Molecular Mechanisms of Pathways in Diabetic Nephropathy Development in Patients with T2DM -A Review
Medha Srivastava1* Bhuvnesh Rai1*
1Department of Molecular Medicine and Biotechnology, Sanjay Gandhi Postgraduate Institute of Medical Sciences, Lucknow, 226014, India
*Corresponding author(s): Medha Srivastava, Bhuvnesh Rai. Department of Molecular Medicine and Biotechnology, Sanjay Gandhi Postgraduate Institute of Medical Sciences, Lucknow, 226014, India
Received: 23 September 2021; Accepted: 30 September 2021; Published: 05 October 2021
Article Information
Citation: Medha Srivastava, Bhuvnesh Rai. Molecular Mechanisms of Pathways in Diabetic Nephropathy Development in Patients with T2DM -A Review. International Journal of Applied Biology and Pharmaceutical Technology 12 (2021): 380-392.
View / Download Pdf Share at FacebookAbstract
Diabetic nephropathy is related to increased mortality in diabetic patients and may be a major reason for end stage renal disease in most countries. The consequence of the combination of metabolic and hemodynamic pathway tends to be diabetic nephropathy. Glucose dependent pathways are activated with in diabetic kidney. This involves growing oxidative stress, and advanced glycated end-product accumulation. The pathogenesis of diabetic nephropathy also includes elevated systemic and intraglomerular pressure and also activation of multiple vasoactive hormone pathways, such as renin-angiotensin and endothelin. The aim of this review analysis is to gather knowledge of the molecular pathways and the factors involved in the pathogenesis of diabetic nephropathy.
Keywords
Diabetic Nephropathy
Nephropathy articles
Article Details
1. Introduction
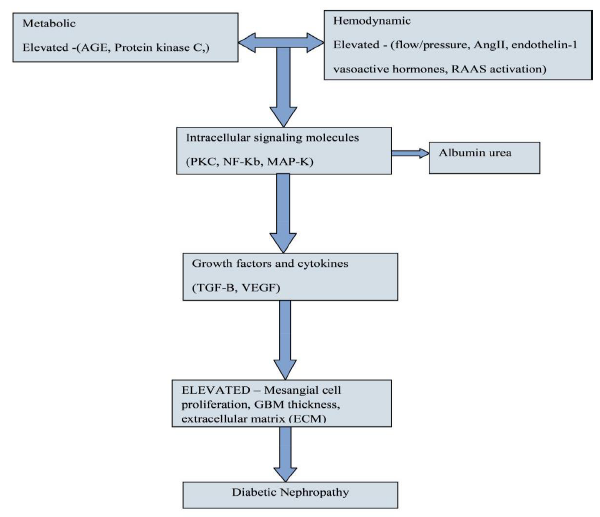
Diabetic nephropathy is also known as Kimmelstiel Wilson Syndrome and was identified in 1936 by Clifford Wilson and Paul Kimmelstiel. Diabetic nephropathy is the product of associations between hemodynamic and metabolic factors [1]. Excessive systemic and intraglomerular strain, together with active multiple vasoactive hormone pathways including renin angiotensin and endothelin [2], is implicated in hemodynamic factors contributing to the production of diabetic nephropathy. These hemodynamic pathways activates intracellenic second messengers such as protein kinase C (PKC), mitogen-activated protein (MAPkinase) [3] and nuclear transcription factors such as the NF-kB and vascular endothelial growth factor, VEGF. Glucose-dependent pathways are most often triggered in the diabetic kidney and lead to increased oxidative stress, renal polyol formation [4] as well as aggregation of advanced glycation end products (AGEs). In addition, the effects of these processes gradually improve the permeability of renal albumins and extracellular matrix, which results in increased proteinuria, glomerulosclerosis and finally tubulointerstitial fibrosis. The impact of diabetic nephropathy on the expanded CKD and ESRD population is massive. Increased multifactorial treatment in patients with type 2 diabetes has resulted in decreased incidence of Controlled Steno Type 2 trials of micro-angiopathy and mortality; yet ESRD is that worldwide [5-7]. In these circumstances it will substantially enhance and encourage the creation of new therapeutically focused strategy through the discovery of inflammation-related molecules and pathways that are critically involved in the progression of diabetic nephropathy [8, 9]. The existing state of work in relation to molecules and pathways involved in diabetic nephropathy and its pathogenesis is discussed and the opportunities for diabetic nephropathy study are outlined in this analysis.
2. History of Diabetic Nephropathy
Diabetic nephropathy has a pathological background distinguished by the prior clinical period in which hyperglycaemia is followed by an improvement in GFR and microalbuminuria (Urinary albumin excretion of urine samples of up to 20–200 mg / min). Lifetime high glomerular flow and pressure and poor hyperglycaemic control eventually cause renal damage, leading to microalbuminuria progression to macroalbuminuria and a decline in the amount of GFR. In 20-40% of patients with type 2 diabetes over a 15-20-year span since the onset of the condition, development to macroalbuminuria (urinary albumin excretion > 200 mg / min) or active nephropathy is reported. Since macroalbuminuria exists, the rate of clearance of creatinine ranges significantly across individuals, with the estimated reduction in untreated individuals of 10 to 12 ml / min per year. The decline of GFR and the growth in final stage renal disease can be accelerated by hypertension and proteinuria [10, 11]. Furthermore, the incidence of cardiovascular disease is increased with microalbuminuria and proteinuria separate marks [12, 13]. While nephropathy typically exists in type 1 and type 2 diabetes, coronary losses in type 2 diseases are more prevalent from the outset. This is probably because Type 2, diagnosis, when macrovascular and microvascular complications are undetected, is usually diagnosed relatively late in the course of the disease.
3. Hemodynamic pathway and their link with metabolic pathway
The early symptoms in both afferent and efferent glomerulin are glomerular hyperfusion and high filtration, which are the result of decreasing resistance. Afferent arteriole is more robust than efferent arteriole. A variety of causes, such as nitrogen oxide, prostanoids, VEGF, TGF-β1, and the rennin angiotensin mechanism, in particular angiotensin II, have been shown to be implicated in this deficient self-regulatory activity. Early hemodynamic changes limit glomerular capillary leakage and overproduction of the mesangial cell matrix as well as distribution and damage to podocytes of the glomerular basement membrane [14]. More mechanical pressure due to these hemodynamic shifts may also contribute to localized release of certain cytokines and growth factors [15]. Vasoactive hormone, like angiotensin II and endotheline, mediate changes in renal hemodynamics. The use of renin-angiotensin-blockers preserves kidney activity and anatomy which leads to the production of diabetic nephropathy. Blockage of the renin-angiotensin - aldosterone pathway by raising its TGF-β1 activation antagonizes the profibrotic activity of Angiotensin II [16]. The analysis of an animal model of diabetic nephropathy further confirms the idea that diabetic nephropathy underlies such profibrotic effects [17]. Proteinuria and strengthened glomerular structures were decreased by temporary (7 weeks) blockage of the renin – angiotensin network in prediabetic rats. In fact, an regulator of angiotensin-converting enzymes is decreased in TGF-β1 serum rates in patients with type-1 diabetes and nephropathy. Shows there is a link between decreased serum-and-urine TGF-β1 rates and the renewal as defined by glomerular filtration rate shifts.
4. Reninangiotensin system in nephropathy
The renin-angiotensin aldosterone system is a hormonal system in the organism that is necessary to control blood pressure and fluid balance (RAAS). The main components of the system are three renin, angiotensin II and aldosterone hormones. The rate of renal blood flow is mostly controlled. Diabetes has been studied intensively. Previous research based on systematic RAS including information and findings were contradictory, motivating, restricting and the system was notified of almost any changes [18]. The controlling variables of systemic RAS as well as the multiple disease stages, many of these divergent observations may be clarified by organisms examined. For various diabetic types, the renal renin content is increased. This was commonly observed, however, compared to plasma renin levels. Suggested release into circulation of damaged rennin, Plasma rennin is reduced. Clinical diabetic nephropathy acivity is attributable to nonenzymatic prorenin glycation with a lower renin transfer. Usually, nephropathy was known as a condition of low renin. However, the behavior of plasma renin does not necessarily reflect the operation of Kidney RAS. The challenge of precise calculation became a further issue, Angiotensin-II (Ang II) plasma, which is important because there could be discordance between the levels of plasma renin and Ang II. More details a big research recently centered on intrarenal RAS. There is ample evidence that local tissue RASs that are regulated are plasma-independent RAS. The glomerular Ang II receptor decrease in the diabetic rat was recorded 3 to 4 weeks after disease induction. Glomerular down regulation Ang II receptors suggest that intra-renal Ang II can be Upgraded. Ang II receptor density in the next tubules has been reduced and supplemented by diabetic rats shows Lowered AT1 receptor and mRNA expression [19]. Recently, AT1 It's been shown that mesangial receptor density has dropped Incubation of the cells into high-glucose media [20].
The biochemical impact of angiotensin II Members by means of different cell-surface receptors In the receptor family 7-transmembrane.The receptor angiotensin can be divided Type 1 (AT1) and 2 pharmacological classes Based on its differential affinities type 2 (AT2) Different antagonists of nonpeptides as ststed above. Learning outcomes With the use of these antagonists, most suggested RAS functions that are classically recognized AT1 receptors are mediated [21]. In contrast The AT2 receptor, however, appears to be working as an AT1 receptor actions detrimental modulator [22-24].
5. Interconnection of Nitric oxide and Reactive oxygen species with RAAS
NO(Nitric oxide), initially referred to as a "endothelial relaxing factor," is a signaling molecule that controls an omnipresent use Large variety of organ and cell roles, like renal hemodynamics, reulatory water and salt. NO is produced, L-arginine amino acid by one of three enzymatically Nitric synthesis: neuronal (NOS1 or nOS) nitric oxide synthesis, Endothelial or 'inducible' (NOS3 or eNOS), (NOS2 or iNOS). Numerous, though not all, intracellular trajectories. The activation of guanylate cyclase is mediated by NO, This raises cyclic guanosine intracellular levels Monophosphate. The mammalian kidney contains all three NOS isoforms the distribution patterns are both distinct and overlapping. Typically a NOS1 in the macula densa and glomerular parietal epithelium is highly expressed both the collection ducts and thin upward limb in the medulla. NOS 2 is found in glomerular endothelia capillaries and afferent and efferent, renal and descending vasa recta arteries, including in proximal tubules and medullary thick ascending limb. In addition to aucurate arteries and vase-recta bundles nos3 is also expressed in tubules, including S3 proximal tubules segments, medullary thick ascending limb, and collecting pipes [25]. The result was conflicting both in vivo and in vitro studies regarding NOS expression and NO diabetes production. In response to hyperglycemia, most, but not all, experiments in cultured cells find a decrease of NO output.
Angiotensin II has direct implications for the kidney as glomerular capillary pressure increases and increase-regulation of the renal wound response components increases. The early study has shown that, in response to high-volume glomerular damage, endothelial cell injury triggers glomerulosclerosis, and involves local up-regularization of angiotensin II synthesis with enhanced expression of TGF-b1 and matrix proteins [26]. The entothelium is synthesized to produce a constituent precursor of nitric oxide (NOS), nitric oxide is an endogenous vasodilator that prevents the development of the vascular smooth muscles and mesangial cell development. Nitric oxide antagonizes the vascular sound and development effects of angiotensin II and therefore lowers the angiotensin converting enzymes (ACE) and angiotensin II of type 1 (AT1).Increased compressive force and cyclic strain increase the NOS activity in endothelial cells, which may be involved in early phases of diabetic nephropathy: high expression of vascular activity of NOS is a homeostatic adaptation to enhanced hemodynamic workloads that can reduce organ failure. Through time, though, the diabetic nephropathy has evidently raising its bioactivity by nitric oxide, A hypertensive design of rat nitric oxide depletion showed that renal lesions were dramatically decreased after treatment with an AT1 receptor blocking agent (ARB) [27]. Throughout this research, the positive benefits of an ARB were independent of blood pressure control as, given a significant reduction throughout systolic blood pressure, hydralazine appeared to enhance renal function. This supports the idea of much more effective anti-hypertensive agents to prevent or avoid renal disease that decreases blood pressure and thus preserve the homeostatic equilibrium of angiotensin II and nitric Oxide in the vessel wall [28, 29]. A functional imbalance of angiotensin II and oxide in hypertensive organ injuries has a major pathogenic role: angiotensin II lowers nitric oxide bio-availability by encouraging oxidative stress. In the translation of molecules that promote the pathogens of mesangial and vascular smooth cell pathology, excess production of superoxide anion in reaction to angiotensin II is of central importance.
Blocking recurrent glomerular and tubulointerstitial damage from RAAS clinically and experimentally [30, 31]. In patients suffering from type1 diabetes, the ACE inhibitors displayed slow down renal decrease, while ARBs exhibited major benefits for both an early stage (microalbuminuria) and at a later stage (proteinuria) of renal deterioration in patients with type 2 diabetes. ARB treatment in the Irbesartan Diabetic Nephropathy in the angiotensin II – Losartan Study (RENAAL) were correlated with substantial proteinuria decreases, independently of blood pressure reduction. The ARB treatments were identified as having a negative effect on blood pressure. In REENAAL, Proteinuria was an important indicator of renal and cardiovascular disease in Occurrences [32]. ARBs was close to ACE Inhibitors when providing for long-term Reno protection in type 2 diabetic patients and initial diabetic nephropathy [33]. Such reports highlight the significance of RAAS blockades as a method to enhance the health performance of renal disease patients.
6. Endothelin-1 (ET-1)
Endothelin-1 (ET- 1) is an extremely active, hypertension-inflammatory, vasoconstrictive peptide [34, 35]. The activity of ET-1 increases with a higher risk of renal disease in diabetes [36]. ET-1 promotes proteinuria and is active in the inflammatory and fibrotic processes of the renal cells [37]. As predicted, in laboratory models of diabetic nephropathy, selected ET-1A receptor blockers were seen to lower both blood pressure and albuminuria [38, 39]. Although, disappointingly, after 4 months of median treatments for elevated cardiovascular potential complications in the avosentan group like (edema, heart failure, cardiovascular death) [40]. The Elevate research work examining the ET antagonist avosentan in conjunction with RAS blockade was reinstated prematurely. However, albuminuria with avosentan has decreased by 40 percent compared to placebo. Related decreases in albuminuria, with moderate edema being the most severe adverse effect, have been found in a smaller sample in which another ET antagonist, atrasenten, was applied to a form 2 RAAS blockade for diabetic nephropathy people [41]. The loss of 50:1 selectivity for the ET1A Receptor with a higher doses of avosentan (> 5 mg daily), resulting in sodium and fluid retention by antagonism to the ET1B receptor, may explain the differences in adverse effects between avosentan and atrasentan [42]. The risk of edema end heart failure in the Evolve study is supposed to be higher in this unwanted effect. In comparison, atrasentan gives a strong degree of ET-1A absorbing selectivity, which contributes to corresponding experiments with a better protection profile [43]. An initial randomized controlled atrasentan clinical trial consisted of 211 adults who received placebo or atrassentan (0.75 mg/1.25 mg) with T2D and DKD on maximum RAAS blocks [44]. Atrasentan-treated patients saw a 35% drop in albuminuria, with around 50 % of patients reporting an albuminuria drop of about 30%. During the 12-week course of the study there's no substantial shift in eGFR. The weight gain and anemia in the atrasentanic groups were indicated, but the edema or heart disease was not greatly affected. Evidence of the more recent analysis of diabetic nephropathy With Atrasentan (SONAR) atrasentan treatment has shown a decrease in the incidence of a standardized renal endpoint versus placebo in more than 2,500 adults with T2D Atrasentan (6.0 percent) vs. placebo (6.9 percent) (doubling of serum creatinine or end-stage kidney disease) [45]. This research was performed in a patient population that was specifically chosen. Despite that, An preliminary 4-week 'enrichment' duration with all atrasentanic patients was subject to randomization – approximately 50 percent with patients were 'respondents' as shown in the earlier study, and only people who had a 30 percent albuminuria change despite fluid retention were randomised. In order not to hit the key endpoint in the pre-specified time period, the SONAR experiment was terminated early, although this was predicted to occur in providing lower event rates overall. If such positive findings can contribute to more therapeutic advancement remains to be seen.
7. Intracellular signaling
NF-KB
In the pathogenesis of diabetic naphropathy NFkB is the most significant transcription factor. One of the relevance of NFkB is to continually be found in cells, even in an inactive state, in regulation of cellular responses. Activating pathways downstream of NFkB does not therefore need a new protein synthesis, allowing NFkB to be a primary respondent to damaging cell stimuli. NFkB dimers are sequestered by a class of inhibitors in cells that are not activated, known asIkBs - NFkb dimers. The linkage of IkBs to NFkB precludes translocation to the nucleus and keeps NFkB inactive. The released NFkB can be entered and the expression NFkB-dependent can be activated after IμB degradation.
A class of inhibitors called IkBs (NFkB Inhibitors) is sequestered in non-stimulated cells. The binding of the IkBs to NFkB impedes its translocation into the nucleus and keeps NFkB inactive in the cytoplasm. Activation of NFκB is initiated by degradation of IκBs in response to extracellular signals, primarily via activation of IκB kinase. The liberated NFkB can reach the nucleus and stimulate NFkB-dependent genes expression following IkB breakdown.
In diabetic nephropathy patients, NFkB is a key part of the interplay between metabolic and hemodynamic variables. In endothelial cells, vascular smooth muscle cells and proximal tubular cells, hyperglycemia can itself activate NFkB. 108,109 Mechanical strain in human mesangial cells leads to single-cyte chemotaxis through a mechanism which depends on NFkB and CCL2. In addition, increasing data suggest that NFkB is important to several linked pathways, including the activation of the renin angiotensin systems, the buildup of AGE and NADPH-related stress, that contribute to the structural and functional alterations found in diabetic nephropathy.
The activation of NFκB and the simultaneous upregulation of NFκB-dependent proinflammatory genes has been demonstrated in biopsy specimens from patients with T2DM and overt nephropathy. NFkB activities are mostly observed in the cortical tubular epithelium cells, and are, to a reduced extent, related to proteinuria and interstitial infiltration in certain glomerular cells (particularly podo cytes).
The importance of NFkb to diabetic nephropathy development has been reaffirmed in the last several years. In vivo investigations, first, it has been established that the evo-flooding of diabetically nephropathically experienced temporary alterations in NFkB activity, and the NFkB appears to be a significant mediator of the angiotensin II effects, predestinally via the angioabluence type-1 receptor II.
Finally, a study of human kidney biopsy samples collected for the European Renal Complementary DNA Bank identified an ‘inflammatory signature’ relating to activation of the NFκB pathway in patients with progressive diabetic nephropathy.A specific NFκB promoter module in conjunction with interferon regulatory factor complexes, termed ‘NFκB_IRFF_01’, is thought to act as a master switch to activate this response.
8. Growth factor and cytokines
TGF beta and VEGF
Several growth factors, cytokines, chemokines and vasoactive agents have been implicated in pathogenesis of diabetic nephropathy. TGF-β, a fibrotic cytokine, plays a central role in the development of renal hypertrophy and accumulation of ECM componenets [46]. In addition, there is increased infilteration of monocytes and macrophages into glomeuli early in diabetes. The release of growth factors and cytokines from these monocytes and macrophages (interlukin-8, monocyte chemotactic peptide-1 etc.) may contribute to promotion of glomerular growth. There is increasing evidence that intrarenal renin-angiotensin system is activated in diabetic nephropathy [47]. There is enhanced expression of Ang II receptors and deceased degradation of Ang II thereby increasing the local effects of Ang II [48] which acts in synergy with hyperglycemia in Free radicals promoting, renal hypertrophy and ECM protein production. The vascular endothelial growth factor (VEGF), platform-derived growth factor (PDGF), connective tissue growth factor (CTGF), and insulin-style growth factor are other variables which contribute to the development of diabetes nephropathy (IGF) [49].
VEGF in nephropathic diabetic Vascular Endothelial Growth Factor (VEGF) is a promising option for endothelial diabetes mediation. VEGF in the renal is generated by glomerular epithelial cells under healthy settings, while mesangial and tube epithelial cells are usually not producing that growth factor. It was demonstrated that during hyperglycemia, overexpression of VEGF occurs through PKC activation [50]. Further, TGF-β1 which is over expressed in kidney also enhances VEGF expression [51]. Moreover, glomerular permeabilization by VEGF might induce both albuminuria and increased mesangial traffic of growth factors from the circulating blood. Hyperglycaemia increases VEGF excretion in the mesangial cell and podocyte via pathways involving PKC and extracellular signal regulated kinase (ERK) [52]. Receptors for VEGF in the glomerulus are found in the endothelial cells and it is thought that this growth factor increases the permeability of the glomerular endothelium and is therefore responsible for the hyperfiltration seen in early diabetic nephropathy. The excision of VEGF in the mesangial cell was also enhanced with mechanical strain imitating the sheer stress generated by hyperfiltration and elevated glomerular pressure. In a study demonstrating this effect it seemed that the effects of shear stress in mesangial cells are mediated via a pathway dependent on PKC and Protein Tyrosine Kinase (PTK) since the combined inhibition of these enzymes completely prevented the increased VEGF excretion in an in vitro experiment [53]. However, MC can also produce VEGF [54] and express VEGF receptors both in vitro and in pathological conditions [55]. Furthermore, VEGF binding to its receptors on MC induces both cell proliferation and collagen expression, providing a possible mechanism by which VEGF may contribute to glomerular hypertrophy/sclerosis [56]. In addition, VEGF potentially stimulate eNOS expression and activity in endothelial cells [57]. TGF-β1 in Diabetic Nephropathy the TGF-β seems to play a central role as a mediator in the pathologic changes in the glomerulus. It has been shown that the AGE formation, PKC activation, angiotensin II, and shear stress increase TGF-β expression [58]. TGF-β is a potent growth factor promoting the deposition of ECM components, such as collagen I, IV and fibronectin. This leads to, the histologically evident glomerular expansion and thickening of the basement membrane. The effects of TGF-β are mediated by the TGF-β receptor type II while the Smad pathway is the downstream intracellular signaling pathway involved in TGF-β signaling [59]. This cytokine play a central role in the development of renal hypertrophy and accumulation of ECM components in diabetes [60]. During hyperglycemia, mesangial and proximal tubular cells synthesise more TGF-β than control. In addition, it has been demonstrated that intracellular glucosamine production resulting from glucose metabolism is responsible for the increased TGF-β1 production in mesangial cells. Several vasoactive factors such as AngII, thromboxane [61] & endithelin-1 [62] may exert part of their growth stimulating and profibrogenic action in diabetic renal diseases to the secondary induction of TGF-β. Furthermore non-enzymatic glycation reactions leading to AGE [63], as well as the early Amatori glucose adducts in proteins such as serum albumin have [64] been shown to stimulate renal expression of TGF-β [65]. Amadori glucose adducts in albumin also increase expression of TGF-β type II receptors m-RNA and protein levels in mesangial cells.
Renal syndromes of nephrpathy
Tubulopathies
Renal Tubular Acidosis/Fanconi Syndrome
Sodium Wasting
Potassium Wasting
Nephrogenic Diabetes Insipidus
Nephrotic Syndrome/Proteinuria
Glomerular Disease
Minimal Change Glomerulonephritis
Focal Segmental Glomerulosclerosis
Membranous Glomerulonephritis
Other
Thrombotic Microangiopathy
Hemolytic uremic syndrome/ Thrombotic thrombocytopenic
purpura
Acute Kidney Injury
Hemodynamic Disturbances
Parenchymal Kidney Disease
Collecting System Disease
Chronic Kidney Disease
Analgesic Nephropathy
Chronic Tubulointerstitial Nephritis
Secondary Progression of Toxin-induced
Kidney Disease
9. Conclusion
Complex relationships between hemodynamic and metabolic routes that contribute to diabetic nephropathy. Although proven therapy used in the appearance of symptoms include strict hyperglycemia regulation and the use of the rennin angiotensin system inhibitors, various metabolic pathway such as advanced glycation and particular protein kinase C isoforms provide essential hope for the future65. A mixture of therapies is expected which directly impact various phases in diabetic nephropathy pathophysiology. The development will need to be more minimized and diabetic nephropathy stopped in future.
Funding
NA
Conflicts of interest
All authors declare no conflict of interest to disclose.
Ethics statement
This study doesn't involve animals or human subjects.
References
- Cooper ME. Interaction of metabolic and haemodynamic factors in mediating experimental diabetic nephropathy. Diabetologia 44 (2001): 1957–1972.
- Hargrove GM, Dufresne J, Whiteside C, Muruve DA & Wong NCW. Diabetes mellitus increases endothelin-1 gene transcription in rat kidney. Kidney Int 58 (2000): 1534–1545.
- Barazi M, Kaur H & Prabhakar S. Diabetic Glomerulopathy. in An Update on Glomerulopathies - Clinical and Treatment Aspects (ed. Prabhakar, S.) (2011).
- Sharma Prabhakar. An Update on Glomerulopathies: Clinical and Treatment Aspects. intechopen (2011).
- Gaede P, Lund-Andersen H, Parving H-H & Pedersen O. Effect of a multifactorial intervention on mortality in type 2 diabetes. N. Engl. J. Med 358 (2008): 580–591.
- Gaede P. et al. Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N. Engl. J. Med 348 (2003): 383–393.
- Gaede P, Vedel P, Parving HH & Pedersen O. Intensified multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: the Steno type 2 randomised study. Lancet Lond. Engl 353 (1999): 617–622.
- Rivero A, et al. Pathogenic perspectives for the role of inflammation in diabetic nephropathy. Clin. Sci. Lond. Engl. 1979 116 (2009): 479–492.
- Navarro-González JF, Mora-Fernández C, Muros de Fuentes M & García-Pérez J. Inflammatory molecules and pathways in the pathogenesis of diabetic nephropathy. Nat. Rev. Nephrol 7 (2011): 327–340.
- UK Prospective Diabetes Study (UKPDS). IX: Relationships of urinary albumin and N-acetylglucosaminidase to glycaemia and hypertension at diagnosis of type 2 (non-insulin-dependent) diabetes mellitus and after 3 months diet therapy. Diabetologia 36 (1993): 835–842.
- Ritz E & Orth SR. Nephropathy in patients with type 2 diabetes mellitus. N. Engl. J. Med 341 (1999): 1127–1133.
- Gall MA, Borch-Johnsen K, Hougaard P, Nielsen FS & Parving HH. Albuminuria and poor glycemic control predict mortality in NIDDM. Diabetes 44 (1995): 1303–1309.
- Tagle R, Acevedo M & Vidt DG. Microalbuminuria: is it a valid predictor of cardiovascular risk? Cleve. Clin. J. Med 70 (2003): 255–261.
- Ziyadeh FN & Wolf G. Pathogenesis of the podocytopathy and proteinuria in diabetic glomerulopathy. Curr. Diabetes Rev 4 (2008): 39–45.
- Wolf G & Ziyadeh FN. Cellular and molecular mechanisms of proteinuria in diabetic nephropathy. Nephron Physiol 106 (2007): p26-31.
- Prabhakar S. An Update on Glomerulopathies: Clinical and Treatment Aspects. (BoD – Books on Demand) (2011).
- Nagai Y, et al. Temporary Angiotensin II Blockade at the Prediabetic Stage Attenuates the Development of Renal Injury in Type 2 Diabetic Rats. J. Am. Soc. Nephrol. JASN 16 (2005): 703–711.
- Wolf G & Ziyadeh FN. The role of angiotensin II in diabetic nephropathy: emphasis on nonhemodynamic mechanisms. Am. J. Kidney Dis. Off. J. Natl. Kidney Found 29 (1997): 153–163.
- Cheng HF, Burns KD & Harris RC. Reduced proximal tubule angiotensin II receptor expression in streptozotocin-induced diabetes mellitus. Kidney Int 46 (1994): 1603–1610.
- Amiri F & Garcia R. Regulation of angiotensin II receptors and PKC isoforms by glucose in rat mesangial cells. Am. J. Physiol 276 (1999): F691-699.
- Timmermans PB, et al. Angiotensin II receptors and angiotensin II receptor antagonists. Pharmacol. Rev 45 (1993): 205–251.
- Hein L, et al. Overexpression of angiotensin AT1 receptor transgene in the mouse myocardium produces a lethal phenotype associated with myocyte hyperplasia and heart block. Proc. Natl. Acad. Sci USA 94 (1997): 6391–6396.
- Ichiki T, et al. Effects on blood pressure and exploratory behaviour of mice lacking angiotensin II type-2 receptor. Nature 377 (1995): 748–750.
- Masaki H. et al. Cardiac-specific overexpression of angiotensin II AT2 receptor causes attenuated response to AT1 receptor-mediated pressor and chronotropic effects. J. Clin. Invest 101 (1998): 527–535.
- Burdeina EV, Nepomnyashchy VN & Kolesnyk NA. THE COMBINATION OF KARTAGENER SYNDROME WITH FOCAL SEGMENTAL GLOMERULOSCLEROSIS: CASE REPORT. Ukr. J. Nephrol. Dial (2015): 64–68.
- Lee LK, Meyer TW, Pollock AS & Lovett DH. Endothelial cell injury initiates glomerular sclerosis in the rat remnant kidney. J. Clin. Invest 96 (1995): 953–964.
- Boffa J-J, et al. Regression of renal vascular and glomerular fibrosis: role of angiotensin II receptor antagonism and matrix metalloproteinases. J. Am. Soc. Nephrol JASN 14 (2003): 1132–1144.
- Zhou M-S, Schulman IH & Raij L. Nitric oxide, angiotensin II, and hypertension. Semin. Nephrol 24 (2004): 366–378.
- Bataineh A & Raij L. Angiotensin II, nitric oxide, and end-organ damage in hypertension. Kidney Int. Suppl. 68 (1998): S14-19.
- Remuzzi A, Fassi A, Bertani T, Perico N & Remuzzi G. ACE inhibition induces regression of proteinuria and halts progression of renal damage in a genetic model of progressive nephropathy. Am. J. Kidney Dis. Off. J. Natl Kidney Found 34 (1999): 626–632.
- Ikoma M, Kawamura T, Kakinuma Y, Fogo A & Ichikawa I. Cause of variable therapeutic efficiency of angiotensin converting enzyme inhibitor on glomerular lesions. Kidney Int 40 (1991): 195–202.
- Raij L. Recommendations for the management of special populations: renal disease in diabetes. Am. J. Hypertens 16 (2003): 46S-49S.
- Barnett AH, et al. Angiotensin-receptor blockade versus converting-enzyme inhibition in type 2 diabetes and nephropathy. N. Engl. J. Med 351 (2004): 1952–1961.
- Yanagisawa M, et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 332 (1988): 411–415.
- Hocher B, et al. Endothelin-1 transgenic mice develop glomerulosclerosis, interstitial fibrosis, and renal cysts but not hypertension. J. Clin. Invest 99 (1997): 1380–1389.
- Shin SJ, Lee YJ & Tsai JH. The correlation of plasma and urine endothelin-1 with the severity of nephropathy in Chinese patients with type 2 diabetes. Scand. J. Clin. Lab. Invest 56 (1996): 571–576.
- Simonson MS & Ismail-Beigi F. Endothelin-1 increases collagen accumulation in renal mesangial cells by stimulating a chemokine and cytokine autocrine signaling loop. J. Biol. Chem 286 (2011): 11003–11008.
- Krum H, Viskoper RJ, Lacourciere Y, Budde M & Charlon V. The effect of an endothelin-receptor antagonist, bosentan, on blood pressure in patients with essential hypertension. Bosentan Hypertension Investigators. N. Engl. J Med 338 (1998): 784–790.
- Boels MGS, et al. Atrasentan Reduces Albuminuria by Restoring the Glomerular Endothelial Glycocalyx Barrier in Diabetic Nephropathy. Diabetes 65 (2016): 2429–2439.
- Mann JFE, et al. Avosentan for overt diabetic nephropathy. J. Am. Soc. Nephrol. JASN 21 (2010): 527–535.
- Kohan DE, et al. Addition of atrasentan to renin-angiotensin system blockade reduces albuminuria in diabetic nephropathy. J. Am. Soc. Nephrol. JASN 22 (2011): 763–772.
- Smolander J, et al. Dose-dependent acute and sustained renal effects of the endothelin receptor antagonist avosentan in healthy subjects. Clin. Pharmacol. Ther 85 (2009): 628–634.
- Opgenorth TJ, et al. Pharmacological characterization of A-127722: an orally active and highly potent ETA-selective receptor antagonist. J. Pharmacol. Exp. Ther 276 (1996): 473–481.
- The Endothelin Antagonist Atrasentan Lowers Residual Albuminuria in Patients With Type 2 Diabetic Nephropathy - PubMed.
- Heerspink HJL, et al. Atrasentan and renal events in patients with type 2 diabetes and chronic kidney disease (SONAR): a double-blind, randomised, placebo-controlled trial. The Lancet 393 (2019): 1937–1947.
- López-Hernández FJ & López-Novoa JM. Role of TGF-β in chronic kidney disease: an integration of tubular, glomerular and vascular effects. Cell Tissue Res 347 (2012): 141–154.
- Urushihara M & Kobori H. Angiotensinogen Expression Is Enhanced in the Progression of Glomerular Disease. Int. J. Clin. Med 2 (2011): 378–387.
- Wysocki J, et al. Targeting the degradation of angiotensin II with recombinant angiotensin-converting enzyme 2: prevention of angiotensin II-dependent hypertension. Hypertens. Dallas Tex 1979 55 (2010): 90–98.
- Makary S, Abdo M, Hassan WA & Tawfik MK. Angiotensin blockade attenuates diabetic nephropathy in hypogonadal adult male rats. Can. J. Physiol. Pharmacol 97 (2019): 708–720.
- Gupta S, et al. Retinoprotective Effects of Moringa oleifera Via Antioxidant, Anti-Inflammatory, and Anti-Angiogenic Mechanisms in Streptozotocin-Induced Diabetic Rats. J. Ocul. Pharmacol. Ther. Off. J. Assoc. Ocul. Pharmacol. Ther 29 (2012).
- Li X, Hu J, Zhang Q, Sun X & Li S. Urocortin 1 improves renal function in rats with streptozotocin-induced diabetes by inhibiting overproduction of TGF-beta 1 and VEGF. Br. J. Pharmacol 157 (2009): 994–1003.
- Moriguchi S, Shioda N, Yamamoto Y & Fukunaga K. Platelet-activating factor-induced synaptic facilitation is associated with increased calcium/calmodulin-dependent protein kinase II, protein kinase C and extracellular signal-regulated kinase activities in the rat hippocampal CA1 region. Neuroscience 166 (2010): 1158–1166.
- de Oliveira S, Silva-Herdade AS & Saldanha C. Modulation of erythrocyte deformability by PKC activity. Clin. Hemorheol. Microcirc 39 (2008): 363–373.
- Shibuya M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes Cancer 2 (2011): 1097–1105.
- Noghero A, et al. Liver X receptor activation reduces angiogenesis by impairing lipid raft localization and signaling of vascular endothelial growth factor receptor-2. Arterioscler. Thromb. Vasc. Biol 32 (2012): 2280–2288.
- Sasaki M, et al. The macrophage is a key factor in renal injuries caused by glomerular hyperfiltration. Acta Med. Okayama 65 (2011): 81–89.
- Lee JH, Chun T, Park S-Y & Rho SB. Interferon regulatory factor-1 (IRF-1) regulates VEGF-induced angiogenesis in HUVECs. Biochim. Biophys. Acta 1783 (2008): 1654–1662.
- Mehta JL. The role of LOX-1, a novel lectin-like receptor for oxidized low density lipoprotein, in atherosclerosis. Can. J. Cardiol 20 (2004): 32B-36B.
- Chen Z, et al. The iron chelators Dp44mT and DFO inhibit TGF-β-induced epithelial-mesenchymal transition via up-regulation of N-Myc downstream-regulated gene 1 (NDRG1). J. Biol. Chem 287 (2012): 17016–17028.
- Mariappan MM. Signaling mechanisms in the regulation of renal matrix metabolism in diabetes. Exp. Diabetes Res (2012): 749812.
- Osaki T, et al. Metabolomic Analyses of Blood Plasma after Oral Administration of D-Glucosamine Hydrochloride to Dogs. Mar. Drugs 10 (2012): 1873–1882.
- Hu Z, Ren L, Wang C, Liu B & Song G. Effect of chenodeoxycholic acid on fibrosis, inflammation and oxidative stress in kidney in high-fructose-fed Wistar rats. Kidney Blood Press 36 (2012): 85–97.
- Jung DH, et al. Extract of Cassiae Semen and its major compound inhibit S100b-induced TGF-beta1 and fibronectin expression in mouse glomerular mesangial cells. Eur. J. Pharmacol 641 (2010): 7–14.
- Li Q, et al. Nuclear translocation of SMAD3 may enhance the TGF-beta/SMADS pathway in high glucose circumstances. Transplant 38 (2006): 2158–2160.
- Rayego-Mateos S, et al. Pathogenic Pathways and Therapeutic Approaches Targeting Inflammation in Diabetic Nephropathy 21 (2020): E3798.


 Impact Factor: * 3.0
Impact Factor: * 3.0 Acceptance Rate: 76.32%
Acceptance Rate: 76.32%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
