Study on Antimicrobial Action of Citric Acid on Campylobacter Jejuni in Reared Chicken Carcass
Marwa AbdElhamed1, Abeed A Saleh2, Elmossalami MK3, Mona M Sobhy4 *
1Researcher at Egypt center research and regenerative medicine
2Prof of Meat Hygiene and Precedence of Damanhur University, Damanhur, Egypt
3Meat Hygiene Dept. Faculty of Vet. Med. Cairo uni Giza, Egypt
4Reproductive diseases research Dept. anim. Reprod. Res. Inst (ARRI), AGR. Giza, Egypt
*Corresponding Author: Mona M Sobhy, Reproductive diseases research Dept. anim. Reprod. Res. Inst (ARRI), AGR. Giza, Egypt
Received: 13 July 2021; Accepted: 23 July 2020; Published: 15 September 2021
Article Information
Citation: Marwa AbdElhamed, Abeed A Saleh, Elmossalami MK, Mona M Sobhy. Study on Antimicrobial Action of Citric Acid on Campylobacter Jejuni in Reared Chicken Carcass. Archives of Veterinary Science and Medicine 4 (2021): 55-66.
View / Download Pdf Share at FacebookAbstract
A total of 200 chicken samples (breast and thigh muscles) obtained from retail broilers at different localities in Egypt. All samples were transported as soon as possible to the lab in Animal Reproduction Research Institute and subjected to bacteriological examination for C. jejuni. All samples examined by conventional methods for isolation and identification of Campylobacter. Samples subjected to standard phenotypic identification of C. jejuni by PCR using specific primers of hippuricase gene. The high incidence of C. jejune breast meat samples was (20%) then thigh samples (14%). The high incidence of C. jejune in breast samples in Giza (22 %) followed by Cairo (18%). The high incidence of C. jejune was in Giza (16 %), and Cairo (12%). All 68 C. jejuni isolates yielded the genus specific (16S rRNA) 63 isolates C. jejuni specific 323 bp. Amplicons 555 bp and 495 bp were confirmed to belong to C. jejuni cytolethal distending toxins showed at 555 bp for CdtC and at 495 bp for CdtB. The virulence genes of C. jejuni (FlaA, virB11) detected at 855 bp and 494 bp respectively. PCR analysis of Campylobacter virulence markers can be utilized as a simple and rapid tool especially when used as profile analysis for a control strategy. The effect of the citric acid was correlated with the level of pH, in chicken muscles in BHI broth at 4ºC after 4 hours of experiment. C. jejune (55.9%) strains was reduction after 30 min of experiment, (76.5%) after one hour, (89.7%) after 2 hours , (97%) after 3 hours and 100% completely reduction after 4 hours at pH changed from 2.5 to 3.5. at the end of experiment. The experiment of inhibitory effect of citric acid in chicken muscles in brain heart infusion (BHI) broth was done on positive muscle samples and pH measurements. Finally, citric acid and their active may be used as an alternative to use of antibiotic performance. Further research is needed regarding understanding mechanism of action, identifying means to standardize the eff
Keywords
<p>Campylobacter jejune; Citric acid; Chicken meat</p>
Article Details
1. Introduction
Campylobacter jejuni is related to the poultry digestive system and to foodborne infection [1]. Campylobacter jejuni is considered the dominant species in chicken [2]. A limited number of studies have reported possible negative health implications in chickens caused by C. jejuni colonization of the gut, therefore this bacterium is considered to have a commensal relationship with chicken [3].
Campylobacteriosis is a significant enterocolitis of people, frequently acquired through consumption of undercooked poultry meat contaminated with C. jejuni [4]. Most human cases of campylobacteriosis are attributed to contaminated poultry [5]. Campylobacter infections in humans are usually characterized by self-limiting watery/bloody diarrhea, abdominal cramps, nausea and fever bacteremia and other extra intestinal complications may develop [6].
Broiler flocks affected with C jejuni, the prevalence can vary from 0% to 100% depending on season (lowest in fall and winter and highest in summer). C jejuni has been found in all areas of poultry production [4]. However, Lynch et al. (2011) [7] , reported that most Campylobacter were isolated from beef chicken (16%). Efforts have been made across the supply chain; including farms, processing plants, retailers, and through educating the public. Collectively, results suggest that Campylobacter is often present in broilers in a poor health condition, therefore suggesting that the mortality rate could be an efficiency marker for farm management practices and biosecurity [8].
Environmental contamination with bird droppings is probably the most common source of infection for dissemination of C jejuni. Campylobacter can be transmitted vertically, either on the surface of eggs or by transovarial transmission [9]. Non-chlorinated water shallow well should be regarded as a possible source. Houseflies can be a transmission source for flocks; equipment and footwear contaminated with feces from an infected source as a vehicle of transmission. Young chicks are easily colonized when exposed to C jejuni and can excrete the organism in the feces for their lifetimes [10].
Contamination of chicken meat with Campylobacter jejuni needs hygienic measures for decontamination in poultry processing. The treatment of chicken meats with plant-derived compound as citric acid, which is recognized as a safe , advisable for minimizing the initial level of Jejuni and extending the shelf life of chicken parts by reducing total microbial number [11]. Therefore, the aim of this work was studied the effect of different dilution of citric acid on the survival of C. jejuni inoculated experimentally in chicken meat samples.
2. Materials and Methods
2.1 Samples
A total of 200 chicken samples (breast and thigh) obtained from retail broilers at different localities in Egypt. All collected samples were transported as soon as possible to Campylobacter lab in Animal Reproduction Research Inst.El-Haram and subjected to bacteriological investigation for C.jejuni.
|
Locality |
Total No. of Chicken Samples |
No. of Chicken Samples |
|
|
Breast muscles |
Thigh muscles |
||
|
Cairo |
100 |
50 |
50 |
|
Giza |
100 |
50 |
50 |
|
Total |
200 |
100 |
100 |
Table (A): Number of chicken meat samples for isolation of C. jejuni.
2.2 Bacteriological examination
2.2.1 Isolation and identification of Campylobacter: A loopful of samples was plated on semisolid Thioglycollate broth (Oxoid) and incubated in microaerophilic atmosphere (5% O2, 10% CO2 and 85% N2) at 37°C and 42°C for 48 -72hrs.
Microscopic examination for the incubated samples was done for detection of Campylobacter microorganisms under phase contrast microscope to examine the characteristic motility. All The isolates were subjected to Gram’s staining and biochemical tests, such as catalase, oxidase, urease, nitrate reduction and indole acetate hydrolysis, hippurate hydrolysis test and susceptibility to Cephalothin and nalidixic acid by the disc diffusion method Holt et al, (1994).
2.3 Molecular characterization of Campylobacter
2.3.1 Oligonucleotide primers used in qPCR: Five pairs of primers were supplied from (Metabion). They have specific sequence and amplify specific products as shown in Table (2).
|
Target gene |
Primer sequence (5'-3') |
Length of amplified product |
Reference |
|
C. jejuni hipO |
ACTTCTTTATTGCTTGCTGC |
323 bp |
Wang et al., 2002 |
|
GCCACAACAAGTAAAGAAGC |
|||
|
FlaA |
AATAAAAATGCTGATAAAACAGGTG |
855 bp |
Datta et al., 2003 |
|
TACCGAACCAATGTCTGCTCTGATT |
|||
|
virB11 |
TCTTGTGAGTTGCCTTACCCCTTTT |
494 bp |
- |
|
CCTGCGTGTCCTGTGTTATTTACCC |
|||
|
CdtB |
GTTAAAATCCCCTGCTATCAACCA |
495 bp |
Bang et al., 2003
|
|
GTTGGCACTTGGAATTTGCAAGGC |
|||
|
cdtC |
TGGATGATAGCAGGGGATTTTAAC |
555 bp |
- |
|
TTGCACATAACCAAAAGGAAG |
Table (B): Oligonucleotide primers sequences.
2.3.2 Extraction of DNA: according to QIAamp DNA mini kit: Twenty μl QIAGEN protease were pipetted into the bottom of a 1.5 ml micro centrifuge tube. 200 μl of the sample was added. 200 μl buffer AL were added to the sample, mixed by pulse vortexing for 15 seconds. The mixture was incubated at 56°C for 10 min.
The 1.5 ml micro centrifuge tube was centrifuged to remove drops from the inside of the lid. 200 μl ethanol (96%) were added to the sample, and mixed again by pulse vortexing for 15 seconds. After mixing, the 1.5 ml micro centrifuge tube was briefly centrifuged to remove drops from the inside of the lid.
The mixture from step 6 was carefully applied to the QIAamp mini spin column (in a 2ml collecting tube) without wetting the rim. The cap was closed, and centrifuged at 8000 rpm for 1 min. The QIAamp mini spin column was placed in a clean 2 ml collection tube, and the tube containing the filtrate was discarded. The QIAamp mini spin column was carefully opened and 500 ml buffer AW1 was added without wetting the rim. The cap was closed, and centrifuged at 8000 rpm for 1 min. The QIAamp mini spin column was placed in a clean 2 ml collection tube, and the tube containing the filtrate was discarded. The QIAamp mini spin column was carefully opened and 500 ml buffer AW2 was added without wetting the rim.
The cap was closed, and centrifuged at full speed for 3 min. The QIAamp mini spin column was placed in a new 2 ml collection tube and the old collection tube was discarded with the filtrate.
Centrifugation at full speed for 1 min was done. The QIAamp mini spin column was placed in a clean 1.5 ml micro centrifuge tube, and the collection tube containing the filtrate was discarded. The QIAamp mini spin column was carefully opened and 100 μl buffer AE were added. The QIAamp mini spin column was Incubated at room temperature (15-25°C) for 1 min, and then centrifuged at 8000 rpm for 1 min [12].
2.3.3 Preparation of duplex PCR master: Mix for each of (C. jejuni hipO), (cdtB and cdtC) and (virB11 and flaA) genes according to GoTaq® Hot Start Green Master Mix.
Table (C): Cycling conditions of the different primers during PCR.
2.3.4 Agarose gel electrophoreses [13]: Electrophoresis grade Agarose (1.5 g) was prepared in 100 ml TBE buffer in a sterile flask, 0.5μg/ml Ethidium bromide was added and mixed thoroughly. Twenty μl of each PCR product samples, negative control and positive control were loaded to the gel. The gel was photographed by a gel documentation system and the data was analyzed.
2.4 Preparation of C. jejuni inoculum
Campylobacter jejuni strains were maintained at 70ºC in BHI broth containing 15% glycerol. During propagation and growth, all plates were incubated at 37ºC in sealed gas jars under microaerobic conditions (5% O2, 10 % CO2, and 85% N2). Cultures were prepared by growing the strains for 3 to 4 days on blood agar base (Oxoid) containing 5% sheep blood. One loopful of each culture was subsequently streaked onto blood agar base plates, which were incubated for 24 h. The cells were harvested with 2 ml of phosphate-buffered saline (Oxoid). The inoculum was diluted based on the observation that the optical density at 600 nm, the inoculum was diluted to approximately 7 logs CFU/ml in either chicken muscle was ready for use [14]).
2.5 Experiment by using Citric acid
2.5.1 Preparation of chicken muscles: Chicken breast and thigh muscles were obtained from retail broilers and stored at low temperatures (5 and 10 degrees C) C. jejuni remained viable in chicken juice for a remarkably longer period of time.
2.5.2 Inhibitory effect of citric acid in chicken meat and BHI broth: Four-milliliter of the inoculum prepared in chicken muscles or BHI broth were transferred into sterile tubes, and 100 ml from a 20% stock solution lemon juice (citric acid) was pipetted into the tubes, resulting in an organic acid concentration of 0.5%. The tubes were immediately stored at 4ºC. After 0, 10, 20, 30 minutes.1, 2, 3 and 4 h of storage, samples were collected to determine populations of C. jejuni. Two replicate experiments were conducted.
2.5.3 PH measurement: The values of pH for all chicken muscles with citric acid were measured after 5 to 10 min and after 1, 2, 3 and 4 h of experiment.
3. Results
3.1 Identification of Campylobacter jejuni by culture method
|
Localities |
Number of examined samples |
Total Positive Campylobacter jejuni % |
Positive C. jejune in breast % |
Positive C. jejune in thigh% |
|
Cairo |
100 |
30 30% |
18 18% |
12 12% |
|
Giza |
100 |
38 38% |
22 22% |
16 16% |
|
Total |
200 |
68 34% |
40 20% |
28 14% |
Table 1: Occurrence of C. jejuni in chicken samples (breast and thigh meat) by conventional method.
|
C. jejuni |
Characteristics |
|
+ + + -- + + + + S R |
Oxidase Catalase Nitrate reduction Urease Hippurate hydrolysis Growth at: 37°C 43°C Growth at 1% glycine Susceptibility to: Nalidixic acid Cephalothin |
Table 2: Biochemical tests to detect C. jejuni.
3.2 Molecular typing of Campylobacter
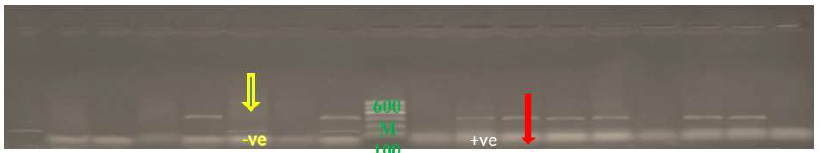
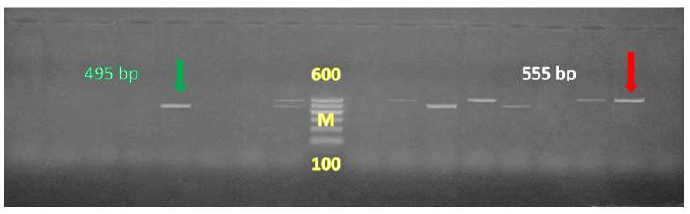
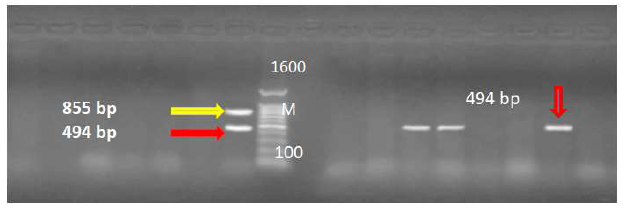
Sixty eight positive samples for C. jejuni were examined by Multiplex PCR for detection of C. jejuni and virulence strains of C. jejuni and cytolethal distending toxins (Figures 1, 2 and 3).
3.3 Inhibitory effect of citric acid in chicken muscles
|
Type of samples |
Positive samples |
15 min |
30 min |
One hr. |
2 hrs. |
3 hrs. |
4 hrs. |
|
Breast meat |
40 |
15 37.5% |
23 57.5% |
30 75% |
35 87.5% |
38 95% |
40 100% |
|
Thigh meat |
28 |
12 42.9% |
15 53.6% |
22 78.6% |
26 92.9% |
28 100% |
28 100% |
|
Total |
68 |
27 39.7% |
38 55.9% |
52 76.5% |
61 89.7% |
66 97% |
68 100% |
Table 3: Relative reduction of C. jejuni strains exposed to Citric acid in BHI at 4 º C.
|
Type of examined samples |
Total Positive C. jejuni |
After 5 min. of Exp. pH |
After 10 min. of Exp. pH |
After 1 hrs. of Exp. pH |
After 2 hrs. of Exp. pH |
After 3 hrs. of Exp. pH |
|
Chicken muscles |
68 |
2.5 |
2.7 |
3 |
3.3 |
3.5 |
Table 4: pH measurements exposed to Citric acid in BHI at 4 º C.
4. Discussion
Consumption of contaminated chicken meat products have been identified as major sources of C. jejuni for humans [15]. Chicken carcass contaminated with C. jejuni during processing by faecal [16, 17]. The cross-contamination between slaughtered flocks and processing of chicken leads to higher contamination with C. jejuni [18]. To reduce human exposure to C. jejuni is by controlling the pathogen at the farm level in the chicken flocks, by effective control measures.
Numerous control strategies have been tested to reduce C. jejuni contamination in chicken, such as farm biosecurity, vaccination, therapy, and the use of feed additives [19]. Non-antibiotic feed additives, such as organic acids and essential oils, were tested for their action on C. jejuni, showing varying efficacy. Essential oil extracts, such as Eucalyptus, Valencia orange and cinnamon, are very potent against C. jejuni in vitro [20]. Organic acids such as formic acid, lactic and acetic acid are bactericidal against C. jejuni in vitro and are also effective at some degree in vivo [21].
The high incidence of C. jejuni in chicken breast muscles samples was (20%) followed by chicken thigh muscles samples (14%) (Table 1). The high incidence of C. jejuni in chicken breast meat samples in Giza (22 %) and Cairo (18%). But, C. jejune in chicken thigh samples was high incidence in Giza (16 %) and Cairo (12%) Table 1. C. jejuni carcass contamination level of 4.7 log CFU/carcass [22, 23]. C. jejuni caecal was contaminated the carcass during slaughtered and evisceration processing [24].
Conventional techniques used for the detection and identification of Campylobacter species are laborious, time-consuming and problems of contamination are often encountered [25, 26]. Polymerase Chain Reaction (PCR) technique is widely used of all molecular techniques, it is highly sensitive, specific and rapid for the detection of food-borne pathogens [27]. All the 68 Campylobacter isolates yielded the genus specific (16S rRNA) 63 isolates C. jejuni specific 323 bp in Figure 1. Amplicons 555 bp and 495 bp were confirmed to belong to C.jejuni cytolethal distending toxins showed at 555 bp for CdtC and at 495 bp for CdtB of the genus Campylobacter (Figure 2).
Therefore, the isolates obtained were further confirmed by PCR analysis whereas most of the strains contained the CDT genes. These results detected by Rajagunalan et al., (2014) [28]. Virulence gene markers were identified by multiplex PCR. The results showed that isolates of C. jejuni contained the flaA gene sequences. The virulence genes of C. jejuni (FlaA, virB11) have shown at 855 bp and 494 bp respectively (Figure 3). The virB11 gene was present only in (14.7%) of the analyzed isolates. These results agreed with Wieczorek and Osek (2011) [29]. PCR analysis detection of Campylobacter virulence markers can be utilized as a simple and rapid tool to recover from different sources, especially when used as profile analysis for a control strategy [30].
The present study analyzed the diversity of C.jejuni population in chicken to monitor the contamination process throughout the farm, slaughter, and post chilling phases. There are several studies in literature assessing the use of plant extracts, as antimicrobials, antioxidants, or digestibility enhancers in animal feeds. Dietary organic acids have been considered as antibacterial nature. Chemically, organic acids used in food animal production described as simple monocarboxylic acids (e.g., formic, acetic, propionic, and butyric acids) or carboxylic acids (e.g., lactic, malic, tartaric, and citric acids) [31].
The effect of the citric acid was correlated with the level of pH, in chicken meat (Table 3). The result was in a greater reduction of C. jejuni in breast and thigh meat in BHI broth at 4ºC after 4 hours of experiment. As of the 68 C. jejuni strains tested, only C. jejune 38 (55.9%) strains was reduction after 30 min of experiment, 52 (76.5%) required one hour for reduction, 61 (89.7%) after 2 hours , 66 (97%) after 3 hours and 100% completely reduction after 4 hours. Survival of C. jejuni in the presence of citric acid observed for stresses at pH 3.5 (Table 4). These findings confirm that citric acid influences C. jejuni [32].
In (Table, 4) found that the antibacterial effect of citric acid on C. jejune on chicken meat. Lowering pH by adding citric acid onto the chicken meat surface, observed a rapid rise in pH within a few minutes, which to some extent neutralized the antibacterial effect with an acceptable taste of the prepared meat.
Perko-Ma¨kela¨ et al. (2000) [33] found that low pH is necessary to obtain a rapid effect for reduction of C. jejuni population in broiler meat reported positive scores with regard to taste and texture. Citric acid is disrupting the outer membrane and initiate cell lysis with an increase in permeability of C. jejuni [34].
Finally, this study shows that citric acid and their active principles may be used as an alternative to the use of antibiotic performance. Further research is needed regarding understanding mechanism of action, identifying means to standardize the effects, improving delivery methods, and increasing their in vivo efficacy.
References
- Cean A, Stef L, Simiz E, et al. Effect of human isolated probiotic bacteria on preventing Campylobacter jejuni colonization of poultry. Foodborne Pathog. Dis 12 (2015): 122-130.
- Corcionivoschi N, Alvarez LA, Sharp TH, et al. Mucosal reactive oxygen species decrease virulence by disrupting Campylobacter jejuni phosphotyrosine signaling. Cell Host Microbe 12 (2012): 47-59.
- Thibodeau A, Fravalo P, Yergeau É, et al. Chicken caecal microbiome modifications induced by Campylobacter jejuni colonization and by a non-antibiotic feed additive. PLoS One 10 (2015): e0131978.
- Lee MD. Avian Campylobacter MSD and the MSD Veterinary Manual. Merck & Co., Inc., Kenilworth, NJ, USA (2019).
- Skarp CPA, Hanninen ML, Rautelin HIK. Campylobacteriosis: The role of poultry meat. Clinical Microbiology and Infection 22 (2016): 103-109.
- Blaser MJ, Engberg J. Clinical aspects of Campylobacter jejuni and Campylobacter coli infections, In Nachamkin I, Szymanski CM, Blaser MJ. (Ed), Campylobacter. ASM Press, Washington, DC (2008): 99-121.
- Lynch OA, Cagney C, McDowell DA, et al. Occurrence of fastidious Campylobacter in fresh meat and poultry using an adapted cultural protocol. Int. J. Food Microbiol 150 (2011): 171-177.
- Oh J, Harper M, Giallongo F, et al. Effects of rumen?protected Capsicum oleoresin on immune responses in dairy cows intravenously challenged with lipopolysaccharide. J Dairy Sci 100 (2017): 1902-1913.
- Georgiev M, Beauvais W, Guitian J. Effect of enhanced biosecurity and selected on-farm factors on Campylobacter colonization of chicken broilers. Epidemiology and Infection 145 (2016): 553-567.
- Ugarte-Ruiz M, Dominguez L, Corcionivoschi N, et al. Exploring the oxidative, antimicrobial and genomic properties of Campylobacter jejuni strains isolated from poultry. Res. Vet. Sci 119 (2018): 170-175.
- Cosansu S, Ayhan K. Effects of lactic and acetic acid treatment on Campylobacter jejuni inoculated onto chicken leg and breast meat during storage at 4°c and - 18°c. Journal of food processing and preservation 34 (2008): 98-113.
- Iroala G, Hernández M, Calleros L, et al. Application of a multiplex PCR assay for Campylobacter fetus detection and subspecies differentiation in uncultured samples of aborted bovine fetuses. J Vet Sci 13 (2012): 371-376.
- Sambrook J, Fritscgh EF, Mentiates F. Molecular coloning. A laboratory manual. Vol Cold spring Harbor Laboratory press, New York (1989).
- Birk T, Gronlund CC, Christensen BB, et al. Effect of Organic Acids and Marination Ingredients on the Survival of Campylobacter jejuni on Meat. Journal of Food Protection 73 (2010): 258-265.
- Thibodeau A, Philippe F, Robert G, et al. Modification of Campylobacter jejuni Broiler Colonization by a Feed Additive Composed of Encapsulated Organic Acids and Essential Oils. Journal of Agricultural Science and Technology A 4 (2014): 853-864.
- Normand V, Boulianne M, Quessy S. Evidence of Cross-Contamination by Campylobacter of Broiler Carcasses Using Genetic Characterization of Isolates. Can. J. Vet. Res 72 (2008): 396-402.
- Ellerbroek LI, Lienau JA, Klein G. Campylobacter in Broiler Flocks at Farm Level and the Potential for Cross-Contamination during Slaughter. Zoonosis Public Health 57 (2010): 81-88.
- Bily L, Petton J, Lalande F, et al. Quantitative and Qualitative Evaluation of Campylobacter Contamination of Turkey Cecal Contents and Carcasses During and Following the Slaughtering Process. J. Food. Prot 73 (2010): 1212-1218.
- Hermans D, Van Deun K, Messens W, et al. Campylobacter Control in Poultry by Current Intervention Measures Ineffective: Urgent Need for Intensified Fundamental Research. Vet. Microbiol 152 (2011): 219-228.
- Nannapaneni R, Chalova VI, Crandall PG, et al. Campylobacter and Arcobacter Species Sensitivity to Commercial Orange Oil Fractions. Int. J. Food Microbiol 129 (2009): 43-49.
- Molatova, Skrivanova E, Bare J, et al. Effect of coated and non-coated fatty acid supplementation on broiler chickens experimentally infected with Campylobacter jejuni. J. Anim. Physiol. Anim. Nutr. (Berl.) 95 (2011): 701-706.
- Allen VM, Bull SA, Corry JE, et al. Campylobacter Contamination of Chicken Carcasses during Processing in Relation to Flock Colonisation. Int. J. Food Microbiol 113 (2007): 54-61.
- Hue O, Allain V, Laisney MJ, et al. Campylobacter Contamination of Broiler Caeca and Carcasses at the Slaughterhouse and Correlation with Salmonella Contamination. Food Microbiol 28 (2011): 862-868.
- Bi P, Cameron AS, Zhang Y, et al. Weather and notified Campylobacter infections in temperate and sub-tropical regions of Australia: an ecological study. Journal of Infection 57 (2008): 317e 23.36.
- Williams LK, Jørgensen F, Grogono-Thomas R, et al. Enrichment culture for the isolation of Campylobacter spp: Effects of incubation conditions and the inclusion of blood in selective broths. Int J Food Microbiol 130 (2009): 131-134.
- Alonso R, Girbau C, Astorga AF. Campylobacter. In Molecular Detection of Human Bacterial Pathogens ed. Liu D (2011): 1125-1139.
- Samosornsuk W, Asakura M, Yoshida E, et al. Evaluation of a Cytolethal Distending Toxin (cdt) Gene-Based Species-Specific Multiplex PCR Assay for the Identification of Campylobacter Strains Isolated from Poultry in Thailand. Microbiol Immunol 51 (2007): 909-917.
- Rajagunalan S, Garima Bisht, Sheetal Pant, et al. Prevalence and molecular heterogeneity analysis of Campylobacter jejuni and Campylobacter coli isolated from human, poultry and cattle, in Pantnagar, India. Veterinarski Arhiv 84 (2014): 493-504.
- Wieczorek K, Osek J. Molecular characterization of Campylobacter isolated from poultry faeces and carcasses in Poland. Acta veterinaria Brno (2011).
- French N. Molecular Epidemiology and Veterinary Public Health Group Enhancing surveillance of potentially foodborne enteric diseases in New Zealand: Human campylobacteriosis in the Manawatu. Palmerston North (NZ) (2008).
- Dibner JJ, Buttin P. Use of organic acids as a model to study the impact of gut microflora on nutrition and metabolism. Journal of Applied Poultry Research 11 (2002): 453-463
- Chan KF, Tran HL, Kanenaka RY, et al. Survival of clinical and poultry-derived isolates of Campylobacter jejuni at a low temperature (4uC). Appl. Environ. Microbiol 67 (2001): 4186-4191.
- Perko-Ma¨kela¨ P, Koljonen M, Miettinen M, et al. Survival of Campylobacter jejuni in marinated and nonmarinated chicken products. J. Food Saf 20 (2000): 209-216.
- Salem LMA, Nashwa O. Khaleafa, et al. Antimicrobial resistance of Campylobacter jejuni isolated from chicken, some animal products and human in Kalyoubia, Egypt, with special reference to its viability. Benha Veterinary Medical Journal 36 (2019): 156-163.

 Impact Factor: * 1.1
Impact Factor: * 1.1 Acceptance Rate: 80.20%
Acceptance Rate: 80.20%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 