Beneficial Effects of Thoracic Epidural Anesthesia on Mortality Rate in Geriatric Patients after Elective Surgery for Colon Cancer: A Study of 215 Consecutive Patients
Simon Moormann1, Marie Luise Rüebsam1, Eva Warnking3, Antje Gottschalk4, Klaus Hahnenkamp2, Hugo Van Aken4, Dietrich Doll5, Olaf Hagemann5, Christian Hönemann5*
1Department of Anaesthesiology and Intensive Care Medicine, Marienhospital Vechta, Vechta, Germany
2Universitätsmedizin Greifswald, Klinik für Anästhesiologie, Greifswald, Germany
3Klinikum Oldenburg, Universitätsklinik für Anästhesiologie / Intensivmedizin / Notfallmedizin / Schmerztherapie, Oldenburg, Germany
4Universitätsklinikum Münster, Klinik für Anästhesiologie, operartive Intensivmedizin und Schmerztherapie, Münster, Germany
5Marienhospital Vechta gGmbH, Abteilung für Anästhesie und Intensivmedizin & Universitätsklinikum Münster, Klinik für Anästhesiologie, Intensivmedizin und Schmerztherapie, Germany
*Corresponding Author: Christian Hönemann, Marienhospital Vechta gGmbH, Abteilung für Anästhesie und Intensivmedizin & Universitätsklinikum Münster, Klinik für Anästhesiologie, Intensivmedizin und Schmerztherapie, Germany
Received: 29 April 2022; Accepted: 05 May 2022; Published: 11 May 2022
Article Information
Citation: Simon Moormann, Marie Luise Rüebsam, Eva Warnking, Antje Gottschalk, Klaus Hahnenkamp, Hugo Van Aken, Dietrich Doll, Olaf Hagemann, Christian Hönemann. Beneficial Effects of Thoracic Epidural Anesthesia on Mortality Rate in Geriatric Patients after Elective Surgery for Colon Cancer: A Study of 215 Consecutive Patients. Anesthesia and Critical care 4 (2022): 104-117.
View / Download Pdf Share at FacebookAbstract
Introduction
Colorectal cancer is a major cause of death in the industrial world. The mortality and morbidity rates depend on postoperative complications and cancer recurrence. Research suggests that regional anesthesia reduces perioperative stress levels, potentially lowering the risk of complications. Patients with perioperative epidural analgesia might have longer survival times compared with patients without perioperative epidural analgesia.
Methods
We compared short and long-term outcome data from 215 patients who underwent open colon cancer surgery by the same surgeon and the same oncologist. Eighty-three patients received general anesthesia plus thoracic epidural analgesia (EPI group), and 132 patients received general anesthesia alone (GA group). Oncological data from a state-wide follow-up database were included. The effects of different perioperative anesthetic techniques on patients’ short and long-term outcomes over 36 months were statistically analyzed (Pearson’s chi-squared test, Student’s t-test, Wilcoxon rank-sum test) as appropriate. A Kaplan-Meier analysis for survival was performed and analyzed by the Wilcoxon rank-sum test.
Results
With the exception of a significantly higher prevalence of arterial hypertension in the EPI group in comparison with the GA group, there were no differences in demographics, tumor staging data, and cancer recurrence rates between the groups. However, mortality rates were significantly different between the groups. Thirty-seven of 132 GA patients (28%) died within 36 months, in comparison with 14 of 83 EPI patients (16.9%, P<0.05). Patients over the age of 70 years in particular significantly benefited from perioperative epidural analgesi
Keywords
<p>PCEA – Patient controlled analgesia, Thoracic epidural anaesthesia, Mortality, Colon cancer, Geriatric patients</p>
Article Details
Abbreviations
EPI = epidural anesthesia
EPI group = group of patients with
epidural and general anesthesia
GA = general anesthesia
GA group = group of patients with general
anesthesia alone
ICU = intensive care unit
kg = kilogram
N = N in TNM refers to the involvement of lymph nodes in cancer.
M = The M refers to whether the cancer has metastasized.
mg = milligram
min = minute
µg = microgram
P = p– value: All hypothesis tests ultimately use a p-value to weigh the strength of the evidence what the data are telling you about the population). A p-value is a number between 0 and 1.
PCEA = patient controlled epidural analgesia
% = percent
SRC = Proto-oncogene tyrosine-protein kinase Src, also known as proto-oncogene c-Src or simply c-Src (a non-receptor tyrosine kinase protein that in humans is encoded
by the SRC gene). This protein phosphorylates specific tyrosine residues in other tyrosine kinases.
TNM = The TNM Classification of of cancer.
1. Introduction
Cancer is a major cause of death in the industrialized world [1]. Cancer treatment is based on multimodal approaches; surgical resection is necessary for most types of solid tumors. The survival period for the patients depends on the grade, the type and stage of the lesion at the primary diagnosis, as well as on the patients’ pre-existing comorbid conditions. Reports indicate that 5% of patients who undergo surgery die within the first year, and the rate is doubled for patients over the age of 65. The primary causes of death include the cancer itself, followed by cardiovascular problems, kidney or liver failure, multiple organ failure, and respiratory failure [2]. Therefore, the challenge for physicians is to find an optimal form of perioperative therapy capable of preventing these causes of morbidity and mortality. To this end, some studies have suggested that regional anesthesia may be a reasonable technique for reducing complications and improving long-term patient outcomes [3]. Thoracic epidural analgesia provides optimal pain therapy for many operations and may, therefore, be able to reduce the morbidity and mortality after major thoracic and abdominal surgery [4]. Compared to general anesthesia, the patient’s immune system is less compromised when combining general anesthesia with regional anesthesia. As a result, data suggest that regional anesthetic techniques may reduce the risk of micrometastases. Patients undergoing cancer resection should, therefore, be able to benefit from epidural analgesia, as it may reduce the risk of recurrent cancer and metastases after cancer surgery [4]. Furthermore, such a method may reduce the mortality rate. It was therefore hypothesized that patients undergoing open surgery for colonic cancer who receive thoracic epidural anesthesia should experience fewer instances of recurrent cancer and has a longer survival period in comparison with patients who received only general anesthesia.
2. Methods
Following approval for the study by the local ethics committee of the General Medical Council of the state of Lower Saxony (Ärztekammer Niedersachsen), prospectively collected data and medical records were routinely analyzed for all patients who had undergone open colon surgery between 1999 and 2007 in St. Mary’s Hospital, Vechta, Germany (St. Marienhospital Vechta). Hospitals can be classified into three groups in relation to colon cancer treatment, based on their annual caseload [5], low-volume (< 30 colon cancer operations per year), medium-volume (30–60 colon cancer operations per year) and high-volume (> 60 colon cancer operations per year) [5]. St. Mary’s belongs to the high-volume group. Patients who underwent elective open colon cancer resection and were over the age of 18 were included in the analysis if oncological follow-up data were prospectively stored in an official electronic database (the Epidemiological Cancer Registry for Lower Saxony, Epidemiologisches Krebsregister Niedersachsen, 215 out of 552 cases). Patients who underwent emergency operations and follow up data were not sampled were excluded. An electronic database was used to determine the baseline demographic variables, the type of anesthesia administered, and the date of surgery. All oncological and follow-up data were taken from the official electronic database (the Epidemiological Cancer Registry for Lower Saxony, Epidemiologisches Krebsregister Niedersachsen). All clinically relevant and potential interacting factors associated with tumor outcome and progression was recorded in a clinical database.
2.1 Anesthesia
The patients underwent elective colonic surgery for tumor resection either under general anesthesia combined with thoracic epidural analgesia (EPI group) or with general anesthesia alone (GA group). In both groups, anesthesia was induced with propofol (Propofol®Lipuro, Braun, Germany) 2–3 mg/kg bodyweight, rocuronium (Esmeron®,Grünenthal GmbH, Germany) 0.6 mg/kg body weight for muscle relaxation, and fentanyl (Fentanyl,®, Jansen GmbH, Germany) (GA group) or sufentanil (Sufentanil®, Actavis GmbH, Germany) (EPI group) for pain control during intubation. Inhaled anesthetics were used in both groups to maintain anesthesia. Whether epidural analgesia should be used was decided by the patient in consultation with the attending anesthesiologist. In the EPI group, the thoracic epidural catheter was inserted preoperatively. Before surgical incision, 10 mL bupivacaine (Bucain-actavis®, Actavis GmbH, Germany) (0.25%) was injected. The epidural analgesia was maintained for 96 hours postoperatively using a patient-controlled epidural analgesia (PCEA) technique with bupivacaine 0.125% plus 0.5 µg/ml sufentanil (rate 5–7 ml/hour, 2 ml bolus on demand, lockout time after bolus 20 min). The patients were examined once per day, or more often if needed, by the hospital’s acute pain service to ensure adequate postoperative analgesia.
2.2 Statistical analysis
The groups were compared using Lee’s Revised Cardiac Risk Index (Table 1 [6]. The data for the patients in the two study groups were compared in relation to all potential confounding factors using Pearson’s chi-squared test, Student’s t-test, and the Wilcoxon rank-sum test, as appropriate. A Kaplan Meyer Analysis was used to investigate effects on mortality for all of the patients as the primary endpoint and statistical difference were calculated by the Wilcoxon rank-sum test. Statistical analyses (t-test, chi-squared, and Wilcoxon Rank Sum tests) were carried out using the IBM PASW statistics program (SPSS Inc. Version 18.0. Chicago: SPSS Inc).
3. Results
Table 2 shows the demographic data for the patients in the two groups, allowing comparison of pre-existing medical conditions. Between 1999 and 2007, 215 patients underwent open colorectal surgery. Eighty-three patients were administered general anesthesia combined with thoracic epidural analgesia (EPI group), and 132 patients underwent surgery with general anesthesia alone (GA group). The demographic data of the patients are shown in table 1.
|
EPI group (n = 83) |
GA group (n = 132) |
P value |
|
|
Male vs. female (n) |
48 vs. 35 |
66 vs. 66 |
0.33 |
|
Age (years) |
67.5 ± 10.75 |
67.3 ± 10.66 |
0.88 |
|
Height (cm) |
171 ± 7.6 |
169 ± 17.4 |
0.27 |
|
Weight (kg) |
79.6 ± 14.6 |
76.4 ± 16.5 |
0.14 |
|
Body mass index (kg/m2) |
26.9 ± 3.75 |
26.3 ± 5.16 |
0.37 |
|
Diabetes mellitus (%) |
18.1 |
16.7 |
0.79 |
|
Hypertension (%) |
67.5 |
46.2 |
>0.01* |
|
Coronary artery disease (%) |
16.9 |
16.7 |
0.97 |
|
History of myocardial infarction (%) |
2.4 |
3 |
0.79 |
|
Chronic obstructive pulmonary disease (%) |
10.8 |
9.1 |
0.67 |
|
Peripheral arterial occlusive disease (%) |
6 |
9.8 |
0.32 |
|
Renal insufficiency (%) |
9.6 |
7.6 |
0.59 |
|
Serum creatinine > 2 mg/dl (%) |
1.2 |
0 |
0.21 |
*Do you have an actual p-value here to report? I would supply it instead.
EPI = general anesthesia plus thoracic epidural anesthesia; GA = general anesthesia alone
Table 1: Demographic data and prevalence of pre-existing conditions in the group with general anaesthesia plus thoracic epidural anesthesia (EPI group) and the group with general anesthesia alone (GA group)
|
1 |
High-risk surgical procedures |
|
2 |
Ischemic heart disease |
|
3 |
History of congestive heart failure |
|
4 |
History of cerebrovascular disease |
|
5 |
Insulin therapy for diabetes |
|
6 |
Preoperative serum creatinine > 2.0 mg/dL |
Table 2: Lee’s revised cardiac risk index [6]. Each risk factor is assigned one point
The anesthesia duration was significantly longer in the EPI group than in the GA group, but the patients in the EPI group were discharged from the intensive-care unit (ICU) and from the hospital significantly sooner than the patients in the GA group (Table 3). The tumor stages in the two study groups were comparable (Figure 1a, P = 0.10). The rates of regional lymph-node metastasis were N0 55.4%, N1 13.3%, and N2 31.3% in the EPI group and N0 55.7%, N1 18.3%, and N2 26.0% in the GA group (Figure 1b, P = 0.44). The rates of distant metastasis were M0 89.2%, M1 10.8%, in the EPI group, and M0 87.9% and M1 12.1% in the GA group (Figure 1c, P = 0.72). Regarding pre-existing conditions, only the rate of hypertension was significantly higher in the EPI group in comparison with the GA group (P = 0.002) (Table 1). Table 2 displays the Lee index with 73.5% of the EPI group and 70.5% of the GA group with one factor; 20.5% of the EPI group and 18.9% of the GA group had two factors; 6.0% of the EPI group and 9.8% of the GA group had three factors, and only 0.8% of the GA group had four factors. The Lee index scores were comparable in the two groups (P = 0.65). No significant differences between the two study groups were noted in relation to the incidence of postoperative surgical or general complications. These complications included 6% of the patients in the EPI group, and 8.4% of those in the GA group developed pneumonia; cerebral strokes occurred in 2.3% of the GA group, and pulmonary embolisms in 1.2% of the EPI group (P = 0.45). Postoperative surgical complications were diagnosed in 8.4% of the EPI group (three anastomotic leakages, two wound dehiscences, one later resection and one other complication) and 6.8% of the GA group (four anastomotic leaks, three wound dehiscences, one case of postoperative peritonitis, and one other complication) (P = 0.66). No significant differences between the GA and EPI groups were noted regarding cancer survival: 21 of the 132 patients in the GA group (15.9%) and eight of the 83 patients in the EPI group (9.6%) died due to recurrences or metastases of colon cancer (P = 0.19). Thirty-seven of the 132 patients in the GA group (28%) died within 36 months compared to 14 of the 83 patients in the EPI group (16.9%, P < 0.05). Figure 2 shows the Cox regression analysis and corresponding P-value. Cox regression analysis in patients who were over the age of 70 at the time of surgery (Figure 3) showed that epidural analgesia significantly reduced patient mortality (P = 0.02).
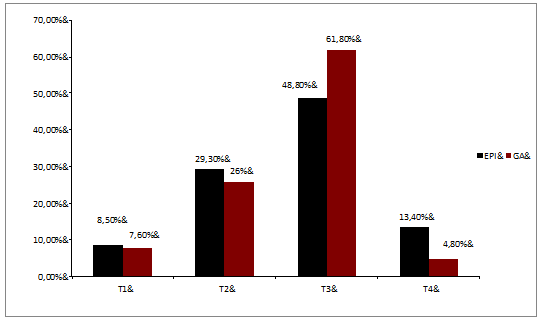
Figure 1a: Distribution of tumor stages in the two study groups. Numbers are shown in percentages for tumor stage
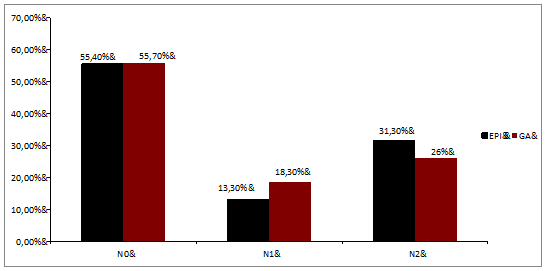
Figure 1b: Distribution of tumor stages in the two study groups. Numbers are shown in percentages for nodal status
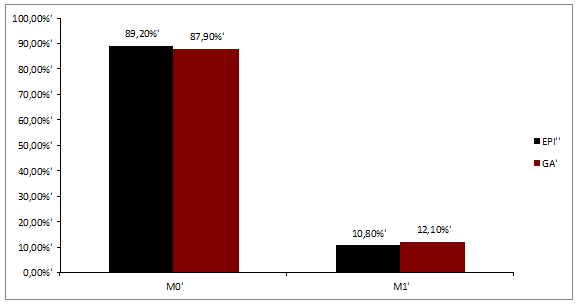
Figure 1c: Distribution of tumor stages in the two study groups. Numbers are shown in percentages for metastatic stage.
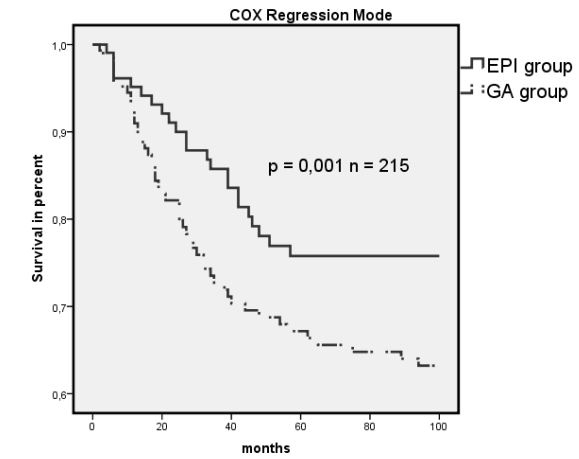
Figure 2: Cox regression survival mode for EPI vs. GA (n = 215). Dashed line indicates GA group, and solid line indicates EPI group.
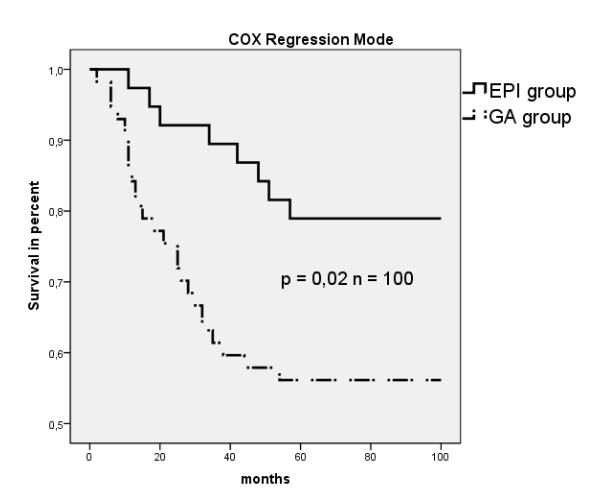
Figure 3: Cox regression survival mode for EPI vs. GA; age > 70 years (n = 100). Dashed line indicates GA group, and solid line indicates EPI group.
4. Discussion
A recent meta-analysis suggested that epidural anesthesia may prove beneficial to the overall survival after cancer surgery, particularly in patients with colorectal cancer [7]. However, the use of epidural analgesia did not further reduce the rate of recurrent cancers. The meta-analysis suggests that epidural anesthesia and analgesia may be associated with improved overall survival in patients with operable cancer, especially colorectal cancer, who undergo surgery [7]. The results of the present study show that administering thoracic epidural analgesia for perioperative analgesia during and after colon cancer resection was associated with a reduced 36-month mortality rate. These results support the findings of the earlier meta-analysis [7]. Patients over the age of 70 at the time of surgery benefitted more from epidural analgesia and survived significantly longer than patients in whom only a balanced anesthetic technique was used. However, after adjusting for confounding variables. no reduction in the incidence of recurrent cancer was noted. Colon cancer is a systemic disease. Despite curative tumor resection, it is important to note that as many as 25–40% of patients who undergo curative tumor resection nevertheless subsequently develop metastatic disease, suggesting that there may be undetected micrometastases that play a pivotal role in relapses. A major cause of relapse is the dissemination of tumor cells shed from the primary carcinoma into the circulation before, during, or after surgery [8]. Due to profound perioperative immunosuppression, the perioperative period appears to be a vulnerable time for potential metastasis in patients with malignancies. This vulnerability is mainly attributed to the suppression of cell-mediated immunity – the first-line defense mechanism against cancer. Immune system depression occurs within hours of surgery, lasts for several days, and is proportional to the extent of surgical trauma [4]. With colorectal surgery, the perioperative and postoperative period is characterized by surgical stress and pharmacological influences; this affects cell-mediated immunity during a vulnerable phase for tumor cell dissemination. In particular, natural killer cells and cytotoxic T cells are suppressed by high levels of stress biomarkers such as epinephrine and norepinephrine [9,10]. Thoracic epidural analgesia blunts the neural transmission of the stress response and avoids negative effects of hypothalamic-pituitary-adrenal activation such as immune suppression [4]. In addition, thoracic epidural analgesia reduces the quantity of anesthetics and analgesics required. These facts appear to be important, as some of these drug types promote immune suppression [4]. Using carefully considered anesthetic techniques may reduce the inhibition of hormonal and cellular immune function and even reduce the pro-angiogenic effect on tumor growth, resulting in fewer tumor recurrences in these oncological patients [4]. Accumulated evidence indicates that inflammation may play a critical role in the initiation, development, growth, and metastasis of cancer [11]. Regional anesthesia may, therefore, have a potential anti-inflammatory effect, attenuating an excessive immune reaction induced by surgical trauma, and it appears to have a beneficial effect on the outcome for the patients [5]. Bupivacaine inhibits prostaglandin E2 (PGE2) E-prostanoid 1 (EP1) receptor signaling at clinically relevant concentrations and affects physiological responses such as fever, inflammation, and hyperalgesia during the perioperative period [12]. Local anesthetics have a time-dependent inhibitory effect on G protein-coupled receptor signaling [13]. Local anesthetics are known to be effective in suppressing neutrophil priming, a process partly responsible for the hyperactive neutrophil response [14]. Local anesthetics can even inhibit thromboxane A2 signaling, a mediator of perioperative myocardial ischemia, vasoconstriction, and thrombosis [15,16]. They also inhibit the activation of human N-methyl-d-aspartate (NMDA) receptors. This effect reduces the hyperalgesia and opiate tolerance that is observed after systemic administration of local anesthetics [17]. Results published by Piegeler et al. indicate that local amide anesthetics may have antimetastatic effects. Underlying this antimetastatic effect is the inhibition of tumor necrosis factor-induced Src activation and intracellular adhesion molecule-1 phosphorylation [18]. This molecular mechanism with amide-type local anesthetics is again independent of the sodium channel blockade. It would be of major interest to determine whether some of the beneficial effects of epidural analgesia may be due to the systemic effects of local anesthetics. Despite these promising results from basic science and theoretical models, however, the results of the present study do not support the view that the perioperative administration of thoracic epidural analgesia leads to less recurrent cancer. This may be due to the familiar problems of retrospective analysis, such as unrecorded confounding factors and changes in the patients’ medical care. This potential deficiency was reduced in the present study, as the surgeon and oncologist were the same throughout the recorded study period. However, it is not known how often the patients may have required additional surgery for other medical conditions, with the same level of surgical stress and the same immunosuppressive effects. Nor is it known whether the patients were receiving medication with statins, beta-blockers, or cyclooxygenase (COX) inhibitors perioperatively, which also appear to have antimetastatic properties [4]. Along with the rapid advances being made in oncological treatment, it will, therefore, be very difficult to prove whether a specific anesthetic technique is really able to reduce the risk of recurrent cancer. However, thoracic epidural analgesia is an important component of fast-track rehabilitation in colon surgery. In addition to faster recovery, some authors have also described a reduction in general morbidity [19,20] with fewer cardiovascular adverse events due to epidural anesthesia and perioperative stress protection, analgesia, and sympathicolysis. The incidence of thrombosis and embolism is reduced. Epidural analgesia reduces the use of systemic opioids, and in combination with splanchnic sympathicolysis, prevents intestinal hypomotility and accelerates convalescence and a return to oral nutrition for the patients. It has also been reported that pain-induced pulmonary atelectasis and pneumonia can be prevented [3,21]. In a population-based study, Cummings et al. [22] compared epidural analgesia and traditional forms of pain management in relation to their effects on survival and recurrent cancer after colectomy. In this large observational study, including 42,151 patients, the 5-year survival rate was 61% in the epidural group and 55% in the non-epidural group [22]. The difference was analyzed using adjusted Cox regression and strongly supports the data obtained in the present study. This study has certain limitations, the most important of which is that it is a small, single-center retrospective study. For conclusive findings on whether regional anesthesia is capable of reducing the risk of tumor relapse, a large, multicenter study with a prospective and randomized design would be needed. In a trial of that type, patients would need to be included within a very short study period in order to reduce the possibility of changes in oncological regimens that would make it difficult to compare the study groups. Prospective trials are currently in progress, but no data have been published so far. The present study excludes the possibility of variations resulting from different participating surgeons and oncologists, and it may, therefore, be of special interest.
5. Conclusions
The data presented here support the theory that epidural analgesia appears to be beneficial for colon cancer patients in reducing the overall mortality rate. The effect is age-dependent; the mortality was reduced particularly in elderly patients (>70 years).
Declarations
Ethics approval
The study was approved by the ethical committee of the General Medical Council of the state of Lower Saxony (letter from Mr. Kay Bogs, Geschäftsführer der Ethikkommission der Ärztekammer Niedersachsen).
Consent to participate
Not applicable.
Consent for publication
All authors revised the final manuscript critically. All authors read and approved the final manuscript.
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author.
Competing interests
None.
Funding
EW, SM, MRL, and CH were supported by Hönemann GbR Research Project (No. TH_PDA_12_Marie). These funding were used to design the study and for data collection, analysis, and interpretation. EW, SM, and MRL received grants for traveling and CH a grant for publication costs.
Authors' contributions
All authors took part in the work and agree with the contents of the manuscript. CH, KH, and HVA provided concepts. EW, SM, CH, MRL conducted data acquisition and data analysis. MRL, DD, OH, AG and CH did the literature review. SM and MRL drafted the initial manuscript equally (first authorship), AG critically revised, and CH submitted the final manuscript (corresponding author).
Acknowledgments
We thank our secretary Mrs. Susanne Stransky for her support over several years of data acquisition and literature research. Dr. Diers and Dr. T. Erhart for their cooperation and support regarding communication with the cancer center in Oldenburg, Germany. We thank the members of our laboratory (Laborarztpraxis Enzenauer, Osnabrück, Germany), Mrs. Sambach - Touré from the Cancer Center Oldenburg, Germany. Your help and support were fundamental for this study.
Clinical trial registration number
(ISRCTN10994336, retrospective registered 07/06/2019)
References
- German Federal Office of Statistics (Statistisches Bundesamt). Krebs immer häufiger Todesursache [press release]. Wiesbaden: Statistisches Bundesamt (2013).
- Monk TG, Saini V, Weldon BC, et al. Anesthetic management and one-year mortality after noncardiac surgery. Anesth Analg 100 (2005): 4-10.
- Freise H, Van Aken HK. Risks and benefits of thoracic epidural anaesthesia. Br J Anaesth 107 (2011): 859-868.
- Gottschalk A, Sharma S, Ford J, et al. Review article: the role of the perioperative period in recurrence after cancer surgery. Anesth Analg 110 (2010): 1636-1643.
- Mroczkowski P, Ortiz H, Penninckx F, et al. European quality assurance programme in rectal cancer are we ready to launch? Colorectal Dis 14 (2012): 960-966.
- Lee TH, Marcantonio ER, Mangione CM, et al. Derivation and prospective validation of a simple index for prediction of cardiac risk of major noncardiac surgery. Circulation 100 (1999): 1043-1049.
- Chen WK, Miao CH. The effect of anesthetic technique on survival in human cancers: a meta-analysis of retrospective and prospective studies. PloS One 8 (2013): e56540.
- Uen YH, Lu CY, Tsai HL, et al. Persistent presence of postoperative circulating tumor cells is a poor prognostic factor for patients with stage I-III colorectal cancer after curative resection. Ann Surg Oncol 15 (2008): 2120-2128.
- Kurosawa S, Kato M. Anesthetics, immune cells, and immune responses. J Anesth 22 (2008): 263-277.
- Ben-Eliyahu S. The promotion of tumor metastasis by surgery and stress: immunological basis and implications for psychoneuroimmunology. Brain Behavior Immun 17 (2003): 27-36.
- Zamarron BF, Chen W. Dual roles of immune cells and their factors in cancer development and progression. Int J Biol Sci 7 (2011): 651-658.
- Honemann CW, Heyse TJ, Mollhoff T, et al. The inhibitory effect of bupivacaine on prostaglandin E(2) (EP(1)) receptor functioning: mechanism of action. Anesth Analg 93 (2001): 628-634.
- Hollmann MW, Herroeder S, Kurz KS, et al. Time-dependent inhibition of G protein-coupled receptor signaling by local anesthetics. Anesthesiology 100 (2004): 852-860.
- Hollmann MW, Kurz K, Herroeder S, et al. The effects of S(-)-, R(+)-, and racemic bupivacaine on lysophosphatidate-induced priming of human neutrophils. Anesth Analg 97 (2003): 1053-1058.
- Hahnenkamp K, Nollet J, Strumper D, et al. Bupivacaine inhibits thromboxane A2-induced vasoconstriction in rat thoracic aorta. Anesth Analg 99 (2004): 97-102.
- Honemann CW, Hahnenkamp K, Podranski T, et al. Local anesthetics inhibit thromboxane A2 signaling in Xenopus oocytes and human k562 cells. Anesth Analg 99 (2004): 930-937.
- Hahnenkamp K, Durieux ME, Hahnenkamp A, et al. Local anaesthetics inhibit signalling of human NMDA receptors recombinantly expressed in Xenopus laevis oocytes: role of protein kinase C. Br J Aanaesth 96 (2006): 77-87.
- Piegeler T, Votta-Velis EG, Liu G, et al. Antimetastatic Potential of Amide-linked Local Anesthetics: Inhibition of Lung Adenocarcinoma Cell Migration and Inflammatory Src Signaling Independent of Sodium Channel Blockade. Anesthesiology (2012).
- Schwenk W, Neudecker J, Raue W, et al. "Fast-track" rehabilitation after rectal cancer resection. Int J Colorectal Dis 21 (2006): 547-553.
- Wilmore DW, Kehlet H. Management of patients in fast track surgery. BMJ 322 (2001): 473-476.
- Kehlet H, Wilmore DW. Multimodal strategies to improve surgical outcome. Am J Surg 183 (2002): 630-641.
- Cummings KC, Xu F, Cummings LC, et al. A comparison of epidural analgesia and traditional pain management effects on survival and cancer recurrence after colectomy: a population-based study. Anesthesiology 116 (2012): 797-806.


 Impact Factor:
* 3.1
Impact Factor:
* 3.1
 Acceptance Rate:
77.58%
Acceptance Rate:
77.58%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
