Thoracolumbar Fascia Enthesopathy as a Cause of Low Back Pain: A Retrospective and Follow-up Study
Ouidade A Tabesh1, Roba Ghossan2, Soha H Zebouni3, Rafic Faddoul4, Michel Revel5, Fouad Fayad6 *
1Department of Rheumatology, Bellevue Medical Center, Saint Joseph University of Beirut, Lebanon.
2Department of Rheumatology, Hotel Dieu de France Hospital, Saint Joseph University of Beirut, Lebanon.
3Department of Radiology, Hotel Dieu de France Hospital, Saint Joseph University of Beirut, Lebanon.
4Higher School of Engineers of Beirut and Saint Joseph University of Beirut, Lebanon.
5Paris Descartes University, Sorbonne Paris Cite, Paris, France.
6Department of Rheumatology, Hotel-Dieu de France Hospital, Saint Joseph University of Beirut, Lebanon.
*Corresponding Author: Dr. F Fayad, Department of Rheumatology, Hotel Dieu De France Hospital, Saint Joseph University of Beirut, Alfred Naccache Street, Achrafieh, Beirut, P.O. Box 166830-Lebanon.
Received: 03 July 2021; Accepted: 12 July 2021; Published: 15 August 2021
Article Information
Citation:
Ouidade A Tabesh, Roba Ghossan, Soha H Zebouni, Rafic Faddoul, Michel Revel, Fouad Fayad. Thoracolumbar Fascia Enthesopathy as a Cause of Low Back Pain: A Retrospective and Follow-up Study. Fortune Journal of Rheumatology 3 (2021): 41-52.
View / Download Pdf Share at FacebookAbstract
Aim: To evaluate the ultrasonographic findings of Thoracolumbar Fascia (TLF) enthesis in patients with low back pain (LBP) due to iliac crest pain syndrome (ICPS).
Method: The ultrasonographic and clinical findings of 60 patients with LBP due to ICPS were compared to those of 30 healthy volunteers with no LBP. Thickness of the TLF was measured with ultrasound (US) at its insertion on the iliac crest.
Results: Forty-eight women and 12 men with a mean age of 42.1 ± 11.3 years were diagnosed with ICPS. In patients, the mean thickness of the TLF was 2.51 ± 0.70mm in affected sides compared to 1.81 ± 0.44mm in the contralateral unaffected sides. The mean thickness difference of 0.82mm between the affected and non-affected sides was statistically significant (95%CI, 0.64-0.99, P<0.0001). In volunteers, the mean thickness of the TLF was 1.6 ± 0.2mm. The mean thickness difference of 0.89mm between the affected sides of patients and volunteers was statistically significant (95%CI, 0.73-1.06, P<0.0001). Forty-two patients who didn’t improve with conservative therapy, received injections of methylprednisolone acetate and 1% lidocaine around the TLF enthesis. All patients reported complete relief of their LBP within 20 minutes of the injections thanks to the lidocaine anesthetic effect. Fifty-six (93.3%) patients were reached by phone for a long-term follow-up. Among them, 33 (58.9%) patients experienced a sustained complete pain relief after a mean follow-up of 45 ± 19.3 months (range, 3-74 months).
Conclusion: Our findings suggest that TLF enthesopathy is a potential cause of nonspecific LBP that can be diagnosed using US.
Keywords
<p>Enthesopathy; Diagnosis; Low back pain; Thoracolumbar fascia; Treatment; Ultrasound</p>
Article Details
1. Introduction
Low back pain (LBP) is a common cause of medical visits, with up to 84% of adults experiencing LBP at some time in their lives [1] The estimated prevalence of LBP worldwide is 9,442/100,000 [2]. While most episodes resolve spontaneously within the first two weeks, LBP can be recurrent or chronic, causing sufficient discomfort to interfere with the ability to work and impair the quality of life [3, 4]. In the United States, the cost of LBP to society was estimated as 100 billion US dollars per year in 2006, with two-thirds of this cost due to lost wages and reduced productivity [5].
Musculoskeletal problems are the most common cause of LBP, with anatomic abnormalities identified on imaging studies in about 10-20% of patients with acute LBP and 10-45% of patients with chronic LBP [6-9]. Abnormalities of the vertebral discs, facet joints, and sacroiliac joints are commonly found in these patients [3]. LBP that is not explained by a specific pathology is called nonspecific low back pain [10].
Iliac crest pain syndrome (ICPS) [11] is a clinical entity characterized by nonspecific LBP, localized tenderness on the medial aspect of the posterior iliac crest, and normal conventional radiographic [12] and magnetic resonance imaging (MRI) [13] studies. ICPS has been attributed to abnormalities of the structures attached to the iliac crest, including the iliolumbar ligament, [14-16] lumbar muscles, and multifidus triangle syndrome [17]. About 53% of patients with nonspecific LBP in general practice and 58% in rheumatology clinics have been reported to have ICPS [18]. The role of thoracolumbar fascia (TLF) abnormalities in patients with LBP is not well described [19].
We evaluated patients with LBP and ICPS in order to better characterize the US findings of TLF attachment on the iliac crest and other clinical findings in these patients. A comparison was made with those of healthy volunteers.
We treated patients with LBP due to TLF enthesopathy with conservative treatment and/or local injections of corticosteroids and lidocaine and reported their outcomes.
2. Methods
2.1 Diagnosis
The medical records of every patient with diagnosed ICPS from April 2013 to June 2019 were reviewed. (Table 1). Body Mass Index (BMI) was defined using standard criteria [20].
|
Characteristics |
Patients |
Healthy Volunteers |
P-Value |
|
Participants, N |
60 |
30 |
|
|
Age, mean ± SD (years) |
42.1 ± 11.3 |
41.2 ± 10.6 |
NS |
|
Gender |
|||
|
Female, N (%) |
48 (80) |
24 (80) |
NS |
|
BMI, mean ± SD (kg/m2)* |
24.2 ± 4.2 |
24.7 ± 4.6 |
NS |
|
Underweight, N (%) |
2 (3.1) |
1 (3.3) |
|
|
Normal weight, N (%) |
35 (56.3) |
15 (50) |
|
|
Overweight, N (%) |
14 (28.1) |
10 (33.3) |
|
|
Obesity, N (%) |
6 (12.5) |
4 (13.3) |
|
|
Duration of symptoms (months) |
NA |
||
|
1-6, N (%) |
27 (45) |
||
|
>6-12, N (%) |
12 (20) |
||
|
>12-24, N (%) |
10 (16.7) |
||
|
>24 , N (%) |
11 (18.3) |
||
|
Localization of back pain |
NA |
||
|
Right, N (%) |
34 (56.7) |
||
|
Left, N (%) |
13 (21.7) |
||
|
Bilateral, N (%) |
13 (21.7) |
||
|
Pain radiation |
NA |
||
|
Buttock, N (%) |
19 (31.7) |
||
|
No radiating pain, N (%) |
36 (60) |
||
|
TLF thickness, mean ± SD (mm)** |
|||
|
13 patients had bilateral pain |
|||
|
A. Side with back pain (n = 73) |
2.51 ± 0.70 |
A vs B: <0.0001 |
|
|
B. Unaffected side (n = 45, 2 missing) |
1.81 ± 0.44 |
||
|
C. Healthy volunteers (n = 60 sides) |
1.6 ± 0.20 |
A vs C: <0.0001 |
*: BMI: Body mass index.
**: Thickness of the Thoracolumbar fascia (TLF) was measured with Ultrasound imaging at its insertion on the postero-medial iliac crest on both left and right sides in patients and volunteers.
Ns: Not significant; NA: Not applicable.
Table 1: Characteristics of patients with Iliac crest pain syndrome (ICPS) and healthy volunteers.
The diagnosis of iliac crest pain syndrome due to TLF enthesopathy was suspected on the basis of chronic LBP, tenderness on palpation of the postero-medial aspect of the iliac crest, absence of any neurologic deficit or other abnormality on physical examination. A provocative test for TLF pathology was performed by having the patient perform lateral bending of the lumbar spine away from the affected side. Systemic rheumatic disease was excluded by history, physical exam, and laboratory testing, including determination of CBC, ESR, CRP, creatinine, SGPT, and SGOT levels.
Ultrasound was performed by a single senior radiologist (SHZ) with extensive experience in the ultrasonography of musculoskeletal disease. A Logiq E9 ultrasound machine with a linear probe ML6-15 MHz (General Electric Company, GE France) was used for all imaging studies. US was performed with the patient in the prone position and a pillow placed under the abdomen to straighten the lumbar lordosis. A transverse scan was performed with the ultrasound transducer initially placed over the iliac crest and then moved medially as the probe was changed to an oblique position (Figure 1).
This allowed the visualization of the erector spinal muscles, covered by a hyperechoic band corresponding to the TLF, the region of interest (ROI) is the insertion of the TLF on the iliac crest (Figure 2). Both left and right sides were examined. The thickness of the TLF was measured at its insertion on the postero-medial iliac crest. The presence of calcifications and cortical irregularity were also noted. Other ultrasonographic signs of enthesopathy, including the presence of TLF hypoechogenicity, iliac enthesophytes or erosions, were not assessed in this retrospective study [21].
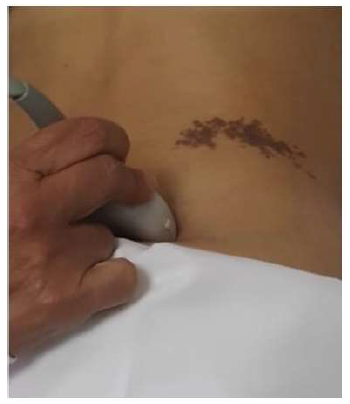
Figure 1: Ultrasound Technique. The ultrasound transducer was initially placed over the iliac crest and moved medially while the probe was rotated to an oblique position to allow visualization of the posterior band of the Thoracolumbar Fascia.
SC = subcutaneous tissue. ES = Erector Spinae muscle.
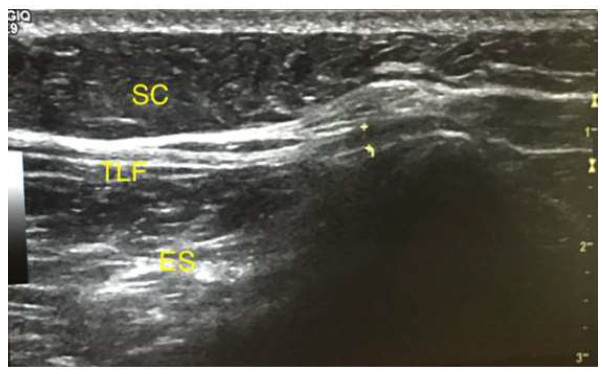
Figure 2: Ultrasound image of the Thoracolumbar Fascia (TLF) in a patient with unilateral Iliac Crest Pain Syndrome. The yellow “+” mark the TLF enthesis. Note the TLF thickening at its insertion on the iliac crest.
2.2 Healthy Controls
Healthy controls with no LBP, matched by age, gender and BMI were prospectively evaluated for the TLF enthesis thickness.
2.3 Treatment
Patients were initially treated with conservative measures after US evaluation, including non-steroidal anti-inflammatory drugs (NSAIDs), analgesics and physical therapy. Patients who failed conservative treatment were treated with one to three monthly injections of 80mg of methylprednisolone acetate (Depo-Medrol®, Pfizer) and 5cc of 1% lidocaine hydrochloride (Lidocaïne®, Laboratoire Renaudin) around the insertion of the TLF at the postero-medial iliac crest. Patients were asked to estimate the percentage of pain improvement 20 minutes after each injection. A positive response to anesthetic injection was scored if the pain intensity improved at least 70% from the baseline pain level reported by the patient when provoked by palpation of the TLF insertion.
All patients were contacted by phone for a follow-up. We noted the long-term improvement and the presence of a pain relapse. For those that we couldn’t reach by phone, we reported their evolution on the last time seen in the clinic after the injection.
The ethical approval for this study was obtained from the ethics committee at Saint Joseph University of Beirut (Ref 2017/97). All methods were carried out in accordance with relevant guidelines and regulations.
2.4 Statistical Analysis
R statistical programming software (Version 1.2.5033) was used for data preprocessing and statistical analysis. Mean and standard deviation (SD) were used for the descriptive presentation of quantitative data. Frequencies were used for the descriptive presentation of categorical data.
Non-parametric Mann-Whitney's test was used to compare TLF thickness between patients affected sides and healthy volunteers’ sides. Non-parametric paired Wilcoxon's test was used to compare TLF thickness between patients affected sides and patients unaffected sides.
An optimal set of predictors was selected for the linear regression used to predict the TLF thickness as the dependent variable. Two optimality criteria were used: (i) maximize the adjusted R2; (ii) minimize the Akaike information criterion (AIC). Both criteria yielded the same set of predictors. The Spearman Correlation Coefficient was used to evaluate the correlation between the improvement after injection and the occurrence of a relapse.
3. Results
3.1 Patients’ characteristics
Forty-eight women and 12 men with a mean age of 42.1 ± 11.3 years (range, 18-63 years) were diagnosed with ICPS (Table 1). The mean BMI was 24.2 ± 4.2 kg/cm2. Twenty patients (35.1%) were overweight or obese. Among the female patients, 28 (58.3%) had a previous pregnancy with a mean number of 1.4 ± 1.4 pregnancies.
Thirty-four (56.7%) patients were affected only on the right side, 13 (21.7%) only on the left side, and 13 (21.7%) on both sides. The duration of symptoms varied between one month and 20 years (median (IQR): 9.5 (3 to 24) months). Thirty-nine (65%) patients were evaluated during their first year of symptoms and 11 (18.3%) after 2 years. All patients had pain in the lower lumbar spine area. Thirty-six (60%) had LBP without irradiation. Nineteen (31.7%) had pain radiating to the buttock, eighteen (30%) to the posterior thigh, six (10%) to the leg and five (8.3%) to the groin. Three men and 10 women (21.7%) had bilateral involvement. These patients had a mean age of 40.9 ± 10.5 years, BMI of 23.7 ± 4.2, similar to patients with unilateral disease.
Fifty-seven (95%) patients underwent a spine and/ or pelvis imaging study (X-ray or MRI). Twenty-seven (45%) patients had plain X-ray imaging of the lumbar spine with or without pelvis and thirty-eight (63.3%) had an MRI of the lumbar spine with or without the pelvis.
3.2 Healthy volunteers’ characteristics
Thirty healthy volunteers with a normal physical exam and no LBP were evaluated (Table 1). There were 24 women and 6 men with a mean age of 41.2 ± 10.6 years and a mean BMI of 24.7 ± 4.6. Fourteen (46.7%) were obese or overweight. Control participants had a similar gender distribution, age, and BMI as ICPS patients (Table 1) and a normal US exam. Among the women volunteers, thirteen (54.2%) had a previous pregnancy with a mean number of 1.2 ± 1.3 pregnancies.
3.3 Ultrasound findings
Passage of the US probe over the affected side reproduced the patients’ typical symptoms of back pain in all ICPS patients on their affected sides. No pain with probe pressure was seen on the unaffected side. US imaging showed thickening of the TLF at its insertion on the postero-medial iliac crest in affected sides in patients (mean thickness, 2.51 ± 0.7 mm; range: 1.2-5 mm; n= 73 sides) (Figure 2). The other US signs that were observed at the insertion of the TLF were calcifications in 10 patients (16.7%) and cortical irregularity in 21 patients (35%). The mean TLF thickness in patients with bilateral involvement was 2.3 ± 0.17 mm (n = 26).
Patients with unilateral ICPS had a thinner TLF enthesis on their contralateral asymptomatic sides (mean thickness, 1.81 ± 0.44 mm; range: 1.0-3.0 mm; n= 45 patient sides, 2 missing values). Mean of the difference affected vs. unaffected sides was 0.81 mm (95% CI, 0.64 -0.99mm, paired Wilcoxon's test, P <0.0001). The mean thickness of the TLF enthesis in healthy volunteers was 1.6 ± 0.2 mm (range: 1.0 - 1.9 mm; n=60 sides). Mean of the difference, affected sides of ICPS patients vs. healthy volunteers was 0.89 mm (95% CI, 0.72 - 1.05 mm, Mann-Whitney's Two Sample test, P <0.0001).
A multiple linear regression was calculated to predict TLF thickness based on gender (Male, Beta = 0.75, P = 0.000284), age (years, Beta=0.013, P=0.036), BMI (kg/cm2, Beta=0.032, P=0.079), degenerative disk disease (Beta=-0.205, P= 0.146), calcifications (Beta=0.28, P=0.15) and cortical irregularities (Beta=0.36, P=0.017). A significant regression equation was found (F (6,50) =10.38, P<0.0001), with an adjusted R2 of 0.501.
3.4 Treatment
Eighteen (30%) patients improved spontaneously or with NSAIDs and/or physical therapy. Forty-two (70%) patients received one to three (median = 2) injections in their affected side at one month-intervals due to persistent or insufficient pain improvement with conservative therapy. Thirty-seven injections in the affected area of the TLF were performed under US-guidance and 59 injections using clinical landmarks. All patients reported complete relief of their LBP within 20 minutes of the injections thanks to the lidocaine anesthetic effect.
3.5 Follow-up
Fifty-six patients were reached by phone for a long-term follow-up. The mean follow-up period was 45 ± 19.3 months (range, 3-74 months). Among them twenty-three (41.1%) reported a relapse of their pain.
Among the 42 patients who received local injections of corticosteroids, 40 had a follow-up. Twenty-three (57.5%) of them experienced a complete pain relief and 25% (10/40) had an improvement from 50 to 90 %. Twenty-one (52.5%) patients had a relapse after the injection. The mean time between the last injection and the relapse was 8.8 ± 7 months (range 1-24 months). We found a negative correlation between the improvement after injections and the occurrence of a relapse (Spearman coefficient -0.47, p <0.05).
3.6 Discussion
The TLF is a deep multilayered connective tissue that covers the erector spinae muscles and separates them from the muscles of the posterior abdominal wall - the quadratus lumborum and psoas major [22]. The TLF is composed of anterior, middle, and posterior layers which fuse caudally and attach to the posterior superior iliac spine and the sacro-tuberous ligament [22]. The posterior layer of the TLF bears most of the stress of forces transferred between the spine, pelvis, and legs [23].
The role of TLF as a source of LBP is not well understood [19] In 2009 and for the first time, Langevin et al. [24] reported abnormalities in the connective tissue structure of the lumbar region in patients with LBP. US imaging showed that affected patients had about 25% greater perimuscular thickness and echogenicity than healthy controls without LBP. In a later study, the shear strain of the TLF was found to be about 16% less in subjects with chronic LBP than in controls [25]. Todorov et al evaluated the attachment of the erector spinae muscle to the posterior iliac crest using US in three studies [26-28].
Patients with unilateral LBP of unidentified cause were found to have increased thickness of the attachment of the erector spinae muscle, more hypoechogenicity, a greater number of calcifications, and more cortical irregularities on the affected side than on the unaffected side and then in healthy volunteers without back pain [27]. It is not certain if the structures observed by Todorov correspond to the TLF enthesis.
Schilder et al injected hypertonic saline or isotonic saline into the erector spinae muscle, the posterior layer of the TLF, or the overlying subcutis of 12 healthy volunteers in order to evaluate the characteristics of pain induced by a simulated injury of the TLF [29]. The TLF was found to be more sensitive to chemical stimulation by hypertonic saline than the underlying erector spinae muscle or overlying subcutis. This pain was mild to moderate in intensity and localized to the site of injection in the TLF and contiguous areas. Radiation of pain to ipsilateral ventral parts of the body was also described. The pain was described as burning, throbbing, and stinging, findings associated with nociceptor nerve fiber stimulation [29].
In addition, the stimulation of the TLF with electrical high-frequency pulses in healthy human subjects was associated with an increase in pain sensitivity [30]. Inducing inflammation in the TLF of rats provoked an increase in the proportion of local neurons responding to stimulation and an increase in the number of nociceptive fibers [31-33]. These findings in animals and humans support the hypothesis that abnormalities of the TLF could result in low back pain.
The most common imaging modalities used to diagnose LBP include CT and MRI [34]. However, US was found to be better than MRI in the diagnosis of enthesopathy of the erector spinae muscles in patients with nonspecific LBP related to ICPS (Sensitivity: US 88% [69-97%] vs. MRI 64% [43-82%]; Specificity: US 80% [60-93%] vs. MRI 80% [59-93%]) [28]. Ultrasound is also safer, cheaper, applicable to a broader range of patients, and more widely available than other imaging modalities [35].
To our knowledge, this is the first study to assess the TLF enthesis abnormalities in patients with LBP. Several elements allow us to suggest the TLF enthesopathy as a cause of LBP. Patients with ICPS treated in our clinic over a 6-year period had significantly thicker TLF enthesis on US in areas with pain than in areas without pain and had significantly thicker TLF enthesis than unaffected healthy volunteers without back pain. About a third of patients responded to conservative therapy and the remaining patients had a high response rate to local injections of lidocaine plus methylprednisolone. All the patients had a positive anesthetic test, which confirms that TLF enthesis is the origin of pain. Furthermore, at the long term follow up, we found that more than half of the patients remained pain free after a conservative treatment and/or local injections.
The positive response to treatment was shown to be negatively associated with the occurrence of a relapse. All these findings together suggest that TLF enthesopathy is a potentially treatable cause of LBP. An appropriate diagnosis with ultrasonography followed by an adequate treatment can relieve the pain of many patients labeled as having chronic nonspecific LBP.
We analyzed the different variables that could be associated with the TLF thickness. We found that the male gender is associated with a thicker TLF enthesis in patients, this could be related to heavier mechanical activities practiced by men compared to women, but these results should be interpreted carefully due to the small number of male patients in our series. The same finding was found with the advanced age, this might be explained by the prolonged mechanical stress on the TLF enthesis with age.
Cortical irregularities were found to be associated with the TLF enthesis thickness, this could reflect the chronicity of the lesions. While an elevated BMI have been reported to be strongly associated with LBP [36], we didn’t find an association between the BMI and the TLF enthesis thickness in our study.
3.7 Limitations
The retrospective nature of the study limited some data retrieval. The VAS pain was not available and the response to treatment was therefore assessed based on the percentage of improvement. The number of patients evaluated was relatively small and US findings were limited to TLF enthesis thickness, calcifications, and cortical irregularities. Other criteria used for the US diagnosis of enthesopathy, such as hypoechogenicity, enthesophytes and erosions, were not available for evaluation [21].
US imaging was only performed using a transverse scan and findings were not confirmed by a longitudinal scan. US imaging was performed by a single radiologist who was not blinded to clinical diagnosis.
4. Conclusion
ICPS patients we examined had a significantly thickened TLF enthesis on their symptomatic side, compared to their asymptomatic side and compared to healthy normal controls with no LBP. Injection of the thickened TLF with steroids and lidocaine was associated with relief of pain.
These findings suggest that TLF enthesopathy is a potential cause of nonspecific LBP that can be diagnosed using US. Prospective trials with larger numbers of patients are needed to confirm our findings.
Disclosures
Ethics
This study was approved by our local Ethics Committee.
Funding
No finding to declare.
Conflict of interest
"We have no Conflict of Interest".
Data availability statement
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
- Hart LG, Deyo RA, Cherkin DC. Physician office visits for low back pain. Frequency, clinical evaluation, and treatment patterns from a U.S. national survey. Spine 20 (1995): 11-19.
- Vos T, Barber RM, Bell B, et al. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, Lancet Lond Engl 386 (2015): 743-800.
- Freburger JK, Holmes GM, Agans RP, et al. The rising prevalence of chronic low back pain. Arch Intern Med 169 (2009): 251-258.
- Sims JA, Moorman SJ. The role of the iliolumbar ligament in low back pain. Med Hypotheses.46 (1996): 511-515.
- Katz JN. Lumbar disc disorders and low-back pain: socioeconomic factors and consequences. J Bone Joint Surg Am. 88 (2006): 2124.
- Nelson NA. The identification of back pain syndromes. In: Back pain, methods for clinical investigations and assessment Eds Hukings DWL and Mulholland RC. Manchester: Manchester University Press; (1986): 13-15.
- Bernard TNJ, Kirkaldy-Willis WH. Recognizing specific characteristics of nonspecific low back pain. Clin Orthop 217 (1987): 266-280.
- Nachemson AL. Advances in low-back pain. Clin Orthop. 200 (1985): 266-278.
- Dillane JB, Fry J, Kalton G. Acute back syndrome-a study from general practice. Br Med J. 2 (1966): 82-84.
- Balague F, Mannion AF, Pellise F, et al. Non-specific low back pain. Lancet Lond Engl. 379 (2012): 482-491.
- Fairbank JC, O’Brien JP. The iliac crest syndrome. A treatable cause of low-back pain. Spine. 8 (1983): 220-224.
- Collee G, Kroon H, Dijkmans BAC, et al. Radiological findings in relation to clinical features in low back pain. 3 (1990): 147.
- Reynierse M, Dijkmans BA, Collee G, et al. Magnetic resonance imaging is normal in the iliac crest pain syndrome. J Rheumatol. 20 (1993): 407-408.
- Collee G, Dijkmans BA, Vandenbroucke JP, et al. A clinical epidemiological study in low back pain. Description of two clinical syndromes. Br J Rheumatol. 29 (1990): 354-357.
- Broudeur P, Larroque CH, Passeron R, et al. Radiological findings in the iliolumbar syndrome J Radiol. 63 (1985): 259-266.
- Maigne JY, Maigne R. Trigger point of the posterior iliac crest: painful iliolumbar ligament insertion or cutaneous dorsal ramus pain? An anatomic study. Arch Phys Med Rehabil. 72 (1991): 734-737.
- Bauwens P, Coyer AB. (1955) The multifidus triangle syndrome as a cause of recurrent low-back pain. Br Med J. 2 (1955): 1306-1307.
- Collee G, Dijkmans BA, Vandenbroucke JP, et al. Iliac crest pain syndrome in low back pain: frequency and features. J Rheumatol. 18 (1991): 1064-1067.
- Stecco C, Stern R, Porzionato A, et al Hyaluronan within fascia in the etiology of myofascial pain. Surg Radiol Anat SRA. 33 (2011): 891-896.
- Healthy Weight. Division of Nutrition, Physical Activity, and Obesity, National Center for Chronic Disease Prevention and Health Promotion [Internet] (2020).
- Wakefield RJ, Balint PV, Szkudlarek M, et al. Musculoskeletal ultrasound including definitions for ultrasonographic pathology. J Rheumatol 32 (2005): 2485-2487.
- Willard FH, Vleeming A, Schuenke MD, et al. The thoracolumbar fascia: anatomy, function and clinical considerations. J Anat. 221 (2012): 507-536.
- Vleeming A, Pool-Goudzwaard AL, Stoeckart R, et al. The posterior layer of the thoracolumbar fascia. Its function in load transfer from spine to legs. Spine. 20 (1995): 753-758.
- Langevin HM, Stevens-Tuttle D, Fox JR, et al. Ultrasound evidence of altered lumbar connective tissue structure in human subjects with chronic low back pain. BMC Musculoskelet Disord 10 (2009): 151.
- Langevin HM, Fox JR, Koptiuch C, et al. Reduced thoracolumbar fascia shear strain in human chronic low back pain. BMC Musculoskelet Disord 12 (2011): 203.
- Todorov PT, Nestorova R, Batalov A. Diagnostic value of musculoskeletal ultrasound in patients with low back pain - a review of the literature. Med Ultrason 1 (2018): 80-87.
- Todorov PT, Batalov A, Kanev P. Ultrasonographic study of erector spinae caudal: enthesopathy could “nonspecific” low back pain become more specific? In: Annals of the Rheumatic Diseases (2013): 153.
- Todorov PT, Kalcheva M, Marinkov A, et al. Low back pain due to enthesopathy of erector spine muscle: a comparative US and MRI study in patients with iliac crest pain syndrome. In: Annals of the Rheumatic Diseases [Internet] (2018): 817.
- Schilder A, Hoheisel U, Magerl W, et al. Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain. Pain 155 (2014): 222-231.
- Schilder A, Magerl W, Hoheisel U, et al. Electrical high-frequency stimulation of the human thoracolumbar fascia evokes long-term potentiation-like pain amplification. Pain 157 (2016): 2309-2317.
- Hoheisel U, Mense S. Inflammation of the thoracolumbar fascia excites and sensitizes rat dorsal horn neurons. Eur J Pain Lond Engl 19 (2015): 419-428.
- Mense S, Hoheisel U. Evidence for the existence of nociceptors in rat thoracolumbar fascia. J Bodyw Mov Ther 20 (2016): 623-628.
- Hoheisel U, Rosner J, Mense S. Innervation changes induced by inflammation of the rat thoracolumbar fascia. Neuroscience 300 (2015): 351-359.
- Carragee EJ, Hannibal M. Diagnostic evaluation of low back pain. Orthop Clin North Am. 35 (2004): 7-16.
- Barnett SB, Ter Haar GR, Ziskin MC, et al. international recommendations and guidelines for the safe use of diagnostic ultrasound in medicine. Ultrasound Med Biol. 26 (2000): 355-366.
- Su, Charles A, Kusin, et al. The Association Between Body Mass Index and the Prevalence, Severity, and Frequency of Low Back Pain. Spine 43 (2018): 848-852.

 Impact Factor:
* 1.7
Impact Factor:
* 1.7
 Acceptance Rate:
74.36%
Acceptance Rate:
74.36%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
