The Systematic Synthesis of Carbon Nanotubes from Aliphatic-Aromatic Compound Mixture Resolves Growth Uniformity and Production Complexity
Sydur Rahman Md*
Department of Basic Science, Lecturer in Chemistry, World University of Bangladesh, Dhaka, Bangladesh
*Corresponding author: Sydur Rahman Md, Department of Basic Science, Lecturer in Chemistry, World University of Bangladesh, Dhaka, Bangladesh
Received: 12 December 2019; Accepted: 07 January 2020; Published: 11 January 2020
Article Information
Citation:
Sydur Rahman Md. The Systematic Synthesis of Carbon Nanotubes from Aliphatic-Aromatic Compound Mixture Resolves Growth Uniformity and Production Complexity. Journal of Nanotechnology Research 2 (2020): 001-009.
DOI: 10.26502/jnr.2688-85210012
View / Download Pdf Share at FacebookAbstract
Nanomaterials exhibit interesting physical properties distinct from both the molecular and broad scales, presenting new opportunities for physico-chemical as well as biomedical researches and applications in various areas of chemistry, biology and medicine. The unique chemical, physical and mechanical properties of carbon nanotubes have stimulated extensive investigation since their discovery in early 1990s by Iijima. Although there have been tremendous advances in the fabrication of CNTs, the integration of these nanostructures into successful applications and large-scale production processes are yet not very smooth. The present research interest focuses on the development of simple and effective way for the synthesis of carbon nanotubes and proposes the new idea of the organic bulk method. Aliphatic-aromatic compound mixture was used as carbon sources. The carbon nanotubes thus produced were very uniform in size and shape.
Keywords
Carbon Nanotubes, CNTs, MWCNTs, CNT Synthesis, Organic Bulk Method
Carbon Nanotubes articles Carbon Nanotubes Research articles Carbon Nanotubes review articles Carbon Nanotubes PubMed articles Carbon Nanotubes PubMed Central articles Carbon Nanotubes 2023 articles Carbon Nanotubes 2024 articles Carbon Nanotubes Scopus articles Carbon Nanotubes impact factor journals Carbon Nanotubes Scopus journals Carbon Nanotubes PubMed journals Carbon Nanotubes medical journals Carbon Nanotubes free journals Carbon Nanotubes best journals Carbon Nanotubes top journals Carbon Nanotubes free medical journals Carbon Nanotubes famous journals Carbon Nanotubes Google Scholar indexed journals CNTs articles CNTs Research articles CNTs review articles CNTs PubMed articles CNTs PubMed Central articles CNTs 2023 articles CNTs 2024 articles CNTs Scopus articles CNTs impact factor journals CNTs Scopus journals CNTs PubMed journals CNTs medical journals CNTs free journals CNTs best journals CNTs top journals CNTs free medical journals CNTs famous journals CNTs Google Scholar indexed journals MWCNTs articles MWCNTs Research articles MWCNTs review articles MWCNTs PubMed articles MWCNTs PubMed Central articles MWCNTs 2023 articles MWCNTs 2024 articles MWCNTs Scopus articles MWCNTs impact factor journals MWCNTs Scopus journals MWCNTs PubMed journals MWCNTs medical journals MWCNTs free journals MWCNTs best journals MWCNTs top journals MWCNTs free medical journals MWCNTs famous journals MWCNTs Google Scholar indexed journals CNT Synthesis articles CNT Synthesis Research articles CNT Synthesis review articles CNT Synthesis PubMed articles CNT Synthesis PubMed Central articles CNT Synthesis 2023 articles CNT Synthesis 2024 articles CNT Synthesis Scopus articles CNT Synthesis impact factor journals CNT Synthesis Scopus journals CNT Synthesis PubMed journals CNT Synthesis medical journals CNT Synthesis free journals CNT Synthesis best journals CNT Synthesis top journals CNT Synthesis free medical journals CNT Synthesis famous journals CNT Synthesis Google Scholar indexed journals Organic Bulk Method articles Organic Bulk Method Research articles Organic Bulk Method review articles Organic Bulk Method PubMed articles Organic Bulk Method PubMed Central articles Organic Bulk Method 2023 articles Organic Bulk Method 2024 articles Organic Bulk Method Scopus articles Organic Bulk Method impact factor journals Organic Bulk Method Scopus journals Organic Bulk Method PubMed journals Organic Bulk Method medical journals Organic Bulk Method free journals Organic Bulk Method best journals Organic Bulk Method top journals Organic Bulk Method free medical journals Organic Bulk Method famous journals Organic Bulk Method Google Scholar indexed journals Nanoscience articles Nanoscience Research articles Nanoscience review articles Nanoscience PubMed articles Nanoscience PubMed Central articles Nanoscience 2023 articles Nanoscience 2024 articles Nanoscience Scopus articles Nanoscience impact factor journals Nanoscience Scopus journals Nanoscience PubMed journals Nanoscience medical journals Nanoscience free journals Nanoscience best journals Nanoscience top journals Nanoscience free medical journals Nanoscience famous journals Nanoscience Google Scholar indexed journals Nanotechnology articles Nanotechnology Research articles Nanotechnology review articles Nanotechnology PubMed articles Nanotechnology PubMed Central articles Nanotechnology 2023 articles Nanotechnology 2024 articles Nanotechnology Scopus articles Nanotechnology impact factor journals Nanotechnology Scopus journals Nanotechnology PubMed journals Nanotechnology medical journals Nanotechnology free journals Nanotechnology best journals Nanotechnology top journals Nanotechnology free medical journals Nanotechnology famous journals Nanotechnology Google Scholar indexed journals bio-chemical articles bio-chemical Research articles bio-chemical review articles bio-chemical PubMed articles bio-chemical PubMed Central articles bio-chemical 2023 articles bio-chemical 2024 articles bio-chemical Scopus articles bio-chemical impact factor journals bio-chemical Scopus journals bio-chemical PubMed journals bio-chemical medical journals bio-chemical free journals bio-chemical best journals bio-chemical top journals bio-chemical free medical journals bio-chemical famous journals bio-chemical Google Scholar indexed journals materials articles materials Research articles materials review articles materials PubMed articles materials PubMed Central articles materials 2023 articles materials 2024 articles materials Scopus articles materials impact factor journals materials Scopus journals materials PubMed journals materials medical journals materials free journals materials best journals materials top journals materials free medical journals materials famous journals materials Google Scholar indexed journals chemicals articles chemicals Research articles chemicals review articles chemicals PubMed articles chemicals PubMed Central articles chemicals 2023 articles chemicals 2024 articles chemicals Scopus articles chemicals impact factor journals chemicals Scopus journals chemicals PubMed journals chemicals medical journals chemicals free journals chemicals best journals chemicals top journals chemicals free medical journals chemicals famous journals chemicals Google Scholar indexed journals
Article Details
1. Introduction
Nanotechnology and Nanoscience are about controlling and understanding matters on submicrometer and atomic scale. By definition they are exciting multidisciplinary fields which involve the design and engineering of objects or tools, characterization, production, and application of structures, devices, and systems by controlled manipulation of size and shape at nanometer (shortly nm) scale. Nanomaterials have sizes ranging from about 1nm up to several hundred nanometers, comparably to many biological macromolecules such as enzymes, antibodies, and DNA plasmids [1]. Materials in this size range exhibit interesting physical properties, distinct from both the molecular and bulk scales presenting new opportunities for biomedical research and applications in various fields including biology and medicine. Among the large variety of nanostructures in the fullerene family, recently one particular member has become the focus of a great deal of scientific and technological attention: the carbon nanotube (shortly CNT). The basic structural unit of CNT is a graphitic sheet (Figure 1) rolled into a cylindrical shape. In practice, we can roughly divide CNTs into two different classes, either by considering their structure or synthesis method, these are single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs) (Figure 1). The first class includes cylinders formed by a single graphitic layer where the typical diameter is 1-1.5nm [2]. Sometimes the diameter reaches to 3-4nm [3]. The second class of tube includes structures formed by the coaxial arrangement of several (2-50) SWCNTs; their external diameter is of the order of 2-100nm [4].
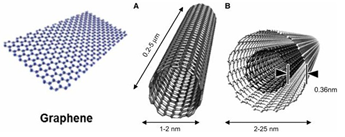
Figure 1: Graphene sheet (left) rolled into cylindrical structure for both SWCNT (A) & MWCNT (B) (schematic diagram).
Since their discovery of CNTs by Iijima [5, 6] there has been extensively studied by researchers in various fields such as chemical, physical, materials and bio-chemical sciences. CNTs have unique nanostructures with remarkably mechanical, thermal and electrical properties, which made them highly attractive for the use as reinforcement in nanotube based composite materials [7, 8]. Another fascinating aspect of CNTs is their cavities, which can be used to incorporate atoms or molecules in order to generate novel compounds or nanostructured materials [9]. Within the family of nanomaterials, CNTs have emerged as new alternative
and efficient tools for transporting and translocating therapeutic molecules [10, 11]. CNT can be functionalized with bioactive peptides, proteins, nucleic acids and drugs, and used to deliver their cargos to cells and organs. Because functionalized CNTs display low toxicity and are not immunogenic [12, 13], such systems hold great potential in the field of bio-nanotechnology and nanomedicine. The ultimate goal of this research work is to develop the preparation technique to synthesize the quality product of CNTs (MWCNTs).
2. Materials and Methods
The chemicals employed in this research work were of analytical graded and were obtained from Sigma Aldrich, England with minimum purity of 99.5%. Water used was doubly distilled throughout the experiment. The aliphatic-aromatic compound mixture was prepared by using an analytical balance with a precision of ± o.1µg. Special care was taken to prevent evaporation and the introduction of moisture into the experimental samples. First, a solution of 40ml of equal aliphatic compound mixture (hexanol + octanol) and 60ml of equal aromatic compound, mixture (benzene + m-xylene) was prepared. 7g (about 15% by weight of carbon sources) of benzalkonium chloride (BZK) and 1g of FeCl3 was then added and the mixture was stirred for 12 hours. BZK is a cationic surfactant which played a role in stabilizing nanoparticles to be formed. 0.5g of hydrazene hydrate was then added as reducing agent and the mixture was stirred again for 24 hours to get densed. In the mean time iron nanoparticles were formed by reduction of the FeCl3 present in this combined densed solution. Then forced pyrolysis was carried out by introducing the obtained solution into a tube furnace with an inert atmosphere by argon gas at 600oC for 20 minutes. Synthesized CNTs thus obtained were then purified and characterized by Energy Dispersive X-Ray spectroscopy (EDX), Scanning Electron Microscopy (SEM), X-Ray Diffraction (XRD), Fourier Transform Infra-red (FTIR) spectroscopic analysis. The tube diameter and morphology of the synthesized CNTs were analyzed by SEM characterization (Figure 2, 3). EDX spectroscopy provided the information about the chemical composition (Figure 4) of synthesized CNTs. XRD (Figure 5) and FTIR (Figure 6) spectra confirms CNT’s growth identity.
3. Results and Discussion
The scanning electron microscope (SEM) uses a focused beam of high-energy electrons to generate a variety of signals at the surface of solid specimens. The signals that derive from electron-sample interactions reveal information about the sample including external morphology (texture) to confirm the size and shape of nanoparticles. The SEM images were taken with an average magnification up to ×100000 at room temperature. The study of the SEM images shows the clear size & shape of CNTs. The SEM images for CNTs are shown in Figure (2, 3) which show dense and clear nanotubes. Distinguishable CNTs are visible at high resolution and the cross section confirms CNTs specific diameter. Obviously, the quality is much better [14, 15]. The diameter of the nanotubes are about 70-95nm on average (Figure 3). The elemental analysis was successfully confirmed by EDX measurement. It’s characterization capabilities are due in large part to the fundamental principle that each element has a unique atomic structure allowing a unique set of peaks in it’s X-ray emission spectrum. From Figure 4 it can be observed that there is a clear abundance of carbon element which sharply supports that CNTs contain only carbons. The abundance was simply detected by the k (alpha) shell electrons at 0.277keV. The carbon percentage by mass and percentage by atomic abundance is 99.34%. Very little amount (0.66%) of unwanted oxygen may come from alcohols. X-ray crystallographer is a tool used for identifying the atomic and molecular structure of a crystal, in which the crystalline atoms cause a beam of incident X-rays to diffract into many specific directions. In Figure 5 X-ray diffraction spectral images and corresponding data were provided for the synthesized CNTs. The height, FWHM (full width at half maxima), d-spacing value and relative intensity at 2θ0 of CNTs give result of high accuracy. The X-ray diffraction image displays the clear sharp peak at 25.72 position which clearly confirms that respective compound is CNT. This higher density of electrons at this position is almost identical for every CNTs (MWCNTs).[16] Fourier Transform Infrared Spectroscopy, also known as FTIR analysis or FTIR Spectroscopy, is an analytical technique used to identify organic, polymeric, and in some cases, inorganic materials. The resulting spectra produce a unique molecular “fingerprint” which can be used to easily screen and scan samples for many different components. FTIR spectra (Figure 6) of synthesized CNTs shows a lot of significant peaks in between 1400- 1650cm-1 which indicates the C-C stretching present in CNTs in ring form (in CNT six carbons ring). Peak in 2368.4cm-1 which might be O=C=O stretching for linear molecules CO2 due to unwanted oxidation of CNTs. Small peaks in 690-760cm-1 are for C-Br band stretching. Unwanted element Br came from KBr used for the FTIR experiments. Peaks present in 2850- 2921cm-1 for O-H bending may come from unreacted alcohols used as carbon source. The above information confirms that growth percentage of CNTs was very good.

Figure 2: SEM image to observe the uniform tube shape of synthesized CNTs.

Figure 3: SEM image also confirms tube diameter as 70-95nm range.

Figure 4: EDX data to observe the elemental confirmation of synthesized CNTs.
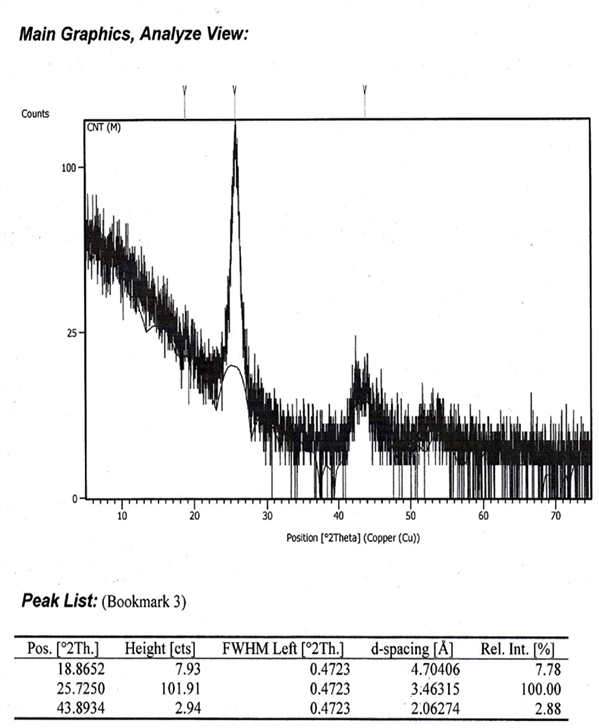
Figure 5: X-ray diffraction spectra and resultant data to confirm growth identity of synthesized CNTs.
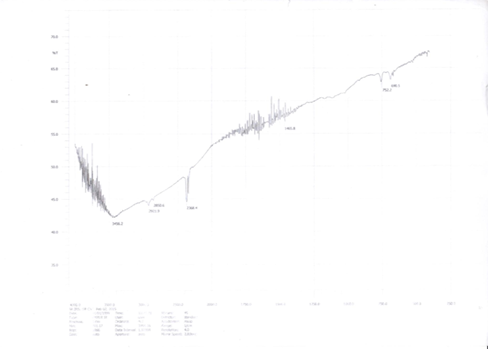
Figure 6: FTIR spectra of synthesized CNTs to observe significant bands.
The above characterizations confirm the significant growth of CNTs with highly uniformed cylindrical structure. The method is capable in monitoring growth directions on the substrate mixtures and synthesizing a large quantity of CNTs. This advanced, integrated and authentic process can resolve the complexity of the uniformed growth of CNTs in large scale.
5. Conclusion
CNTs are low-dimensional Sp2 carbon nanomaterials, exhibit many unique physical and chemical properties that are interesting in a wide range of areas including nanomedicine. Large scale production processes of CNTs depend on the understanding of several fundamental issues, which are yet to be addressed. The carbon arc discharge method is a technique that produces a complex mixture of components and requires further purification to separate the CNTs from the soot and the residual catalytic metals present in the crude product which is very much complicated.[17, 18] Producing CNTs in high yield depends on the uniformity of the plasma arc and the temperature of the deposit forming on the carbon electrode that is difficult to maintain. Another formulation technique of CNTs is laser vaporization method.[19, 20] Arc discharge and laser vaporization methods involve evaporating the carbon source, so it has been unclear how to scale up production to the industrial level using these approaches. The role played in the chemical vapor deposition (CVD) of CNTs is not yet fully understood.[21] The simplistic view that only plays a catalytically passive role in the formation of CNTs requires examination. In this research work, selective growth of CNTs (MWCNTs) by organic bulk method using aliphatic- aromatic compound mixture has been studied. Evidences of self-assembled carbon nanostructures are presented, In addition, CNTs growth was assumed to be possible from other carbon sources like free long chain hydrocarbons. The integration of CNTs in this manufacturing may solve the growth uniformities too.
References
- Zhao K, Peng R, Li D. Separation of nanoparticles by a nano-orifice based DC dielectrophoresis method in a pressure-driven flow. Nanoscale 8 (2016): 18945-18955.
- Yu F, Yang M, Li F, et al. The growth mechanism of single walled carbon nanotubes with a controlled diameter. J Physica 44 (2012): 2032-2040.
- Chen G. Diameter control of single-walled carbon nanotube forests from 1.3-3.0 nm by arc plasma deposition. J S reports 4 (2014): 3804.
- US Research nanometarials, inc (2016).
- Ajayan P M, Iijima S. Capillarity-induced filling of carbon nanotubes. J Nature 361 (1993): 333.
- Ajayan P M, Ebbesen T W, Ichihashi T S, et al. Opening carbon nanotubes with oxygen and implications of filling. J Nature 362 (1993): 522.
- Anantram P M, Yang L, Han J, et al. Subhash S Electro-mechanical properties of Carbon Nanotubes. Ryerson University, Toronto, Canada, ResearchGate (1998).
- Wang X, Jiang Q, Xu W, et al. Effect of carbon nanotube length on thermal, electrical and mechanical properties of CNT/bismaleimide composites. Carbon 53 (2013): 145-152.
- Svrcek V. Functionalization of carbon nanotubes with luminescent silicon nanocrystals upon nanosecond laser processing in liquid media. Intech 305 (2010): 8568.
- Liu Z, Robinson J T, Tabakman S M, et al. Carbon materials for drug delivery & cancer therapy. J Mater Tod 14 (2011): 316-323.
- Jawahar N, Surendra E, Krishna K R. A Review on Carbon Nanotubes: A Novel drug Carrier for Targeting to Cancer Cells. J Pharm Sci & Res 7 (2015): 141-154.
- Yang S T, Wang X. Long term accumulation and low toxicity of single walled carbon nanotubes in intravenously exposed mice. J Toxicol Lett 181 (2008): 182-189.
- Gottardi R, Douradinha B. Carbon nanotubes as a novel tool for vaccination against infectious diseases and cancer. J Nanobiotechnol (2013).
- Carbon nanotube Wikipedia (2016).
- Hoyos-Palacio L M. Catalytic effect of Fe, Ni, Co and Mo on the CNTs production. IOP Conf Ser, Mater Sci Eng (2014).
- Nie P, Min C, Song H, et al. Preparation and tribiological properties of polyimide/carboxyl-functionalized multiwalled carbon nanotube nanocomposite films under sea water lubrication. ResearchGate 58 (2015): 7.
- Keidar M, Shashurin A, Volotskova O, et al. Mechanism of carbon nanostructure synthesis in arc plasma. J Physics of Plasmas 17 (2010): 057101/1-057101/9.
- Volotskova O, Fagan J A, Huh J Y, et al. Tailored distribution of single-wall carbon nanotubes from arc plasma synthesis using m agnetic fields. J ACS Nano 4 (2010): 5187-5192.
- Marchiori R, Braga W F, Mantelli M B H, et al. Analytical solution to predict laser ablation rate in a graphitic target. J Mater Sci 45 (2010): 1495-1502.
- Stuerzl N, Lebedkin S, Malik S, et al Preparation of 13C single-walled carbon nanotubes by pulsed laser vaporization. J Basic Solid State Phys 246 (2009): 2465-2468.
- Kumar M, Zhao X, Ando Y, et al. Carbon nanotubes from camphor by catalytic CVD. J Molec Cryst and Liq Cryst 387 (2002): 117-121.

 Impact Factor: * 2.9
Impact Factor: * 2.9 Acceptance Rate: 78.36%
Acceptance Rate: 78.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 