Mucormycosis, COVID-19 and Acute Lymphoid Leukemia: Case Report
Edgar Laurencio Cordero-Hernández1, Carlos Martínez Murillo1, Gilberto Barranco-Lampón1, Adán Germán Gallardo Rodríguez1, Javier Araiza Santibañez2, Itzel Arias-Castro3, Adrián Santoyo-Sánchez4, Christian Omar Ramos-Peñafiel1*
1Department of Hematology, General Hospital of Mexico, Mexico City, Mexico
2Department of Dermatology, General Hospital of Mexico, Mexico City, Mexico
3Department of Medicine, Autonomous University of Sinaloa, Sinaloa, Mexico
4Departement of Experimental Medicine, Autonomous University of Mexico, Mexico
*Corresponding author: Christian Omar Ramos Peñafiel, Department of Hematology, General Hospital of Mexico, Mexico City. C.P. 06726, Mexico
Received: 01 February 2022; Accepted: 09 February 2022; Published: 25 May 2022
Article Information
Citation: Edgar Laurencio Cordero-Hernández, Carlos Martínez Murillo, Gilberto Barranco-Lampón, Adán Germán Gallardo Rodríguez, Javier Araiza Santibañez, Itzel Arias-Castro, Adrián Santoyo-Sánchez, Christian Omar Ramos-Peñafiel. Mucormycosis, COVID-19 and Acute Lymphoid Leukemia: Case Report. Archives of Internal Medicine Research 5 (2022): 192-198.
View / Download Pdf Share at FacebookAbstract
Background: During the era of COVID-19, an increase has surged in the number of cases of mucormycosis; the use of high doses of steroids, and states of immunosuppression such as diabetes are the main related risk factors. In the treatment of acute leukemia, it is recommended to reduce the dose of chemotherapy, postponing highly myeloablative schemes and the use of caution with monoclonal antibodies.
Case presentation: We present the case of a patient (25 years-old male) with acute lymphoblastic leukemia and COVID-19 confirmed infection who developed rhinocerebral mucormycosis during the induction stage.
Conclusion: In the age of COVID-19, taking samples through a nasal swab should be avoided in individuals with risk of infection from mucormycosis, such as diabetes, cancer, or individuals treated with high doses of steroids.
Keywords
<p>COVID-19, Mucormycosis, Leukemia, Immunosuppression</p>
Article Details
1. Introduction
At the beginning of 2021, the critical moment of the pandemic was located in Latin America; countries like Mexico and Brazil recorded a significant rise in the mortality associated with individuals with recog-nized risk factors such as diabetes and hypertension [1, 2]. Then population with cancer was recognized as vulnerable since the first studies in China, requiring the modification of cytotoxic schemes, delaying surgeries and deferring doctor visits [3, 4]. In leukemia, it was recognized that the mortality associated with COVID-19 is potentially higher than 30%, limiting the efficacy of schemes with conven-tional chemotherapy or target therapies (venetoclax, FLT-3 inhibitors, IDH1/2 inhibitors), due to the delay in administration, in spite of the patients being asymptomatic [5]. In acute lymphoblastic leukemia, cooperative groups agree that highly immunosup-pressor regimens should be used with caution, as well as the combination of monoclonal antibodies, and even transplant of hematopoietic progenitors should be delayed [6, 7]. With COVID-19, different bacterial (mainly Staphylococcus, Escherichia coli, Pseudomonas aeruginosa), viral (cytomegalovirus) and fungal infections are still fund among the main causes of complications related with treatment, and a polymerase chain reaction test should be applied in all cases with fever [8, 9]. Mucormycosis (zygomy-cosis) is a fungal infection caused by mucoral fungi (Rhizopus oryzae is the most common) that greatly affects immunocompromised individuals with high mortality [10]. Among the main risk factors for its development are neutropenia (60%) and acute leukemia (65%), with diagnosis from suspicion in more than half the cases and 40% performed post-mortem [11]. In our hospital, Bonifaz et al. recorded the experience of 35 years, reporting 214 cases of mucormycosis, the majority in patients with diabetes (76.6%), followed by individuals with hematological neoplasia (15.4%) [12]. Recently, a rapid rise in cases of mucormycosis was reported in individuals with COVID-19, associated mainly with uncontrolled diabetes mellitus (94%) or patients in critical state [13]. Sen et al. reported the series of six cases of patients with COVID-19 and mucormycosis that required antifungal treatment (liposomal amphot-ericin, posaconazol) and debridement treatment of necrotic tissue, with a time of appearance of symp-toms associated with mucormcosis of 15.6 ± 9.6 (3-42) days [14]. We present the case of a patient with acute lymphoblastic leukemia submitted to induction chemotherapy who presented COVID-19 infection and later nasal swab showed infection by rhino-cerebral mucormycosis.
2. Case Presentation
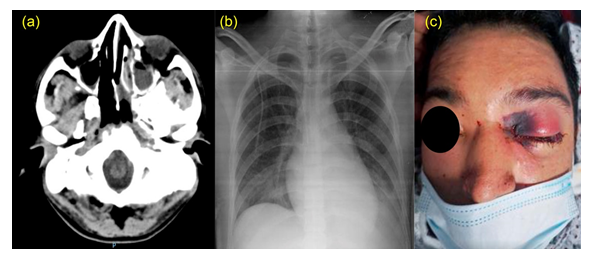
Twenty-five year old male patient with history of insulin-dependent diabetes mellitus diagnosed since adolescence, in prolonged action insulin treatment with adequate adherence to treatment, entered to Department of Hematology for a study of leukocytes of 35 x109/L with 80% atypical lymphocytes, blood smear was performed, analysis of bone marrow identified acute lymphoblastic leukemia precursor B, habitual risk, negative Philadelphia chromosome, deciding to initiate induction stage with scheme of CALGB 10403. During pre-treatment with high doses of steroids, he developed lack of glycemic control, requiring an adjustment to insulin dose and receiving chemotherapy dose on day +1 and +8 (vincristine and doxorubicin), due to hyperglycemia, the use of asparaginase was deferred. On day +13 of treatment, neutrophil count was 0.1 x109/L, he presented fever, and empirical antimicrobial therapy was initiated for febrile neutropenia. Due to the persistence of fever and oxygen desaturation, the polymerase chain reaction (PCR) test for SARS-CoV2 was requested, taking the sample by naso-pharingeal swab. Chemotherapy treatment was suspended until the fever improved. At 24 hrs from the swab, the patient showed rhinorrhea in the left nasal passage and pain, beginning with an increase in volume, and a tomographic study was taken, showing rhinosinusitis (Figure 1a); his thorax X-ray showed data suggesting pneumonia (Figure 1b), he continued antibiotic and antifungal treatment with fluconazole.
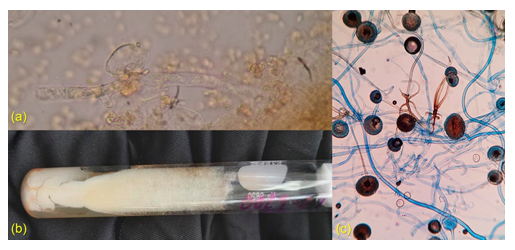
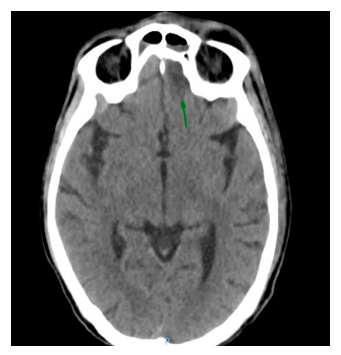
During follow-up, the patient persisted with fever, presenting swelling, erythema and intense pain at the left nasal rim (Figure 1c), progressing rapidly in the following 48 hours. A culture was requested of nasal exudate, and direct visualization of fungi with KOH (potassium hydroxide) at 10% positive for Rhizopus arrhizus (Figures 2). Our institution does not have other diagnostic tools for fungi such as PCR or sequencing methods. Treatment was begun based on amphotericin B; he persisted with fever and data of neurological deterioration, the tomograph after 5 days from the start of treatment showed cerebral affect-tation compatible with rhino-cerebral mucormycosis (Figure 3). The patient showed progressive deteri-oration, requiring ventilator support, and dying 7 days after start of symptoms. PCR results for SARS-CoV2 at 72 hrs from the swab were positive.
3. Discussion
Infectious processes are among the main compli-cations related with cancer treatment, especially with high-intensity chemotherapy. In leukemia, the use of high doses of steroids, especially during induction therapy, or comorbidities such as diabetes mellitus, are among the main risk factors for developing mucormycosis [15, 16]. During the last year, an increase has been observed in cases of mucorm-ycosis, but associated with COVID-19, especially in areas such as India [17, 18]. There is a precedent in leukemia; Zurl et al. reported the case of a patient with acute myeloid leukemia, aged 53 years, who developed pulmonary invasive mucormycosis; among his risk background were obesity (body mass index of 34 kg/m2), depression, and being under prophylactic treatment with voriconazol 400 mg once a day, after induction on day +54 of treatment, he developed a positive result for SARS CoV2 through nasopharyngeal swab; at 5 days he presented respiratory manifestations, entering on intensive care unit and diagnosed with Rhizopus microsporus infec-tion through DNA sequencing methods [19]. Upon analyzing the different cases of COVID-19 chara-cterized by pain and increase in volume of the orbital region [20-22], it has been agreed that the use of steroids is the main risk factor for developing rhino-cerebral mucormycosis. Other factors such as pro-longed stay in an intensive care unit, ventilator support, pronation and finally the use of nasal points with high oxygen flow are factors that contribute to the colonization of fungal infections in individuals with COVID-19 [23]. Timely identification of mani-festations suggesting mucormycosis (edema, pain, erythema, nasal flow), Access to diagnostic tools (cultures, polymerase chain reaction test, sequencing methods), and the timely start of antifungal therapies (liposomal amphotericin, voriconazol) are fundamen-tal to avoid fulminant cases in patients with COVID-19 [24-26]. Finally, in our experience, individuals with leukemia, especially under immunosuppressor treatment with high doses of steroids, are susceptible to opportunist infections such as mucormycosis. In the age of COVID-19, other factors have been added, such as pronation, nasal flow with high-flow oxygen, and especially we must consider that techniques such as nasal swab are a critical factor for mechanical rupture and fungal colonization. Throughout the pan-demic, complications have been described in the technique, including skull perforation, cerebrospinal fluid fistula or mucous lesion; knowing this, adequate training for taking samples is fundamental (including videos on YouTube), especially in individuals with risk factors such as cancer or diabetes [27, 28].
4. Conclusions
In conclusion, individuals with lymphoblastic leuke-mia have an important risk of developing mucorm-ycosis, and the taking of routine nasal samples may be deferred, or preferentially other sites such as the oral cavity should be used.
References
- COVID-19 Map - Johns Hopkins Corona-virus Resource Center (2020).
- Chen T, Wu D, Chen H, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: Retrospective study. BMJ 368 (2020): m1091.
- Moujaess E, Kourie HR, Ghosn M. Cancer patients and research during COVID-19 pandemic: A systematic review of current evidence. Crit Rev Oncol Hematol 150 (2020): 102972.
- Liu C, Zhao Y, Okwan-Duodu D, et al. COVID-19 in cancer patients: risk, clinical features, and management. Cancer Biol Med. Cancer Biol Med 17 (2020): 519-527.
- Paul S, Rausch CR, Jain N, et al. Treating Leukemia in the Time of COVID-19. Acta Haematol 144 (2021): 132-145.
- Alhuraiji A, Eldadah S, Alfraih F, et al. Optimal Management of Acute Lympho-blastic Leukemia (ALL) in Adult Patients During the Novel Coronavirus Disease 2019 (COVID-19) Pandemic. Gulf J Oncol 1 (2020): 7-18.
- Taub JW, Ge Y, Xavier AC. COVID-19 and childhood acute lymphoblastic leukemia. Pediatr Blood Cancer. John Wiley & Sons, Ltd 67 (2020): e28400.
- Boyd K, Parcell B, Tauro S. Immuno-suppression in hematological cancer patients with Covid-19 —Uncomplicated infections but delayed viral clearance?. Leuk Res 96 (2020): 106407.
- Cooksley T, Font C, Scotte F, et al. Emerging challenges in the evaluation of fever in cancer patients at risk of febrile neutropenia in the era of COVID-19: a MASCC position paper. Support Care Cancer 29 (2021): 1129-1138.
- Hassan MIA, Voigt K. Pathogenicity patterns of mucormycosis: epidemiology, interaction with immune cells and virulence factors. Med Mycol 57 (2019): S245-S56.
- Krcmery V, Kunova E, Jesenska Z, et al. Invasive mold infections in cancer patients: 5 years’ experience withAspergillus, Mucor, Fusarium and Acremonium infections. Sup-port Care Cancer 4 (1996): 39-45.
- Bonifaz A, Tirado-Sánchez A, Hernández-Medel ML, et al. Mucormycosis at a tertiary-care center in Mexico. A 35-year retrospective study of 214 cases. Mycoses 64 (2021): 372-380.
- John TM, Jacob CN, Kontoyiannis DP. When Uncontrolled Diabetes Mellitus and Severe COVID-19 Converge: The Perfect Storm for Mucormycosis. J Fungi 7 (2021): 298.
- Sen M, Lahane S, Lahane T, et al. Mucor in a Viral Land: A Tale of Two Pathogens. Indian J Ophthalmol 69 (2021): 244.
- Greene J, Pak J, Tucci V, et al. Mucor-mycosis in immunochallenged patients. J Emergencies, Trauma Shock 1 (2008): 106.
- Skiada A, Lanternier F, Groll AH, et al. Diagnosis and treatment of mucormycosis in patients with hematological malignancies: guidelines from the 3rd European Confer-ence on Infections in Leukemia (ECIL 3). Haematologica 98 (2013): 492-504.
- Verma DK, Bali RK. COVID-19 and Mucormycosis of the Craniofacial skeleton: Causal, Contributory or Coincidental? J Maxillofac Oral Surg 20 (2021): 165-166.
- Ahmadikia K, Hashemi SJ, Khodavaisy S, et al. The double-edged sword of systemic corticosteroid therapy in viral pneumonia: A case report and comparative review of influenza-associated mucormycosis versus COVID-19 associated mucormycosis. Mycoses (2021): 13256.
- Zurl C, Hoenigl M, Schulz E, et al. Autopsy Proven Pulmonary Mucormycosis Due to Rhizopus microsporus in a Critically Ill COVID-19 Patient with Underlying Hema-tological Malignancy. J Fungi 7 (2021): 88.
- Maini A, Tomar G, Khanna D, et al. Sino-orbital mucormycosis in a COVID-19 patient: A case report. Int J Surg Case Rep 82 (2021): 105957.
- Saldanha M, Reddy R, Vincent MJ. Paranasal Mucormycosis in COVID-19 Patient. Indian J Otolaryngol Head Neck Surg (2021).
- Veisi A, Bagheri A, Eshaghi M, et al. Rhino-orbital mucormycosis during steroid therapy in COVID-19 patients: A case report. Eur J Ophthalmol (2021): 1120672-12110094.
- Sarkar S, Gokhale T, Choudhury S, et al. COVID-19 and orbital mucormycosis. Indian J Ophthalmol 69 (2021): 1002.
- Karimi-Galougahi M, Arastou S, Haseli S. Fulminant mucormycosis complicating coronavirus disease 2019 (COVID-19). Int Forum Allergy Rhinol 11 (2021): 1029-1030.
- Waizel-Haiat S, Guerrero-Paz JA, Sanchez-Hurtado L, et al. A Case of Fatal Rhino-Orbital Mucormycosis Associated With New Onset Diabetic Ketoacidosis and COVID-19. Cureus (2021).
- Dallalzadeh LO, Ozzello DJ, Liu CY, et al. Secondary infection with rhino-orbital cerebral mucormycosis associated with COVID-19. Orbit (2021): 1-4.
- Zedtwitz-Liebenstein K. Correctly perform-ed nasal swabs. Infection (2021).
- Itamura K, Wu A, Illing E, et al. YouTube Videos Demonstrating the Nasopharyngeal Swab Technique for SARS-CoV-2 Speci-men Collection: Content Analysis. JMIR Public Heal Surveill 7 (2021): e24220.


 Impact Factor: * 3.6
Impact Factor: * 3.6 Acceptance Rate: 78.21%
Acceptance Rate: 78.21%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 