Establishment and Qualification of A Competitive Enzyme-Linked Immunosorbent Assay for the Measurement of Heparin in Plasma Products
Andrea Engelmaier, Christoph Zlabinger, Alfred Weber*
Baxalta Innovations GmbH, a Takeda Company, Pharmaceutical Sciences, Industriestraße 67, Vienna, A-1220, Austria
*Corresponding Author: Alfred Weber, Baxalta Innovations GmbH, a Takeda Company, Pharmaceutical Sciences, Industriestraße 67, Vienna, A-1220, Austria
Received: 06 October 2020; Accepted: 16 October 2020; Published: 22 October 2020
Article Information
Citation: Citation: Andrea Engelmaier, Christoph Zlabinger, Alfred Weber. Establishment and Qualification of A Competitive Enzyme-Linked Immunosorbent Assay for the Measurement of Heparin in Plasma Products. Journal of Analytical Techniques and Research 2 (2020): 110-122.
View / Download Pdf Share at FacebookAbstract
Abstract
Heparin is used in plasma fractionation processes to prevent activation of coagulation factors. Consequently, intermediates and final products have to be measured for their heparin content. Established chromogenic heparin assays rely on measurement of the accelerated inhibitory action of antithrombin against thrombin or activated factor X. These assays, however, show limitations, especially at the low sample dilutions required to provide adequate sensitivity. These limitations, potentially biasing results, originate from the specific sample matrix including proteases or protease inhibitors or from the test sample’s color. To overcome these shortcomings, a commercially available competitive enzyme-linked immunosorbent assay (ELISA) was established, described so far to measure heparin in plasma only. Briefly, heparin contained in the sample competed with the binding of a peroxidase-labelled heparin binding protein to the wells of a heparin-coated plate. The six-point calibration curve ranging from 0.03 to 7.14 IU heparin/mL showed acceptable accuracy and reproducibility. Test samples were diluted at least 1/6 in normal human citrated plasma. Spike-recovery was carried out by adding heparin to obtain a concentration of 0.26 IU/mL in the test dilution of the respective sample. Acceptable recovery was found in all sample types investigated (final immunoglobulin G product and process intermediates, albumin and α1-proteinase inhibitor), while intra-and inter-run precision data showed relative standard deviations lower than 10%. The ELISA’s limit of quantification was 0.18 IU/mL, thus demonstrating required sensitivity. In summary, the competitive heparin ELISA allowed accurate, precise and sensitive heparin measurement in specific plasma protein matrices, which interfere with chromogenic heparin assays.
Keywords
<p>Heparin measurement; Competitive ELISA; Plasma products and intermediates</p>
Article Details
1. Introduction
The sulfated polysaccharide heparin [1] is added during plasma fractionation to inhibit undesired activation of the coagulation system. Heparin exerts its potent anticoagulant activity by binding via a specific pentasaccharide sequence to the heparin binding site of antithrombin [2]. The conformational change of antithrombin [3,4] caused by the binding of full-length heparin enhances at physiological ionic strength the second order rate constants for thrombin (FIIa) and activated factor X (FXa) inhibition by 4,300 and 580-fold [5], respectively. In addition to this well understood antithrombin-mediated anticoagulative effect, direct heparin binding has been described for the coagulation factors IX [6], factor XI [7], von Willebrand factor [8] and many other plasma proteins. As a consequence of these multiple interactions, it seems reasonable to measure residual heparin concentrations in intermediates and the final plasma protein products. Since the original assignment of heparin’s biological activity, which was defined by Howell in 1923 as minimum heparin amount to maintain the fluidity of cats’ blood for 24 hours at 0°C [9], various heparin assays have been described. First assays used non-anticoagulated blood with all supply issues associated. Citrated bovine plasma was then applied for the heparin assay by Reinert and Winterstein in 1930 [10], while the use of citrated sheep plasma, described by Kuizenga et al in 1943 [11], became later the basis of the USP assay. Nevertheless, except for the glass surface, these assays did not include any clot-promoting agents, thus resulting in long clotting times. Addition of tissue extracts called thromboplastin or thrombokinase and CaCl2 to the test mix, first described for the prothrombin time assay by Quick [12], accelerated the clotting time of citrated plasma to less than 30 sec. This methodical advancement was implemented for a heparin assay in 1941 [13], and subsequently a similar method using citrated sheep plasma was described by Adam and Smith [14]. This method became the basis of the British Pharmacopeia heparin assay. Of note, the use of thromboplastin reagents requires heparin concentrations in the range of 1-3 U/mL for prolonging clotting times, while activated partial thromboplastin time reagents allow measurement of heparin levels of as little as 0.1 U/mL. Basically, theses clotting-based methods were used up to the 1980s. In 1973, Yin et al [15] and Denson and Bonnar [16] described the anti-FXa method, in which residual FXa was initially measured by a clotting method. With the advent of the chromogenic substrates, low molecular weight peptides that release the chromogen p-nitroanilide after amidolytic cleavage, chromogenic methods were developed. Both the chromogenic FIIa and FXa heparin determination rely on the chromogenic measurement of residual FIIa/FXa activity after inhibition with heparin-free antithrombin under conditions, where the inhibition rate is strictly dependent on the heparin concentrations present. Recently, the establishment and validation of efficient assays for anti-FIIa and anti-FXa activities of heparin has been described [17]. As an orthogonal approach, not based on the measurement of residual amidolytic activity, a surface plasmon resonance method (SPR) was described [18]. In principle, the heparin concentration in the test sample determines the degree of the competition of antithrombin binding to heparin, immobilized on the chip. The heparin concentrations obtained correlated well with the results of the chromogenic FIIa and FXa assays.
Despite their recognized suitability and widespread use, chromogenic heparin assays are nevertheless subject to certain limitations when applied for the measurement of samples other than heparin preparations or plasma samples with high heparin levels. Such limitations include the presence of proteases in the test sample which add to the residual FIIa/FXa activity, which negatively correlates with the heparin concentration. Consequently, presence of proteases can cause heparin concentrations to be underestimated. On the other hand, the presence of protease inhibitors can result in the opposite effect or at high concentrations even fully prevent running the assay. Finally, sample coloration could also cause assay interference especially at low heparin levels requiring the sample measurement at low assay dilutions. As sensitive heparin measurement down to 0.2 IU/mL was required in samples types showing the limiting features mentioned above, an alternate method for the heparin measurement was sought.
To address these short comings, we identified a commercially available competitive heparin ELISA, so far described only for use in human plasma and adapted and qualified it for the measurement of low heparin levels in plasma protein samples, which could not be accurately measured with chromogenic FIIa/FXa assays. Furthermore, ELISA performance data including accuracy, precision and linearity were evaluated and heparin stability studies were carried out in human plasma matrix.
Abbreviations
A1PI: α1-proteinase inhibitor; ARA: ARALAST NP, α1-plasma-derived-proteinase inhibitor concentrate; ELISA: enzyme-linked immunosorbent assay; hSA: human serum albumin; IgG: immunoglobulin G; MSD: minimal sample dilution; OD: optical density; PBS: phosphate-buffered saline; PBST: phosphate-buffered saline with 0.05% Tween 20; PptG: precipitate G, an intermediate of the GAMMAGARD Liquid/KIOVIG manufacturing process; GGL: GAMMAGARD Liquid/KIOVIG, plasma-derived intravenous immunoglobulin preparation; RSD: relative standard deviation; RT: room temperature (+18 to +26°C); SPR: surface plasmon resonance; USP: United States Pharmacopeia.
2. Materials and Methods
2.1 Material
The competitive ELISA for unfractionated heparin was from Lifespan Technologies (K-2100; Utah, US). A plate coated with heparin, a heparin-binding detection reagent and the reagents required for the color reaction are supplied with the kit. The pooled normal human plasma CCN-10 from Precision BioLogic (CoaChrom, Vienna, Austria) was used as the dilution medium for the calibration curve and the samples. The USP reference standard for unfractionated heparin 1304005 with an assigned heparin concentration of 357 IU/mL (0.18 IU corresponds to 1 μg) was used to prepare the assay calibration curve. All other chemicals were of highest analytical grade. In particular, KCl, NaCl, KH2PO4, and Na2HPO4×2 H2O were from VWR (Vienna, Austria) and Tween 20 (EIA grade) was from Bio-Rad (Vienna, Austria). Phosphate-buffered saline (PBS; 0.8% [g/v] NaCl, 0.02% KCl, 0.02% KH2PO4, 0.126% Na2HPO4×2H2O) and PBS with Tween (PBST; PBS with 0.05% [g/v] Tween 20 were used. Five lots of the intravenous immunoglobulin (IgG) preparation GAMMAGARD LIQUID (GGL) process intermediate precipitate G (PptG), four lots of the α1-proteinase inhibitor (A1PI) preparation ARALAST NP (ARA), five lots of 20% human serum albumin (hSA) preparation and one lot of 25% albumin were used. In addition, one lot Cohn Pool, representing the starting material of the fractionation process, and each one lot of Cohn II+III precipitate and Cohn V precipitate, representing process intermediates with enriched IgG and albumin concentrations, respectively, were used.
2.2 Description of the ELISA
The USP reference standard for unfractionated heparin with a labelled heparin concentration of 357 IU/mL was used for the construction of the calibration curve. This curve was obtained by preparing triplicates starting at the dilution 1/50 and adding five serial 1+2 dilutions, using the pooled normal human plasma CCN-10 as the dilution medium and as blank. Thus, the calibration curve covered a heparin concentration range from 0.03 to 7.14 IU/mL. Test samples were diluted 1/6 followed by serial 1+2 dilutions if required. Ten μL of the triplicate dilutions of samples/standard were loaded to the wells. Immediately after the plate loading, 90 μL/well of the detection conjugate were added. Using the Grant bio plate shaker PHMP-4 (Szabo; Vienna, Austria) the plate was shaken at room temperature (RT) and 500 rpm for 60 min. The incubation was stopped by washing the plate with the Biotek plate washer ELx405 (Szabo; Vienna, Austria). For the colour reaction, 100 μL/well reagent was added; the plate was incubated at RT for 20 min, before the colour reaction was stopped by the addition of 50 μL/well stopping solution. Measurement was done at 450 nm (reference wavelength 620 nm) with the Biotek ELISA reader EL 808 (Szabo; Vienna, Austria). The data evaluation was based on the 4-parameter algorithm (blank-included), calculated with the ELISA reader software GEN 5 (Biotek). Only optical densities (ODs) of the samples within the range of the six-point calibration curve were evaluated on the calibration curve. Figure 1 shows a schematic representation of the assay.
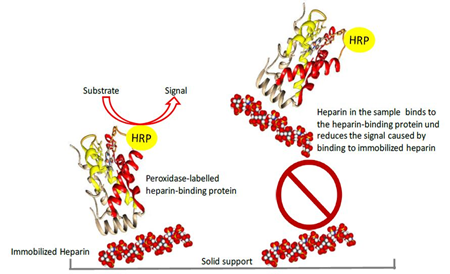
Figure 1: Basic principle of the competitive heparin ELISA
The peroxidase labelled heparin-binding protein binds to plate-immobilized heparin. Heparin in the sample binds to the labelled heparin-binding protein in solution and thus reduces the signal obtained in the competitive ELISA. HRP indicates peroxidase conjugated to the heparin-binding protein.
3. Results
The result section first presents the calibration curve data followed by the results supporting the definition of the minimum sample dilution for the three sample types of interest. Then, assay performance data including accuracy, precision and linearity are shown for relevant plasma protein solutions. Finally, the data of heparin stability studies carried out in plasma at RT and addressing repeated freezing-thawing are presented.
3.1 Definition of the Calibration Curve
As running the competitive ELISA includes the manipulation of volumes of as low as 10 μL and the incubation of only 100 μL/well, plate homogeneity was investigated first. Results confirmed adequate conditions as the means (n=12) determined for the eight rows of the plate showed relative standard deviations (RSDs) of not more than 4.9%. This allowed to exclude the presence of positional effects. Next, the calibration curve model was established. The attempt of using a log-log calibration curve failed because of the unacceptable high relative total error of 40.2% observed. Therefore, a 4-parameter fitting approach was applied to obtain the six-point standard curve ranging from 0.03 to 7.14 mIU/mL heparin. Figure 2 shows the mean calibration curve, obtained on 15 different days together with the relative agreement of the back-fitted ODs of the six calibration curve standards. The curve’s mean ODs ranged from 1.226 (0.03 mIU/mL) to 0.262 (7.14 mIU/mL) with RSDs of the individual means not exceeding 16.1%. These RSDs were in the magnitude of that determined for the mean assay blank, which was 1.459 and had an RSD of 12.8%. This permitted to explain the RSDs observed for the assay standards by changes in general assay conditions rather than by changes directly affecting the calibration curve preparation. The quality of fit was expressed by the coefficient of variance, which was at least 0.998, and by the back-fitting approach. The relative mean agreements of the six calibration curve standards ranged from 95.4% to 104.6% with all individual values within a 100 ± 22% range. Only one of 90 values deviated more than 20% from its nominal concentration. The RSDs of the mean agreements did not exceed 11.1%, ranging from 2.9% to11.1%. Reasonably, the lowest RSDs were determined for the three heparin concentrations defining the center of the calibration curve. All in all, the data confirmed that the assay calibration curve could be constructed accurately, precisely and reproducibly.
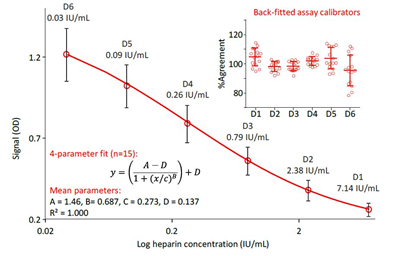
Figure 2: Calibration curves and agreement of back-fitted assay calibrators
The mean four-parameter fit calibration curve ranging from 0.03 to 7.14 mIU/mL, obtained in 15 independent runs on different days is shown. Error bars mark the single standard deviations of the means. The insert shows the agreement of the back-fitted assay calibrators as a percent of their respective nominal concentrations.
3.2 Definition of Minimal Sample Dilutions (MSDs)
The assay was designed by the manufacturer for the matrix of human plasma, so it was reasonable to dilute test samples of interest composed of matrices other than human plasma by using a human plasma preparation as diluent. Table 1 shows data for the IgG-containing GGL processintermediate PptG, the 20% hSA preparation and the purified A1PI preparation ARALAST NP. These samples were diluted 1/2,1/3,1/4,1/6,1/8 and 1/10 using pooled normal plasma and spiked with 0.5 IU heparin/mL. Interestingly, despite obvious different matrices of the three samples the recovery of spiked heparin increased with the samples’ dilutions starting at the dilution of 1/2 with not more than 46.0% and reaching an acceptable level at the dilution of 1/6, while using higher dilutions again resulted in decreasing recoveries. These data provided the basis for selecting the MSD 1/6 as the adequate sample measurement dilution. This selection was further confirmed by constructing heparin calibration curves in 1/6-diluted PptG, 20% hSA and ARA samples. The securves were compared to that obtained in human plasma (Figure 3). Essentially, these curves further confirmed the suitability of the dilution 1/6 using normal human plasma as diluent for the samples’ measurement, since the slopes of the curves prepared in the three matrices were highly similar and differed by not more than 3.5% from that obtained for the pure reference plasma preparation. These data provided unambiguous evidence that the matrices of the three plasma protein solutions did not interfere with the competitive heparin ELISA. The different composition of the three samples had no influence on the assay using the sample dilution of 1/6.
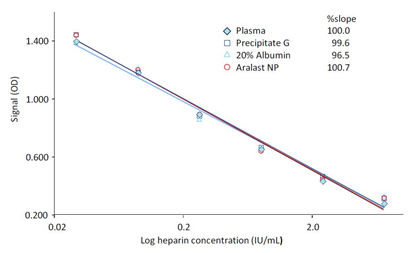
Figure 3: Calibration curves in test sample matrices
Instead of using human plasma as diluent for the heparin standards, 1/6-diluted samples of precipitate G, ARALAST NP and a 20% human serum albumin solution were used for the construction of the calibration curve. The insert shows the slopes of the log-log regression curves calculated between the dilutions and the blank-corrected ODs of the samples.
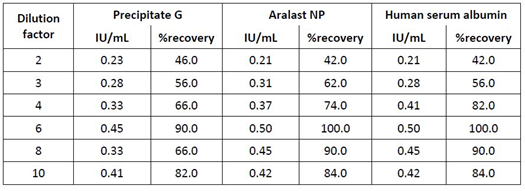
Table 1: Definition of the minimum dilution for three sample types containing proteases and protease inhibitors and showing color.
Remarks: Samples of precipitate G, ARALAST NP and human serum albumin (20%) were spiked with a nominal heparin concentration of 0.5 IU/mL and measured using the dilution given. Human plasma pool was used for the dilution. %recovery was calculated according to the formula (found concentration/nominal concentration) ×100.
3.3 Assay Performance Characteristics
3.3.1 Accuracy Investigation
Assay accuracy was investigated by spike-recovery at the assay’s lower quantification limit. In particular and common to all individual experiments, the 1/4-diluted sample was mixed with two parts of the assay standard containing 0.79 IU heparin/mL resulting in a final heparin level of 0.26 IU/mL. The mean recovery results are presented in Table 2. The non-spiked samples were shown to contain heparin levels below 0.18 IU/mL, representing the assay’s lower limit of quantification (data on file). The starting material of the Cohn fractionation process, termed Cohn Pool, demonstrated a mean recovery of 98.7% (n=6) with individual values ranging from 95.4% to 105.9%. The two intermediates of the Cohn fractionation process, namely Cohn II+III precipitate and Cohn Fraction V precipitate, in which IgG and albumin are enriched for the further downstream processing, showed mean recoveries of 91.7% and 110.2% (n=6), respectively. The recovery determined for the GGL downstream intermediate PptG ranged from 92.3% to 107.7% resulting in a mean recovery of 99.1% (n=6). Similarly, acceptable recoveries were determined for the final products GGL (7 lots), ARALAST NP (4 lots) and hSA at concentrations of 20% (4 lots) and 25% (1 lot, 6 independent measurements). In essence, the individual recoveries ranged from 80.8% to 115.1% with all mean recoveries within the 100 ± 10%, which qualified the assay for use using the EMA guidelines for bioanalytical assay validation as basis for the evaluation [19].
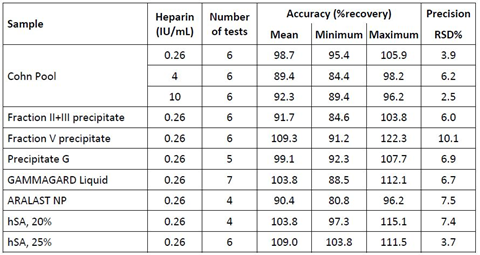
Table 2: Accuracy and precision data
Remarks: Spiked heparin concentrations are shown in the column heparin. Neat samples showed heparin concentrations below the assay’s limit of quantification (0.18 IU/mL). %recovery was calculated according to the formula (found concentration/nominal concentration) ×100. Precision relates to the variance seen within one run. Results comply with the recommendations given by the EMA guideline for the validation of bioanalytical methods [19].
3.3.2 Precision Investigation
Samples of the Cohn Pool starting material, spiked with three heparin concentrations, of three process intermediates and four final products were subjected to repeatability analysis (n=6). The mean RSDs are presented in Table 2. Spiked Cohn Pool was measured at the three heparin concentrations 0.26, 4 and 10 IU/mL with acceptable RSDs ranging from 2.5 to 6.2%. The RSDs for all other samples did not exceed 10.2% with this particular RSD found for Cohn Fraction V, known to contain ethanol up to 5%. These RSDs obtained were similar to those determined for an anti-FXa assay [17], where RSDs ranging from 3.1 to 8.8% were reported for the measurement of heparin standards.
In a separate approach, assay precision was addressed by the results obtained for the repeated measurement of an assay control sample with a nominal heparin concentration of 2 IU/mL spiked to human plasma and stored frozen at ≤-15°C. Intra-run precision could be estimated by two experiments, both based on eight measurements in one run, resulting in RSDs of 5.8% and 8.5%. The mean over the ten independent runs had an RSD of 8.1%, being an estimate for the inter-run precision of the assay.
3.3.3 Linearity Investigation
Assay linearity was investigated in human plasma addressing the heparin concentration range from 0.05 to 2 IU/mL. The regression curve between the heparin concentration and the reciprocal value of the dilution was linear over the whole range. Comparing nominal versus found heparin concentrations resulted in a linear regression curve with a slope of 0.99 and a y-intercept of -0.008. These data confirmed the linearity of the competitive ELISA. Figure 4 shows the data in form of two plots.
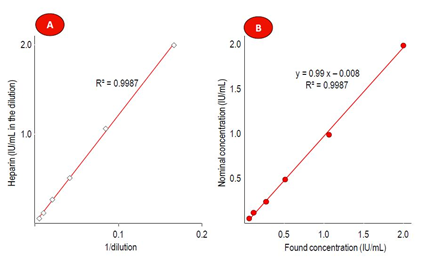
Figure 4: Results of the linearity investigation
Part A shows the linear regression curve between found heparin concentrations and the reciprocal value of the plasma sample dilutions. Part B relates the found with the expected heparin concentrations, also showing the linear regression curve between both parameters. In both cases, the coefficient of determination R² is shown.
3.3.4 Stability Investigations
Two stability studies attempted to address assay robustness in terms of heparin stability in human plasma: a freezing-thawing study and a RT stability study. Both studies aimed to detect possible freezing or time-dependent interactions of heparin with heparin-binding plasma proteins, which could impact the assay. The reference plasma pool was spiked with 1 and 10 IU heparin/mL and subjected to three consecutive freezing-thawing cycles with one cycle consisting of at least 12 h freezing at ≤ -15°C and 1 h thawing at RT. Table 3 shows the results indicating that both heparin concentrations showed stability on repeated freezing-thawing for up to three times, when a 100 ± 20% stability criterion was applied for the evaluation [19]. For the RT stability study, the reference plasma pool was spiked with 10 IU heparin/mL and kept at RT for up to 48 h. Samples were taken at time zero and after 0.25, 0.5, 1, 1.5, 2, 3, 4, 6, 8, 24, 28 and 48 h. These samples were snap frozen at ≤ -60°C and their heparin concentrations were measured in one run. The results presented in Table 4 confirmed the stability of heparin, spiked to human plasma and kept at RT for 48 h, since all the relative concentrations measured at all sampling times differed by less than ± 20% from the initially measured or the nominal concentration [19].
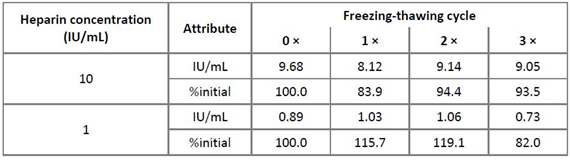
Table 3: Results of the freezing-thawing study
Remarks: Relative heparin concentrations are given as a percent of the concentration measured for the sample subjected to the freezing-thawing cycles.
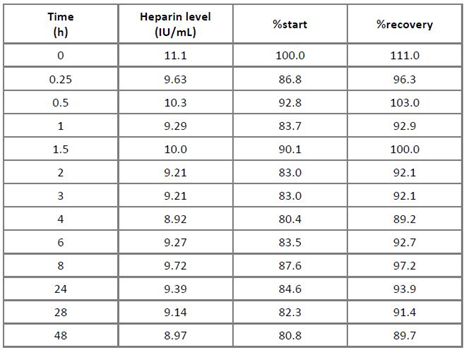
Table 4: Stability of heparin in human plasma at room temperature
Remarks: The columns “%start” and “%recovery” relate the found heparin concentrations to those determined for the sample subjected to the stability study.
4. Discussion
Due to its pronounced anticoagulative activity, the sulfated polysaccharide heparin represents an important auxiliary material for the fractionation of human plasma. In first line, heparin is used to avoid unwanted activation of the human coagulation system, which is primarily achieved by intensively enhancing the activity of the serine protease inhibitor antithrombin. Consequently, addition of heparin requires its measurement. Despite the plethora of heparin assays developed and successfully used over the time, the measurement of heparin remains challenging when heparin has to be measured in protein matrices other than human plasma or purified heparin preparations. This is especially true when measurement of low heparin concentrations or even the proof of heparin removal during a purification process is required. Basically, two groups of heparin assays can be discriminated: the first measures inhibition of clot formation, triggered by either thromboplastin or by contact activators, while the second measures residual activity of the coagulation proteases FIIa/FXa in the presence of purified essentially heparin-free antithrombin, in most cases by using appropriate chromogenic substrates. The chromogenic anti-FIIa/FXa assays have become the methods of choice for many applications because of their sensitivity and convenience. Apart from these basic approaches, only few methods for the measurement of heparin have been reported. Recently, the SPR-based assay represented one of these few examples. Like the chromogenic methods, also this method relies on the interaction of heparin with antithrombin. Antithrombin, added to the test sample, is allowed to bind to heparin, immobilized onto a chip. Binding is measured by SPR. Heparin dose-dependently reduces the antithrombin binding to immobilized heparin. This assay was demonstrated to be suitable for the measurement of heparin preparations, but data for its performance in protein-containing sample matrices were not provided so far. Good agreement with the results obtained with chromogenic anti-FIIa/FXa assays was demonstrated for the measurement of purified heparin preparations.
Despite their adequate performance, chromogenic anti-FIIa/FXa assays nevertheless exhibit certain constraints, caused by test sample matrix. Thus, the presence of active proteases or protease inhibitors can potentially impact assay accuracy, especially when sensitive heparin determination is required. This potential bias relates to the underlying measurement principle, i.e. the determination of residual amidolytic activity after antithrombin inhibition. Such detrimental matrix effects can be detected only in samples which are absolutely devoid of heparin, but except using high sample dilutions there will be no remediation of this interference. Similarly, the sample’s color could falsify the results obtained with a chromogenic FIIa/FXa assay. The heparin ELISA described here circumvents these restrictions of chromogenic FIIa/FXa assays. Primarily designed for measurement of heparin concentrations in human plasma, the ELISA was adapted to be used for samples making high demands on the chromogenic FIIa/FXa assays. Basically, this adaption was achieved by using human plasma as sample diluent. This approach altered the assay milieu by the particular sample matrix to an extent, which had no obvious influence on the assay performance. Thus, the 1/6-dilution proved adequate for all sample types investigated.
Among these three sample types, ARALAST NP represents the sample, in which high protease inhibitor concentration causes failure of the chromogenic FIIa/FXa assay, even though neutrophil elastase is the primary target of α1-proteinase inhibitor and FIIa/FXa are inhibited to a much lesser extent. Similarly, the GGL intermediate PptG contains significant levels of the protease inhibitor α2-macroglobulin apart from moderate levels of amidolytic activities. Both features impacted the chromogenic heparin measurement and prevented successful assay qualification. Finally, therapeutically used human serum albumin preparations are available at concentrations of 20% and 25%. These yellowish colored solutions require a sample dilution of 1/100 to remove the color’s impact on the assay, but this increases the assay’s limit of quantification so that it can no longer be used to claim absence of heparin. For all three cases, the suitability of the heparin ELISA for the measurement of such challenging samples was unambiguously demonstrated by the data of the spike-recovery studies. Furthermore, the low levels of heparin spiked demonstrated accuracy close to assay’s limit of detection, which was 0.18 IU/mL. These data were further corroborated by the precision data, which complied with recent recommendations for bioanalytical methods [19].
The results of the two stability studies further confirmed the suitability of the competitive ELISA for the heparin measurement in plasma protein-containing test samples. Human plasma contains many heparin-binding proteins with different avidities to heparin, which could interfere with the heparin binding of the labelled protein, being the basis of this competitive ELISA. The finding that neither repeated thawing-thawing nor prolonged incubation at RT showed substantial changes in the heparin concentrations measured for spiked plasma, highlights the robustness of the method in terms of preanalytical factors like storage or freezing-thawing of test samples. It furthermore confirms the strong heparin avidity of the labelled heparin binding protein, as binding was not substantially impaired by competition even after prolonged incubation at RT.
5. Conclusion
The data presented confirmed the successful adaption and qualification of a commercially available competitive heparin ELISA for the measurement of heparin concentrations in plasma protein solutions, in which chromogenic FIIa/FXa heparin assays could not be carried out. This enabled for increased process- and product understanding and knowledge. Certainly, the use of this ELISA is not restricted to the measurement of plasma protein solutions, but it could be applied also for the measurement of recombinantly manufactured protein solutions whenever required.
Acknowledgments
The authors thank Hubert Ritter for skillful technical assistance and Susanne Brunner and Sabine Riedler for review of the experimental data. The work was sponsored by Baxalta Innovations GmbH, a Takeda company.
Conflict of Interest:
All authors are full time employees of Baxalta Innovation GmbH, a Takeda company.
References
- Lane DA, Ulf L. Heparin: chemical and biological properties, clinical applications (1989).
- Jin L, Abrahams JP, Skinner R, et al. The anticoagulant activation of antithrombin by heparin. Proceedings of the National Academy of Sciences 94 (1997): 14683-14688.
- Whisstock JC, Pike RN, Jin L, et al. Conformational changes in serpins: II. The mechanism of activation of antithrombin by heparin. Journal of Molecular Biology 301 (2000): 1287-1305.
- Hricovíni M, Guerrini M, Bisio A, et al. Conformation of heparin pentasaccharide bound to antithrombin III. Biochemical Journal 359 (2001): 265-272.
- Olson ST, Björk I, Sheffer R, et al. Role of the antithrombin-binding pentasaccharide in heparin acceleration of antithrombin-proteinase reactions. Resolution of the antithrombin conformational change contribution to heparin rate enhancement. Journal of Biological Chemistry 267 (1992): 12528-12538.
- Buyue Y, Misenheimer TM, Sheehan JP. Low molecular weight heparin inhibits plasma thrombin generation via direct targeting of factor IXa: contribution of the serpin-independent mechanism. Journal of Thrombosis and Haemostasis 10 (2012): 2086-2098.
- Zhao M, Abdel-Razek T, Sun MF, et al. Characterization of a heparin binding site on the heavy chain of factor XI. Journal of Biological Chemistry 273 (1998): 31153-31159.
- Sobel M, Soler DF, Kermode JC, et al. Localization and characterization of a heparin binding domain peptide of human von Willebrand factor. Journal of Biological Chemistry 267 (1992): 8857-8862.
- Howell WH. The purification of heparin and its presence in blood. American Journal of Physiology-Legacy Content 71 (1925): 553-562.
- Reinert M, Winterstein A. Contribution to the study of heparin. Archives Internationales De Pharmacodynamie ET DE Therapie 62 (1939): 47-68.
- Kuizenga MH, Nelson JW, Cartland GF. The bioassay of heparin preparations. American Journal of Physiology-Legacy Content 139 (1943): 612-616.
- Quick AJ. The prothrombin in haemophilia and in obstructive jaundice. J Biol Chem 109 (1935): 73–74.
- Macintosh FC. A method for estimating the potency of heparin preparations. Biochemical Journal 35 (1941): 770-775.
- Adams SS, Smith KL. The use of sulphated whole blood in the assay of heparin. Journal of Pharmacy and Pharmacology 2 (1950): 836-846.
- Yin ET, Wessler S, Butler JV. Plasma heparin: a unique, practical, submicrogram-sensitive assay. The Journal of Laboratory and Clinical Medicine 81 (1973): 298-310.
- Denson KE, Bonnar J. The measurement of heparin. Thrombosis and Haemostasis 30 (1973): 471-479.
- Suzuki T, Ishii-Watabe A, Hashii N, et al. The establishment and validation of efficient assays for anti-IIa and anti-Xa activities of heparin sodium and heparin calcium. Biologicals 41 (2013): 415-423.
- Zhao J, Liu X, Malhotra A, et al. Novel method for measurement of heparin anticoagulant activity using SPR. Analytical Biochemistry 526 (2017): 39-42.
- European Medicines Agency: Guideline on bioanalytical assay validation (2011). EMEA/CHMP/EWP/192217/2009.


 Impact Factor: * 2.8
Impact Factor: * 2.8 Acceptance Rate: 77.30%
Acceptance Rate: 77.30%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 