High-Definition 4K-Three-Dimensional Exoscope in Spinal Surgery: A New Era of Visualization in Microsurgery?
Stefano Peron*, Angelo Rusconi, Andrea Cividini, Marco Sicuri, Paolo Roccucci, Roberto Stefini
Neurosurgical Unit, Department of Neurosciences, ASST Ovest Milanese - Legnano Hospital, Legnano, Italy
*Corresponding Author: Stefano Peron, Neurosurgical Unit, Department of Neurosciences, ASST Ovest Milanese - Legnano Hospital, Via Papa Giovanni Paolo II, 20025 Legnano, Italy
Received: 18 June 2021; Accepted: 25 June 2021; Published: 09 July 2021
Article Information
Citation:
Stefano Peron, Angelo Rusconi, Andrea Cividini, Marco Sicuri, Paolo Roccucci, Roberto Stefini. High-Definition 4K-Three-Dimensional Exoscope in Spinal Surgery: A New Era of Visualization in Microsurgery?. Journal of Spine Research and Surgery 3 (2021): 058-070.
View / Download Pdf Share at FacebookAbstract
In the last years, the use of exoscopes in microsurgery has been proposed due to their greater and sharper intraoperative magnification. The main advantages of these devices, in addition to the high definition and magnification, are better comfort for surgeons and the possibility to involve the entire surgical team in the procedure. The aim of this study is to demonstrate feasibility and advantages of using the exoscope in spinal surgery. The exoscope is equipped with an external orbital camera on an articulated arm, controlled manually or via a foot control. The position of the exoscope, including the camera and the two monitors (55-inch and 31-inch), in the operating room (OR) depends on the spinal procedure. The surgical procedures performed with the exoscope were assessed by each surgeon immediately afterwards through a questionnaire with nine questions on the characteristics of the instrumentation. From September 2019 to March 2021, we performed 99 spinal surgical procedures using the exoscope instead of the operating microscope. In all but one evaluation question, the score obtained was equal to or greater than 4, in a range from 1 (very dissatisfied) to 5 (very satisfied). In no case was it necessary to shift from the exoscope to the microscope due to limitations or difficulties in viewing anatomical details. The exoscope is an effective tool in spinal surgery and is able to replace the microscope in microsurgical procedures, offering very high-quality and detailed anatomical visualization, allowing surgeons to operate in a comfortable position.
Keywords
Exoscope, ORBEYE, 4K-3D Images, Spinal Microsurgery
Exoscope articles Exoscope Research articles Exoscope review articles Exoscope PubMed articles Exoscope PubMed Central articles Exoscope 2023 articles Exoscope 2024 articles Exoscope Scopus articles Exoscope impact factor journals Exoscope Scopus journals Exoscope PubMed journals Exoscope medical journals Exoscope free journals Exoscope best journals Exoscope top journals Exoscope free medical journals Exoscope famous journals Exoscope Google Scholar indexed journals ORBEYE articles ORBEYE Research articles ORBEYE review articles ORBEYE PubMed articles ORBEYE PubMed Central articles ORBEYE 2023 articles ORBEYE 2024 articles ORBEYE Scopus articles ORBEYE impact factor journals ORBEYE Scopus journals ORBEYE PubMed journals ORBEYE medical journals ORBEYE free journals ORBEYE best journals ORBEYE top journals ORBEYE free medical journals ORBEYE famous journals ORBEYE Google Scholar indexed journals 4K-3D Images articles 4K-3D Images Research articles 4K-3D Images review articles 4K-3D Images PubMed articles 4K-3D Images PubMed Central articles 4K-3D Images 2023 articles 4K-3D Images 2024 articles 4K-3D Images Scopus articles 4K-3D Images impact factor journals 4K-3D Images Scopus journals 4K-3D Images PubMed journals 4K-3D Images medical journals 4K-3D Images free journals 4K-3D Images best journals 4K-3D Images top journals 4K-3D Images free medical journals 4K-3D Images famous journals 4K-3D Images Google Scholar indexed journals Spinal Microsurgery articles Spinal Microsurgery Research articles Spinal Microsurgery review articles Spinal Microsurgery PubMed articles Spinal Microsurgery PubMed Central articles Spinal Microsurgery 2023 articles Spinal Microsurgery 2024 articles Spinal Microsurgery Scopus articles Spinal Microsurgery impact factor journals Spinal Microsurgery Scopus journals Spinal Microsurgery PubMed journals Spinal Microsurgery medical journals Spinal Microsurgery free journals Spinal Microsurgery best journals Spinal Microsurgery top journals Spinal Microsurgery free medical journals Spinal Microsurgery famous journals Spinal Microsurgery Google Scholar indexed journals intraoperative articles intraoperative Research articles intraoperative review articles intraoperative PubMed articles intraoperative PubMed Central articles intraoperative 2023 articles intraoperative 2024 articles intraoperative Scopus articles intraoperative impact factor journals intraoperative Scopus journals intraoperative PubMed journals intraoperative medical journals intraoperative free journals intraoperative best journals intraoperative top journals intraoperative free medical journals intraoperative famous journals intraoperative Google Scholar indexed journals Spinal surgery articles Spinal surgery Research articles Spinal surgery review articles Spinal surgery PubMed articles Spinal surgery PubMed Central articles Spinal surgery 2023 articles Spinal surgery 2024 articles Spinal surgery Scopus articles Spinal surgery impact factor journals Spinal surgery Scopus journals Spinal surgery PubMed journals Spinal surgery medical journals Spinal surgery free journals Spinal surgery best journals Spinal surgery top journals Spinal surgery free medical journals Spinal surgery famous journals Spinal surgery Google Scholar indexed journals cranial surgery articles cranial surgery Research articles cranial surgery review articles cranial surgery PubMed articles cranial surgery PubMed Central articles cranial surgery 2023 articles cranial surgery 2024 articles cranial surgery Scopus articles cranial surgery impact factor journals cranial surgery Scopus journals cranial surgery PubMed journals cranial surgery medical journals cranial surgery free journals cranial surgery best journals cranial surgery top journals cranial surgery free medical journals cranial surgery famous journals cranial surgery Google Scholar indexed journals lumbar disc surgery articles lumbar disc surgery Research articles lumbar disc surgery review articles lumbar disc surgery PubMed articles lumbar disc surgery PubMed Central articles lumbar disc surgery 2023 articles lumbar disc surgery 2024 articles lumbar disc surgery Scopus articles lumbar disc surgery impact factor journals lumbar disc surgery Scopus journals lumbar disc surgery PubMed journals lumbar disc surgery medical journals lumbar disc surgery free journals lumbar disc surgery best journals lumbar disc surgery top journals lumbar disc surgery free medical journals lumbar disc surgery famous journals lumbar disc surgery Google Scholar indexed journals lumbar hemilaminectomy articles lumbar hemilaminectomy Research articles lumbar hemilaminectomy review articles lumbar hemilaminectomy PubMed articles lumbar hemilaminectomy PubMed Central articles lumbar hemilaminectomy 2023 articles lumbar hemilaminectomy 2024 articles lumbar hemilaminectomy Scopus articles lumbar hemilaminectomy impact factor journals lumbar hemilaminectomy Scopus journals lumbar hemilaminectomy PubMed journals lumbar hemilaminectomy medical journals lumbar hemilaminectomy free journals lumbar hemilaminectomy best journals lumbar hemilaminectomy top journals lumbar hemilaminectomy free medical journals lumbar hemilaminectomy famous journals lumbar hemilaminectomy Google Scholar indexed journals spine surgeons articles spine surgeons Research articles spine surgeons review articles spine surgeons PubMed articles spine surgeons PubMed Central articles spine surgeons 2023 articles spine surgeons 2024 articles spine surgeons Scopus articles spine surgeons impact factor journals spine surgeons Scopus journals spine surgeons PubMed journals spine surgeons medical journals spine surgeons free journals spine surgeons best journals spine surgeons top journals spine surgeons free medical journals spine surgeons famous journals spine surgeons Google Scholar indexed journals
Article Details
1. Introduction
Spinal surgery, such as cranial surgery, has been using operating microscope (OM) for years, especially when magnification of anatomical structures is needed as is normal during decompression procedures on the spinal cord and nerve roots, with both anterior and posterior approach, as well as removal of spinal and intradural tumors. Many spinal surgeries require a magnified view of the surgical field and anatomical details, which are traditionally provided by the OM. The 1970s mark the beginning of the use of the OM for spinal surgeries mainly by surgeons such as Caspar, Yasargil and Williams, who understood its advantages in lumbar disc surgery [1, 2, 3]. In recent years, thanks to significant developments in screen-based technology with head-up displays, telescopes (also known as exoscopes), have also been introduced in surgery as a viable alternative or in addition to microscopic vision. The use of the exoscope has been proposed for different microsurgical procedures, and in particular in cranial and reconstructive surgery [4-10]. So far, only a few neurosurgeons have described their experience with the exoscope in cranial surgery and, even less in spinal surgery. The use of exoscopes in spinal surgery has been reported in the literature only for degenerative cases using a microscope with a hybrid exoscopic function using either 4K-2D or HD-3D resolution. In addition, there are reports of dedicated exoscopes with 2D-HD and 3D-HD resolution [11-15]. However, the use of a combined 4K-3D image of an exoscopic system in both vertebral and intradural spinal procedures has not yet been described. We have assessed the advantages and disadvantages of a 4K-3D exoscope, namely the ORBEYE, in different spinal procedures, comparing it to traditional OM.
2. Material and Methods
We performed a prospective, non-randomized analysis of all patients undergoing spinal surgery in our department, over a period of 12 months, using the exoscope (ORBEYE 4K-three-dimensional (3D) exoscope, by Sony Olympus Medical Solutions Inc., Tokyo, Japan) for both degenerative diseases and intradural tumors. The use of the exoscope in all surgical procedures was approved by the Healthcare Authority of ASST Ovest Milanese, which did not deem necessary the approval of the Institutional Review Board as patients were treated with approved diagnostic and therapeutic procedures according to generally accepted standards of care. Each patient included in the study gave his or her informed written consent for the use of the exoscope during surgery and for the publication of anonymous radiological and intraoperative images. All surgical procedures, where the exoscope was used during all or at least a part of them, were performed by 8 senior neurosurgeons with extensive experience in spinal surgery and in the use of the OM. The surgeons evaluated each spinal procedure performed with the exoscope immediately afterwards with a questionnaire with 9 questions on the characteristics of the instrumentation (Table 1). The purpose of the questionnaire was to assess the advantages and disadvantages of the exoscope compared to the OM. Scale-based questions, i.e. 1 to 7, used a 1 to 5 rating scale, where 1 corresponded to very dissatisfied and 5 to very satisfied. Question number 8 concerned the spatial organization of the OR with all the devices located, i.e. the exoscope equipment (the camera and the two monitors), as well as the surgical, anesthesiologic, radiological and, if required, neurophysiological monitoring equipment. Question number 9 regarded the need or not to shift from vision with the exoscope to that with the OM due to difficulties in anatomical visualization or surgical manipulation.
2.1 Exoscope characteristics
The exoscope is equipped with an external 4K-3D orbital camera with a versatile semi-robotic arm, controlled manually or through a foot control that allows positioning above the operating field in multiple angles and with a very high zoom. An optical motorized continuous zoom function is provided to quickly zoom as needed, with an instant digital zoom for further magnification and extra-detailed vision. In particular, the combination of optical and digital zoom results in magnification ranging from 1.1 to 26.2x. Illumination consists of an LED panel with the light being transmitted via fiber optics to the camera head. Focal lengths and fields of view range from 220 to 550 mm and from 7.5 to 171 mm, respectively. In this way, the surgeon is able to keep the camera very far from the operating field, with greater ease of movement for him and the assistant. In addition to the camera, there are two medical 4K-3D monitors with 55-inch and 31-inch, respectively. The monitors can be positioned anywhere required inside the operating room (OR) for the convenience of both surgeons and scrub nurses. Polarized 3D glasses are required for viewing the monitor images in 3D. Specific parameters of brightness, color and saturation, e.g. slight yellow saturation and red attenuation, have been set on the exoscope and used in all spinal surgeries.
2.2 OR setting
In the OR, the arrangement of the staff, surgical and radiological equipment and the exoscope, including camera and two monitors, depends on the position of the patient and the location of the pathology. In case of posterior approaches, the surgeon and the assistant are on opposite sides of the patient with the surgeon looking at the 55-inch monitor and the assistant at the 31-inch one, with each one placed in front of each surgeon (Figure 1). Scrub nurses look at one of the two monitors based on their positioning with respect to the patient. Similarly, the other members of the surgical team, such as other surgeons, residents, students, nurses, or neurophysiology technicians, can look at one of the two monitors depending on their position in the OR. In case of anterior approaches, such as anterior cervical discectomy and fusion (ACDF), the 55-inch monitor is positioned lateral or at the head of the patient according to the position and the preference of the surgeon. The OR setting is schematically visualized for easier understanding (Figure 2).
|
Very dissatisfied |
Very satisfied |
||||
|
1. Were the 3D image quality and brightness of operative field of the exoscope superior to that of OM? |
1 |
2 |
3 |
4 |
5 |
|
2. Was it easy to zoom and focus with the exoscope? |
1 |
2 |
3 |
4 |
5 |
|
3. Was it easy to perform surgery by looking at a monitor? |
1 |
2 |
3 |
4 |
5 |
|
4. Was it comfortable to operate wearing 3D glasses? |
1 |
2 |
3 |
4 |
5 |
|
5. Could the surgery be performed in a comfortable position? |
1 |
2 |
3 |
4 |
5 |
|
6. Was it easy to perform surgery from the position of an assistant? |
1 |
2 |
3 |
4 |
5 |
|
7. Was the exoscope useful, if any, as an educational tool? |
1 |
2 |
3 |
4 |
5 |
|
8. Were the OR setting and space management comfortable with the exoscope? |
1 |
2 |
3 |
4 |
5 |
|
9. Was it necessary to switch from the exoscope to the conventional microscope? |
yes |
no |
|||
|
If yes, why? |
|||||
Table 1: Questionnaire.
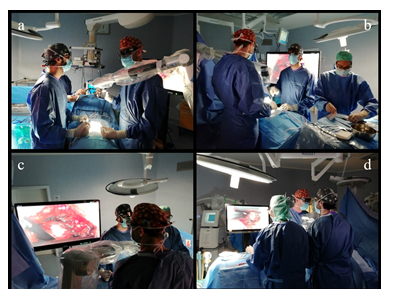
Figure 1: OR setting for lumbar hemilaminectomy. The camera is positioned very distant above the operating field, allowing the surgeons excellent freedom of movement (a,b). The surgeon and assistant, wearing polarized 3D glasses, operate by looking respectively at the 55-inch (c) and 31-inch (d) monitors placed in front of them.
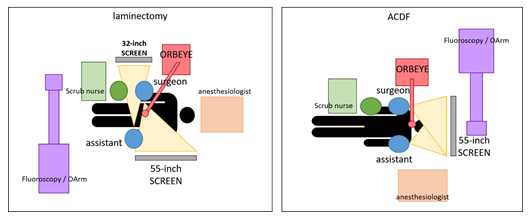
Figure 2: According to the preferences of the surgeon and the procedure performed, and different positioning of the OR staff, the ORBEYE and other radiological or navigation devices inside the OR are possible. Standard settings for laminectomy (left) and for ACDF (right) are shown.
3. Results
During a 18-month period between September 2019 and March 2021, 99 spinal microsurgeries were performed at the Department of Neurosurgery of ASST West Milan-Legnano Hospital using the exoscope instead of the OM. The procedures are summarized in Table 2. No intraoperative complications related to the instrum-entation were observed. In our series, there were no cases of superficial or deep surgical site infection. There were also no cases of CSF leakage requiring surgical revision.
Although it is beyond the scope of this publication to demonstrate the difference in operating times between the OM and the exoscope, we nevertheless highlight that, in 18-month period between February 2018 and August 2019, the total number of spinal procedures performed with a OM with integrated 4K technology, sorted by the same pathology, was approximately equal (103 vs. 99). This indicates that the operating times, including intra- and perioperative times, do not significantly different between the two technologies. In all questions of the questionnaire, the score obtained was equal to or greater than 4, with the exception of question 2 where the average score was 3, especially when choosing to use the focus manually or with the foot excluding autofocus. The results of the questionnaire are summarized in Figure 3. With regards to question 8, the placement of the exoscope and other equipment in the OR did not represent an encumbrance, but rather encouraged the participation of medical and paramedical staff in the surgery.
In the same way, considering question 9, in no case did exoscopic vision or handling create difficulties in visualizing anatomical details with the need to change to the OM. There was a short learning curve aimed at losing the OM-trained behavior, such as following the body of the microscope to look into the eyepieces, and gaining a new proprioceptive scheme in surgical manipulation on head-up displays. All surgeons claimed that the learning curve for the exoscope was faster than that for OM, with a minimum of 10 procedures performed with the exoscope sufficient to obtain good manual skills.
|
Pathology |
Procedure |
Number |
|
Lumbar degenerative disease (stenosis) |
hemi-/laminectomy |
33 |
|
Cervical disc herniation |
ACDF |
14 |
|
Lumbar disc herniation |
removal |
28 |
|
Intradural tumor |
hemi-/laminectomy and removal |
24 |
Table 2: Spinal surgical procedures performed with 4k-3D exoscope.
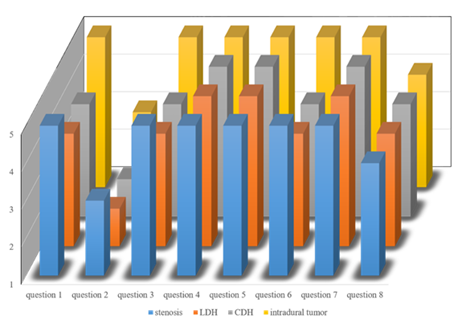
Figure 3: Diagram showing the results of the questionnaire with the individual questions on the X-Axis, average ratings (ranging from 1 representing “Very dissatisfied” to 5 representing “Very satisfied”) on the Y-Axis and the grouping of the procedures (stenosis, LDH, CDH, intradural tumor with colors as indicated in legend) on the Z-Axis.
4. Discussion
Several decades ago, the advent of the OM revolutionized the world of surgery, allowing the surgeon, to visualize, illuminate, and enlarge anatomical details at the same time like never before. This contributed to considerably reducing surgical risks with significant advantages for the patient in terms of clinical outcomes. From the late 1970s, but especially between the 1980s and, even more, the 1990s, the OM increasingly became an important tool in spinal surgery and is now used by default in many spinal procedures where magnification is required or desirable, as in the case of e.g.: cervical and lumbar herniectomies or laminotomy [16-17]. As with many surgical approaches, which, compared to the past focus more on pursuing a goal of minimal invasiveness, developments in technology in the surgical field are often a response to the requests of patients, who ask for even greater attention to detail, as well as better clinical results [18]. In all areas, in fact, technology has always had a great appeal to users. Many companies on the international market compete to find solutions with the greatest impact in terms of both aesthetics and functionality, and, especially in medicine, of efficacy and outcomes.
Belykh makes it very clear how, to lead towards the goal of better outcomes, there is the need for improved illumination, optimal stereoscopic visualization of the details of small structures in hidden areas, in particular through minimally invasive approaches, as well as for a technology allowing smooth, rapid, and well-controlled movements. In fact, if better viewing of the surgical target can be achieved through well-planned patient positioning and surgical approach, optimal illumination and magnification depend only on the visualization device [19]. In recent years, the exoscope has been proposed as a possible alternative to the OM offering superior ergonomics, ease of use, and depth of focus [6-8]. These systems aim to combine the advantages of the microscope with those of the endoscope, and at the same time trying to overcome the limits of both, such as the poor ease of movement and ability to adapt to the operating room setting, the limits in the depth of field and poor ergonomics for surgeons [20]. The advent of the exoscope has the potential to represent an epochal change and the beginning of a new way of performing surgery, and thus a real step forward in technological innovation in spinal neurosurgery. If we look at the past, a similar change could be compared to the introduction of the microscope and, more recently, of the endoscope in cranial and spinal surgery. The advantages of a new technique in surgery and, in our case, of a new technology, are very difficult to demonstrate and, at least at the beginning, widespread adoption by the surgical community may take a long time. The introduction of the OM in spinal surgery represented a paradigm shift some ten years ago [21-22].
In addition, the introduction of the endoscope in ventricular surgery and hydrocephalus and, more recently, in skull base and spinal surgery, was accompanied by initial skepticism. Even today its use, due to advances in microneurosurgery, is still subject of controversial discussion among neurosurgeons [23-24]. As Barkun well described, the adoption of an innovation into standard surgical practice can be defined by the increase in the number of surgeons doing the procedure over time, which will occur until it is either accepted or rejected [18]. There is always a starting point in technological innovation when adoption is low and involves a small number of surgeons. The final step occurs when the adoption of that technology suddenly involves an increasing number of people (i.e. surgeons) until reaching a tipping point. Barkun specifies what the characteristics of these stages are, including the level of refinement of the new technique, need for ethical disclosure, number of patients on whom it is being carried out, and the number and characteristics of surgeons who are using it [18]. At the moment, some publications in the literature have described procedures performed with an exoscopic system, but only on degenerative vertebral pathologies. In particular, Ariffin described the use of a OM with associated optional 4K 2D or HD 3D technology (Kinevo 900 Carl Zeiss Meditec AG, Jena, Germany) in 69 tubular-access minimally invasive spine surgeries (MISS) [12]. Muhammad et al. presented their experience on 8 cranial and spinal (degenerative) procedures with a 2D exoscope (Synaptive Medical Modus V), while Barbagallo described 2 cases of ACDF with a 3D high-definition exoscope (Vitom 3D, Karl Storz, Tuttlingen, Germany) [13, 14].
In all studies, the exoscope was appreciated for its advantages in ergonomics, magnification, depth of field, reducing the amount of refocusing, and teaching. In particular, Belykh stresses how, once the surgeon has become accustomed to operating while looking at a 3D projection of the surgical site rather than the actual surgical site, the exoscope provides additional freedom of movement, easy positioning and target finding, and greater degrees of surgical freedom [19]. On the other hand, the lack of stereoscopy is reported as one of the main limits, possibly due to the lack of 3D visualization. Another important disadvantage reported by various authors is the difficulty in assisting the surgeon from a position of the assistant [9]. In fact, when the assistant is positioned less than 180° in relation to the surgeon, the surgical field on the monitor has a rotated view, creating considerable difficulties in orientation and manipulation on the operating field. Instead, with the OM assistants can orientate the viewing angle according to their position. This is not possible with the exoscope, which allows you to rotate the image only 180° from the surgeon's vision. This might explain the rating in question 6 with an average of 3 for LDH and CDH.
However, this disadvantage can be eliminated in spinal surgery, where surgeons are usually positioned on both sides of the patient. In this case, assistants can look at the monitor facing them with an image rotated 180°, exactly suited to their position, ensuring comfortable viewing and precise manipulation. Murai et al. performed an observational study with a survey on advantages and disadvantages of the ORBEYE in microsurgery [9]. They performed 22 procedures, including three laminectomies. No complications were observed. In their opinion, thanks to the ORBEYE the surgeon could operate in a comfortable position, which was much better than with the OM in terms of ergonomic features. However, they concluded that, in their limited experience, the exoscope is unable to replace the microscope in all operations. We used the exoscope for different spinal procedures. Our series of spinal surgical procedures with the exoscope is currently the largest and most complete reported in the literature. In most cases, surgery was performed with the two surgeons on both sides of the patient. This allowed positioning the two monitors facing each other, the 55-inch monitor in front of the surgeon and the 31-inch monitor in front of the assistant, allowing the latter to also have optimal vision. In some procedures on the cervical spine, based on the comfort and preference of the surgeon, the monitor was placed behind the patient's head. However, both surgeons were able to look at the same monitor, without image rotation problems or discomfort in terms of manipulation on the operating field.
Moreover, the OM frequently requires the surgeon to assume unergonomic body postures to keep the surgical site in view, sometimes for prolonged periods of time. This can cause discomfort and, even worse, musculoskeletal disorders of the cervical and lumbar spine, as well as of the shoulders, leading to chronic pain and osteoarthritis [1]. The exoscope allows for neutral cervical spine posture by placing the monitor at eye level, directly in front of the surgeon, or at any angle required. This ensures an upright and neutral posture, avoiding long hours spent in a fixed position looking through a microscope, or with the head bent over the patient, with or without loupes [7, 8, 10]. In all cases, by carrying out the procedure in an upright position, surgeons avoid keeping their head flexed on the patient or on the microscope lenses. This may result in considerably increased comfort for both surgeons. This advantage is even more evident when the surgeon has to look and work in the most uncomfortable and hidden corners with an unfavorable inclination. In these cases, using the OM, the surgeon is forced to oblique and consequently to assume a position that involves twisting of the neck and back. In similar situations, and when using the exoscope, surgeons can change viewing angles easily by simply tilting the camera head, and without having to change posture. This offers a significant advantage in terms of the surgeon’s comfort.
In our opinion, the high-resolution 4K-3D images of nervous and vascular structures provided by the exoscope make surgery more accurate and precise, with optimal perception of depth and colors as well as magnification (Figure 4). To demonstrate this, we used a personal feedback questionnaire to all spinal surgeons of our team currently using the exoscope. The questionnaire was aimed at collecting opinions on utilization of the exoscope. The aim was to identify the advantages as well as to highlight any limitations or difficulties and to transform them into suggestions for improvement of this technology. The high score obtained by the questionnaire (i.e. 4 or more in 6 of 7 questions) confirms considerable appreciation for this device, in particular for image quality and comfort in operating in an upright position looking at 3D monitors. Another key advantage of the exoscope is that it enables involving the entire surgical team in the procedure, sharing information and suggestions with other surgeons, neurologists, pathologists, or nursing staff.
In fact, the exoscope offers high quality, 4K-3D images for everyone who watches the 55-inch or 31-inch monitor using special polarized glasses. This advantage can also be used for educational purposes for students and residents, because the use of external monitors and glasses gives trainees the same high-resolution view as the surgeon [7]. Furthermore, the exoscope, allows more space around the operating table and the patient. This is especially useful in procedures that require multiple equipment, in particular navigation devices normally requiring moving the microscope away in order to give the navigation system’s camera an unobstructed view of the surgical site (Figure 5). In this regard, Yoon's observation concerning possible involuntary errors due to fractions of a second in which the surgeon, manipulating an instrument, for example a navigated instrument, looks away from the operating field to direct the view towards the monitor in front of him, is interesting. Yoon correctly notes that in certain situations, as in our case during the removal of spinal cord tumors, these errors can lead to minor tissue damage but with major consequences for the patient [25].
This risk is significantly reduced with our exoscope since, thanks to the Picture-in-Picture (PiP) or Picture-out-Picture (PoP) mode, and it is possible to check, whenever necessary, the position of the navigated instrument directly on the 55-inch monitor, without ever taking your eyes off the operating field (Figure 6). The operative environment and surgical and radiological equipment in the operating room are reported to be among the potential modifiable risk factors for surgical site infection [26]. However, in our series, there were no cases of superficial or deep infection, despite use of a large amount of equipment, with (i.e. camera) and without (i.e. two monitors and recording system) draping, in addition to any navigation systems or equipment for neurophysiological monitoring, set up inside the OR during spinal exoscopic surgery (Figure 2). As is obvious for all new technologies, a learning curve is also required for the exoscope. In our opinion, for example, the focusing and zoom system, whether used with manual adjustment, which requires the surgeon to move the hand even far away from the operating field to reach the controls on the camera (while it is much easier with the microscope where the controls are placed on the knobs next to the eyepieces), or with the foot control, is not easy to learn, therefore requiring some training. This may also justify the average score of 3 in question 2.
However, in our opinion, as in the literature, with 10 procedures the exoscope has a shorter learning curve than the OM [6, 10, 11]. Among the aspects that are new to the surgeon we noticed a difficulty in gaining a proprioceptive ability when manipulating the instruments by looking at the operating field projected on the 3D monitor, which might be the reason for the average rating of 3 for LDH and CDH in question 3. This aspect is particularly highlighted when working at high magnification and when the surgeon needs frequent and rapid movements of moving away from and approaching the surgical target. Our experience shows a greater difficulty, compared to OM, in returning with the instruments to the operating field with a feeling both subjective, by the surgeon, and objective, by observers, of a relative lesser safety in reaching the target. This occurred in all our surgeons who have used the exoscope and was more evident in the initial stages of the surgical procedure, considerably decreasing during the same, as a sign of a general ease of adaptation to the new technology. This limit can be even more easily overcome for those who frequently perform endoscopic surgical procedures, as they are used to operating with their view directed onto a monitor and not directly on the operating field or into the microscope’s eyepieces. In any case, this ability is rapidly acquired with the increase in the total performed procedures.
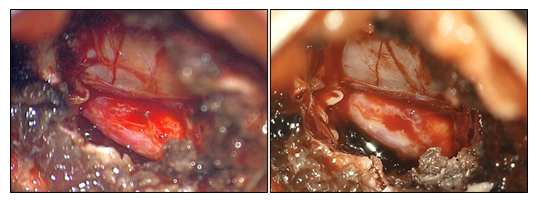
Figure 4: Microscopic (left) and exoscopic (right) vision during operation for lumbar disc herniation. Greater sharpness and more faithful reproduction of anatomical details can be noticed. In this procedure, as in all spinal procedures, a color setting with red attenuation has been used for personal preference of the surgeons.
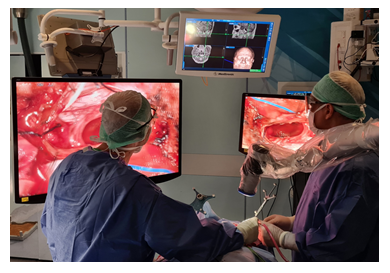
Figure 5: A case of cervical meningioma operated on with the Orbeye. The surgeon and assistant can operate comfortably by looking at the two monitors placed in front of them. The large working space around the operating field allows the surgeon to use the navigation devices with great ease and freedom of movement.
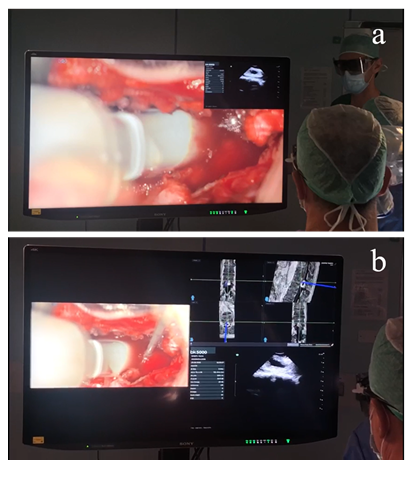
Figure 6: During the removal of an intradural tumor (i.e. lumbar neurinoma) the PiP (a) or PoP (b) mode can be used to check, all on the 55-inch monitor, the position of the tumor with the navigated instrument and with the ultrasound probe, even simultaneously. In this way, the surgeon never has to look away from the operating field.
5. Conclusions
Our questionnaire highlights a very good opinion of the exoscope in terms of image quality, comfort in operating looking at 3D monitors, and postural comfort for surgeons. Although a relatively small number of 99 cases represents a limited first experience with this new device, we firmly believe that the exoscope can be very effective in spinal procedures where high magnification is required, replacing the OM.
In particular, our exoscope offers 4K-3D images with very high-quality and detailed anatomical visualization and allows surgeons to operate in a comfortable position. The possibility of involving all the OR staff in the procedure is another significant advantage, as well as being fundamental for both in terms of sharing key decisions and for educational purposes. The learning curve, compared to that of the OM, is faster and the difficulty highlighted in acquiring a proprioceptive skill in operating looking at the head-up monitors can be easily overcome by increasing the number of procedures performed.
Conflicts of Interest
The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
References
- Caspar W. A new surgical procedure for lumbar disc herniation causing less tissue damage through a microsurgical approach. Adv Neurosurg 4 (1977): 74-77.
- Yasargil MG. Microsurgical operation of herniated lumbar disc. Adv Neurosurg 7 (1977): 81.
- Williams RW. Microlumbar discectomy: a conservative surgical approach to the virgin herniated lumbar disc. Spine 3 (1978): 175-182.
- Ahmad FI, Mericli AF, DeFazio MV, et al. Application of the ORBEYE three-dimensional exoscope for microsurgical procedures. Microsurgery 40 (2020): 468-472.
- Langer DJ, White TG, Schulder M, et al. Advances in Intraoperative Optics: A Brief Review of Current Exoscope Platforms. Oper Neurosurg (Hagerstown) 19 (2020): 84-93.
- Takahashi S, Toda M, Nishimoto M, et al. Pros and cons of using ORBEYE™ for microneurosurgery. Clin Neurol Neurosurg 174 (2018): 57-62.
- Mamelak AN, Danielpour M, Black KL, et al. A high-definition exoscope system for neurosurgery and other microsurgical disciplines: preliminary report. Surg Innov 15 (2008): 38-46.
- Krishnan KG, Schöller K, Uhl E. Application of a Compact High-Definition Exoscope for Illumination and Magnification in High-Precision Surgical Procedures. World Neurosurg 97 (2017): 652-660.
- Murai Y, Sato S, Yui K, et al. Preliminary Clinical Microneurosurgical Experience With the 4K3-Dimensional Microvideoscope (ORBEYE) System for Microneurological Surgery: Observation Study. Oper Neurosurg (Hagerstown) 16 (2019): 707-716.
- Sack J, Steinberg JA, Rennert RC, et al. Initial Experience Using a High-Definition 3-Dimensional Exoscope System for Microneurosurgery. Oper Neurosurg (Hagerstown) 14 (2018): 395-401.
- Kwan K, Schneider JR, Du V, et al. Lessons Learned Using a High-Definition 3-Dimensional Exoscope for Spinal Surgery. Oper Neurosurg (Hagerstown) 16 (2019): 619-625.
- Ariffin MHM, Ibrahim K, Baharudin A, et al. Early Experience, Setup, Learning Curve, Benefits, and Complications Associated with Exoscope and Three-Dimensional 4K Hybrid Digital Visualizations in Minimally Invasive Spine Surgery. Asian Spine J 14 (2020): 59-65.
- Muhammad S, Lehecka M, Niemelä M. Preliminary experience with a digital robotic exoscope in cranial and spinal surgery: a review of the Synaptive Modus V system. Acta Neurochir (Wien) 161 (2019): 2175-2180.
- Barbagallo GMV, Certo F. Three-Dimensional, High-Definition Exoscopic Anterior Cervical Discectomy and Fusion: A Valid Alternative to Microscope-Assisted Surgery. World Neurosurg 130: e244-e250.
- Oren J, Kwan K, Schneider J, et al. Minimally Invasive Navigated Foraminal Discectomy via Contralateral Approach Using a 3-Dimensional 4K High-Definition Exoscope: 2-Dimensional Operative Video. Oper Neurosurg (Hagerstown) 19 (2020): E188.
- Damodaran O, Lee J, Lee G. Microscope in modern spinal surgery: advantages, ergonomics and limitations. ANZ J Surg 83 (2013): 211-214.
- Grochulla F. Surgical Microscopy in Spinal Surgery. Springer, Berlin, Heidelberg (2012).
- Barkun JS, Aronson JK, Feldman LS, et al. Evaluation and stages of surgical innovations. Lancet 374 (2009): 1089-1096.
- Belykh EG, Zhao X, Cavallo C, et al. Laboratory Evaluation of a Robotic Operative Microscope - Visualization Platform for Neurosurgery. Cureus 10 (2018): e3072.
- Chakravarthy V, Sheikh S, Schmidt E, et al. Imaging Technologies in Spine Surgery. Neurosurg Clin N Am 31 (2020): 93-101.
- Donaghy RM. The history of microsurgery in neurosurgery. Clin Neurosurg 26 (1979): 619-625.
- Kriss TC, Kriss VM. History of the operating microscope: from magnifying glass to microneurosurgery. Neurosurgery 42 (1998): 899-908.
- Abbott R. History of neuroendoscopy. Neurosurg Clin N Am 15 (2004): 1-7.
- Cavallo LM, Somma T, Solari D, et al. Endoscopic Endonasal Transsphenoidal Surgery: History and Evolution. World Neurosurg 127 (2019): 686-694.
- Yoon JW, Chen RE, Kim EJ, et al. Augmented reality for the surgeon: Systematic review. Int J Med Robot 14 (2018): e1914.
- Basques BA, Golinvaux NS, Bohl DD, et al. Use of an operating microscope during spine surgery is associated with minor increases in operating room times and no increased risk of infection. Spine 39 (2014): 1910-1916.


 Impact Factor: * 3.123
Impact Factor: * 3.123 Acceptance Rate: 75.30%
Acceptance Rate: 75.30%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 