A Clinicopathological Study: Expression of ER, PR and HER/2neu in Endometrial Carcinoma
Jasmeen Kaur1*, Anil Suri2, Manjit Kaur3
1Assistant Professor, Department of Pathology, AIIMS, Punjab, India
2Department of Pathology, Associate Professor, Government medical college, Punjab, India
3Department of Obstretics and Gynaecology, Associate Professor, Government medical college, Punjab, India
*Corresponding Author: Jasmeen Kaur, Assistant Professor, Department of Pathology, AIIMS, Bathinda, Punjab, India
Received: 14 November 2021; Accepted: 26 November 2021; Published: 31 January 2022
Article Information
Citation:
Jasmeen Kaur, Anil Suri, Manjit Kaur. A Clinicopathological Study: Expression of ER, PR and HER/2neu in Endometrial Carcinoma. Obstetrics and Gynecology Research 5 (2022): 41-53.
View / Download Pdf Share at FacebookAbstract
Endometrial carcinoma is a common invasive malignancy of the female genital tract. Investigation of endometrial immunomarkers involved in carcinogenesis may influence early detection and treatment, with direct impact on prognosis by increasing life expectancy. It can often be difficult to distinguish higher grade endometrioid carcinomas from serous carcinomas. Use of immunohistochemistry as an adjunct has been the subject of a number of recent studies.
Aims and Objectives: To study the association of endometrial carcinoma with relation to age, parity and other presenting feature. To determine the association between the expression of ER, PR, and Her-2/neu and clinicopathological features of endometrial carcinoma patients. To correlate the endometrial histopathology with immunohistochemistry.
Material and methods: The present study comprised of analysis of 50 cases of endometrial carcinoma received in the Department of Pathology, Government Medical College, Patiala from January 2018 to May 2020. Routine Haematoxylin and Eosin and on confirmed cases of endometrial carcinoma , IHC for ER, PR and HER-2/neu was performed.
Results: Endometrioid carcinomas showed maximum incidence around the age of 65 with G1 tumours - 56%, G2 - 12%, G3 - 18 % and non Endometrioid were mostly serous and clear representing 14% cases. The predominance of grade-I endometrial carcinoma was found in 56% followed by grade-II in 12% and grade-III in 18% cases. Statistical analysis showed that the degree of differentiation significantly correlated (very highly significant) significant) with histology (p value .000). Out of 50 cases of EMC, 32(64%) cases showed ER positivity. PR positivity was seen in 30(60%) cases and HER-2/neu immunehistochemical expression was positive in 11(22%) cases.
Conclusion: A decrease of the hormonal receptors expression, ER and PR was observed in parallel with
Keywords
<p>Endometrial Carcinoma, Progesterone Receptor, Estrogen Receptor</p>
Article Details
1. Introduction
Endometrial carcinoma is a common invasive mali-gnancy of the female genital tract. Investigation of endometrial immunomarkers involved in carcinogen-nesis may influence early detection and treatment, with direct impact on prognosis by increasing life expectancy [1]. The endometrium is in a constant state of flux, cycling between the proliferative, secretory and menstrual phases. Changes in hormone levels are reflected at the cytological level, but overall endometrial homeostasis is achieved by maintaining a balance between proliferation and apoptosis. Endometrial cell behaviour is primarily governed by the ovarian steroids [2]. Abnormal uterine bleeding and infertility are among the commonest conditions for which patients seek advice in the gynecological outpatient department [3]. Abnormal uterine bleeding may be the result of systemic diseases, functional disturbances or local anatomic disturbances [4]. Endometrial carcinoma typically occurs in elderly patients, 80% of the patients being postmenopausal at the time of diagnosis, the important presenting complaint is abnormal bleed in postmenopausal women [5]. In the developed countries, endometrial carcinoma is the fourth most common cancer, ranking behind breast, lung and colorectal cancer, and eighth leading cause of death from malignancy in women. Overall, about 2-3% of women develop endometrial cancer during their lifetime [6].
1.1 Role of IHC in diagnosis and metastasis of EMC
The majority (60-70%) of endometrial cancers express estrogen receptor. Typically, estrogen-receptor-positive endometrial tumors are associated with a more favorable outcome. Despite this, there is often a discrepancy between estrogen receptor expre-ssion and clinical outcome of the disease [7]. HER-2/neu oncogene encodes a 185 transmembrane glycol-protein. It possesses tyrosine kinase activity [8]. Immunohistochemical staining correlates with gene amplification It can often be difficult to distinguish higher grade endometrioid carcinomas from serous carcinomas. Use of immunehistoche-mistry as an adjunct has been the subject of a number of recent studies [9, 10]. ER and PR status in metastatic endometrial cancer has a predictive value in determining response to hormonal therapy, which supports the use of these assays in the management of patients with metastatic disease [11]. Immunohisto-chemistry by itself may not be an ideal technique to help in the pathological distinction of endometrioid from non-endometrioid carcinomas. It has been shown that the qRT-PCR assessment of a small panel of estrogen induced genes can help to identify low grade endometrioid carcinomas that recur [12].
2. Subjects and Methods
The study was comprised of analysis of 50 endometrial biopsies and total abdominal hyster-ectomy of endometrial carcinoma received in the Department of Pathology, Government Medical College, Patiala from January 2016 to May 2019. This study included 50 histopathologically confirmed cases of endometrial carcinoma diagnosed from endometrial biopsies from endometrial curettage or hysterectomy specimens. The biopsy tissue and hysterectomy specimens were fixed in 10% formalin solution for 6-24 hours and were grossly examined for any abnormality. Sections were stained with routine Haematoxylin and Eosin (H and E) and were examined under the microscope. Deep cuts and serial sections were taken wherever necessary. Simultan-eously, after processing, 1-2 paraffin blocks selected (confirmed cases of endometrial carcinoma) for IHC and sections of 4 microns thickness were cut using microtome, immunohistochemical staining for ER, PR and HER-2/neu was performed.
Antibodies used, Source, clone, antigen and localisation
|
Antibody |
Source |
Clone |
Antigen |
Localisation |
|
ER |
Mouse monoclonal |
ID5 |
Estrogen Receptor |
Nuclear |
|
PR |
Mouse Monoclonal |
PgR636 |
Progesterone Receptor |
Nuclear |
|
HER-2/neu |
Rabbit monoclonal |
EP3 |
c-erbB-2 protein |
Cell membrane |
3. Observations
The study titled “The Clinicopathological Study: Expression of ER, PR and HER-2/neu in endometrial carcinoma” was conducted on 50 cases of endo-metrial carcinoma received in the Department of Pathology, Government Medical College, Patiala; In the study, 8% were in the age group of <40 years, 40% were in the age group of 51-60 years and 20% were in the age group of 61-70 years of age. The maximum cases of EMC were in the age group of 51-60 years (40%) i.e. 7th decade of life. Also there were 45 (90%) cases were menopausal at the time of diagnosis whereas 5 (10%) were premenopausal at the time of diagnosis. Also 40 (80%) cases presented with post menopausal bleeding. 5 (10%) cases presented with post-menopausal discharge and 5 (10%) cases presented with pre-menopausal bleeding and discharge. The most common histopathological pattern of endometrial carcinoma was endometrioid (Table 1) seen in 42 (84%) patients and non endo-metrioid (Table 2) (Figure 4-5) was observed in 8 (16%) patients However, histopathological examina-tion of endometrium in 50 cases of endometrial carcinoma revealed 28 (56%) were grade-I (well differentiated) (Figure 1), 6 (12%) were grade-II (moderately differentiated) (Figure 2), 8 (16%) were grade-III poorly differentiated carcinomas whereas 8 (16%) cases were diagnosed as high grade (Table 2) (Figure 3) endometrial carcinoma.
In the study ER receptor positivity was seen in 32 (64%) cases, 9 (18%) out of 32 cases showed mild nuclear positivity, other 9 (18%) showed moderate nuclear positivity and 14 (28%) showed strong ER receptor nuclear positivity (Figure 6-7) whereas 18 (36%) were negative for ER receptor. In the study PR receptor positivity was seen in 30 (60%) cases. 15 (30%) out of 30 showed mild nuclear positivity, 3 (6%) showed moderate nuclear positivity and 12 (24%) showed strong PR receptor nuclear positivity (Figure 8) whereas 20 (40%) cases were negative for PR receptor. In the study out of 50 cases 39 (78%) were negative for HER-2/neu expression whereas 11 (22%) were positive out of which 4 (8%) were scored as 1+ HER-2/neu positivity, 6 (12%) were scored as 2+ and 1 (2%) were scored 3+.
According to table 3, well differentiated carcinoma, 3 (10.7%) cases were negative, 4 (14.3%) cases show-ed mild positivity, 8 (28.6%) cases showed moderate positivity and 13 (46.4%) cases showed strong positive (Figure 6). In Moderately differ-entiated carcinoma, 1 (16.7%) case was negative, 4 (66.7%) cases showed mild ER positivity and 1 (16.7%) case showed moderate positivity (Figure 7). All poorly differentiated carcinoma were negative for ER expre-ssion whereas in serous/clear endometrial carcinoma, 6 (75.4%) were negative, 1 (12.5%) case showed mild positivity and 1 (12.5%) case showed strong positivity for ER. The p value was 0.000 which is highly significant. The table 4 depict relation of grade and PR expression in endometrial carcinoma. In well differentiated carcinoma, 4 (14.3%) cases were nega-tive, 11 (39.3%) cases of showed mild positivity, 3 (10.7%) cases showed moderate positivity and 10 (35.7%) cases showed strong positivity for PR (Figure 8). In moderately differentiated carcinoma, 1 (16.7%) case was negative, 4 (66.7%) cases showed mild positivity and 1 (16.7%) case showed strong PR positivity. All 8 (100%) cases of poorly differentiated carcinoma was negative for PR expression whereas in serous/clear endometrial carcinoma, 7 (87.5%) cases were negative, 1 (12.5%) case showed strong PR positivity (Figure 9-10). The p value was 0.000 which is highly significant. The below tables depict relation of grade and HER-2/neu expression in endo-metrial carcinoma. In well differentiated carcinoma, 26 (92.2%) were negative, 1 (3.6%) case showed 1+ score and 1 (3.6%) case showed 2+ score (Figure 11). In moderately differentiated carcinoma, 5 (83.3%) cases were negative, 1 (16.7%) case showed 1+ score whereas in poorly differentiated carcinoma, 5 (62.5%) cases were negative, 2 (25%) cases showed 1+ score and 1 (12.5%) case showed 2+ score. In serous/clear and endometrial carcinoma, 3 (37.5) cases were negative, 4 (50%) cases showed 2+ score and 1 (12.5%) showed 3+ score (Figure- 12-13). The p value was 0.003 which is highly significant.
|
Expression |
Frequency |
Percent |
||
|
ER |
PR |
ER |
PR |
|
|
Negative |
18 |
20 |
36 |
40 |
|
Mild |
9 |
15 |
18 |
30 |
|
Moderate |
9 |
3 |
18 |
6 |
|
Strong |
14 |
12 |
28 |
24 |
|
Total |
50 |
50 |
100 |
100 |
Table 1: Estrogen Receptor Expression in Patients of Endometrial Carcinoma (N=50).
|
Scoring |
Frequency |
Percent |
|
Negative |
39 |
78 |
|
1+ |
4 |
8 |
|
2+ |
6 |
12 |
|
3+ |
1 |
2 |
|
Total |
50 |
100 |
Table 2: Her-2/Neu Receptor Expression in Emc.
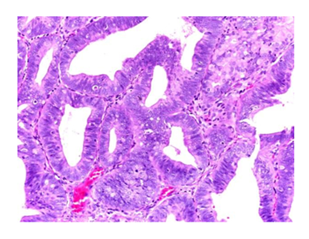
Figure 1: Photomicrograph showing well differentiated endometrioid carconima (H&E X 100).
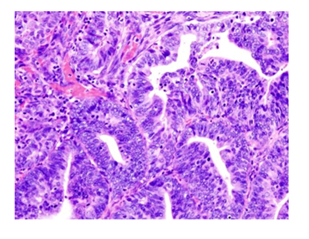
Figure 2: Photomicrograph showing moderately differentiated endometrioid carconima (H&E X 100).
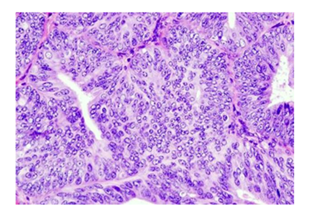
Figure 3: Photomicrograph showing poorly differentiated endometrioid carconima (H&E X 400).
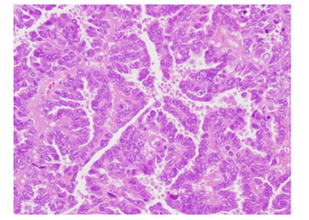
Figure 4: Photomicrograph showing serous endometrial carconima (H&E X 100).
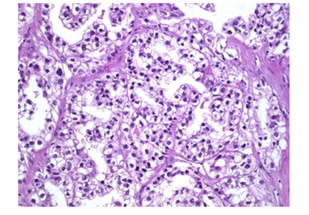
Figure 5: Photomicrograph showing clar cell endometrial carconima (H&E X 400).
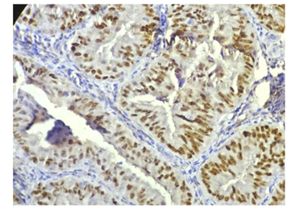
Figure 6: Photomicrograph showing well differentiated endometrioid carconima, strong positive for ER (H&E X 400).
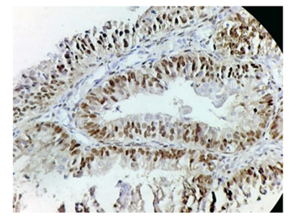
Figure 7: Photomicrograph showing moderately differentiated endometrioid strong positive for ER (H&E X 40).
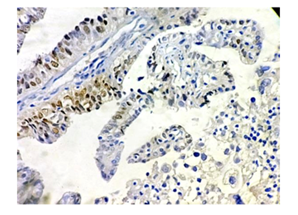
Figure 8: Photomicrograph showing moderately differentiated endometrioid carcinoma, mild positive for PR .(H&E X 400).
|
Grade |
Estrogen Expression |
Total |
p value |
Sig. |
|||
|
Negative (%age) |
Mild(%age) |
Moderate (%age) |
Strong (%age) |
||||
|
WD |
3 (10.7) |
4 (14.3) |
8 (28.6) |
13 (46.4) |
28 (100) |
0 |
HS |
|
MD |
1 (16.7) |
4 (66.7) |
1 (16.7) |
- |
6 (100) |
||
|
PD |
8 (100) |
- |
- |
- |
8 (100) |
||
|
Serous/clear |
6 (75.4) |
1 (12.5) |
- |
1 (12.5) |
8 (100) |
||
|
Total |
18 (36) |
9 (18) |
9 (18) |
14 (28) |
50 (100) |
||
Table 3: Relation of Grade and Er Expression in Emc.
|
Grade |
Progesterone Expression |
Total |
p value |
Sig. |
|||
|
Negative (%age) |
Mild (%age) |
Moderate (%age) |
Strong(%age) |
||||
|
WD |
4 (14.3) |
11 (39.3) |
3 (10.7) |
10 (35.7) |
28 (100) |
0 |
HS |
|
MD |
1 (16.7) |
4 (66.7) |
- |
1 (16.7) |
6 (100) |
||
|
PD |
8 (100) |
- |
- |
- |
8 (100) |
||
|
Serous/clear |
7 (87.5) |
- |
- |
1 (12.5) |
8 (100) |
||
|
Total |
20 (40) |
15 (30) |
3 (6) |
12 (24) |
50 (100) |
||
Table 4: Relation of Grade and Pr Expression in Emc
|
Negative (%age) |
1+(%age) |
2+(%age) |
3+(%age) |
||||
|
WD |
26 (92.9) |
1 (3.6) |
1 (3.6) |
- |
28 (100) |
0.003 |
S |
|
MD |
5 (83.3) |
1 (16.7) |
- |
- |
6 (100) |
||
|
PD |
5 (62.5) |
2 (25) |
1 (12.5) |
- |
8 (100) |
||
|
Serous/clear |
3 (37.5) |
- |
4 (50) |
1 (12.5) |
8 (100) |
||
|
Total |
39 (78) |
4 (8) |
6 (12) |
1 (2) |
50 (100) |
Table 5: Relation of Grade and Her-2/Neu Receptor Expression.
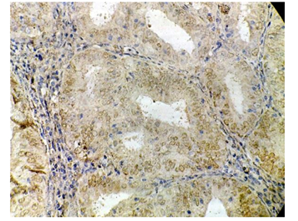
Figure 9: Photomicrograph showing serous type of endometrial carcinoma, strong positive for PR (H&E X 400).
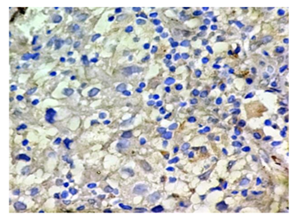
Figure 10: Photomicrograph showing clear cell carcinoma, negative for PR (H&E X 400).
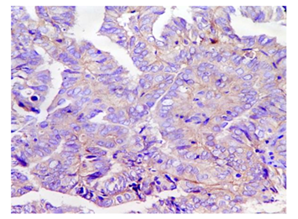
Figure 11: Photomicrograph showing well differentiated endometrioid carcinoma, negative for HER-2/neu (H&E X 400).
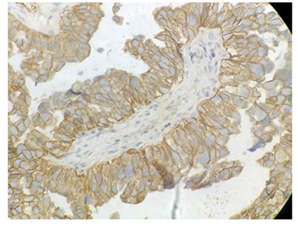
Figure 12: Photomicrograph showing serous type of endometrial carcinoma, strong positive for HER-2/neu (H&E X 400).
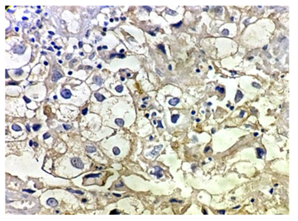
Figure 13: Photomicrograph showing clear cell endometrial carcinoma, positive for HER-2/neu (H&E X 400).
4. Discussion
The results of histopathological and immunohisto-chemical examination were compiled and compared with the other studies available in the literature. In the present study, 10% patients were premenopausal and 90% patients were postmeno-pausal which was also seen with the study of Maniketh et al [13]. These studies observed that the most of the patients with EMC were postmenopausal. The studies show expression of estrogen receptor in endometrial carcinoma.In these studies, ER receptor positivity varied from 59.3% to 86% which is comparable with the present study which shows estrogen receptor positivity in 64% cases of endometrial carcinoma. Stoian et al [16] reported 86.30% cases to be ER positive (Table 6). The studies show expression of progesterone receptor in endometrial carcinoma. In these studies, PR receptor positivity range from 65% to 84.4%. The present study showed progesterone receptor positivity in 60% cases which is consistent with the study of Srijaipracharoen et al. [14] who reported 65.7% cases with PR positivity. The present study showed the expression of HER-2/neu receptor in 22% cases of endometrial carcinoma. The results were inconsistent with the above studies probably because of less number of cases and because of the inclusion of both Endometrioid (of all grades) and non Endometrioid carcinoma in the present study.
|
Authors and year of study |
Estrogen Receptor |
|
|
Negative (%age) |
Positive (%age) |
|
|
Suthipintawong et al. [14] |
23.10% |
76.90% |
|
Srijaipracharoen et al. [15] |
40.70% |
59.30% |
|
Stoian et al. [16] |
13.40% |
86.30% |
|
Maniketh et al. [13] |
26.70% |
73.30% |
|
Present Study |
36% |
64% |
Table 6: Estrogen Receptor Positivity in Endometrial Carcinomas.
|
Authors and year of study |
Progesterone Receptor |
|
|
Negative (%age) |
Positive (%age) |
|
|
Suthipintawong et al. [14] (2008) |
27.7% |
72.3% |
|
Srijaipracharoen et al. [15] (2010) |
34.3% |
65.7% |
|
Stoian et al. [16] (2011) |
18.9% |
81.1% |
|
Maniketh et al. [13] (2014) |
15.6% |
84.4% |
|
Present Study |
40% |
60% |
Table 7: Progesterone Receptor Positivity in Endometrial Carcinoma.
|
Authors and year of study |
HER-2/neu |
|
|
Negative (%age) |
Positive (%age) |
|
|
Suthipintawong et al [14] |
89.2% |
10.8 |
|
Srijaipracharoen et al [15] |
97.2% |
2.8% |
|
Present Study |
78% |
22% |
Table 8: Her-2/Neu Receptor Positivity in Endometrial Carcinomas.
4.1 Summary
- The maximum incidence (74%) of cases was observed in the age group of 51-70 years and post menopausal status (90%).
- The histopathological study revealed the clear predominance of Endometrioid carcinomas (84%) than Non-Endometrioid ones (16%).
- Endometrioid carcinomas showed maximum incidence around the age of 65 with G1 tumours - 56%, G2 - 12%, G3 - 18 % and non Endometrioid were mostly serous and clear representing 14% cases.
- A decrease of the hormonal receptors expression, ER and PR was observed in parallel with the decreased histological degree of differentiation, the lowest values occurring in the case of Endometrioid G3 carcinomas and were absent in Non-Endometrioid carcinomas. This finding may be of a particular clinical importance because almost half of poorly differentiated endometrial carcinomas contain estrogen/ progesterone receptors and they might benefit from a progesterone therapy.
- PR immunoexpression showed statistically significant association with parity, presenting symptom, type, and grade of EMC. Here by, making it an independent prognostic factor.
- The oncoprotein immunoexpression HER-2/neu was positive in 22% cases, mostly in the Non-Endometrioid (62.5%) and Endometrioid G3 (37.5%) carcinomas (intense and uniform positivity), G1 (7.2%) and G2 (16.7%) Endometrioid tumours were rarely positive (weak and focal positive), implying that for endometrioid carcinomas the number of positive cases decreased proportionally with the increasing degree of differentiation, thus defining a subset of endometrial carcinomas with a highly aggressive behavior.
5. Conclusion
The analysis of HER-2/neu expression and of hormonal receptors (ER and PR) showed an inverse relationship. Thus, in the case of Endometrioid tumours (G1 and G2) the ER and PR expression was the highest, while HER-2/neu expression was low; compared with the Endometrioid tumours of high-level (G3) and Non-Endometrioid ones which showed low ER and PR and high expression of HER2/neu. It may be inferred that immunohisto-chemical determination of ER, PR and HER-2/neu on curettage fragments of patients with endometrial carcinomas G1 and G2 allows the recognition of patients with favorable prognosis who may respond to progerterone therapy.
References
- Stoian SC. Clinical, histopathological and immunohistochemical study of endometrial carcinomas. University of Medicine and Pharmacy Craiova (2011): 2.
- McCluggage W. Glen Benign Diseases of the Endometrium. In : Blaustein’s Pathology of Female genital tract (6th edition). Springer Science & Business Media (2011): 307.
- Mehrotra VG, Mukherjee K, Pandey M, et al. Functional uterine bleeding: A study of 150 cases. JOGI 32 (1972): 680.
- Dallenbach-Hellweg The Histopathology of the Endometrium Springer (1975): 83-235.
- Rose PG. Endometrial carcinoma. N Engl J Med 335 (1996): 640-649.
- Berek et al, Berek & Novak’s Gynaecology, Lippinkot Williams & Wilkins (2007): 1349-1377.
- Assikis VJ, Bilimoria MM, Muenzner HD, et al. Mutations of the Estrogen Receptor in Endometrial Carcinoma: Evidence of an Association with High Tumor Grade. Gynecologic Oncology 63 (1996): 192-199.
- Murphy CG, Modi S. HER2 breast cancer therapies: a review. Biologics 3 (2009): 289-301
- Schlosshauer PW, Ellenson LH, Soslow RA. Beta-catenin and E-cadherin expression patterns in high-grade endometrial carcinoma are associated with histological subtype. Mod Pathol 15 (2002): 1032-1037.
- c-erbB-2/HER2. Concentrated and Prediluted Rabbit Monoclonal Antibody: Catalogue CME 342 A, B. Biocare Medical (2021).
- Temkin SM, Fleming G. Current Treatment of Metastatic Endometrial. Cancer Control 16 (2009): 38-45.
- Westin SN, Broaddus RR, Deng L, et al. Molecular clustering of endometrial carcinoma based on estrogen induced gene expression. Cancer Biol Ther 8 (2009): 2126-2135.
- Maniketh I, Ravikumar G, Crasta JA, et al. Estrogen and Progesterone Receptor Expression in Endometrioid Endometrial Carcinomas: A Clinicopathological Study. Middle East Journal of Cancer 5 (2014): 67-73.
- Suthipintawong C, Wejaranayang C, Vipupinyo C. Prognostic significance of ER, PR, Ki67, c-erbB-2, and p53 in endometrial carcinoma. J Med Assoc Thai 91 (2008): 1779-1784.
- Srijaipracharoen S, Tangjitgamol S, Tanvanich S, et al. Expression of ER, PR, and Her-2/neu in endometrial cancer: a clinicopathological study. Asian Pac J Cancer Prev 11 (2010): 215-220.
- Stoian SC, Simionescu C, Margaritescu C, et al. Endometrial carcinomas: correlation between ER, PR, Ki67 status and histopathological prognostic parameters. Rom J Morphol Embryol 52 (2011): 631-636.


 Impact Factor: * 3.2
Impact Factor: * 3.2 Acceptance Rate: 76.63%
Acceptance Rate: 76.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 