Effective Tumor Control Following Re-Irradiation Combined with Superficial Hyperthermia of Inoperable, Locally Recurrent Breast Cancers
Markus Notter1,2*, Andreas R. Thomsen3,4, Peter Vaupel3,4
1Radiation Oncology Lindenhofspital Bern, Bern, Switzerland
2Member of the Swiss Hyperthermia Network
3Department of Radiation Oncology, University Medical Center, University of Freiburg, Freiburg, Germany
4German Cancer Consortium (DKTK), Partner Site Freiburg and German Cancer Research Center (DKFZ), Heidelberg, Germany
*Corresponding author: Markus Notter, Radiation Oncology Lindenhofspital Bern, Bern, Switzerland.
Received: 17 January 2022; Accepted: 25 January 2022; Published: 29 January 2022
Article Information
Citation:
Notter Markus, Thomsen AR, Vaupel P. Effective Tumor Control Following Re-Irradiation Combined with Superficial Hyperthermia of Inoperable, Locally Recurrent Breast Cancers. Journal of Women’s Health and Development 5 (2022): 050-056
DOI: 10.26502/fjwhd.2644-28840076
View / Download Pdf Share at FacebookAbstract
Inoperable, locally Recurrent Breast Cancer (LRBC) in previously irradiated regions is a challenging disease to manage. Superficial Hyperthermia (sHT) in combination with Re-Irradiation (re-RT) offers an effective local tumor control with a total re-RT dose of 20 Gy. The low toxicity enables the application of repeated re-irradiations, especially in recurrent lymphangiosis carcinomatosa. This technology can also be applied with other superficial tumor types, e.g., in primary or radiation-associated angiosarcoma or skin metastasis.
Keywords
<p>Superficial Hyperthermia; Re-Irradiation; Hyperthermia</p>
Article Details
Summary
Inoperable, locally Recurrent Breast Cancer (LRBC) in previously irradiated regions is a challenging disease to manage. Superficial Hyperthermia (sHT) in combination with Re-Irradiation (re-RT) offers an effective local tumor control with a total re-RT dose of 20 Gy. The low toxicity enables the application of repeated re-irradiations, especially in recurrent lymphangiosis carcinomatosa. This technology can also be applied with other superficial tumor types, e.g., in primary or radiation-associated angiosarcoma or skin metastasis.
1. Background
Breast cancer patients presenting with inoperable, locally recurrent tumors suffer from extreme psychological and physiological stress. This stress is exacerbated by the appearance of the disease as well as by any symptoms associated with the extension of the disease such as pain, ulceration, bleeding, constriction and shortness of breath (cancer en cuirasse), lymph edema and unpleasant odor. The need for therapeutic intervention for these patients is high, but therapeutic options for recurrences in previously irradiated regions are very limited. An additional re-irradiation with effective dosage is often contra-indicated and is related to a high risk of severe side effects. Frequently, these heavily pretreated regions are resistant to systemic therapies, and the potential benefit of these therapies must then be weighed against possible side effects. Oftentimes, the goal of effective local tumor control is then abandoned.
1.1 Hyperthermia and radiation therapy
The combination of Radiotherapy (RT) with sHT provides an evidence based, effective therapeutic option for these patients. Mild Hyperthermia (HT) with tissue temperatures between 39-43°C is an efficient radio-sensitizer and can enhance antitumor immune responses. This combination provides the opportunity to significantly reduce the radiation doses and consequently, radiation toxicity. This effect is mainly achieved by (a) increased tumor perfusion, (b) improved tissue oxygenation, (c) inhibition of DNA-repair and by (d) fostering immunological effects. Therefore, HT is indicated specifically in previously irradiated target volumes, where unfavorable, hypoxic conditions are commonly present. A meta-analysis; published in 2016, demonstrated the efficiency of the combined sHT/re-RT protocol with regards to LRBC [1]. The application of this treatment scheme is no longer considered an “outsider method” and should be utilized in cases with limited therapeutic options. Bearing this in mind, it is important then that physicians and patients know that this treatment is a viable option for LRBC.
1.2 Improved technical methods
A novel technical method using thermography-controlled water-filtered infrared-A superficial hyperthermia (wIRA-sHT) provides a contact-free application of energy to large surfaces with heterogeneous body contours [2]. The heating of the surface is automatically limited to 43°C, and thus the risk of burns and blistering in scars or skin transplants is practically eliminated.
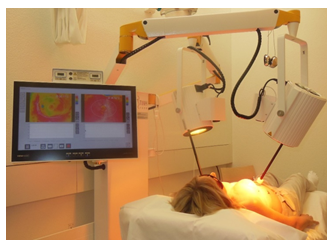
Figure 1: Therapeutic setting of water-filtered infrared-A superficial hyperthermia (hydrosun®TWH1500, Hydrosun Medizintechnik, Muellheim, Germany) in a bilateral chest wall recurrence of a breast cancer patient.
As a well-tolerated treatment protocol we recommend the following procedure: weekly wIRA-sHT for 45 – 60 min, immediately followed by re-RT. Single irradiation dose is 4 Gy, with a total dose of 20 Gy, 1x/week [3]. This treatment allows for an outpatient setting.
2. Results
A retrospective analysis of 201 patients with inoperable, previously irradiated LRBC treated with the aforementioned protocol was published in 2020 [4]. The size of local recurrences was recognized as one of the most important prognostic factors. Therefore, a novel size classification was proposed and the treatment results were related to the tumor extensions. Response rates were as follows: in Class I (largest diameter ≤ 10 cm): CR (complete remission) = 76%, PR (partial remission) = 24%; in Class II (>100 cm2, but limited to the ipsilateral chest wall): CR = 61%, PR = 36%, NC (no change) = 3%; in Class III (extension beyond ipsilateral chest wall): CR = 36%, PR = 61%, PD (progressive disease) = 2%; and in Class IV (extension from both chest wall to the back, classical “cancer en cuirasse”): CR 2%, PR 83%, NC 10%, PD 5%. The overall response rate for the patients in this analysis was 95%, which means that only a small percentage did not report any clinical benefit. In 2/3 of the patients with CR, the local control could be maintained and in more than half of the patients with PR, a local progression free situation could be achieved during life time. However, this is despite above-average previous radiation doses and tumor extensions, in comparison to other studies. In addition, the re-RT dose used was the lowest one ever reported in the literature (see Table 1).
|
Authors & year |
n |
HT technique used |
Total RT-dose (Gy) |
CR |
HT related toxicity ≥ G2 |
|
a. Randomized trials |
|||||
|
Vernon et al. [5] |
56 |
100 – 1000 MHz |
32 |
38 % (RT) 78% (HT-RT) |
23% |
|
Jones et al. [6] |
39 |
434 MHz |
30 – 66 |
24 % (RT) 68% (HT-RT) |
21% |
|
b. Non-randomized trials / retrospective analyses |
|||||
|
Oldenborg et al. [7] |
414 |
434 MHz |
32 (20 – 40) |
58% |
13% |
|
Linthorst et al. [8] |
248 |
434 MHz |
32 |
70% |
23% |
|
Bakker et al. [9] |
262 |
434 MHz |
32 |
n.r. |
26% |
|
Notter et al. [4] |
201 |
wIRA |
20 |
43% (2-76%)* |
0.5% |
Table 1: Results of combined superficial hyperthermia & re-irradiation in locally recurrent breast cancer. n = number of patients treated, HT = hyperthermia, RT = radiotherapy, MHz = (megahertz, microwave technique), wIRA = water-filtered infrared-A, CR = complete remission, n.r. = not reported, * remission according to size classes of macroscopic disease.
2.1. Side effects
The reported side effects of this treatment protocol were very low with 114 (57%) of the 201 patients presenting with no acute side effects at all, 65 (33%) with acute skin reactions grade I, and 4 (2%) with acute skin reactions grade II. Chronic reactions like hyperpigmentation grade I (26%) and new teleangiectasias grade II (3%) were observed in 56 patients. This low toxicity allows physicians to use this treatment scheme multiple times, even several times in the same formerly treated region. This could be advantageous for the management of lymphangiosis carcinomatosa (involvement of lymphatic drainage system of the skin by cancer cells) with the tendency to recur again and again.
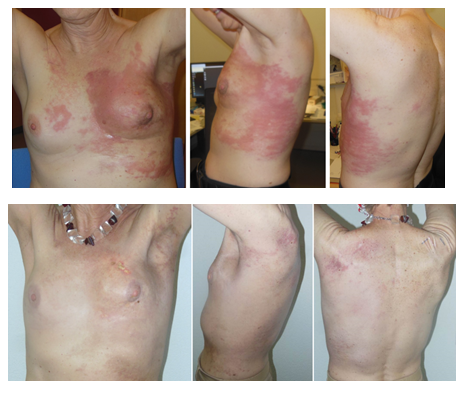
Figure 2: Upper panel: Cancer en cuirasse before combined treatment (January 14, 2020). Lower panel: Result 4 ½ weeks after completion of combined treatment of anterior and left lateral chest wall, back and supraclavicular fossa, each region with 5 x 4 Gy 1x/week and wIRA-sHT (June 24, 2020).
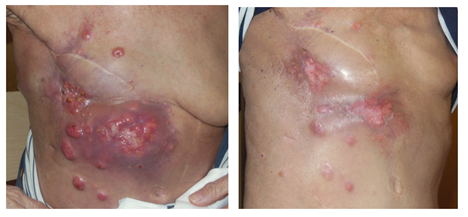
Figure 3: Locally recurrent breast cancer. Left panel: before combined treatment (March 1, 2017). Right panel: 6 weeks after 5 x 4 Gy 1x/week and wIRA-sHT. Distinct regression and relief of pain (July 5, 2017)
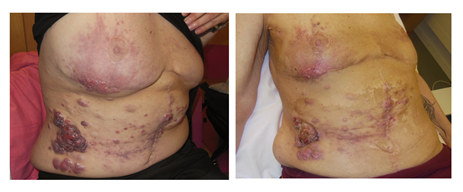
Figure 4: Metastatic lesions of an advanced ovarian cancer within the abdominal wall, skin metastases and extension to the right breast. Resistance to different attempts with chemotherapy. Left panel: before combined treatment (October 22, 2019). Right panel: 4 weeks after 5 x 4 Gy 1x/week and wIRA-sHT: distinct relief of pain, drying of the ulcerating lesions, higher comfort and reduced unpleasant odor. Good palliative effect (January 7, 2020)
Ou3.tlook
Comparative confirmatory phase III trials comparing sHT/re-RT versus best supportive care (without tumor-directed therapy) are not feasible for LRBC due to patient related factors and ethical considerations [10]. Patients with heavily pre-treated recurrences may refuse randomization, may also insist on the immediate start of treatment with the intention of effective local tumor control or distinct relief of symptoms. Comparative studies with sHT/re-RT with reduced doses versus re-RT alone using conventional
doses would either exclude or compromise those patients who are at risk of unacceptable cumulative toxicity. Moreover, the highly individual differences in lesions sizes, type and number of pre-treatments, resistances to other therapies, comorbidities etc. impede randomization into comparable groups. It is stated, that sHT immediately followed by low dose re-RT is an acceptable treatment protocol for therapeutic consideration [11]. Patients with microscopic disease after resection of a LRBC with R1 margins (non-radical operation) or with a high risk of local recurrence due to limited resection margins, also profit from the combined sHT-re-RT [4,12]. The presented treatment schedule can also be used to treat other superficial tumor entities, e.g., primary skin cancer (melanoma and non-melanoma tumors), Merkel-cell carcinoma, cutaneous lymphomas, skin metastasis, primary or radiation-associated angiosarcomas etc. The latter of the tumor entities, is a rare disease that has also been proven to have a clinical benefit following a sHT/re-RT treatment scheme [13].
References
- Datta NR, Puric E, Klingbiel D, et a Hyperthermia and radiation therapy in locoregional recurrent breast cancers: a systematic review and meta-analysis. Int J Radiat Oncol Biol Phys 94 (2016): 1073-1087.
- Vaupel P, Piazena H, Müller W, et a Biophysical and photobiological basics of water-filtered infrared-A hyperthermia of superficial tumors. Int J Hyperthermia 35 (2018): 26-36.
- Notter M, Piazena H, Vaupel P. Hypofractionated re-irradiation of large-sized recurrent breast cancer with thermography-controlled, contact-free water-filtered infrared-A hyperthermia. Int J Hyperthermia 33 (2017): 227-236.
- Notter M, Thomsen AR, Nitsche M, et al. Combined wIRA-hyperthermia and hypofractionated re-irradiation in the treatment of locally recurrent breast cancer: Evaluation of therapeutic outcome based in a novel size classification. Cancers 12 (2020): 606.
- Vernon CC, Hand JW, Field SB, et al. Radiotherapy with or without hyperthermia in the treatment of superficial localized breast cancer: results from five randomized controlled trials. International collaborative hyperthermia group. Int J Radiat Oncol Biol Phys 35 (1996): 731-744.
- Jones EL, Oleson JR, Prosnitz LR, et al. Randomized trial of hyperthermia and radiation for superficial tumors. J Clin Oncol 23 (2005): 3079-3085.
- Oldenborg S, Griesdorn V, van Os R, et al. Reirradiation and hyperthermia for irresectable locoregional recurrent breast cancer in previously irradiated area: size matters. Radiother Oncol 117 (2015): 223-228.
- Linthorst M, Baaijens M, Wiggenraad R, et al. Local control rate after the combination of re-irradiation and hyperthermia for irresectable recurrent breast cancer: results in 248 patients. Radiother Oncol 117 (2015): 217-222.
- Bakker A, Kolff WM, Holman R, et al. Thermal skin damage during re-irradiation and hyperthermia is time-temperature dependant. Int J Radiat Oncol Biol Phys 2017, 98, 392-399
- Thomsen AR, Vaupel P, Grosu AL, et al. Hyperthermia plus re-irradiation in the management of unresectable locoregional recurrence of breast cancer in previously irradiated sites. J. Clin. Oncol 38 (2020): 3576-3577.
- Buchholz TA, Ali S, Hunt KK, et al. Reply to Thomsen A. et al. J Clin Oncol 38 (2020): 3577.
- Linthorst M, Geel ANV, Baaijens M, et al. Re-irradiation and hyperthermia after surgery for recurrent breast cancer. Radiother Oncol 109 (2013): 188-193.
- Notter M, Stutz E, Thomsen AR, et al. Radiation-associated angiosarcoma of the breast and chest wall treated with thermography-controlled, contactless wIRA-hyperthermia and hypofractionated re-irradiation. Cancers 13 (2021): 3911.


 Impact Factor: * 3.4
Impact Factor: * 3.4 Acceptance Rate: 78.89%
Acceptance Rate: 78.89%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 