Menopausal Status and MTHFR Gene Polymorphism in the Etiopathogenesis of Osteoporosis
Yasovanthi Jeedigunta1*, Shehnaz Sultana2, Balakrishna Nagalla1, Raghunath Manchala1, Rajender Rao Kalashikam1
1Department of Endocrinology and Metabolism, National Institute of Nutrition, Jamai Osmania, Telangana, India
2Institute of Genetics and Hospital for Genetic Diseases, Osmania University, Telangana, India
*Corresponding author: Dr. Yasovanthi Jeedigunta, Department of Endocrinology and Metabolism, National Institute of Nutrition, Jamai Osmania, Hyderabad – 500 007, Telangana, India
Received: 08 June 2020; Accepted: 17 July 2020; Published: 01 August 2020
Article Information
Citation:
Yasovanthi Jeedigunta, Shehnaz Sultana, Balakrishna Nagalla, Raghunath Manchala, Rajender Rao Kalashikam. Menopausal Status and MTHFR Gene Polymorphism in the Etiopathogenesis of Osteoporosis. Journal of Women’s Health and Development 3 (2020): 211-221.
DOI: 10.26502/fjwhd.2644-28840032
View / Download Pdf Share at FacebookAbstract
Background: Osteoporosis is a skeletal disorder characterized by low bone mass with consequent increase in bone fragility and fracture. The interplay between genetics and environment has a crucial role in determining the bone mineral density. Many studies showed that the genetic polymorphisms of MTHFR gene and its impact in the development of numerous human diseases. Normal MTHFR activity may help maintain the pool of circulating folate and methionine and possibly prevent of homocysteine. It has been shown that high serum homocysteine concentration may weaken bone by interfering with collagen cross-linking, thereby increasing the risk of osteoporosis. Therefore, the present study was aimed to investigate the role of MTHFR C677T gene polymorphism and its influence on BMD in pre and postmenopausal osteoporotic women of Indian ethnicity.
Methods: In this study 427 osteoporotic women and 460 age matched controls were included. MTHFR C677T gene polymorphism was assessed by PCR-RFLP method. Total ALP, Bone specific ALP Total acid phosphatase, TRAP, Calcium and Phosphorus was measured by Bergmeyer et al method.
Results: The frequency of TT genotype and T allele was more in pre- and postmenopausal osteoporotic women in comparison with controls. The logistic regression analysis to understand the risk assessment showed that the TT genotypes were 2.7 times (95% CI 1.1-1.6) and CT 1.7 times (95% CI 1.3-2.2 ) were at higher risk of osteoporosis in comparison to CC genotypes .This was found true even after adjustment for menopausal status.
Conclusions: This study showed that increased bone turnover is not only restricted to postmenopausal women indicating the role of MTHFR gene variations in determining osteoporosis in both pre and post-menopausal South Indian women from Telangana.
Keywords
<p>Bone mineral density, Genotypes, Polymorphism, Postmenopausal, Premenopausal, Osteoporosis</p>
Article Details
1. Introduction
Osteoporosis is a skeletal disorder characterized by low bone mass and micro architectural deterioration of bone tissue with consequent increase in bone fragility and fracture. The major predictor of osteoporosis is bone mineral density which is the net result of peak bone mass achieved in early adulthood or bone loss later in life or can be a combined effect[1]. The interplay between genetics and environment have a crucial role in determining the bone mineral density. Methylenetetrahydrofolatereductase a candidate gene which is playing a key role in the production of homocysteine and has been suggested as one of the genetic factors in regulating bone mass [2, 3, 4, 5]. The MTHFR gene is located on Chromosome 1 at 1p36.3 which is a member of nuclear hormone receptor super family that modulates the transcription of target genes. Many studies showed that the genetic polymorphisms of MTHFR gene and its impact in the development of numerous human diseases. A point mutationC to T substitution at nucleotide 677 that converts an alanine to a valine residue is responsible for the synthesis of a thermo labile form of MTHFR [6-9]. Further this change in the MTHFR gene produces a form of methylenetetrahydrofolatereductase that has reduced activity at higher temperatures (thermo labile). The thermolabile variant of the MTHFR enzyme which is less effective and associated with an increase in total homocysteine levels [10].
The high serum homocysteine concentration may weaken bone by interfering with collagen cross-linking, lowering bone mineral density thereby increasing the risk of osteoporosis. Many studies have been reported that the allelic variants of the gene encoding MTHFR recognized by HinfI restriction endonuclease have been associated with bone density [11, 12, 13]. Studies have also shown that this polymorphism was associated with an increased risk of low bone mineral density leading to osteoporotic fractures [14]. Some other studies have failed to confirm this association [3, 12, 15]. The discrepancy among the studies in establishing the association between MTHFR gene polymorphisms and osteoporosis could be due to the differences in populations, genetic make-up and ethnicity. However, there are no studies on these lines to understand the association of this genetic variant with osteoporosis in women of Indian ethnicity. Therefore, the present study has been taken up with an aim to assess the role of MTHFR C677T (rs1801133) gene polymorphism in the development of osteoporosis in pre-and postmenopausal South Indian women from Telangana.
2. Material and Methods
2.1 Subjects
For this study the ethical committee clearance was obtained from the institute. Subjects were recruited after taking informed consent with an assurance of confidentiality. Blood samples were collected from427 osteoporotic (premenopausal n=180 and postmenopausal n=247) women. As a control group 460 healthy women (normal BMD), age matched were recruited from the same demographic area (premenopausal n=206 and postmenopausal n=254). Women were considered postmenopausal if they had no menstruation for at least 12 months. Women with regular menses, non-pregnant and non-lactating at the time of sample collection were considered as premenopausal. All the subjects underwent Bone Mineral Density (BMD) measurements by Dual Energy X-ray Absorptiometry (DEXA) at lumbar spine (vertebrae L1–L4). On the basis of T-score of DEXA, they were classified as osteoporotic or normal according to WHO classification of osteoporosis (WHO 1994). A structured questionnaire was used to collect the detailed information on age, medical history and life style of all the subjects studied. None of the subjects were alcoholic or smokers and they were on their daily routine. Subjects with endocrinological disorders (such as hyperthyroidism, hypo- and hyperparathyroidism, diabetes mellitus, chronic disorders of liver and kidney, other skeletal diseases, Paget's disease, osteogenesis imperfecta, rheumatoid arthritis, systemic lupus erythematosus, ankylosing spondylitis and rheumatic arthritis) were excluded from the study. Subjects who were on medications known to affect bone density and metabolism (corticosteroids and anticonvulsants), or women with hormone replacement therapy, premature menopause were also excluded.
2.2 BMD measurements
Bone Mineral Density (BMD) was measured from anterior–posterior lumbar spine (L1–L4) scanning with Dual Energy X-ray Absorptiometry (DEXA, Lunar GE Medical Systems, Prodigy 6.80, DF+10243). Subjects were categorized according to the World Health Organization (WHO) definition osteopenia was diagnosed at −2.5
2.3 Biochemical markers
Blood samples were collected after an overnight fasting. Serum calcium and phosphorus levels were measured using atomic absorption spectroscopy. Total alkaline phosphatase, Bone specific alkaline phosphatase (BALP) and tartrate resistant acid phosphatase was measured by the Bergmeyer et al. (1974) method.
2.4 PCR-RFLP analysis
Genomic DNA was isolated from the whole blood samples using TKM1 method and genotypes were detected by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) analysis (Lahiri and Nurberger 1991; Frosst et al., 1995). The polymorphic locus of MTHFR gene was amplified using 5’TGAAGGAGAAGGTGTCTGCGGGA-3’as forward and5’AGGACGGTGCGGTGAGAGTG -3’ as reverse primer. PCR (Thermo cycler; Bio-Rad) was performed through 40 cycles by the following steps: denaturation at 94 °C for 30 sec, annealing at 61 °C for 30 sec. and extension at 72 °C for 1 min. The 198 bp amplicon was subjected for digestion with HinfI restriction endonuclease, (Fermentas Glen Burnie, MD) for 14 hours. Digested PCR products along with 100 bp marker were analyzed on 3% agarose gel. The genotyping was done on the basis of the presence or absence of Hinf site: CC=198 bp, CT=198 bp, 175 bp and 23 bp, and TT =175bpand 23 bp, lengths respectively (Figure-1).
2.5 Statistical analysis
Hardy–Weinberg equilibrium was first tested for MTHFR C677T genotypes. Association between the genotypes and osteoporosis was examined by using odds ratio (OR) with 95% confidence interval (CI) and χ2 analysis was carried out using SPSS software ver. 15 (Chicago, IL). All the statistical tests were 2-sided and were considered significant at p<0.05. Allelic frequencies were calculated according to the number of different alleles observed and the total number of alleles examined.
3. Results
In this study we found that the BMI and BMD were low in osteoporotic subjects in pre and postmenopausal women when compared to controls (p<0.05) (Table1). The mean ages of the study subjects were 39.5±4.4 and 57.7±4.6 years in pre- and postmenopausal women respectively. It was observed that the percentage of individuals with family history was more in osteoporotic group in comparison with the controls. There was no significant difference depicted with respect to age at menarche (Table 1).
|
Variables |
Premenopausal women |
Postmenopausal women |
||
|
Controls |
Osteoporotics |
Controls |
Osteoporotics |
|
|
Number of Subjects |
206 |
180 |
254 |
247 |
|
Age (years) |
37.36 ± 4.18 |
39.51 ± 4.23 |
55.73 ± 4.39 |
57.74 ± 6.33 |
|
BMI (kg/m2) |
25.92 ± 3.79 |
23.61 ± 3.72 |
25.46 ± 3.31 |
24.07 ± 4.88 |
|
LS BMD (T score) |
-0.42 ± 0.30 |
-2.74 ± 0.29 |
-0.78 ± 0.26 |
-2.89 ± 1.01 |
|
LS BMD (Z score) |
-0.52 ± 0.27 |
-2.91 ± 0.35 |
-0.15 ± 0.23 |
-2.93 ± 0.45 |
|
LS BMD (g/cm2) |
1.12 ± 0.11 |
0.88 ± 0.07 |
0.969 ± 0.839 |
0.768 ± 0.129 |
|
Age at menarche (Years) |
12.52 ± 0.53 |
12.53 ± 0.52 |
12.51 ± 0.51 |
12.78 ± 0.61 |
|
Age at menopause (Years) |
- |
- |
46.94 ± 5.44 |
45.98 ± 5.51 |
|
Years since menopause (Years) |
- |
- |
8.17 ± 6.42 |
9.25 ± 6.34 |
|
Family History (%) |
- |
43.3 |
- |
37.8 |
Data are mean ± SD
Table 1: Characteristics of the study population according to menopausal status.
The biochemical markers bone alkaline phosphatase, total acid phosphatase and bone acid phosphatase were significantly higher in osteoporotic subjects in both the groups when compared to their respective controls. The serum calcium and phosphorus levels were comparable in both the groups studied (Table 2).
|
Variables |
Pre- menopausal women |
Post-menopausal women |
||
|
Controls |
Osteoporotics |
Controls |
Osteoporotics |
|
|
Total ALP (IU/L) |
73.93 ± 5.90 |
73.68 ± 9.21 |
99.14 ± 6.17 |
99.35 ± 7.28 |
|
Bone ALP (IU/L) |
24.24 ± 3.18 |
40.40 ± 5.45* |
34.88 ± 4.3 |
42.47 ± 7.46* |
|
Total Acid Phosphatase (IU/L) |
7.24 ± 0.37 |
10.48 ± 1.99* |
8.64 ± 1.33 |
10.24 ± 1.54* |
|
Bone acid phosphatase (IU/L) |
3.25 ± 0.44 |
5.18 ± 1.53* |
3.60 ± 0.53 |
6.24 ± 0.76* |
|
Calcium (mg/dl) |
9.90 ± 0.47 |
9.10 ± 0.41 |
9.29 ± 0.33 |
9.33 ± 0.64 |
|
Phosphorus (mg/dl) |
3.52 ± 0.23 |
3.58 ± 0.33 |
4.21 ± 0.29 |
4.44 ± 1.80 |
Data are mean ± SD,*P≤ 0.05 using ANOVA
Table 2: Bone Biochemical markers according to menopausal status.
The percentage distribution of MTHFR C677T genotype in pre-menopausal controls were 64.1% of ‘CC’, 34.45% of ‘CT’ and 1.45% of ‘TT’ and the allele frequency of ‘C’ was 0.81 and ‘T’ was 0.19. In osteoporotic it was 61.1% of ‘CC’, 35.6% of ‘CT’ and 3.3% of ‘TT’ and the allelic frequency of ‘C’ was 0.78 and ‘T’ was 0.21. On the other hand, in post-menopausal controls it was 64.8% of ‘CC’, 36.2% of ‘CT’ and 2% of ‘TT’ and the frequency of “C” allele 0.8 and ‘T’ allele 0.2. In osteoporotic, the genotypes were 59.5% of ‘CC’, 37.3% of ‘CT’ and 3.2% of ‘TT’ and their allelic frequencies were ‘C’ 0.78 and ‘T’ 0.21 respectively (Table-3).
|
Study group |
MTHFR Genotypes |
Allelic frequencies |
|||||
|
Premenopausal women |
CC |
CT |
TT |
Total |
C |
T |
Total |
|
Controls |
132(64.1) |
71(34.45) |
3(1.45) |
206 |
335(0.81) |
77(0.19) |
412 |
|
Osteoporotic |
92(51.1) |
81(45.0) |
7(3.9) |
180 |
265(0.70) |
95(0.30) |
360 |
|
Postmenopausal women |
|||||||
|
Osteoporotic |
122(49.4) |
117(47.4) |
8(3.2) |
247 |
361(0.70) |
133(0.30) |
494 |
Table 3: Genotypic and allelic frequencies of MTHFR C677Tgenotypes according to menopausal status.
Parenthesis (percentage)
- χ2=0.83 (2df), p=0.36 for genotypes; χ2=0.56 (1df), p=0.4 for allelic frequency, OR for T vs. C=1.16 (95% CI: 0.81–1.65), p=0.4 in premenopausal women.
- χ2=0.41 (2df), p=0.51 for genotypes; χ2=0.48 (1df), p=0.4 for allelic frequency, OR for T vs. C=1.11 (95%
CI: 0.82–1.51), p=0.4 in postmenopausal women.
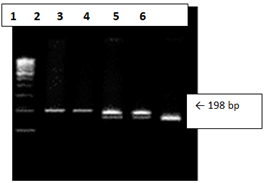
Figure 1: Gel Picture Showing MTHFR C677T Genotypes.
Lane 1 : 100 bp -marker.
Lane 2 : 198 bp-PCR product
Lane 3 : 198 bp -Homozygous (CC).
Lane 4, 5 : 198,175 bp- Heterozygous (CT)
Lane 6 : 175 bp-Homozygous (TT)
|
MTHFR C677T genotypes |
|||||
|
Variables |
CC |
CT |
TT |
||
|
(n=503) |
(n=361) |
(n=23) |
|||
|
Age (years) |
48.40 ± 10.1 |
49.18 ± 10.7 |
49.17 ± 11.3 |
||
|
BMI ( kg/m2) |
24.83 ± 4.02 |
24.72 ± 3.5 |
24.33 ± 4.12 |
||
|
BMD (T-score) |
-1.40 ± 1.2 |
-1.81 ± 1.2 |
-2.03 ± 1.1* |
||
|
Calcium (mg/dl) |
9.16 ± 1.5 |
9.13 ± 1.8 |
9.28 ± 1.6 |
||
|
Phosphorus (mg/dl) |
3.95 ± 0.8 |
4.04 ± 1.1 |
3.99 ± 1.5 |
||
|
TALP (IU/L) |
88.03 ± 16.17 |
89.17 ± 15.8 |
87.07 ± 16.2 |
||
|
BALP (IU/L) |
36.46 ± 10.0 |
37.98 ± 9.5 |
37.48 ± 7.9 |
||
|
Total Acid phosphatase (IU/L) |
9.13 ± 2.27 |
9.50 ± 2.10 |
9.4 ± 1.52* |
||
|
TRAP(IU/L) |
4.26 ± 2.02 |
4.43 ± 3.18 |
3.82 ± 2.1 |
||
Data are mean ± SD, *P ≤ 0.05 using Anova
Table 4: Association of Biochemical markers according to MTHFR genotypes in all the Study subjects.
The MTHFR C677T genotypes and its allelic frequencies when compared between controls and osteoporotic it was noticed that the TT genotype and T allele frequency was significantly more in the osteoporotic in both the pre- and post-menopausal women. However, when compared between osteoporotic of both the groups the study could not find any significant difference. Further it was also noticed that the serum calcium, phosphorus, bone ALP, and TRAP did not differ among the genotypes (Table -4). Interestingly it was noticed that the TT genotype individuals of controls as well as osteoporotic subjects were having significantly low BMD (p<0.05) in comparison to CC and CT genotypes (Table -4). The logistic regression analysis to understand the risk assessment showed that the TT genotypes were 2.7 times (95% CI 1.1-1.6) and CT 1.7 times (95% CI 1.3-2.2) were at higher risk of osteoporosis in comparison to CC genotypes. This was found true even after adjustment for menopausal status.
4. Discussion
Studies have reported that the gene polymorphisms play an important role in controlling the Methylenetetrahydrofolatereductase enzyme activity and leads to elevated homocysteine level which is a risk factor for low BMD and fracture [16, 17]. The present study showed that low BMI and BMD in osteoporotic subjects in pre and postmenopausal women aged 39.5 ± 4.4 and 57.7 ± 4.6 years respectively are in accordance with earlier reports [18-20]. Moreover, the demographic data on family history of low BMD have shown that genetics play an important role in determining the bone mass (Table 1). It was also noticed that significantly increased levels of bone alkaline phosphatase and bone acid phosphatase in osteoporotic indicating high bone turnover which is quite common in post-menopausal women [21]. Interestingly the present study noticed this condition even in pre-menopausal women.
However, to the best of our knowledge this study first time showed the increased levels of bone turn over markers even in the pre-menopausal women indicating high bone remodeling. The frequency of the MTHFR 677T allele varies substantially in different regions of the world and among ethnic groups [22-24]. A north-to-south increase of allele 677T prevalence has been observed in Europe [25]. In contrast, a north-to-south decrease of TT genotype was reported in China [26]. It has been reported that the TT genotype was found in about 8%–9% of the Caucasian population [27]. On the other hand, Chinese populations showed a higher proportion of TT genotype (17%–40%) [28]. More interestingly, the distribution of the MTHFR genotype may modify its effect on the plasma homocysteine [29]. All these findings strengthen the importance of studying the influence of this polymorphism in different populations. So far, to the best of our knowledge this is the first study to determine the association of C-T polymorphism of HinfI genotypes with osteoporosis and also to correlation with BMD in both pre- and post-menopausal women from South Indian population, Telangana.
In this study it was observed that the frequency of the TT genotype and the T-allele are 2.4% and 0.20% respectively in all the subjects studied which is comparable to a previous study from this region on cardiovascular diseases [30-32]. Also our study on MTHFR C677T genotypic and allelic frequencies were very similar to those observed in Asian Koreans and Japanese [24, 33]. The low frequency distribution of TT genotype in our study population observed may be due to racial and ethnic differences. The studies on causal mechanisms underlying the relationship between MTHFR genotypes and BMD not yet clear. However, the present study found a significant relation of TT genotype with BMD (Table-4). The TT genotype individuals were having lower BMD in comparison with CC and CT genotype individuals irrespective of menopausal status. When risk assessment was calculated by logistic regression analysis it was found that TT genotypes were at higher risk of osteoporosis in comparison to CC genotype individuals and this condition does not vary in both groups. These results were in agreement with studies on post-menopausal women where it was noticed that a significant association between the MTHFR TT genotype and low lumbar spine and total body BMD [34]. whereas others studies could not find any association with BMD [12, 35]. The discrepancy in frequency distribution of MTHFR genotypes and its association with BMD could be due to racial differences coupled with environmental factors. Further this study supports the role of genetics in pathology of osteoporosis.
5. Conclusion
MTHFR gene polymorphism was associated with BMD in the present study subjects and TT genotype individuals are at 2.7 time’s higher risk of osteoporosis in comparison to CC genotypes irrespective of menopausal status. This study also showed that increased bone turnover is not only restricted to postmenopausal women indicating the role of MTHFR gene variations in determining osteoporosis.
Conflict of Interest
All the authors declare no conflict of interest
Acknowledgements
The authors are thankful to Indian Council of Medical Research (ICMR), New Delhi for financial support.
References
- Turner RT, Riggs BL, Spelsberg TC. Skeletal effects of estrogen. Endocr. Rev 15 (1994): 275- 300.
- Jorgensen HL, Madsen JS, Madsen B, Saleh M M A, Abrahamsen B, Fenger M, et al. Association of a common allelic polymorphism (C677T) in the methylene tetrahydrofolatereductasegene with a reduced risk of osteoporotic fractures. A case control study in Danish postmenopausal women. Calcif Tissue Int 71 (2002): 386-392.
- Agueda L, Urreizti R, Bustamante M, Jurado S, Garcia-Giralt N, Díez-Pérez A, et al. Analysis of three functional polymorphisms in relation to osteoporosis phenotypes: replication in a Spanish cohort. Calcif Tissue Int 87 (2010): 14-24.
- Huang QY, Li GH, Kung AW. Multiple osteoporosis susceptibility genes on chromosome 1p36 in Chinese. Bone 44 (2009): 984-988.
- Kiel DP, Demissie S, Dupuis J, Lunetta KL, Murabito JM, Karasik D. Genome-wide association with bone mass and geometry in the Framingham Heart Study. BMC Med Genet 8 (2007): S14.
- Goyette P, Sumner JS, Milos R, Duncan A M, Rosenblatt DS, Matthews RG, et al. Human methylenetetrahydrofolatereductase: Isolation of cDNA mapping and mutation identification. Nat Genet 7 (1994a): 551.
- Rozen R. Genetic predisposition to hyperhomocysteinemia: deficiency of methylenetetrahydrofolatereductase (MTHFR). Thromb Haemost 78 (1997): 523-526.
- Frosst P, Blom H J, Milos R, Goyette P, Sheppard C A, Matthews R G, et al. A candidate genetic risk factor for vascular d H J isease: a common mutation in methylenetetrahydrofolatereductase. (Letter). Nat Genet 10(1995): 111-113.
- Goyette P, Pai A, Milos R, Frosst P, Tran P, Chen Z, et al. Gene structure of human and mouse methylenetetrahydrofolatereductase (MTHFR). Mamm Genome 9 (1998b): 652-656.
- Kluijtmans LAJ, Young IS, Boreham CA, Murray L, McMaster D, McNulty H, et al. Genetic and nutritional factors contributing to hyperhomocysteinemia in young adults. Blood 101 (2003): 2483-2488.
- Baines M, Kredan MB, Usher J, Davison A, Higgins G, Taylor W, et al. The association of homocysteine and its determinants MTHFR genotype, folate, vitamin B12 and vitamin B6 with bone mineral density in postmenopausal British women. Bone 40 (2007): 730-736.
- Abrahamsen B, Madsen JS, Tofteng CL, Stilgren L, Bladbjerg EM, Kristensen SR, et al. Are effects of MTHFR (C677T) genotype on BMD confined to women with low folate and riboflavin intake? Analysis of food records from the Danish osteoporosis prevention study. Bone 36 (2005): 577-583.
- Bathum L, von BornemannHjelmborg J, Christiansen L, Madsen JS, Skytthe A, Christensen K. Evidence for an association of methylene tetrahydrofolatereductase polymorphism C677T, an increased risk of fractures: results from a population-based Danish twin study. Osteoporos Int 15 (2004): 659-664.
- Gjesdal CG, Vollset SE, Ueland PM, Refsum H, Meyer HE, Tell GS. Plasma homocysteine, folate, and vitamin B 12 and the risk of hip fracture: the hordaland homocysteine study. J Bone Miner Res 22 (2007): 747-756.
- Villadsen MM, Bunger MH, Carstens M, Stenkjaer L, Langdahl BL. Methylenetetrahydrofolatereductase (MTHFR) C677T polymorphism is associated with osteoporotic vertebral fractures, but is a weak predictor of BMD. Osteoporos Int 16 (2005): 411-416.
- MasatakaShiraki, TomohikoUrano, Tatsuhiko Kuroda Mitsuru Saito, Shiro Tanaka, Mariko Miyao-Koshizuka Satoshi Inou. The synergistic effect of bone mineral density and methylenetetrahydrofolatereductase (MTHFR) polymorphism (C677T) on fractures, J Bone Miner Metab 26 (2008): 595-602.
- Clara Gram Gjesdal, Stein Emil Vollset, Per MagneUeland, Helga Refsum, Christian A Drevon, Ha kon K Gjessing, et al. Tell. Plasma Total Homocysteine Level and Bone Mineral Density. Arch Intern Med 166 (2006): 88-94.·
- Cummings SR, Nevitt MC, Browner WS, Stone K, Fox KM, Ensrud KE, et al. Risk factors for hip fractures in white women. New England Journal of Medicine 332 (1995): 767-773.
- Reid IR, Ames R, Evans MC, Sharpe S,Gamble G, France JT, et al. Determinants of total body and regional bone mineral density in normal post menopausal women : a key role for fat mass. Journal of Clinical Endocrinology and Metabolism 75 (1992): 45-51.
- Christiansen C, Riis BJ. New methods for identifying “at risk” patients for osteoporosis. Clinical Rheumatology 8 (1989): 52-55.
- Garnero P, Sornay-Rendu E, Chapuy MC, Delmas PD. Increased bone turnover in late postmenopausal women is a major determinant of osteoporosis, J Bone Miner Res 11 (1996): 337-349.
- Gueant-Rodriguez RM, Gueant JL, Debard R, Thirion S, Hong LX, Bronowicki JP, et al. Prevalence of methylenetetrahydrofolatereductase 677T and 1298C alleles and folate status: a comparative study in Mexican, West African, and European populations. Am J Clin Nutr 83 (2006): 701-707.
- Hegele RA, Tully C, Young TK, Connelly PW. V677 mutation of methylenetetrahydrofolatereductases and cardiovascular disease in Canadian Inuit. Lancet 349 (1997): 1221-1222.
- Pepe G, Venegas OC, Giusti B, Brunelli T, Marucci R, Attanasio M, et al. Heterogeneity in world distribution of thermolabile C677T mutation in 5,10-methylenetetrahydrofolate reductase. Am J Hum Genet 63 (1998): 917-920.
- Botto LD, Yang Q. 5,10-Methylenetetrahydrofolate reductase gene variants congenital anomalies: a HuGE review. Am J Epidemiol 151 (2000): 862-877.
- Zhu J, Ren A, Hao L, Pei L, Liu J, Zhu H, et al. Variable contribution of the MTHFR C677T polymorphism to non-syndromic cleft lip and palate risk in China. Am J Med Genet A 140 (2006): 551-557.
- Linda Sharp, Julian. Little Polymorphisms in Genes Involved in Folate Metabolism and Colorectal Neoplasia: A HuGE Review American Journal of Epidemiology 159 (2004): 423-443.
- Crider KS, Zhu JH, Hao L, Yang QH, Yang TP, Gindler J, et al. MTHFR 677C->T genotype is associated with folate and homocysteine concentrations in a large, population-based, double-blind trial of folic acid supplementation. Am J Clin Nutr 93 (2011): 1365-1372.
- Kumar J Das, Sharma SK, Karthikeyan P, Ramakrishnan G, Sengupta L. Homocysteine levels are associated with MTHFR A1298C polymorphism in Indian population. J.Hum.Genet 50 (2005): 655-663.
- Salden A, Keeney S, Hay CR, Cumming AM, et al. The C677T MTHFR variant and the risk of venous thrombosis. (Letter). Br J Haematol 99 (1997): 472.
- Sell SM, lugema. Development of a highly accurate, rapid PCR-RFLP genotyping assay for the methylenetetrahydrofolatereductase gene. Genet Test 3 (1999): 287-289.
- Rosenblatt DS. Inherited disorders of folate transport and metabolism. In: Scriver CR, Beaudet AL, Sly WS, et al, eds. The metabolic and molecular bases of inherited disease. New York, NY: McGraw-Hill Book Company (1995): 3111-28.
- Schneider JA, Rees DC, Liu YT, Clegg JB. Worldwide distribution of a common methylene tetra hydro folate reductase mutation. Am J Hum Genet 62 (1998): 1258-1260.
- Miyao M, Morita H, Hosoi T, Kurihara H, Inoue S, Hoshino S, et al. Association of methylenetetrahydrofolatereductase (MTHFR) polymorphism with bone mineral density in postmenopausal Japanese women Calcif Tissue Int 66 (2000): 190-194.
- Golbahar J, Hamidi A, Aminzadeh MA, Omrani GR. Association of plasma folate plasma total homocysteine but not methylenetetrahydrofolatereductase C667T polymorphism, with bone mineral density in postmenopausal Iranian women: a cross-sectional study. Bone 35 (2004): 760-765.
- Bergmeyer HU, Gawehn K, Grassl M. In Methods of Enzymatic Analysis (Bergmeyer, H.U. ed.) 2nd ed., Academic Press, New York 518 (1974): 856-860.
- Lahiri DK , Nurberger JI. A rapid non-enzymatic method for preparation of HMW DNA from blood for RFLP studies, Nuc. Acid Res 19 (1991): 5444.
- Consensus development conference: diagnosis prophylaxis and treatment of osteoporosis. Am J Med 94 (1994): 646-650.


 Impact Factor: * 3.4
Impact Factor: * 3.4 Acceptance Rate: 78.89%
Acceptance Rate: 78.89%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 