The Beauty and Simplicity of Using Peritoneal Dialysis in Acute Kidney Injury Secondary to COVID-19 Infection: A Case Series and Literature Review
Abdullah Al-Hwiesh1*, Khadija M. Alshehabi1, Amani Alhwiesh1, Abdelgalil Moaz Mohammed1, Nadia Al-Audah2, Eman alsaleh2, Lamees Alayoobi1, Hend A. Aljenaidi1, Ibrahiem Saeed Abdul-Rahman1
1Nephrology Division, Department of Internal Medicine, King Fahd Hospital of the University, Imam Abdulrahman Bin Faisal University, Saudi Arabia
2Department of Pathology, Dammam Central Hospital, Saudi Arabia
*Corresponding Author: Abdullah Al-Hwiesh, Professor and Consultant of Nephrology, King Fahd Hospital of the University Al-Khobar, 40246, Saudi Arabia
Received: 25 January 2021; Accepted: 16 February 2021; Published: 08 March 2021
Article Information
Citation: Abdullah Al-Hwiesh, Khadija M. Alshehabi, Amani Alhwiesh, Abdelgalil Moaz Mohammed, Nadia Al-Audah, Eman alsaleh, Lamees Alayoobi, Hend A. Aljenaidi, Ibrahiem Saeed Abdul-Rahman. The Beauty and Simplicity of Using Peritoneal Dialysis in Acute Kidney Injury Secondary to COVID-19 Infection: A Case Series and Literature Review. Archives of Nephrology and Urology 4 (2020): 031-042.
DOI: 10.26502/anu.2644-2833034
View / Download Pdf Share at FacebookAbstract
Since the declaration that coronavirus disease (COVID-19) is an ongoing pandemic disease, the health care system worldwide is under enormous pressure. COVID-19 has infected more than 63 million patients and caused more than 1.5 million deaths until December 20, 2020 and the number of cases is rapidly increasing. The mortality rate of COVOD -19 worldwide is 6.3%. Here we report our case series of six patients who were treated with automated peritoneal dialysis with a favourite outcome. To the best of our knowledge, few reports in the literature have discussed the use of peritoneal dialysis in acute kidney injury secondary to COVID-19 infection.
Keywords
<p>COVID19; AKI; Automated peritoneal dialysis</p>
Article Details
1. Introduction
Since the World Health Organization (WHO) has declared that COVID-19 is a pandemic disease on March 11.2020 [1], The global health care system was under enormous pressure [1, 2]. The COVID-19 cases worldwide have exceeded 63 million and number of death was surpassing 1.5 million cases. In June 2020, the WHO has announced that the disease could run a second wave, which may be more severe than the first one [2, 4]. The mortality rate of COVID-19 worldwide is 6.37%, whereas in the Kingdom of Saudi Arabia (KSA) is 0.2%; the variation of this mortality rate is uncertain. However, this could be attributed to proactive and effective implementation of stringent measures from the Ministry of Health (MOH) to contain the spread of COVID-19 [3-5], different sub genotype affecting different areas, potential attenuation of its effect due to environmental factors or people in KSA have some kind of immunity against this sub genotype; this matter needs to be explored in future trial. The prevalence of acute kidney injury (AKI) in patients with COVID-19 is unknown; it varies from 0.5 to 29% [6]. Although diffuse alveolar damage and acute respiratory failure were the main features of COVID-19, the involvement of other organs needed to be studied [7]. AKI occurs in about 5% of hospitalized patients and up to 50% in intensive care units (ICU). The mortality rate from AKI is extremely high, despite the presence of COVID-19 infections [6].
The incidence of AKI in Wuhan in China was 5.1% out of 701 COVID-19 positive patients [6]. The incidence of AKI was significantly higher in patients with elevated baseline serum creatinine (11.9%) than in patients with normal baseline values (4.0%) [6]. Another study conducted in Wuhan hospital in China showed that AKI occurred in 27% in 85 COVID-19 positive patients [7]. Older people (age >60 years) and those with coexisting hypertension, and heart failure are at higher risk of developing AKI [7]. Postmortem kidney pathological examination of six patients demonstrated severe acute tubular injury, prominent lymphocyte infiltration, detection of viral antigen in tubular epithelial cells, complement C5b-9 deposition and macrophage infiltration [7]. The lymphocyte and immune cell infiltration found in COVID-19–induced AKI is likely an important pathophysiologic factor in kidney injury and repair [8]. Management of AKI includes identifying the etiological factor, treating reversible causes such as volume depletion and hypotension, removing any active insults to minimize new injury, identifying and treating the complications that may eventually require renal replacement therapy (RRT) [9]. Peritoneal dialysis (PD) is not commonly used for treating AKI. Nevertheless, it should not be discarded as a useful therapeutic modality for AKI patients. PD offers several advantages over hemodialysis (HD), including its technical simplicity, excellent cardiovascular stability, absence of extracorporeal circuit, low risk of bleeding and hydro-electrolyte imbalance and most importantly preservation of residual renal function. However, PD has some limitations as it needs an intact peritoneal cavity, and it carries risks of peritoneal infections and protein losses [10, 11]. We hereby reporting our successful experience of using APD in six patients with AKI secondary to COVID-19 pneumonia who were treated with tidal PD with favourite outcome. To the best of our knowledge, there are scarcity of reports that have discussed the usefulness of PD in AKI secondary to COVID-19.
2. Brief presentation for Six Cases
2.1 Case 1
A 34-year-old Saudi lady known case of diabetes mellitus complicated by diabetic nephropathy, hypertension, and dilated cardiomyopathy with reduced ejection fraction (EF) 48%. She was admitted to our hospital due to cough and shortness of breath. Vital signs: BP 130/65 mmHg, pulse 100 beats/min and temperature 38.5oC. Chest exanimation showed bronchial breathing mainly at the right side. Cardiovascular examination: normal S1+S2 no added sound or murmur. Abdominal examination showed multiple scars of previous C/S. Nasopharyngeal and throat swab for COVID-19 was positive. Urgent flexible double cuff Tenckhoff PD catheter was inserted uneventfully at bedside, under local anesthesia using Dr. AlHwiesh technique [30], due to severe uremia and metabolic acidosis. Tidal peritoneal dialysis 70% of 25L per day was started instantly, using Physioneal 1.36% 15L and 2.27% 10L and occasionally 3.8% as clinically indicated, each fill 1.5L and it was gradually increased until 2L. Each PD session lasted for 24 hours, and sessions were repeated daily until correction of acidosis and uremia was achieved or urine output more than 1000 ml per day. The patient received 1 gm of Vancomycin prior to catheter insertion as prophylaxis per our protocol. She was started initially on Ceftriaxone 2 gm IV, Azithromycin 500 mg OD, Oseltamivir 75 mg OD. Lopinavir/Ritonavir 200 mg/50 mg Q12hr. Patient’s condition dramatically improved, and she was discharged against medical advice after ten days from admission.
2.2 Case 2
A 57-year-old Saudi lady known case of type II diabetes mellitus for 15 years with diabetic nephropathy and retinopathy, hypertension, ischemic heart disease, and stage III chronic renal failure. She was admitted to our hospital with frequent episodes of watery diarrhea, nausea, repeated attacks of vomiting and high-grade fever 39.5oC. On examination: she looked ill, dehydrated, with dry mucous membrane. Vital signs: BP 120/65 mmHg, pulse 110 beats/min and temperature 39.5oC. Chest auscultation revealed bilateral crepitation (Figure 1). The patient had normal cardiovascular examination with normal heart sounds and no added sounds or murmur. Abdominal examination showed multiple scars of previous Cesarean Section. Nasopharyngeal and throat swab for COVID-19 was positive. PD was started instantly uneventfully similar to the previous case. Ten days later, she was discharged from hospital with good urine output and off dialysis.
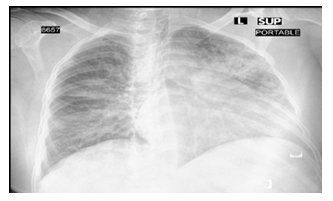
Figure 1: CXR-Bilateral pulmonary infiltration.
2.3 Case 3
A 62-year-old Saudi female known case of hypertension, cardiomyopathy with reduced EF 36%, dyslipidemia and psoriasis presented to the ER with symptoms of cough, shortness of breath, subjective fever, and diarrhea for two days. She gave a history of exposure to a relative confirmed to have COVID-19. The patient was vitally stable, but her chest examination was remarkable for the presence of bilateral crepitations. Her chest X-ray showed: Bilateral peripheral multifocal opacities with lower zone predominance and bilateral mild pleural effusion more on the left side, (Figure 2). The patient was admitted as a case of COVID positive Pneumonia. During her hospital course, she was intubated and transferred to the ICU where she was kept there for six days. She also developed AKI and PD was started uneventfully like the previous cases. Her condition improved and after eight sessions of PD her urine output increased to 1500 and PD was discontinued.
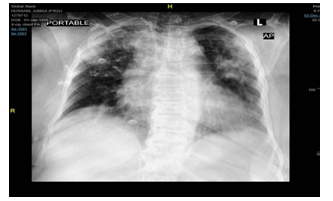
Figure 2: Bilateral peripheral multifocal opacities.
2.4 Case 4
A 55-year-old Saudi lady known case of dilated cardiomyopathy, atrial fibrillation, class IV congestive heart failure. She was admitted due to decompensated heart failure and generalized anasarca with deterioration in her renal function. Nasopharyngeal and throat swab for COVID-19 was positive. She was started initially on intravenous loop diuretics, but she did not improv and she developed catheter-related septicemia and became anuric. On examination, she looked ill and in respiratory distress. Vital signs showed blood pressure of 80/50 mmHg, pulse 90 beats/min, oxygen saturation 88 on room air and her weight was 55 kg. Cardiovascular examination showed raised jugular venous pressure, normal S1+ S2 with S3 gallop. Chest examination was consistent with bilateral pleural effusion mainly at the right side (Figure 3). PD catheter was inserted uneventfully, and urgent dialysis was started similar to the previous cases, with dramatic improvement of her symptoms. After seven sessions of PD with average ultrafiltration of 1.7 L/day and improvement in her urine output, PD was discontinued. Her latest investigations prior to discharge from hospital were: BUN 32 mg/dl, Creatinine 2.1 mg/d, CO2 32 mEq/L and Hemoglobin 11.8 gm/dL.
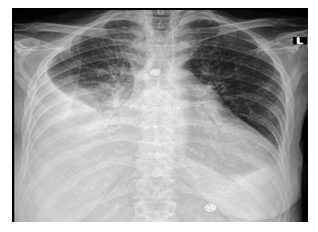
Figure 3: CXR-Bilateral pleural effusion and pulmonary congestion.
2.5 Case 5
A 59-year-old Saudi lady known case of diabetes mellitus with diabetic nephropathy, hypertension and coronary artery disease was admitted due to dyspnea and productive cough with high-grade fever. On examination, she was conscious, oriented and in respiratory distress. Vital signs: temperature 39oC, pulse 98 beats/min, BP 140/86 mmHg, oxygen saturation 88% on 4L nasal cannula and her weight is 75 Kg. On chest examination, there was fine bilateral crepitations up to middle zone. Her initial investigations showed BUN 100, Creatinine 8 and chest x-ray consistent with bilateral infiltration (Figure 4). She was intubated and PD catheter was inserted uneventfully, and PD immediately started as previous cases. After one week, she was extubated, but was still dialysis-dependent with average ultrafiltration of 1300 to 1500 per day and residual renal function of 300 ml per day.
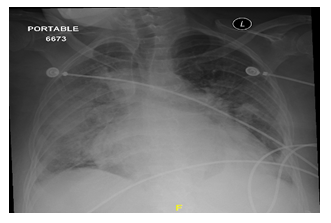
Figure 4: CXR-Bilateral infiltration.
2.6 Case 6
An 80-year-old Saudi male who was known to have diabetes mellitus, hypertension, history of multiple strokes and stage IV chronic renal failure presented to the ER with vomiting and diarrhea associated with high-grade fever. On examination patient looked ill. Vital signs were as follows: BP 90/55 mmHg, pulse 110 beats/min and temperature 39oC. The chest was clear on percussion and auscultation. Nasopharyngeal and throat swab for COVID-19 was positive. Owing to deterioration of his renal function and severe metabolic acidosis, PD was started promptly like the other cases and was discharged after three weeks in a stable condition. Six weeks later, he was readmitted with a new onset cerebrovascular stroke. The patient was intubated, and his condition deteriorated, and he was succumbed to his illness after one week from ICU admission. For all the six cases, PD went smooth without infectious or mechanical complications. Table 1 depicts the important investigations at admission and five days after PD commencement. Table 2 describes the demographic data and the outcomes.
|
P 1 |
P2 |
P3 |
P4 |
P5 |
P6 |
|
|
Age |
34 |
57 |
62 |
57 |
59 |
80 |
|
Sex |
F |
F |
F |
F |
F |
M |
|
DM |
Yes |
Yes |
yes |
Yes |
yes |
Yes |
|
HTN |
Yes |
Yes |
Yes |
Yes |
yes |
Yes |
|
CVD |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
|
CVA |
No |
No |
No |
Yes |
Yes |
Yes |
|
Dyslipidemia |
No |
No |
Yes |
Yes |
Yes |
Yes |
|
Intubation |
No |
No |
yes |
No |
No |
Yes |
|
Dialysis dependent |
Yes |
NO |
No |
No |
yes |
Yes |
|
Death |
No |
No |
NO |
No |
No |
yes |
P: Patient, DM: Diabetes Mellitus, HTN: Hypertension, CVD: Cardiovascular, Disease, CVA: Cerebrovascular Accident
Table 1: All the six cases, PD went smooth without infectious or mechanical complications.
BUN: Blood Urea Nitrogen, UF: Ultrafiltration, RBS: Random Blood Sugar , K : Potassium.
Table 2: The demographic data and the outcomes.
3. Discussion
Human coronaviruses (HCoVs) can be classified into highly pathogenic and low pathogenic coronaviruses. The low pathogenic HCoVs usually affect upper respiratory tract causing mild flu like illnesses whereas, highly pathogenic HCoVs, such as COVID 19, SARS-COV and MERS-COV usually affect lower respiratory tract causing severe pneumonia that can be complicated with acute lung injury and acute respiratory distress syndrome leading to high morbidity and mortality rate [12]. The outbreaks of SARS-COV in 2002 and MERS-COV in 2012 have shown that coronaviruses are more severe and fatal when they infect human being [13]. The prevalence of AKI in patients with COVID-19 is unknown, initial reports from China and Italy found that the prevalence of AKI in patients admitted due to COVID-19 was ranging from 0.5% to 29% [14, 15]. In a retrospective observational cohort study, Hirsh et al. reported the prevalence of AKI in 5449 patients admitted with COVID-19 to be 46% which increased to 89.7% in patients who required mechanical ventilation; 14.3% of them needed RRT. All patients who required dialysis were treated either by intermittent hemodialysis or continuous renal replacement therapy, none of them were started on PD. Among 285 patients who required RRT; 157 died and 108 are still in hospital with 90.8% requiring RRT [13]. The risk factors for AKI included older age, diabetes mellitus, hypertension, cardiovascular diseases, use of mechanical ventilators and the need for vasopressor medications [15]. The mechanism of COVID-19 induced AKI is multifactorial including, acute tubular necrosis induced directly by COVID-19 infection and replication at the proximal tubular cell, complement mediated mechanism leading to tubular-interstitial fibrosis, epithelial cell apoptosis and cytokine release syndrome [7-18]. Several studies have shown that patients infected with COVID-19 exhibit marked lymphopenia particularly cytotoxic lymphocyte (CD8+), increase neutrophil count and significant elevation in pro-inflammatory mediators in particular high levels of cytokines and chemokines such as interleukin (IL)-2, IL-6, and IL-10. This cytokine storm may have a significant role in AKI in COVID-19 patients [19-20].
There is scarcity in the literature that implicates the use PD in AKI related to COVID-19 [5, 13, 15]. AKI is a common complication in patients treated in the intensive care unit (ICU). Since 1990, continuous veno-venous therapies have gained popularity in ICU settings and become the modality of choice for AKI patients [19]. PD is used infrequently for treating patients with AKI [20]. The mortality rate reaches up to 50-60 % in patients who developed AKI in ICU settings [21], regardless of dialysis type. PD modality should not be discarded since it is a valuable therapeutic option for patients who develop AKI in ICU settings. In our recent randomized trial, 125 ICU patients with AKI and multiorgan failure; were randomly allocated to 62 patients on continuous veno-venous hemodiafiltration (CVVHDF) and 63 patients on tidal PD (TPD). The survival rate at 28 days was significantly better in patients who were treated with TPD when compared to CVVHDF; 69.8% vs 46.8%, P< 0.01 and the recovery of kidney function occurred in 35.5% and 60.3% respectively. The median time of renal recovery and ICU stay was shorter with TPD [11]. Dianeal Ponce et al. performed a randomized clinical trial on 120 patients with AKI, comparing high volume PD (60 patients) with intermittent HD (60 patients). The mortality rate did not differ significantly between the two groups (58% for high volume PD vs 53% for intermittent HD) and the rate of renal recovery was similar for both groups. However, high volume PD was associated with shorter recovery time 7.2 ± 2.5 vs 10 ± 4.7 days [22]. ISPD guidelines for using PD in AKI clearly stated that PD provides an acceptable treatment option in patients with AKI [23]. PD offers several advantages over HD, such as being a simple, safe, and cost-effective renal replacement therapy method. It can correct AKI induced metabolic abnormalities, electrolyte and acid-base disturbances as well as volume overload in an ICU setting [24, 25]. Acute PD is highly preferable for patients with cardio-renal syndromes, hemodynamically unstable patients, conditions where systemic anticoagulation needs to be avoided and in cases with difficult vascular access creation [26]. Due to growing evidence of hypercoagulability state in critically ill COVID-19 patients, and the increased frequency of dialysis interruption during CRRT leading to inadequate dialysis, PD appears to be the preferred modality in those particular patients [27, 28].
However, PD may have some potential limitations in certain cases such as in severe life-threatening hyperkalemia, acute pulmonary edema and previous multiple abdominal surgeries where extracorporeal blood purification method is preferable [26]. PD is not commonly used for treating AKI in COVID-19 patients [15-29]. This could be due to lack of competency and knowledge by intensivists and nephrologists working in ICU, the inability to insert PD catheter urgently, and the myths associated with PD. It could also be related to PD adequacy not meeting the target in certain conditions such as in patients with hyper-catabolism state. In this case series, we report the successful outcomes of our six patients with AKI secondary to COVID-19 who were treated with tidal PD. To the best of our knowledge, there are limited case reports in the literature of using PD for AKI secondary to COVID-19 infection.
4. Conclusion
As COVID-19 causes profound cytokine storm and immunopathology transformations during the early stage of infection which corresponds to high morbidly and mortality [18]; there is high body of evidence indicating that PD has the capability to remove small and middle size molecules [27, 28]. Thus, the removal of these cytokines and inflammatory mediators by PD could explain the successful and accelerated recovery in our patients, and this need to be explored more in further studies.
Acknowledgements
The authors are pleased to express their great gratitude and appreciation to the ICU staff and peritoneal dialysis nurses at King Fahad University Hospital in Al Khobar for their valuable support during the patients’ hospitalization.
Author Contributions
Dr. Khadija was responsible for collecting the cases and drafting the case presentation. Dr. Amani and Dr. Abdelgalil performed literature review and drafted the discussion. Prof. Alhwiesh, Dr. Lamees, Dr. Hend and Dr. Nadia contributed in writing and reviewing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was obtained from Imam Abdulrahman bin Faisal University Review Board of Medical Center, and all written consents were taken from the patients.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
- Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID 19) Hospital Preparedness Assessment Tool. CDC (2020).
- World Health Organization. Coronavirus disease (COVID-19) dashboard (2020).
- World Health Organization. Coronavirus disease (COVID-19) dashboard (2020).
- Ministry of Health. COVID-19 Guidelines. Media Center (2020).
- Al-Hwiesh AK, Mohammed AM, Elnokeety M, et al. Successfully treating three patients with acute kidney injury secondary to COVID-19 by peritoneal dialysis: Case report and literature review. Peritoneal Dialysis International 40 (2020): 496-498.
- Cheng Y, Luo R, Wang K, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney International 97 (2020): 829-838.
- Diao B, Feng Z, Wang C, et al. Human kidney is a target for novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. MedRxiv (2020).
- Rabb H, Griffin MD, McKay DB, et al. Inflammation in AKI: current understanding, key questions, and knowledge gaps. Journal of the American Society of Nephrology 27 (2016): 371-379.
- UpToDate . Uptodate.com. 2020 [cited 29 May 2020]. Available from: https://www.uptodate.com/contents/overview-of-the-management-of-acute-kidney-injury-aki-in-adults#!
- Seria M, Al-Safar N, Dalal A-S, et al. Acute kidney injury secondary to rhabdomylosis and automated peritoneal dialysis. Global Advanced Research Journal of Medicine and Medical Sciences 3 (2014): 147-151
- Al-Hwiesh A, Abdul-Rahman I, Finkelstein F, et al. Acute kidney injury in critically ill patients: a prospective randomized study of tidal peritoneal dialysis versus continuous renal replacement therapy. Therapeutic Apheresis and Dialysis 22 (2018): 371-379.
- Channappanavar R, Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. InSeminars in immunopathology 39 (2017): 529-539.
- Hirsch JS, Ng JH, Ross DW, et al. Acute kidney injury in patients hospitalized with COVID-19. Kidney International 98 (2020): 209-218.
- Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. The Lancet Respiratory Medicine 8 (2020): 475-481.
- El Shamy O, Sharma S, Winston J, et al. Peritoneal dialysis during the coronavirus 2019 (COVID-19) pandemic: acute inpatient and maintenance outpatient experiences. Kidney Medicine (2020).
- Su H, Yang M, Wan C, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney International 98 (2020): 219-227.
- Meng XM, Nikolic-Paterson DJ, Lan HY. Inflammatory processes in renal fibrosis. Nature Reviews Nephrology 10 (2014): 493.
- Liu J, Li S, Liu J, et al. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EbioMedicine 55 (2020): 102763.
- Bellomo R, Mansfield D, Rumble S, et al. A Comparison of Conventional Dialytic Therapy and Acute Continuous Hemodiafiltration in the Management of Acute Renal Failure in the Critically III. Renal Failure 15 (1993): 595-602.
- Hyman A, Mendelssohn DC. Current Canadian approaches to dialysis for acute renal failure in the ICU. American Journal of Nephrology 22 (2002): 29-34.
- Góes CR, Berbel MN, Balbi AL, et al. Approach to the metabolic implications of peritoneal dialysis in acute kidney injury. Peritoneal Dialysis International 35 (2015): 397-405.
- Gabriel DP, Caramori JT, Martim LC, et al. High volume peritoneal dialysis vs daily hemodialysis: a randomized, controlled trial in patients with acute kidney injury. Kidney International 73 (2008): S87-S93.
- Cullis B, Abdelraheem M, Abrahams G, et al. Peritoneal dialysis for acute kidney injury. Peritoneal Dialysis International 34 (2014): 494-517.
- Ponce D, Berbel MN, de Goes CR, et al. High-volume peritoneal dialysis in acute kidney injury: indications and limitations. Clinical Journal of the American Society of Nephrology 7 (2012): 887-894.
- Sharma SK, Manandhar D, Singh J, et al. Acute peritoneal dialysis in Eastern Nepal. Peritoneal Dialysis International 23 (2003): 196-199.
- Chionh CY, Soni SS, Finkelstein FO, et al. Use of peritoneal dialysis in AKI: a systematic review. Clinical Journal of the American Society of Nephrology 8 (2013): 1649-1660.
- Alberici F, Delbarba E, Manenti C, et al. on behalf of the “Brescia Renal COVID Task Force. Management of patients on dialysis and with kidney transplant during SARS-COV-2 (COVID-19) pandemic in Brescia, Italy. Kidney Int Rep 5 (2020): 580-585.
- Tang N, Li D, Wang X, et al. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. Journal of Thrombosis and Haemostasis 18 (2020): 844-847.
- Srivatana V, Aggarwal V, Finkelstein FO, et al. Peritoneal dialysis for acute kidney injury treatment in the United States: brought to you by the COVID-19 pandemic. Kidney360 (2020): 10-34067.
- Al-Hwiesh AK. Percutaneous peritoneal dialysis catheter insertion by a nephrologist: a new, simple, and safe technique. Peritoneal Dialysis International: Journal of the International Society for Peritoneal Dialysis 34 (2014): 204.


 Impact Factor: * 3.3
Impact Factor: * 3.3 Acceptance Rate: 73.59%
Acceptance Rate: 73.59%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 