Effectiveness of Water Gradient Technology for Preventing Meibomian Gland Loss with Silicone Hydrogel-Soft Contact Lens Use
Hisataka Fujimoto1, *, Shintaro Ochi2, Yasushi Inoue2, Junichi Kiryu1
1Department of Ophthalmology, Kawasaki Medical School, 577 Matsushima, Kurashiki, 701-0192 Okayama, Japan
2Inoue Eye Clinic, 1-14-31 Uno, Tamano, 706-0011 Okayama, Japan
*Corresponding author: Hisataka Fujimoto, Department of Ophthalmology, Kawasaki Medical School, 577 Matsushima, Kurashiki, 701-0192 Okayama, Japan
Received: 18 October 2021; Accepted: 26 October 2021; Published: 10 November 2021
Article Information
Citation:
Hisataka Fujimoto, Shintaro Ochi, Yasushi Inoue, Junichi Kiryu. Effectiveness of Water Gradient Technology for Preventing Meibomian Gland Loss with Silicone Hydrogel-Soft Contact Lens Use. Journal of Ophthalmology and Research 4 (2021): 301-312.
DOI: 10.26502/fjor.2644-00240049
View / Download Pdf Share at FacebookAbstract
Purpose: Soft contact lenses (SCLs) are effective for refractive error correction, but prolonged wear results in discomfort and discontinuation. This study examined whether the water gradient technology in delefilcon A-based SCLs (which are silicone hydrogel (SH) SCLs) prevents meibomian gland (MG) injury.
Methods: This retrospective observational study included a total of 48 asymptomatic SCL users as follows: 23 delefilcon A users (46 eyes) and 29 narafilcon A users (58 eyes) (comparator conventional SCL group) without ocular comorbidities and previous use of ophthalmic treatment, except for anti-allergic eye drops or artificial tears. Data on the MGs were collected by infrared meibography. Measurements of the bare eye were recorded as follows: at visit 1 (v1) and at visit 2 (v2) after SCLs were worn for ≥5 h; v2 occurred 30 ± 5 days after v1.
Results: The area of the MG in the lower eyelid was significantly reduced at v2 compared to that at v1 in the narafilcon A group (46.6% at v1 and 44.5% at v2; P = 0.015). The area of the lower eyelid MG was not significantly reduced in the delefilcon A group (43.1% at v1 and 43.8% at v2; P > 0.05).
Conclusion: The water gradient technology used in delefilcon A may significantly reduce MG loss induced by SH-SCLs. The use of water gradient technology might alleviate MG injury caused by SHSCL use, prevent pathological tear dynamics, and improve lens performance.
Keywords
<p>Antibiotic resistance; Enterobacteria; Self-medication; Resuscitation</p>
Article Details
1. Introduction
Recent studies in corneal physiology have enabled the development of soft contact lenses (SCLs) with improved component materials, design, and production processes. SCLs are thus currently the safest, most sought after, and effective interventions for refractive error correction [1]. However, continuous and extended SCL use is associated with adverse effects and undue stress on the cornea mainly due to issues related to oxygen permeability [2], corneal neovascularization [3], and tear film abnormality and endothelial disorders [4].
Silicone hydrogel (SH) SCLs were developed two decades ago to overcome the limitations associated with SCLs and conventional hydrogel lenses [5]. SH-SCLs have distinct surface and mechanical properties that allow for increased permeability of oxygen, reduced protein deposition, and mild frictional interactions, though they show increased lipid deposition and less favorable elastic properties (stiffness) [6]. Several investigations have also established that SH-SCLs reduce the level of limbal inflamations [7], alleviate contact lens-associated keratitis [8] and increase the ocular surface oxygen supply compared to conventional hydrogel SCLs [9]. Despite these advantages, symptoms associated with contact lens discomfort (CLD) persist in SH-SCLs [10]. One of the reasons for CLD in SH-SCLs is friction between the SCL surface and the conjunctiva. This surface friction also induces epitheliopathy [11].
The frictional interactions of SCLs are partially influenced and may be mitigated by the lipid components of tears secreted by the meibomian glands (MGs). MG dysfunction results in pathological tear film conditions such as a shorter break-up time, decreased contact lens surface coverage by the tear film during the overall interblink period, and greater exposure of the contact lens surface to the atmosphere during blinking [12].
Dailies Total1 (DT1) (delefilcon A) was developed as a water gradient SH-SCL whose lens surface has distinct properties designed to minimize surface problems associated with SH-SCLs. It has a core silicone hydrogel material comprising 33% water that can transit to the outer surface layer (which has 80% water) [13]. The surface has a low compression modulus and includes water gradient technology that enables the surface to be lubricated and facilitates high oxygen permeability [14]. The present study investigates whether the water gradient structure of the delefilcon A lens (a SH-SCL) contributes to improving the condition of the MGs with reference to tear film quality; the delefilcon A lens is compared to the narafilcon A lens, which is a conventional SH-SCL.
2. Materials and Methods
2.1 Study Design
This retrospective observational study included 48 asymptomatic patients who used SCLs daily. The study was approved by the Institutional Review Board of Kawasaki Medical School Hospital (approval number 3403-01). Informed consent was obtained from all the study participants. The study adhered to the tenets of the Declaration of Helsinki and was performed according to Good Clinical Practice (GCP). The identity of the sponsor of the study (Alcon) was masked to the assessors until the completion of the study. The study observation period was defined as the date from which the patient started receiving care at the study site until the date at which the last data point was measured; the study was conducted between May 1, 2019 and February 29, 2020. On-site training was conducted to ensure uniform data collection, assessment, and compliance with GCP.
2.2 Study participants and visits
A retrospective review of the clinical records (including video records) of 48 SH-SCL users was conducted for potential participants who were able to achieve a clinically acceptable fit with each of the study lenses. Asymptomatic participants who were ≥20 years of age with far-corrected vision of ≥0.8 in both eyes, those wearing contact lenses for at least five days a week for greater than 5 h a day, and regular users of SCL were included from one site (Inoue eye clinic) in Japan.
Contact lens wearers without active ocular surface disorders such as epithelial disorders, infections, conjunctivitis, ocular inflammatory disease, and nasolacrimal duct obstruction were selected. As an exception, those with a dry eye with a short break-up time were included in the study since such symptoms are often evident in contact lens users and hold clinical significance; these symptoms can potentially be targeted effectively with delefilcon a use. Individuals with a history of ocular surface surgery or those considered unsuitable by the principal investigator were excluded from the study. To minimize the intervention in this retrospective study, subjects were enrolled as delefilcon A or narafilcon A habitual users. The 48 SH-SCL users included a consecutive series of 23 delefilcon A habitual users and 25 narafilcon A habitual users who met the above criteria.
2.3 Study groups and data collection
To minimize bias, the eligible patients were enrolled in a blinded manner to one of two study groups: the delefilcon A group which used DT1 lenses (Alcon Laboratories, Inc., Fort Worth, Texas, USA), and the narafilcon A group which used 1-Day Acuvue TruEye (TE) lenses (Johnson & Johnson Vision); the lenses were allotted in a consecutive series. Delefilcon A lenses are SH-SCLs with a water gradient structure and a surface with high lubricity [15]. The core has low water content (33%), similar to other conventional SH-SCLs [16]. We chose narafilcon A as the representative conventional control SH-SCL because its core component also has low water content (46%) [17]. Both are daily disposable SH-SCLs [18]. The water content in narafilcon A is constant throughout the lens, while the water gradient technology in delefilcon A creates a low water content core and a high water content surface [17]. Several previous studies have also compared delefilcon A with narafilcon A [19-22].
We included the following background data: age, sex, power (diopter), Schirmer’s test (mm), central corneal thickness (CCT, µm), and ophthalmic solution use; we also included the following data elements measured on the bare eye: non-invasive tear breakup time (NIBUT) (seconds) using the tear interferometer DR-1α (Kowa, Tokyo, Japan), tear meniscus height (TMH) (mm), subjective dryness estimated by the visual analog scale (VAS; minimum 0, maximum 100), and ocular higher-order aberrations (HOAs).
The MG area within the lower eyelid was measured by infrared meibography using Keratograph 5M (Oculus, Wetzlar, Germany). The area of the MG structure was extracted by automatic threshold identification using ImageJ software, and analyzed as described previously [23, 24].
The data were categorized as visit-1 (v1) and visit-2 (v2) data for each patient based on the chronology of availability. For data based on the qualification tests and bare eye measurements, the time that SCLs were worn to the hospital, removed, and washed was set as ≥30 min. Data were collected for clinical v1 after the tests, and observation parameters were measured without SCL-wearing. For clinical v2, the following data were collected: data from tests and observation parameters measured after wearing SCLs for ≥5 h after 30 ± 5 days of SCL use following v1. Between v1 and v2, SCLs were used 5-12 hours per day, 5-7 days per week. Room temperature and humidity were maintained at 23–25 °C and 30%–40%, respectively.
For the NIBUT, the three videos captured using the tear interferometer DR-1α were analyzed in a masked manner, and the three break-up times were averaged.
TMH and CCT were obtained using anterior segment optical coherence tomography (AS-OCT; CASIA 2, Tomey Corporation, Nagoya, Japan). Analysis software (Tomey Corporation, Nagoya, Japan) was used to identify and digitize the anterior corneal or SCL surfaces, posterior corneal surfaces, and tear meniscus areas. The digitization tasks were performed by researchers who were blinded to the group allocation of the participants. For TMH and CCT, three measurement values were obtained and averaged.
The following ocular HOAs were measured: total HOA, coma, trefoil, spherical, tetrafoil, and 2nd order astigmatism aberrations through a 6 mm pupil using a Shack—Hartmann wavefront sensor in the KR-1W Wavefront Analyzer (TOPCON, Tokyo, Japan). The HOA values (RMS; in μm) measured every 1 to 10 s after each blink were averaged.
All measurements were obtained from bilateral eyes and were recorded.
2.4 Statistical analysis
This study was designed to assess the difference between the delefilcon A and narafilcon A groups in terms of tear film dynamics. The sample size was planned as per the number needed for evaluating the null hypothesis of no difference between the two groups with the specified probability. Data distributions were assessed for normality using the Shapiro-Wilk test. Based on the assumption that all the data on the outcomes of interest followed a parametric distribution, the two-sample independent t-test was used to analyze all data except for data on the following: male/female ratio, ophthalmic solution use ratio. A paired t-test was performed to compare the MG area between v1 and v2. For the male/female, and ophthalmic solution use data, the Pearson’s chi-squared test was used. All statistical analyses were performed using SPSS version 25.0 (IBM SPSS Statistics for Windows, version 25.0; IBM Corp., Armonk, NY USA).
3. Results
3.1 Enrollment
In this study, a total of 48 patient records were reviewed, and 96 eyes of 48 regular SH-SCL users were enrolled into two groups. These were 23 regular users (46 eyes) of delefilcon A-based DT1 and 29 regular users (58 eyes) of narafilcon A-based TE. Among the 29 narafilcon a participants, four participants (eight eyes) also used delefilcon A and were included in the delefilcon A group. For the four participants who used both lenses, the v1 and v2 measurements were recorded for an additional day after 60 days, and were compared to the measurements of individuals who exclusively used either delefilcon A or narafilcon A.
The study of three patients who did not appear for v2 was discontinued after v1. The three patients included two participants from the delefilcon a group and one participant from the narafilcon a group. There were no reports of discontinuation due to adverse events.
3.2 Patient characteristics and area of the lower eyelid MGs
The study comprised a total of nine men and 39 women; the average age was 34.6 ± 9.8 years and 32.5 ± 8.6 years in the delefilcon A and narafilcon A groups, respectively. There were no significant differences between the study groups in the baseline parameters and the studied parameters (bare eye measurements) (Table 1). Apart from artificial tears or anti-allergic eye drops, no other ophthalmic solutions were used. The distributions of the SCL products that the participants used most frequently within one month before v1 were also not significantly different between the groups (P = 0.07, Jonckheere-Terpstra test, Table 2).
The area of the lower eyelid MG (Figure 1) was significantly reduced at v2 compared that at v1 in the narafilcon A group (46.6% at v1 and 44.5% at v2; P = 0.015, paired t-test, Figure 2A). Such a reduction was not observed in the delefilcon A group (43.1% at v1 and 43.8% at v2; P > 0.05, paired t-test, Figure 2B).
Table 1: Comparison of the participant background data between the two silicone hydrogel-soft contact lens (SH-SCL) groups
|
Narafilcon A (in narafilcon A) Average ± SD |
Delefilcon A (in delefilcon A) Average ± SD |
*P value delefilcon A vs. narafilcon A |
|
|
Age (years) |
32.5 ± 8.6 |
34.6 ± 9.8 |
0.25 |
|
Female ratio |
0.83 |
0.70 |
0.28^ |
|
CCT (μm) |
533.7 ± 32.2 |
530.7 ± 31.2 |
0.64 |
|
Power (diopter) |
−3.71 ± 1.47 |
−3.30 ± 1.50 |
0.17 |
|
Schirmer’s test (mm) |
20.7 ± 12.8 |
17.5 ± 10.9 |
0.20 |
|
Artificial tear use |
20.7% (6/29) |
43.5% (10/23) |
0.08^ |
|
Anti-allergic use |
3.5% (1/29) |
8.7% (2/23) |
0.42^ |
|
NIBUT (DR-1α) (in seconds) |
6.9 ± 2.9 |
6.7 ± 3.2 |
0.71 |
|
TMH (mm) |
0.25 ± 0.07 |
0.27 ± 0.07 |
0.20 |
|
Subjective dryness (VAS score) |
53.8 ± 23.1 |
49.6 ± 23.1 |
0.36 |
|
HOA (RMS; in μm) |
0.39 ± 0.13 |
0.39 ± 0.18 |
0.95 |
Data are expressed as the mean ± standard deviation.
*P values were calculated using a two-sample independent t-test in all instances except for the female ratio and ophthalmic solution use ratio, where the Pearson chi-squared test was used (^).
CCT, central corneal thickness; HOA, higher-order aberration; NIBUT, non-invasive tear break-up time; SD, standard deviation; SH-SCL, silicone hydrogel-soft contact lens; TMH, tear meniscus height; VAS, visual analog scale
Table 2: Comparison of the frequently used soft contact lenses (SCLs) between the two silicone hydrogel-soft contact lense (SH-SCL) groups within one month before the first visit (v1)
|
Narafilcon A (in the narafilcon A group) Total 58 eyes (%) |
Delefilcon A (in the delefilcon A group) Total 46 eyes (%) |
|
|
1-Day Acuvue TruEye (TE) (narafilcon A) |
7 (12.1%) |
6 (13.0%) |
|
Dailies Total 1 (delefilcon A) |
0 (0%) |
10 (21.7%) |
|
Myday (stenfilcon A) |
8 (13.8%) |
8 (17.4%) |
|
Medalist FreshFit (balafilcon A) |
4 (6.9%) |
0 (0%) |
|
Biofinity (comfilcon A) |
4 (6.9%) |
0 (0%) |
|
2-week Aqualox (samfilcon A) |
2 (3.4%) |
2 (4.3%) |
|
2-week Acuvue Oasis (senofilcon A) |
4 (6.9%) |
2 (4.3%) |
|
2-week Acuvue Advance (galyfilcon A) |
2 (3.4%) |
0 (0%) |
|
1-day Premio (asmofilcon A) |
2 (3.4%) |
0 (0%) |
|
1-day Acuvue Oasis (senofilcon A) |
0 (0%) |
4 (8.7%) |
|
1-day Aqualox (samfilcon A) |
4 (6.9%) |
0 (0%) |
|
1-day Acuvue Moist (etafilcon A) |
6 (10.3%) |
0 (0%) |
|
1-day Bio true (nesofilcon A) |
10 (17.2%) |
10 (21.7%) |
|
1-day Acuvue Define (etafilcin A) |
2 (3.4%) |
0 (0%) |
|
1day Pure (HEMA) |
1 (1.7%) |
0 (0%) |
|
1-day Medalist (hilafilcon B) |
2 (3.4%) |
2 (4.3%) |
|
Unknown |
0 (0%) |
2 (4.3%) |
The distributions of the SCL products that the participants used most frequently within one month before v1 were not significantly different (P = 0.07, Jonckheere-Terpstra test). HEMA, 2-Hydroxyethyl Methacrylate
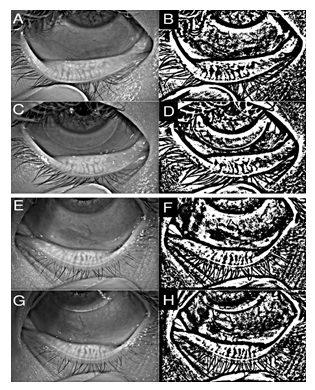
Figure 1: Representative measurements of eyelid meibomian gland (MG) area (%)..
Representative measurements of the lower eyelid MGs by meibography on Keratograph 5M at the first visit (vl) before narafilcon A silicone hydrogel-soft contact lens (SH-SCL) use (A);
digitized image from A (B);
meibography image at the second visit (v2) after one month of narafilcon A SH-SCL use (C);
digitized image from C (D):
The MG area in B was 47.57% in the total lower eyelid, which was reduced to 42.96% in D. Representative measurements for lower eyelid MG by meibography on Keratograph 5M at vi before delefilcon A use (E);
digitized images from E (F);
meibography at v2 after one month of delefilcon A SH-SCL use (G);
digitized image from G (H).
The MG area in F was 58.31% in the total lower eyelid, which was not reduced in H, at 64.59%.
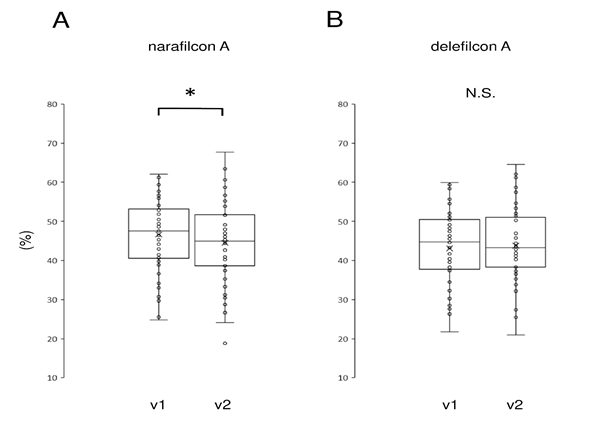
Figure 2: Comparison of meibomian gland (MG) area in the lower eyelid measured by infrared meibography
A reduction in MG area (in %) was observed in the narafilcon A group (A) but not in the delefilcon A group (B). The horizontal lines in the box and whisker plots represent the median values, and the bottom and top of the boxes represent the lower and upper quartiles, respectively. The “x” represents the mean and the bars represent the minimum and maximum values within 1.5 times the lower and upper quartiles, respectively.
* P < 0.05, paired t-test
NS, not significant
4. Discussion
This study assessed whether the water gradient structure of the delefilcon A lens has an impact on the MG. Overall, we observed that there was no damage to the MGs with the use of the delefilcon A lenses with a water gradient structure.
SH-SCLs have been developed to improve the properties of SCLs [25]. SCH-SCLs show high oxygen permeability [26] and resistance to protein deposition due low water content [27]. However, SH-SCLs are hydrophobic and thus have low affinity with the tear-film [28]. In addition, lipids tend to deposit on SHs [29].
SH-SCLs have certain unique limitations [30]. These limitations arise primarily because SHs are relatively hard [31, 32]. For this reason, “soft” SH contact lenses (SH-SCLs) have been developed, and are known to be relatively effective [33]. However, discomfort is a major residual complication with SH-SCLs [34]. Tear film thinning results in an increase in evaporation, and induces SH-SCL surface friction [36]. DT1 was developed to overcome these adverse properties of SH-SCLs; the water gradient technology employed in the DT1 lenses minimizes the surface friction issues [19].
MGs are the major organs that secrete the lipid layer of the tear film [24]. The lipid layer of the tear film prevents evaporation of the aqueous component of the tear film, thus stabilizing the tear dynamics and the homeostasis of the cornea and the conjunctiva [36]. The loss of MBs induced by the conventional SH-SCLs in this study might be due to surface friction which is characteristic of SH-SCLs [37]. A vicious cycle is initiated where MG loss due to conventional SH-SCLs may lead to instability of the lipid layers, which also results in increased surface friction [38].
Delefilcon A is a daily disposable water gradient contact lens marketed as DAILIES TOTAL1® by Alcon Laboratories The delefilcon A lenses possess the characteristics of both conventional hydrogel lenses and SH lenses. These lenses have a SH core with a water content of 33% ± 2% and a surface water content of 84.6% [13]. They have a total surface thickness of 5.9 ± 0.8 μm, including a 1–2 μm transition zone and a 4–5 μm outer surface layer with a low compression modulus; there is a significant modulus gradient in the zone of transition [13]. These features result in a water gradient that transitions from the core with a lower water content to the outermost hydrophilic gel layer with a higher water content. Consequently, compared to narafilcon A (the conventional SH counterpart with 46% water content), delefilcon A has higher oxygen permeability and a higher tensile modulus. Additionally, the water gradient technology has been reported to reduce the surface friction of SH-SCLs [39]. In this study, we observed that there was no physical damage to the MGs in the delefilcon A group, while there was significant MG damage in the narafilcon A group. This suggests that the water gradient technology was useful in preventing MB damage and maintaining ocular surface health during SH-SCL usage.
The main limitations of the present study included its retrospective and observational nature. Therefore, no randomization was performed. To minimize bias, the MG area measurements were obtained in a blinded manner. In addition, to reduce the probability of bias, a consecutive series of individuals was included in both the delefilcon A and narafilcon A groups with characteristics that matched the inclusion criteria. In this study, artificial tears or anti-allergic ophthalmic solutions were used in some patients, who could have impacted the results on tear dynamics; however, the distribution of participants did not differ between the delefilcon A and narafilcon groups. Our results need to be verified in a prospective, randomized, masked study in the future.
In conclusion, the loss of MGs induced by SH-SCL usage, presumably due to stress due to SCL wear, was significantly reduced in the delefilcon a group; this implies that the water gradient structure of the delefilcon a lens leads to reduced surface friction. Thus, the use of the delefilcon a lens can alleviate the abnormal MG conditions that result in pathological tear film conditions among contact lens wearers. Delefilcon A can maintain normal tear film conditions that may in turn improve lens performance.
Acknowledgements
The advice, discussions, and criticisms provided by T. Yamashita and Y. Setoguchi that greatly benefited this project are gratefully appreciated. This study was funded in part by an unrestricted investigator-initiated grant from Alcon Japan Ltd. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Funding Information: This research was funded in part by an unrestricted investigator-initiated grant from Alcon Japan Ltd (Grant Number: 42166553).
Commercial relationships disclosure: Hisataka Fujimoto, Alcon (F), Otsuka (F), Kowa (F, P), Santen (F), Novartis (F); Shintaro Ochi, Santen (F); Yasushi Inoue, Santen (F); Junichi Kiryu, (F); Kowa (F), Alcon (F), Novartis (F), Otsuka (F).
Word count: 2486 words (excluding title page, abstract, reference list, tables, and figure legends)
References
- Inoue T, Maeda N, Young LS, et al. Epithelial pigment slide in contact lens wearers: a possible marker for contact lens-associated stress on the corneal epithelium. Am J Ophthalmol 131 (2001): 431–437.
- Chan WK, Weissman BA. Corneal pannus associated with contact lens wear. Am J Ophthalmol 121 (1996): 540–546.
- Holden BA, Mertz GW. Critical oxygen levels to avoid corneal edema for daily and extended wear contact lenses. Invest Ophthalmol Vis Sci 25 (1984): 1161–1167.
- Fujimoto H. Noninvasive measurement of tear film break-up time in eyes with high-water-content contact lenses. Kawasaki Medical Journal 46 (2020): 87-
- Dumbleton KA, Woods CA, Jones LW, Fonn D. Comfort and adaptation to silicone hydrogel lenses for daily wear. Eye Contact Lens 34 (2008): 215–223.
- Chirila TV. An introduction to ophthalmic biomaterials and their application through tissue engineering and regenerative medicine. Biomat Regenerat Med Ophthalmol. Wood head Publishing (2010): 1–13.
- Fonn DP, Situ P, Simpson T. Hydrogel lens dehydration and subjective comfort and dryness ratings in symptomatic and asymptomatic contact lens wearers. Optom Vis Sci 76 (1999): 700–704.
- Efron N, Morgan PB. Rethinking contact lens-associated keratitis. Clin Exp Optom 89 (2006): 280–298.
- Maldonado-Codina C, Morgan PB, Schnider CM, Efron N. Short-term physiologic response in neophyte subjects fitted with hydrogel and silicone hydrogel contact lenses. Optom Vis Sci 81 (2004): 911–921.
- Nichols JJ, Jones L, Nelson JD, Stapleton F, Sullivan DA, Willcox MD. The TFOS international workshop on contact lens discomfort: introduction. Invest Ophthalmol Vis Sci 54 (2013): TFOS1–TFOS6.
- Rohit A, Willcox M, Stapleton F. Tear lipid layer and contact lens comfort: a review, Eye Contact Lens 39 (2013): 247–253.
- Pruitt J, Qiu Y, Thekveli S, Hart R. Surface characterization of a water gradient silicone hydrogel contact lens (delefilcon A). Invest Ophthalmol Vis Sci 53 (2012): 6107.
- Tighe BJ. A decade of silicone hydrogel development: surface properties, mechanical properties, and ocular compatibility. Eye Contact Lens 39 (2013): 4–12.
- Fujimoto H, Ochi S,Yamashita T, Inoue Y, Kiryu J. Role of the water gradient structure in inhibiting thin aqueous layer break in silicone hydrogel-soft contact lens. Transl Vis Sci Technol 10 (2021): 5.
- Dunn AC, Urueña JM, Huo Y, et al. Lubricity of surface hydrogel layers. Tribol Lett 49 (2013): 371–378.
- Belda-Salmerón L, Ferrer-Blasco T, Albarrán-Diego C, Madrid-Costa D, Montés-Micó R. Diurnal variations in visual performance for disposable contact lenses. Optom Vis Sci 90 (2013): 682–690.
- Montés-Micó R, Belda-Salmerón L, Ferrer-Blasco T, Albarrán-Diego C, García-Lázaro S. On-eye optical quality of daily disposable contact lenses for different wearing times. Ophthalmic Physiol Opt 33 (2013): 581–591.
- Varikooty J, Keir N, Richter D, Jones LW, Woods C, Fonn D. Comfort response of three silicone hydrogel daily disposable contact lenses. Optom Vis Sci 90 (2013): 945–953.
- Varikooty J, Schulze MM, Dumbleton K, et al. Clinical performance of three silicone hydrogel daily disposable lenses. Optom Vis Sci 92 (2015): 301–311.
- Dursch TJ, Liu DE, Oh Y, Radke CJ. Fluorescent solute-partitioning characterization of layered soft contact lenses. Acta Biomater 15 (2015): 48–54.
- Wolffsohn JS, Mroczkowska S, Hunt OA, Bilkhu P, Drew T, Sheppard A. Crossover evaluation of silicone hydrogel daily disposable contact lenses. Optom Vis Sci 92 (2015): 1063–1068.
- Rex J, Knowles T, Zhao X, Lemp J, Maissa C, Perry SS. Elemental composition at silicone hydrogel contact lens surfaces. Eye Contact Lens 44 (2018): S221–S226.
- Arita R, Suehiro J, Haraguchi T, Shirakawa R, Tokoro H, Amano S. Objective image analysis of the meibomian gland area. Br J Ophthalmol 98 (2014): 746–755.
- Choi YJ, Park SY, Jun I, et al. Perioperative ocular parameters associated with persistent dry eye symptoms after cataract surgery. Cornea 37 (2018): 734–739.
- Alvord L, Court J, Davis T, et al. Oxygen permeability of a new type of high Dk soft contact lens material. Optom Vis Sci 75 (1998): 30–
- Efron N, Morgan PB, Cameron ID, Brennan NA, Goodwin M. Oxygen permeability and water content of silicone hydrogel contact lens materials. Optom Vis Sci 84 (2007): 328–337.
- Morgan PB, Efron N, Helland M, et al. Twenty first century trends in silicone hydrogel contact lens fitting: an international perspective. Cont Lens Anterior Eye. 2010;33(4):196–198.
- Jones L, Senchyna M, Glasier MA et al. Lysozyme and lipid deposition on silicone hydrogel contact lens materials. Eye Contact Lens 29 (2003): S75–S79.
- Malet F, Pagot R, Peyre C, et al. Subjective experience with high-oxygen and low-oxygen permeable soft contact lenses in France. Eye Contact Lens 29 (2003): 55–59.
- Dumblenton K. Noninflammatory silicone hydrogel contact lens complications. Eye Contact Lens 29 (2003): S186–S189.
- Zhao Z, H Fu, Skotnitsky CC, Sankaridurg PR, Willcox MDP. IgE antibody on worn highly oxygen-permeable silicone hydrogel contact lens from patients with contact lens-induced papillary conjunctivitis (CLPC). Eye Contact Lens 34 (2008): 117–121.
- Dumbleton K. Adverse events with silicone hydrogel continuous wear. Cont Lens Anterior Eye 25 (2002): 137–146.
- Horst CR, Brodland B, Jones LW, Brodland GW. Measuring the modulus of silicone hydrogel contact lenses. Optom Vis Sci 89 (2012): 1468–1476.
- Dumbleton K, Woods CA, Jones LW, Fonn D. The impact of contemporary contact lenses on contact lens discontinuation, Eye Contact Lens 39 (2013): 93–99.
- Dumbleton K, Guillon M, Theodoratos P, Patel T. Diurnal variation in comfort in contact lens and non-contact lens wearers, Optom Vis Sci 93 (2016): 820–827.
- Llorens-Quintana C, Garaszczuk IK, Szczesna-Iskander DH. Meibomian glands structure in daily disposable soft contact lens wearers: a one-year follow-up study. Ophthalmic Physiol Opt 40 (2020): 607-616.
- Alghamdi WM, Markoulli M, Holden BA, Papas EB. Impact of duration of contact lens wear on the structure and function of the meibomian glands. Ophthalmic Physiol Opt 36 (2016): 120-31.
- Arroyo-Del AC, Fernández I, Novo-Diez A, Blanco-Vázquez M, López-Miguel A, González-García MJ. Contact Lens Discomfort Management: Outcomes of Common Interventions. Eye Contact Lens 47 (2021): 256-264.
- Eftimov P, Yokoi N, Peev N, Georgiev GA. Relationships between the material properties of silicone hydrogels: Desiccation, wettability and lubricity. J Biomater Appl 35 (2021): 933-946.


 Impact Factor: * 1.2
Impact Factor: * 1.2 Acceptance Rate: 79.45%
Acceptance Rate: 79.45%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
