Congenital Hypertrophy of the Right Ventricle Successfully Treated with Very Low Dose Beta Blockers
Michael Emeis, MD1, Fatima I Lunze, MD2,3,4, Oliver Miera, MD2, Felix Berger, MD, PhD2,3,4, Rainer Rossi, MD, PhD1, Boris Schmitt, MD, PhD2,3,4,5 *
1Department of Pediatrics, Vivantes-Klinikum Neukölln, Berlin, Germany
2Department of Congenital Heart Defects and Pediatric Cardiology, German Heart Center Berlin, Germany
3Department of Pediatrics, Division of Cardiology, Charité University Medicine Berlin, Germany
4DZHK (German Centre for Cardiovascular Research), partner site Berlin, Germany
5BCRT-Berlin-Brandenburg Center for Regenerative Therapies Berlin, Germany
*Corresponding Author: Dr. Boris Schmitt, Department of Congenital Heart Defects and Pediatric Cardiology German Heart Center Berlin, Germany, Augustenburger Platz 1, 13353 Berlin, Germany
Received: 28 August 2020; Accepted: 07 September 2020; Published: 15 December 2020
Article Information
Citation: Michael Emeis, Fatima I Lunze, Oliver Miera, Felix Berger, Rainer Rossi, Boris Schmitt. Congenital Hypertrophy of the Right Ventricle successfully Treated with Very Low Dose Beta Blockers. Cardiology and Cardiovascular Medicine 4 (2020): 760-765.
View / Download Pdf Share at FacebookAbstract
A late preterm baby girl presented with feeding problems and desaturation. Septic work-up was negative, however. Severe right ventricular cardiac hypertrophy is revealed by echocardiography. Based on recent scientific findings on cardiodynamics and the inner antagonism in myocardial architecture, the baby is treated with very low-dose beta blockers instead of higher standard dosage. The therapy leads to immediate improvement in right ventricular function and reduction of right ventricular hypertrophy. Oxygen supply could be stopped, normal feeding was possible. Here we report the first case of very low-dose beta blocker therapy in a neonate for resolution of myocardial inner antagonism.
Keywords
<p>Right ventricular hypertrophy; Metoprolol</p>
Article Details
1. Case
1.1 Patient
A late preterm female baby born at 36+3 weeks of gestation (birth weight 2750 g) and good postnatal adaptation (APGAR 9/10/10) presented on day four with feeding problems. Laboratory values disclosed a slight elevation of inflammatory markers (leukocytes 14.1/nl, CRP 15.9 mg/l), the trancutaneous oxygen saturation did not exceed 92%, clinically there were no signs of respiratory distress.
The admitting clinic started an antibiotic therapy and transferred the baby to our level I perinatal center for further evaluation.
On admission we saw a 5 day-old female baby in stable conditions without signs of infant respiratory distress syndrome (IRDS), no heart murmur or abdominal clinic.The weight was 2450g. Feeding was possible by gastric tube only. The oxygen saturation level of 93% was maintained by the administration of oxygen, 30% with a flow of 4 liters/min.
1.2 Lab results
Blood count: Leucocytes 17.1/nl, Hemaglobin 17.8 g/dl, Hematocrit 0.48 l/l, Thrombocytes 464/nl, C-reactive protein 13.7 mg/l. Cerebrospinal fluid (CSF): Leucocytes 4/µl, protein 521.7 mg/l, Lactate 14 mg/del, Glucose 55 mg/dl. No detection of bacteria or viruses in the CSF nor in the blood samples. Blood gas analyses were normal for pH, pCO2, pO2, HCO3, base excess, electrolytes, lactate, glucose.)
1.3 Echocardiography
Regular anatomy with concordance of atria, ventricles and vessels. Enlarged right atrium with bulging of the interatrial septum (IAS) the left. Right to left shunting via the interatrial septum. Normal size of left atrium with regular pulmonary venous return.
Immense hypertropy of the anterior wall of the right ventricle (+8.2 z), hardly any cavity of the right ventricle detectable. Obvious diastolic restriction of the right ventricle due to hypertrophia. Tricuspid valve (TV) competent, narrow right ventricular outflow tract (RVOT) with endsystolicaly touching walls. Narrow pulmonary valve (PV; diameter 7 mm) and pulmonary artery (PA) with no signs of restriction. Right and left pulmonary artery (RPA and LPA) appeared small but with no obvious restriction. No persistent Ductus arteriosus (PDA).
Mitral Valve (MV), left ventricle (LV), left ventricular posterior wall (LVPW), outflow tract (LVOT) and aortic valve (AV) were aorta, aortic arch and isthmus normal.
§- decline from baseline to discharge demonstrated with ´´-``; RV = right ventricle, LV = left ventricle, TAPSE – tricuspid Annular Plane Systolic Excursion, VTI – Velocity Time Integral.
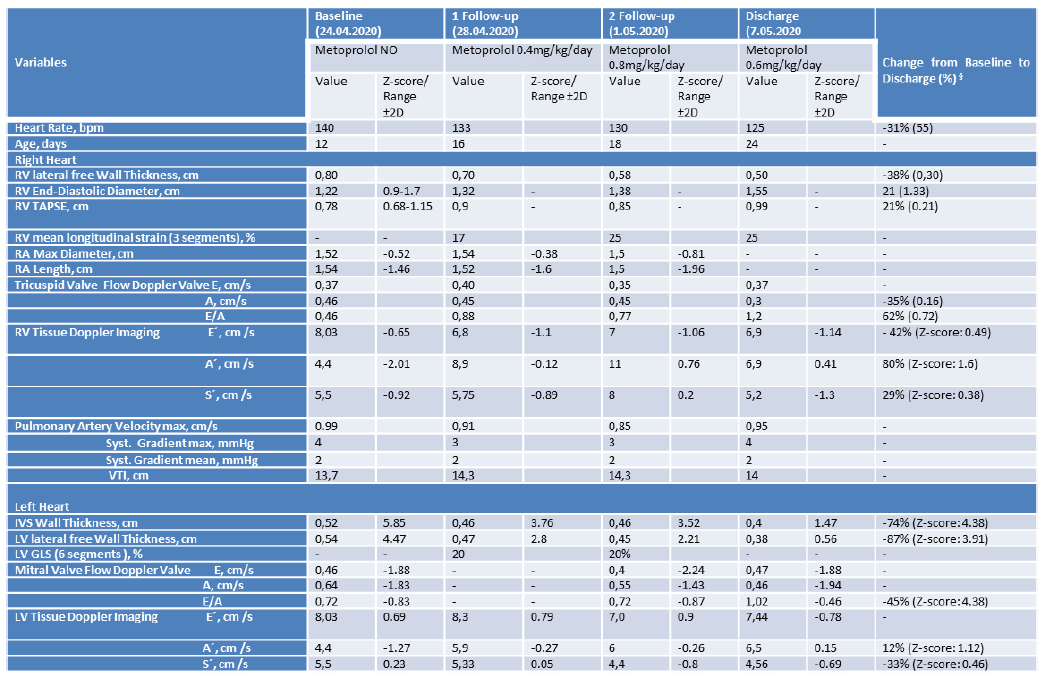
Figure 1: Serial Echocardiogram of parasternal short axis with right ventricular (RV) free wall thickness and end-diastolic diameter at baseline prior Metoprolol administration (A) and shortly after low dose Metoprolol administration at day 4 and 12 of follow-up (B and C, respectively): A – Baseline, 12th day of life; B – Metoprolol 0.4 mg/kg/day, 16th day of life; C – Metoprolol 0.6 mg/kg/day, 24th day of life.
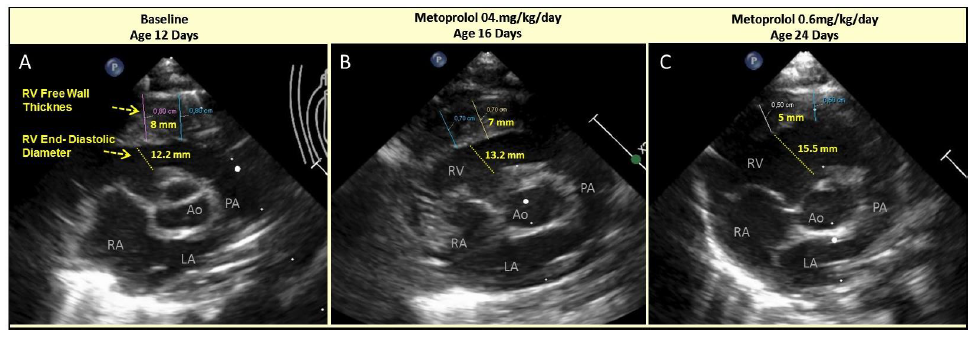
Figure 2: Serial Echocardiogram of apical 4 chamber view with right ventricular (RV) free wall, interventricular septum and left ventricular (LV) free wall thicknesses at baseline prior Metoprolol administration (A) and shortly after low dose Metoprolol administration at day 4 and 12 of follow-up (B and C, respectively): A – Baseline, 12th day of life; B – Metoprolol 0.4 mg/kg/day, 16th day of life; C – Metoprolol 0.8 mg/kg/day, 18th day of life.
1.4 First line treatment
Antibiotics (Ampicillin and Gentamycin) were administered intravenously, feeding happened via gastric tube. Supplemental oxygen was given to improve oxygen saturation to >95%.
1.5 Cardiac treatment
The Department of Pediatric Cardiology, German Heart Center Berlin (DHZB) recommended beta blocker medication at standard dosis in order to lower the heart frequency and to buy time for the decision if a biopsy of the myocardium would be messesary. Over the next days the situation clinically improved slightly without specific medication except of oxygen application. Oxygen requirement decreased and the ability to drink improved somewhat. However, echo findings remained unchanged with severe RV hypertrophy.
So beta-blocker medication was started. However, the dosage of beta-blockers were significantly reduced when compared to clinical standards. Based on the findings of Schmitt et al. Only 20% of the recommended dose of Metoprolol were given every 6 hours (0.1 mg/kg 4 times per day).
The resulting plasma level after 4 days was <2 mcg/l. We increased the dose to 0.2 mg/kg every 6 hours, still with a resulting plasma level of <2 mcg/l. Based on echocardiographic findings an improvement in right ventricular function together with a decrease in RV hypertrophy was noticed. Within a few days the patient was discharged with a reduced dose of Metoprolol of 0.2 mg/kg every 8 hours.
1.6 Clinical course
After starting the medication of very low-dose Metoprolol, we documented a rapid improvement of physical activity within days. Oxygen and respiratory support were not needed any longer and a weight gain was registered (+260 g). After 6 days of treatment discharge was possible. All laboratory values were normal. After one week at home the newborn was re-examined by echocardiography. An impressive improvement of the echo findings went along with a physically absolute normal appearance of the baby.
2. Discussion
2.1 Conversion from Esmolol to Metoprolol
In the publication of Schmitt et al. [1] the doses with major influence on improving myocardial function range from 5 to 25 mcg/kg/min Esmolol. In contrast, the regular dose of Esmolol for the treatment of adults and children comprises a loading dose of 500 mcg/kg followed by 25 to 300 mcg/kg/min [2].
For Metoprolol the regular loading dose is 100 mcg/kg followed by 1-5 mcg/kg/min, or oral application of 0.5-1-2 mg/kg every 6-12 hours [2]. In the publication of Schmitt et al. The lowest given dose per minute of esmolol was only 20% (i.e. (5 mcg/kg/min) of the regularly recommended dose after loading [1].
2.2 Antagonistic organizations of myocardial structure and function
The heart muscle´s motion pattern has essentially been due to a matched interaction of a pair of intramural antagonistic forces. The heart muscle´s three-dimensional architecture generates both constrictive and dilating forces which are simultaneously active during systolic ventricular depletion [3-5].
The prevailing tangential fiber aggregates exclusively drive ventricular depletion. The obliquely intruding and extruding fibers hinder systolic wall thickening [6]. This antagonistic principle helps in stabilizing ventricular shape and protecting the heart muscle and the capillary network from “over-contraction”. It gives the first kick to early diastolic ventricular filling. Being part of the intrinsic afterload, it contributes to myocardial stiffness. In addition, it confines myocardial microcirculation by increasing intramural pressure [7].
By use of needle force probes it has been shown that the heart muscle´s tangential netting drives ventricular constriction. In contrast, an auxotonic force is generated by the intruding and extruding components (endo-epicardially netting). They sustain ventricular dilation. The most outstanding features of the auxotonically contracting oblique myocardial netting are its two times higher sensitivity to “low dose catecholamines” [8] than that in the tangential netting, resulting in its particularly high sensitivity to low dose ß-blockade. With rising doses of positive and negative inotropics, this typical asymmetrical sensitivity in both netting components disappears.
2.3 Potential ways to Antagonistic mismatch
The concept of intrinsic antagonism suggests various potential mechanisms to be involved in early postnatal right ventricular hypertrophy. If clinical investigations do not show any hemodynamic resistance to right ventricular outflow, we must assume, one of the intrinsic afterloads to be responsible. Hypertrophy becomes a self-perpetuating process into both netting components which continues until contractility at least in one of both components is turned down. The appropriate action is to confine auxotonic forces in the oblique transversal netting by low dose ß-blockade. Once the blockade of transmural netting has started, intrinsic after load of tangential netting progressively vanishes hence progress in hypertrophy finds an end.
Having taken into account this theoretical and experimental knowledge we started a clinically sub-therapeutic treatment with beta-blockade. Within a view days the thickness of the myocardium changed substantially without altering systolic function. The functional echo data and the clinical course improved accordingly.
In this example, we transferred theoretical assumptions and experimental findings to practical use with obvious benefit to the neonatal patient.
Disclosures
None.
References
- Schmitt B, Li T, Kutty S, Khasheei A, Schmitt KRL, et al. Effects of incremental beta-blocker dosing on myocardial mechanics of the human left ventricle: MRI 3D-tagging insight into pharmacodynamics supports theory of inner antagonism. Am J Physiol - Hear Circ Physiol 309 (2015).
- Shann F. Drug Doses. 17th ed. Melbourne (2017).
- Lunkenheimer PP, Redmann K, Kling N, Jiang X, Rothaus K, et al. Three- dimensional architecture of the left ventricular myocardium. Anat Rec A Discov Mol Cell Evol Biol 288 (2006): 565-578.
- Schmitt B, Fedarava K, Falkenberg J, Rothaus K, Bodhey NK, et Three-dimensional alignment of the aggregated myocytes in the normal and hypertrophic murine heart. J Appl Physiol 107 (2009).
- Anderson RH, Sanchez-Quintana D, Redmann K, Lunkenheimer PP. How are the myocytes aggregated so as to make up the ventricular mass? Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu (2007): 76-86.
- Lunkenheimer PP, Redmann K, Florek J, Fassnacht U, Cryer CW, et The forces generated within the musculature of the left ventricular wall. Heart 90 (2004): 200-207.
- Pp L, Redmann K, Hoffmeister A, Niederer P, Stephenson R, et al. The Ventricular Structure Functioning as an Antagonistic Continuum The Ventricular Structure Functioning as an Antagonistic Continuum (2017).
- Lunkenheimer PP, Redmann K, Cryer CW, Batista RV, Stanton JJ, et Beta-blockade at low doses restoring the physiological balance in myocytic antagonism. Eur J cardio-thoracic Surg 32 (2007): 225-230.


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 