Osteogenesis Imperfecta-Serine Replacing Glycine in the COL1A1 Gene-A New Establishment in Genetics
Usman Tauseef1, Mohsina Ibrahim1, Muhammad Sohaib Asghar2*, Abubakar Tauseef2, Maryam Zafar2, Uzma Rasheed3, Gul Muhammad Memon3, Mohammed Akram3
1National Institute of Child Health, Karachi, Pakistan
2Dow University of Health Sciences, Karachi, Pakistan
3Liaquat National Hospital and Medical College, Karachi, Pakistan
*Corresponding Author: Muhammad Sohaib Asghar, Dow University Hospital, Dow University of Health Sciences, Karachi, Pakistan
Received: 14 March 2020; Accepted: 27 March 2020; Published: 03 April 2020
Article Information
Citation:
Usman Tauseef, Mohsina Ibrahim, Muhammad Sohaib Asghar, Abubakar Tauseef, Maryam Zafar, Uzma Rasheed, Gul Muhammad Memon, Mohammed Akram. Osteogenesis Imperfecta: Serine Replacing Glycine in the COL1A1 Gene-A New Establishment in Genetics. Fortune Journal of Rheumatology 2 (2020): 061-066.
View / Download Pdf Share at FacebookAbstract
Osteogenesis Imperfecta (OI) is a genetic disorder of bone fragility. In most cases, genetic testing is not usually done either due to a lack of availability or unaffordability. In our study, we looked for a rare gene variant in a patient with Osteogenesis Imperfecta type 1. Our patient who was a 6-month-old baby boy presented to us with multiple bone fractures, blue sclera, umbilical hernia with the absence of dentinogenesis Imperfecta, and hypercalcemia. A gene sequencing came out to be COL1A1 positive having a nucleotide change of glycine acid mutation replaced by serine, which was a unique feature in a patient of OI, and as Glycine was critical for COL1A1 gene, its replacement leads to the patient presented as a homozygous case of OI. OI is usually diagnosed on a history of recurrent spontaneous fractures and characteristic features like blue sclera, short stature, and deformities of long bones or spine.
Keywords
<p>Osteogenesis, Mutation, Bone Disease, Genetic, Congenital Abnormalities, Deformities</p>
Article Details
1. Introduction
Osteogenesis Imperfecta is a rare genetic disorder described by Lobstein as Fragilatus Osseum which was later renamed as osteogenesis Imperfecta by Vrolik. Its incidence is 1 in 10000-20000 births [1-3]. It manifests as bone fractures, blue sclera, dentinogenesis Imperfecta, hearing impairment, hyperlaxity of skin and ligaments bone deformities and short stature. The clinical presentation varies depending on the type of OI, as it has been classified into eight subgroups. The most fatal one is type 2 which is perinatally lethal. Type 1 is inherited in autosomal dominant form and present with fractures and blue sclera as present in our case. The severity of the disease depends on the location of mutation on a specific polypeptide chain and also on the type of amino acid substitution. Medical treatment includes Bisphosphonate and antiresorptive agents but surgical intervention is required if medical therapy fails. There is no definitive cure for OI to date.
2. Case Presentation
Our patient is a six months old baby boy of Asian descent who presented to us with a history of recurrent fractures and lower limb deformity since birth and painful bony swelling at the right leg for 2 days. In birth history, the patient was a booked case at a tertiary care hospital, when at 28 weeks of gestation parents were informed that bowing of legs was noted in antenatal ultrasound scan but no fracture or deformity of bone was observed at that time. The baby was born at a tertiary care hospital via vacuum-assisted vaginal delivery and got fractures of the skull, right ulna, right femur and left humerus (Figure 1, 2).
At that time doctors raised the suspicion of genetic bone disorder and fractures were managed conservatively along with intravenous Pamidronate infusion, calcium, and vitamin -D supplements in syrup form along with paracetamol for pain relief. The patient was not diagnosed until the patient developed another spontaneous fracture of the femur at 1 month of life, after which further evaluation of the patient was done. The child was developmentally normal. Parents were first cousins and their elder son was alive and healthy. On examination, the patient had blue sclera and a bony deformity at the left leg. He was vitally stable and his length and weight were less than 5th percentile for the age. The laboratory findings were: Hemoglobin: 11.4g/dL, the mean corpuscular volume of 70.7fL, Total leukocyte count: 15,400 cells per microlitre of blood with lymphocytosis, Platelets of 6,96,000 per microlitre of blood.
Serum calcium was in the upper limit of normal whereas serum magnesium, 25 hydroxyl-vitamin D, parathyroid hormone (PTH), alkaline phosphatase (ALP) levels all were within normal limit. The differential considerations included rickets, child abuse, Ehler Danlos syndrome and Osteogenesis Imperfecta. Patient's DEXA scan was performed which turned out to be insignificant after which molecular and cytogenetic testing was carried out which showed mutation of COL1A1 gene in homozygous state with replacement of glycine by serine at codon 338 of COL1A1 protein (Figure 3), which was not associated with most cases of OI and patient was diagnosed as a case of Osteogenesis Imperfecta on the basis of clinical presentation and genetic testing.
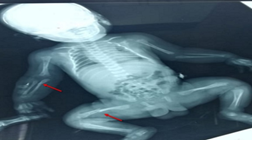
Figure 1: Showing fractures at radius, ulna, and femur (Arrows).
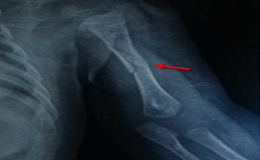
Figure 2: Showing fracture at humerus (Arrow).

Figure 3: Gene testing report.
3. Discussion
Osteogenesis Imperfecta (OI) is a genetic disorder comprising a heterogeneous group of diseases. It is a pleiotropic disorder of bone fractures ranging from a slight fracture to prenatal fracture. Also, the presence of blue sclera, dentinogenesis Imperfecta (DI), hyperlaxity of ligaments and skin, hearing impairment, short stature, and bone deformities are evident by the studies. It is a rare disorder, with an incidence rate of 1 in 10,000-20,000 births [1]. Lobstein called it as “osteopsathyrosis idiopathica”, “fragilitus osseum” and hereditary fragility of bone, these names are further replaced by brittle bone disease and fragile bone disease. Vrolik in 1845 renamed the disease Osteogenesis Imperfacta, as being called in the literature [2, 3].
OI has a wide range of presentations of dentinogenesis Imperfecta. Type II is autosomal recessive and is usually lethal in the newborn period with multiple congenital fractures, small deformed limbs, and severe lung disease. Type III and IV are the intermediate phenotypes with type III being the most severe non-lethal form which can be congenital or can cause multiple fractures ultimately leading to severe bone deformity. Type IV, is an intermediate phenotype of OI, but similar to type I. in 1979, Sillence, et al.[4] classified the disorder into four different categories: Type I; dominantly inherited OI with blue sclera as was seen in our patient, type II; perinatally lethal, deforming OI with crumpled femora, type III; progressively deforming OI with normal sclera and type IV; dominantly inherited OI with normal sclera. Type I is a mild form with recurrent fractures and blue sclera, which was further classified into two different sub-categories on the basis classified into sub-group A and B based on the presence and absence of dentinogenesis Imperfecta.
In the last couple of decades, due to an advancement in the field of histology and molecular biology Ward et al classified it further into four more sub-groups from type V to type VIII [5, 6]. Type V, moderate deforming with normal teeth along with the presence of blue sclera; type VI, moderate disease with fish scale pattern of bone lamellation which was unique to this category along with normal sclera and teeth; type VII, which is clinically similar to type II, with the exception that the patients have a smaller head and normal sclera; and type VIII, patients exhibit defects in growth and mineralization. Plotkin re-classified OI secondary to mutations in type I pro-collagen genes and “syndromes resembling osteogenesis Imperfecta” other than the involvement of type I pro-collagen genes [7].
Several factors played an important role in determining the severity of OI, which also includes amino acid substitution [8]. More than 90% of patients with OI exhibit heterozygous mutations in collagen α-1(I) chain (COL1A1) or collagen α-2(I) chain (COL1A2), genes encoding two types I pro-collagen α chains, pro-α1(I) and pro-α2. Translated collagen α chains include N- and C-terminals and a core triple-helical domain of the Gly-X-Y triplet repeat unit. The C-terminus is involved in the reorganization and assembly of type I collagen. N-terminal cleavage results in the formation of the α-1 chain which is terminated by an N-telopeptide [9, 10]. All N-termini adhere to the surface of the fibrils preventing further accretion and limiting fibril diameter. Genetics not only plays an important role in causing OI, but environmental factors can also act as a triggering factor leading to the onset of OI, including smoking in the pregnant female as shown in the article, which was not seen in our patient [11, 12]. COL1A1 and COL1A2 are ~18 kb and 38 kb long, respectively and are located in 17q21.3-17q22, 7q21.3-7q22.3, respectively, both with >50 exons in contrast to our subject which includes only 16 the location of the mutation on a specific polypeptide chain, also a mutation in any particular exons.
In our study, our patient who was a 6-month-old baby boy, presented to us with multiple bone fractures, blue sclera, umbilical hernia with the absence of dentinogenesis Imperfecta, and his blood calcium level was done which came out to be 10.16 mg/dl, the upper limit of normal laboratory range. A blood sample was sent for gene sequencing which came out to be COL1A1 positive having a nucleotide change of c.1012G>A, and amino of p.Gly338Ser revealing that the amino acid on 338 positions which was supposed to be glycine acid mutation was replaced by serine in our case, which was a unique thing to be found in the patient of OI, and as Glycine was critical for COL1A1 gene, its replacement leads to the patient presented as a homozygous case of OI.
After the patient being diagnosed with OI, an evaluation was done by the multidisciplinary team which includes Physiotherapy, rehabilitation, and orthopedic surgery. The goal of multimodality therapy is to attain a normal quality of life for the patient [13]. The medical treatment usually starts with oral and intravenous Bisphosphonate (BPs) being followed by antiresorptive agents. Clinical trials have indicated the effectiveness of BPs in the improvement of Bone Mass Density and the alleviation of symptoms that the patient was exhibiting [12]. Furthermore, GH and PTH analogs can also be used to manage the refractory cases but before their usage, the patient should be analyzed as these drugs might have a wide range of adverse effects on the human body [12]. Surgical intervention is only used if medical therapy fails in the patient. In our case, no deformities were observed and the patient gave a positive response to Pamidronate, calcium and Vitamin D, so surgical intervention was not required. Additionally, after the patient was discharged from hospital, Physiotherapy and physical activity were advised to augment the patient to improve the quality of life by preventing the formation of contracture or atrophy of the muscles of the lower limbs [14].
There is no doubt that medical treatment has led to some miraculous improvement of the children with OI, ultimately, but unfortunately, it is not a definitive cure. A definitive has been hypothesized which includes the elimination of the mutated gene. The Gene therapy approach is being investigated as a potential for future treatments of OI [15]. Until then, palliative treatment remains the only option.
Financial support statement
This work is not supported by any sponsors. No funding required in this study.
Declaration of conflict of interest
The authors declare no conflicts of interest with this article's content.
References
- Raunch F, Glorieux FH. Osteogenesis imperfecta. Lancet 363 (2004): 1377-1385.
- Brailsford JF. Osteogenesis Imperfecta. The British Journal of Radiology 16 (1943): 129-136.
- Robichon J, Germain JP. Pathogenesis of osteogenesis imperfecta. Canad. Med. Ass. J 99 (1968): 975-979.
- Glourieux FH. Osteogenesis imperfecta. Best Practice and Research Clinical Rheumatology 22 (2008): 85-100.
- LM Ward, F Rauch, R Travers, et al. Osteogenesis Imperfecta Type VII: An Autosomal Recessive Form of Brittle Bone Disease. Bone 31 (2002): 12-18.
- Glorieux FH, Rauch F, Plotkin H, et al. Type V Osteogenesis Imperfecta: A New Form of Brittle Bone Disease. J Bone Miner Res 15 (2000): 1650-1658.
- Plotkin H. Syndromes with congenital brittle bones. BMC Pediatrics 4 (2004): 1-6.
- Byers PH, Wallis GA, Willing MC. Osteogenesis imperfecta: Translation of mutation to phenotype. J Med Genet 28 (1991): 433-442.
- Forlino A, Cabral WA, Barnes AM, et al. New perspectives on osteogenesis imperfecta. Nat Rev Endocrinol 7 (2011): 540-557.
- Boot-Handford RP, Tuckwell DS. Fibrillar collagen: The key to vertebrate evolution? A tale of molecular incest. BioEssays 25 (2003): 142-151.
- Starr SR, Roberts TT, Fischer PR. Osteogenesis imperfecta primary care. Pediatr Rev 31 (2010): 54-64.
- Monti E, Mottes M, Fraschini P, et al. Current and emerging treatments for the management of osteogenesis imperfecta. Ther Clin Risk Manag 6 (2010): 367-381.
- Engelbert RH, Pruijs HE, Beemer FA, et al. Osteogenesis imperfecta in childhood: treatment strategies. Arch Phys Med Rehabil 79 (1998): 1590-1594.
- Zeitlin L, Fassier F, Glorieux FH. Modern approach to children with osteogenesis imperfecta. J Pediatr Orthop B 12 (2003): 77-87.
- Niyibizi C, Wang S, Mi Z, et al. Gene therapy approaches for osteogenesis imperfecta. Gene Ther 11 (2004): 408-416.

 Impact Factor:
* 1.7
Impact Factor:
* 1.7
 Acceptance Rate:
74.36%
Acceptance Rate:
74.36%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
