerulosclerosis in a Kidney Transplant Recipient Who Smokes. AJKD 50 (2007): 830-833.
- Wu J, Yu S, Tejwani V, et al. Idiopathic nodular glomerulosclerosis in Chinese patients: a clinicopathologic study of 20 cases. Clin Exp Nephrol 18 (2014): 865-875.
- Nash DA, Rogers PW, Langlinais PC, et al. Diabetic glomerulosclerosis without glucose intolerance. Am J Med 59 (1975): 191.
- Kanwar YS, Garce´s J, Molitch ME. Occurrence of intercapillary nodular glomerulosclerosis in the absence of glucose intolerance. Am J Kidney Dis 15 (1990): 281.
- da Silva EC, Saldanha LB, Pestalozzi MSC, et al. Nodular diabetic glomerulosclerosis without diabetes mellitus. Nephron 62 (1992): 289.
- UchidaT, Oda T, Watanabe A, et al. Idiopathic nodular glomerulosclerosis in a never-smoking, normotensive, non-obese, normal-glucose-tolerant middle-aged woman. Clin Kidney J 5 (2012): 445-448.
- Gonzalo A, Navarro J, Mampaso F, et al. Nodular glomerulosclerosis without glucose-intolerance: Long-term follow-up. Nephron 66 (1994): 481.
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 37 (2014): S81-S90.
- Owda AK, Abdallah AH, Haleem A, et al. De novo diabetes mellitus in kidney allografts: nodular sclerosis and diffuse glomerulosclerosis leading to graft failure. Nephrol. Dial. Transplant 14 (1999): 2004-2007.
- Wojciechowski D, Onozato ML, Gonin J. Rapid onset of diabetic nephropathy in three renal allografts despite normoglycemia. Clinical Nephrology 71 (2009): 719-724.
- Prasad N, Gupta P, Jain M, et al. Outcomes of De Novo Allograft Diabetic Nephropathy in Renal Allograft Recipients. Experimental and clinical transplantation 11 (2013).
- Kuppachi S, Idris N, Chander PN, et al. Idiopathic nodular glomerulosclerosis in a non-diabetic hypertensive smoker- case report and review of literature. Nephrol Dial Transplant 21 (2006): 3571- 3575.
- López-Revuelta K, Méndez Abreu AA, Gerrero-Márquez C, et al. Diabetic Nephropathy without Diabetes. J. Clin. Med 4 (2015): 1403-1427.
- Li W, Verani RR. Idiopathic nodular glomerulosclerosis: a clinicopathologic study of 15 cases. Human Pathology 39 (2008): 1771-1776.
- Alchi B, Nishi S, Iguchi S, et al. Recurrent light and heavy chain deposition disease after renal transplantation. Nephrol. Dial. Transplant 20 (2005): 1487-1491.
- Resuli M, Thomsen Nielsen F, Gimsing P, et al. Multiple myeloma in a kidney transplanted patient primarily diagnosed with monoclonal gammopathy of unknown significance (MGUS)- related nephropathy. Clin Kidney J 6 (2013): 445-446.
- Ganeval D. Kidney involvement in light chain deposition disease. In: Minotti L, D'Amico G, Ponticelli C, eds. The Kidney in Plasma Cell Dyscrasias. Kluwer Accademic Publishers, Durdrect, Boston and London (1988): 221-228.
- Pozzi C, Fogazzi GB, Banfi G, et al. Renal disease and patient survival in light chain deposit disease. Clin Nephrol 43 (1995): 281-287.
- Hall CL, Peat SDS . Light chain deposit disease: a frequent cause of diagnostic difficulty. Nephrol. Dial. Transplant 16 (2001): 1939-1941.


 Impact Factor: * 3.3
Impact Factor: * 3.3 Acceptance Rate: 73.59%
Acceptance Rate: 73.59%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 