The Predictive Value of Urinary Progesterone Metabolite PdG Testing in Pregnancy Outcomes
Amy Beckley1*, Alexandra Koudele2, John Park3, Gary Levy4, Aimee Eyvazzadeh5,Joshua Klein6
1MFB Fertility, Inc, 5485 Conastoga Ct Suite 201, Boulder, CO 80301, USA
25485 Conastoga Ct Suite 201, Boulder, CO 80301, USA
3Carolina Conceptions, 2601 Lake Dr 301, Raleigh, NC 27607, USA
4Fertility Cloud, Inc, 2100 Geng Rd, Palo Alto, CA 94303, USA
55401, Dr Aimee, Norris Canyon Road, Suite, 106, USA
6Extend Fertility, 200 W 57th St 1101, New York, NY 10019, USA
*Corresponding Author: Amy Beckley, MFB Fertility, Inc, 5485 Conastoga Ct Suite 201, Boulder, CO 80301, USA
Received: 06 September 2022; Accepted: 12 September 2022; Published: 22 September 2022
Article Information
Citation: Amy Beckley, Joshua Klein, John Park, Aimee Eyvazzadeh, Gary Levy, Alexandra Koudele. The predictive value of urinary progesterone metabolite PdG testing in pregnancy outcomes. Obstetrics and Gynecology Research 5 (2022): 194-198
View / Download Pdf Share at FacebookAbstract
Background: Progesterone is essential for establishing and maintaining a pregnancy. However, premature atresia of the corpus luteum (CL) or sub-optimal progesterone secretion after ovulation may result in the decreased ability to establish or maintain a pregnancy. The current limitations of serum hormone testing fail to give a complete picture of progesterone production across the luteal phase required for pregnancy.
Results: Of the 185 enrolled women, 172 had complete cycles. Of the 172 women who demonstrated complete cycles, 54 (31.4%) women reported pregnancies. Thirty-five (64.8%) of these pregnancies resulted from a PdG positive cycle with a miscarriage rate of 14.3% (5/35). Nineteen pregnancies (35.2%) resulted from PdG negative cycles with a miscarriage rate of 89.5% (17/19). The association of a negative or positive cycle with pregnancy outcome was significant (two-sided p=0.0001). Positive PdG cycles are associated with increased pregnancy rates and lower odds of a first-trimester pregnancy loss.
Conclusion: A consideration for luteal phase support with progesterone or ovulation induction in women with negative PdG cycles can be of benefit to increase pregnancy rates. Furthermore, the use of LH and PdG testing prior to conception can be used as a screening tool to potentially identify women who are at risk of subfertility and at a higher risk of first-trimester pregnancy loss related to low progesterone.
Keywords
<p>Progesterone; Implantation; Pregnanediol-3a-glucuronide (PdG); Luteal phase; Pregnancy; Miscarriage</p>
Article Details
Background
The luteal phase of the menstrual cycle is the period of time from ovulation to pregnancy or menstruation. It is initiated with a surge of luteinizing hormone (LH) triggering ovulation; the process by which the dominant follicle ruptures and releases an oocyte to begin the journey to the endometrium. The remnants of the dominant follicle then transform into the corpus luteum, which begins to secrete progesterone. In this way, progesterone dominates the latter half of a women’s menstrual cycle. Progesterone prepares the endometrium for implantation and is essential for the maintenance of a healthy pregnancy [1].
The corpus luteum continues to secrete progesterone until the placenta takes over this role, usually between 6-10 weeks of gestation. The maintenance of serum progesterone levels is essential to the health and success of a pregnancy [2]. However, if endogenous progesterone production is insufficient, the resulting lower serum progesterone levels during the luteal phase are associated with implantation failure and early miscarriage [3]. This is sometimes referred to as luteal phase defect (LPD) and was first mentioned as a possible cause for infertility in a 1949 study conducted by Georgeana Seegar Jones Y et al [4].
Despite over 70 years of research, there is still a lack of consensus regarding the etiology and diagnosis of LPD [5]. Clinically, LPD manifests as a luteal phase lasting less than 10 days and/or prolonged spotting before the expected day of menstruation. LPD has been associated with early pregnancy loss and infertility [6].
Traditionally, serum progesterone levels are measured during the mid-luteal timeframe, approximately 7 days past ovulation. It has been suggested that optimal serum progesterone is considered to be >10 ng/mL [6]. However, the biggest limitation of progesterone monitoring is the pulsatile release from the corpus luteum, with fluctuations from 2 to 40 ng/mL during 24 hours in the same healthy subject [6]. The frequency and amplitude of these fluctuations are present during the entire luteal phase, making a single value potentially uninterpretable and thus unhelpful in diagnosing or excluding LPD. Recently, pregnanediol-3a-glucuronide (PdG), the urinary metabolite of progesterone, has been proven to be a useful marker for monitoring mid-luteal activity in a non-invasive manner.[7] Pregnanediol-3a-glucuronide tests are a non-invasive solution for hormone monitoring utilizing first-morning urine. Pregnanediol-3a-glucuronide test strips turn positive at a threshold of 5 μg/mL in urine, the equivalent of about 10 ng/mL of progesterone in blood [7] and the associated mobile application can record and interpret the results. The utilization of PdG tests could present providers with a better diagnostic tool to identify ovulation disorders that result in suboptimal progesterone production and provide personalized treatment options to many women struggling to conceive or maintain a pregnancy.
Methods
Trial design
This study was a prospective and observational study that utilized urinary PdG hormone levels to confirm ovulation and used a mobile application diagnostic device to track PdG levels and pregnancy outcomes. The study sought to assess if certain patterns of fertility hormone levels over the whole ovulation cycle correlated with different pregnancy outcomes. The protocol, informed consent form, and all patient materials were approved by the Solutions IRB. The study was conducted in compliance with ICH GCP, the Declaration of Helsinki, and applicable laws and regulations.
Participants
Study participants were recruited from a database of established users currently using the mobile application to log their LH and PdG results. Women were eligible for study participation if they were over the age of 18 and had tracked at least one cycle utilizing the Proov® diagnostic device and mobile application. Subjects were invited to participate in this clinical trial by email. All participants were provided with an informed consent form prior to study participation. Only subjects who provided informed consent were registered into the study. Subjects were compensated $10 for their time.
Intervention
Proov® is a Class I in vitro diagnostic device cleared by the FDA for identification of urinary PdG and thus confirming ovulation at home. The urine test strips can be paired with a mobile application to facilitate recording of daily results. The mobile application has a user interface that captures, interprets the test strip, and reports the hormone concentration. The device measures the level of luteinizing hormone (LH) and pregnanediol-3-glucuronide (PdG) in urine to predict and confirm ovulation. A rise in PdG values above 5 μg/mL following an LH surge confirms the occurrence of ovulation and the beginning of the luteal phase. The number of days with sustained PdG values above 5 μg/mL were recorded. Subjects without a PdG raise were categorized as anovulatory. Study participants were instructed to conduct daily testing with first-morning urine for up to 5 cycles. All women used LH tests (sensitivity of 25 mIU/mL) and PdG tests (sensitivity of 5 μg/mL) and standardized LH and PdG testing instructions. Our PdG analysis used daily, first-morning urine samples on 4 consecutive days, collected between 7 and 10 days past peak fertility, where peak fertility was considered to be the day of the first urine LH value of 25 mIU/mL or above. The first day of a cycle was defined as the first day of bleeding occurring during menstruation. A complete cycle was considered when a peak day (LH positive test) was identified, and all 4 days of PdG testing (7-10 days after peak day) were completed. Pregnancy was defined as a positive home pregnancy test. Miscarriage is defined as a pregnancy that was lost prior to the 14th week of gestation. Subjects were free to share their results with their physicians to guide pharmacologic therapy or gain insight into infertility concerns.
Statistical analysis
Data was analyzed by Fisher’s exact test. We anticipated that at least 42 subjects would need to be enrolled for the study to have 80% power with a type one error rate of 0.05 to detect a difference in pregnancy rates. The historical incidence rate of pregnancy was 20% per cycle and the expected incidence rate in the study group was anticipated to be 5%. All continuous variables were summarized as means with their associated range. Categorical variables were reported as absolute values and percentages.
Results
Study participants
A total of 185 women were enrolled in the study between February and August 2021. Of these women, 183 reported that they were actively trying to conceive and the other two were not actively avoiding or trying (Table 1). Table 1 details the demographics of the study population. Of note, there were 3 participants that were aged 45 or above, all actively trying to conceive. One did not report a pregnancy during the study time period while the other two did report pregnancies. Only 169 women enrolled reported both height and weight, and the average recorded Body Mass Index (BMI) was 28.7, ranging between 18.1 and 54.9. Of these women, 62 (36.7%) were classified as obese, with a BMI exceeding 30. At the time of study registration, 53 participants were already diagnosed as infertile. Additionally, 64 of the respondents reported trying to conceive for over 6 months. Therefore, 117 (68%) of the respondents were considered to be subfertile. A total of 328 cycles were tracked from 178 women. Table 2 details the number of cycles tracked per participant (Table 2).
|
Characteristic |
N = 185 |
|
Age, mean, range |
33.8 (21 – 47) |
|
BMI, mean, range (n = 169) |
28.7 (18.1 – 54.9) |
|
> 30, n, % |
62 (36.7) |
|
Hispanic, Latino, or Spanish Origin |
|
|
Yes |
22 (7) |
|
Preferred not to answer |
2 (1.1) |
|
Race, n* |
|
|
White |
155 |
|
Black/African American |
14 |
|
Asian |
13 |
|
Native American/Native Alaskan |
5 |
|
Native Hawaiian/Pacific Islander |
2 |
|
Preferred not to answer |
9 |
|
Smoker or family member smokes, n, % |
17 (9.1) |
|
Actively trying to conceive, n, % |
183 (98.9) |
|
< 6 months |
70 (38.3) |
|
7 – 12 months |
55 (30.1) |
|
> 13 months |
58 (31.7) |
|
Reported irregular cycles |
33 (17.7) |
|
Reported cycle history, n, % |
|
|
Cycle length 25 – 30 days |
140 (75.7) |
|
Cycle length 31 – 36 days |
26 (14.1) |
|
< 25 or > 36 days |
9 (4.9) |
|
Not regular |
10 (5.4) |
|
Prior diagnosis of infertility |
53 (28.6) |
|
PCOS |
12 (6.5) |
|
Luteal phase defect |
2 (1.1) |
|
Male factor |
8 (4.3) |
|
Diminished ovarian reserve |
6 (3.2) |
|
Endometriosis |
5 (2.7) |
|
Unilateral tubal occlusion |
1 (0.5) |
|
Recurrent pregnancy loss |
4 (2.2) |
|
Unexplained |
15 (8.1) |
|
Baseline concomitant medications |
|
|
Progesterone |
5 |
|
Letrozole |
1 |
|
Clomiphene |
8 |
|
Clomiphene + Letrozole |
2 |
|
Letrozole + Progesterone |
8 |
|
Letrozole + Progesterone + HCG Trigger |
6 |
|
HCG Trigger + Progesterone |
2 |
|
Clomiphene + Progesterone + HCG Trigger |
2 |
|
Letrozole + HCG Trigger |
2 |
|
Clomiphene + Progesterone |
4 |
|
Clomiphene + HCG Trigger |
1 |
* Some subjects identified as more than one race.
Table 1: Subject demographics
|
Number of Cycles Tracked |
Number of Women |
|
1 |
86 |
|
2 |
49 |
|
3 |
29 |
|
4 |
13 |
|
5 |
1 |
Table 2: Cycles tracked
Hormone assessments
Of the 185 enrolled women, 172 reported complete cycles. There were 81 (47.1%) of the 172 women who reported one or more negative PdG cycles (Table 3). The vast majority of the women reporting negative cycles, 67 of the 81, were from the 117 subfertile respondents. We analyzed the data to identify risk factors for negative PdG cycles as shown in Table 3. Since over 35 years old is considered advanced maternal age and age has shown to impact fertility, we divided the respondents into above or below 35. Of the 81 women reporting negative cycles, 26 women were over 35 years old and 55 women were 35 or under. Fifty percent of the women under 35 had a negative cycle while 45% of women over 35 had a negative cycle. Being overweight increased the risk of negative PdG results by 1.73 fold (almost twice). Sixty two women in the study were classified as obese with a BMI ≥ 30 and 66.1% of women with high BMI had a negative PdG cycle whereas only 38.3% of normal BMI women had a negative cycle. No correlation was found between low AMH levels and negative PdG results. Seventy-six women reported their anti-müllerian hormone (AMH) results. Other participants had not yet received or were unsure of their results. AMH was considered low if under 1 and normal if 1 or above. Sixty-two women had normal AMH results and 14 women had low AMH results. Of the normal AMH women, 33 had a negative Proov cycle and 29 had a positive Proov cycle. Of the women low AMH, six had a negative Proov cycle and eight had a positive Proov cycle. Therefore, low AMH does not seem to increase the likelihood of low PdG levels after ovulation.
|
Negative Cycle |
|
|
Age 35 or under (n=120) |
55 (45.8%) |
|
36 and over (n=52) |
26 (50%) |
|
BMI 30 or over (N = 62) |
41 (66.1%) |
|
BMI under 30 (N = 107) |
41 (38.3%) |
|
AMH < 1 (N = 14) |
6 (42.9%) |
|
AMH > 1 (N = 62) |
33 (53.2%) |
|
Diagnosis of infertility (N = 53) |
30 (56.6%) |
|
On fertility medication (N = 78) |
9 (11.5%) |
Table 3: Distribution of negative cycles
Fertility medications
During study participation, 78 women (45.35%) were on one or more fertility medications for ovarian stimulation, ovulation induction or luteal phase support (Table 4). Table 4 lists the fertility medications utilized by subjects during trial participation. Fourty-one women were on medication at the start of the study and were using LH and PdG results to monitor their cycles while on the fertility medication(s). The other 37 of these women had a negative cycle which was presented to a physician to prescribe fertility medications. Of these 37 women, 14 of them reported both a positive cycle and positive pregnancy after the treatment was started. Of interest, eight of these women required only the addition of progesterone. Table 5 lists the medications used in the positive pregnancies (Table 5). Out of the 172 women who participated in the study, 54 women reported pregnancies. Thirty-five of these pregnancies resulted from a positive cycle and the miscarriage rate for pregnancies resulting from positive cycles was 14.3% (5/35). Fourteen of these 35 pregnancies were medicated due to a history of loss or negative PdG results. Only 19 pregnancies resulted from negative PdG cycles, and the miscarriage rate of these pregnancies was 89.5% (17/19), an increase of 75% over positive cycles (figure 1). In the two pregnancies reported in women 45years or over, one was a negative PdG cycle and resulted in miscarriage while the other was a positive cycle and resulted in a pregnancy.
|
Medication |
N = 78 |
|
Progesterone |
22 |
|
Clomiphene or Letrozole |
14 |
|
Clomiphene or Letrozole + Progesterone |
25 |
|
Clomiphene or Letrozole + Progesterone + HCG Trigger |
10 |
|
HCG Trigger + Progesterone |
3 |
|
Clomiphene or Letrozole + HCG trigger |
4 |
Table 4: Ovulation medications used during the trial
|
Medication |
N = 14 |
|
Progesterone |
8 |
|
Letrozole |
2 |
|
Clomiphene |
2 |
|
Clomiphene + Letrozole + Progesterone + HCG Trigger |
1 |
|
HCG Trigger + Progesterone |
1 |
Table 5: Medications used during positive pregnancies
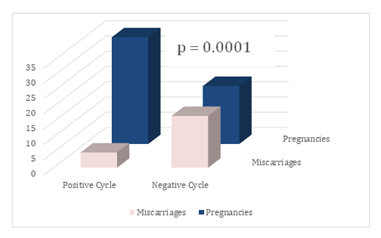
Figure 1: Pregnancies and Miscarriages by PdG Cycle Type. Utilizing Fisher’s exact test, the association of a negative or positive PdG cycle with pregnancy outcome was significant (two-sided p= 0.0001)
Discussion
Traditional predictors of fertility such as age and cycle regularity were not found to directly impact pregnancy outcomes. However, this study demonstrates a strong association between PdG cycle status and pregnancy outcomes. Urine PdG levels following ovulation at or above 5 μg/mL sustained for over 2 days were associated with a higher rate of clinical pregnancies. We observed that pregnancies resulting from a positive cycle were significantly less likely to result in a miscarriage. While age and AMH levels were not associated with negative PdG cycles, an association with BMI was significant. More specifically, obesity was a significant risk factor of insufficient progesterone elevation. While infertility and subfertility are well documented in obese women[9], this new information may contribute to the overall understanding of the underlying pathology and possible treatment options. In this study, daily hormone monitoring helped providers manage fertility medications and identify causes of infertility. As a result of hormone monitoring, 37 women were started on pharmacologic therapy and 14 reported successful pregnancies after the start of therapy. Eight of these women required only mid-luteal progesterone support. While this study did generate information for further hypothesis testing, it does have some weaknesses that could be addressed in future studies. About a quarter of the study population was on one or more fertility medications at the time of study registration. This could have caused the under-identification of negative cycles. As a result, a full assessment of who is at greatest risk of negative cycles was not possible. In addition, only one complete cycle was tracked for about 48% of the population. This resulted in an inability to understand luteal health trends in individuals and across the full population. Another weakness noted is the short duration of the hormone tracking. Many women had identified that they had a negative PdG testing cycle and were prescribed ovulation-inducing medication at the start of the next cycle. Further studies that track women for more cycles will be needed to fully assess the ability of certain medications to correct for potential luteal phase insufficiency. Further research should be conducted to better understand the frequency of negative cycles as tested via urinary PdG across various populations and their impact on pregnancy outcomes. This research should also seek to more fully characterize populations more likely to have negative cycles to make it easier to identify patients who may benefit from ovulation induction or progesterone supplementation.
Conclusions
At-home urinary PdG testing over four days offers a non-invasive method of monitoring mid-luteal activity and can be used as a marker of luteal health. It is a more patient-friendly way to assess for progesterone deficiency and provides a more complete picture of luteal function when compared to a brief snapshot of one serum progesterone level. Adding Proov PdG testing to clinical workups for female infertility may augment treatment plans and monitoring during treatment in order to ultimately increase pregnancy rates and decrease miscarriage rates.
Declarations
Ethics approval
We want to thank all the women and men involved in collection of the data analyzed in this work. The protocol, informed consent form, and all patient materials were approved by the ethics committee, chair Ayn O’Reilly, from Solutions IRB (IRB Registration # IORG0007116).
Consent to participate
All participants included in the study have consented to participate by written consent.
Consent for publication
All authors have given consent for publication.
Availability of data and material
Data is freely available on Clinicaltrials.gov, NCT05033366. Please contact Amy Beckley (amy@mfbfertility.com) to request data.
Competing interests
Beckley is the CEO and founder of MFB Fertility, Inc. A. Koudele is an employee of MFB Fertility, Inc. J. Park, J. Klein, and A. Eyvazzadeh are medical advisors for MFB Fertility.
Funding
The design of the study and collection, analysis, and interpretation of data and in writing the manuscript were all funded by MFB Fertility, Inc. the makers of Proov branded of PdG and LH test strips.
Authors’ contributions
A.B. designed and led the study, A.K. performed all the data analysis, J.P, J.K and A.E. provided medical oversight of the trial and helped with study design.
Acknowledgments
We would like to thank Maddie Barbera, Evyn White, and Oana Gharbi for their thoughtful review and editing of this manuscript.
Trial registration
NCT05033366. Registered 02 September 2021, https://clinicaltrials.gov/ct2/show/NCT05033366
References
- Sonntag B, Ludwig M. An integrated view on the luteal phase: diagnosis and treatment in subfertility. Clinical Endocrinology 77 (2012): 500-507.
- Daya S. Luteal support: Progestogens for pregnancy protection. Maturitas 65 (2009): S29-S34.
- Current clinical irrelevance of luteal phase deficiency: a committee opinion. Fertility and Sterility 103 (2015): e27-e32.
- Jones HW Jr. Luteal-phase defect: the role of Georgeanna Seegar Jones. Fertility and Sterility 90 (2008): e5-e7.
- Palomba S, Santagni S, La Sala GB. Progesterone administration for luteal phase deficiency in human reproduction: an old or new issue? J Ovarian Res 8 (2015): 77-77.
- Shah D, Nagarajan N. Luteal insufficiency in first trimester. Indian J Endocrinol Metab 17 (2013): 44-49.
- Ra, Z. SF, Gentzschein E, et al. Urinary progesterone and pregnanediol. Use for monitoring progesterone treatment. The Journal of reproductive medicine 42 (1997).
- Facts & Figures | Fertility Statistics. https://extendfertility.com/your-fertility/fertility-statistics-by-age/
- Silvestris E, De Pergola G, Rosania R, et al. Obesity as disruptor of the female fertility. Reprod Biol Endocrinol 16 (2018): 22-22.


 Impact Factor: * 3.2
Impact Factor: * 3.2 Acceptance Rate: 76.63%
Acceptance Rate: 76.63%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 