Negative Effect of Biventricular Pacing on Right Ventricular Function: A Case Report
Riccardo Maragna1,2, MD, Alessandra Scoccia1, MD, Fabrizio Tundo1, MD, Laura Fusini1, MD, Piergiuseppe Agostoni, MD, PhD1,3, Carlo Vignati1,3,*, MD.
1Centro Cardiologico Monzino, IRCCS, via C. Parea 4 Milan, Italy
2Cardiovascular Disease Fellowship Program, University of Milan, Italy
3Department of Clinical Sciences and Community Health – Cardiovascular Section, Università degli Studi di Milano, Milan, Italy
*Corresponding Author: Carlo Vignati, MD, Centro Cardiologico Monzino, IRCCS. Via Parea 4, 20138, Milan, Italy
Received: 21 April 2020; Accepted: 28 April 2020; Published: 22 September 2020
Article Information
Citation: Riccardo Maragna, Alessandra Scoccia, Fabrizio Tundo, Laura Fusini, Piergiuseppe Agostoni, Carlo Vignati. Negative Effect of Biventricular Pacing on Right Ventricular Function: A Case Report. Cardiology and Cardiovascular Medicine 4 (2020): 574-578.
View / Download Pdf Share at FacebookKeywords
<p>Biventricular pacing; Heart failure; Right ventricular function; Left ventricular assist device; QRS duration</p>
Article Details
1. Introduction
Cardiac resynchronization therapy (CRT) devices are commonly implanted for the treatment of heart failure refractory to pharmacological therapy. However, almost 30% of candidates continue to fail to respond (“non-responder”) or even worsen after implantation (“negative-responder”), and this is due to different mechanisms.
2. Case Report
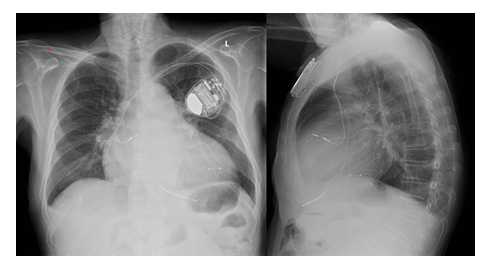
We present the case of a negative effect of biventricular pacing on right ventricular (RV) function. A 67-year-old man with heart failure (HF), left ventricular ejection fraction (LVEF) =35% due to ischaemic cardiomyopathy, in NYHA class II-III, with LBBB (QRS 160 msec) underwent cardiac resynchronization therapy (CRT-D bipolar LV lead positioned in lateral coronary vein branch, targeting a hypokinetic wall, Figure 1). At follow-up, several hospitalizations for recurrent episodes of HF were recorded, with a progressive worsening of right ventricular function.
Patients was transferred to our Unit in December 2014 with inotropic support (dobutamine infusion).
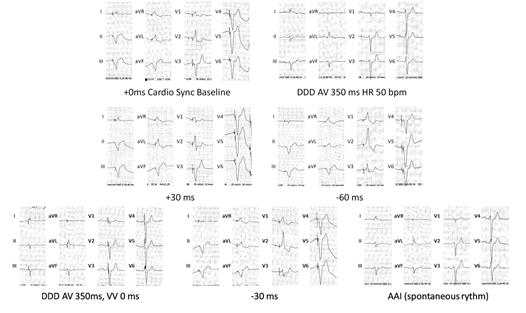
The ECG showed HR 59bpm with biventricular pacing (Figure 2).
Transthoracic echocardiography (TTE) showed severely dilated left atrial volume (110ml/mq), severely dilated and hypokinetic LV with apex akinesia, LVEF =15%. RV dysfunction (TAPSE 13mm, DTIs 6cm/s, shortening fraction 11%), with pulmonary hypertension (sPAP 59mmHg). The patient was considered not eligible for left ventricular assist device (LVAD) due to RV dysfunction.
CRT optimization was attempted, testing different programs of LV-RV interval (figure 2), without any QRS narrowing. We turned off the biventricular stimulation prolonging the AV delay to allow intrinsic conduction to ventricles, thus obtaining a slight narrowing of QRS to pre-existing LBBB morphology with a duration of 160msec. TTE showed no acute improvement of LV or RV function. The patient was dismissed with the following therapy: Bisoprolol 2.5mg BID, Furosemide 50mg BID, Spironolactone 100mg, Amiodarone 200mg, and Warfarin. One month later, the patient was readmitted to our hospital for clinical evaluation. TTE showed severely dilated atrial volume (102 ml/mq), severely dilated and hypokinetic LV with apex akinesia, LVEF = 13%. Increase of filling pressure (E/e’=8, restrictive pattern, pulmonary diastolic gradient 21mmHg). RV with normal ejection fraction (TAPSE=18mm, DTIs 9cm/s), shortening fraction (35%) and normal Myocardial Performance Index (0.25-0.3). sPAP 55mmHg.
Therefore, following RV recovery, the patient underwent successful LVAD implantation. Five years later, the patient is in NYHA class II, in good clinical conditions.
3. Discussion
According to the guidelines of European Society of Cardiology, CRT is recommended for symptomatic patients with QRS duration ≥150 msec and LBBB QRS morphology and with LVEF ≤35% despite optimal medical therapy. Positive clinical effects of CRT are mediated by a significant reduction of LV volumes (reverse remodeling), improvement of prognosis and quality of life [1].
When the patient underwent CRT was in II - III NYHA class, in sinus rhythm with reduced EF (35%), with QRS duration of 160 msec and LBBB morphology (despite not fulfilling all the Strauss criteria [2]).
However, over two decades after the introduction of CRT, ∼30% of candidates continue to fail to respond (“non-responder”) or even worsen after implantation (“negative-responder”) [3, 4]. Main causes may be related to the wrong selection of the patient (pacing an underlying narrow QRS; RV severe dysfunction), anatomo-functional characteristics (lacking of coronary venous branches in the pacing target area with suboptimal LV pacing; wide fibrotic scar in the site of LV pacing with delayed conduction), or pacing programming issues (biventricular pacing < 90%; suboptimal A-V or V-V timing). Moreover, several retrospective studies have postulated harmful effect of CRT on the RV and on the tricuspid valve. In particular, insertion of a lead across the tricuspid valve can be complicated by perforation of valve leaflets, interference with papillary muscle function and adhesion of valve tip to the device due to fibrous tissue 5. All these mechanisms can develop, in a subset of patients with remodeled RV, a mechanical increase in tricuspid regurgitant volumes with consequent further RV dilatation, and haemodynamic deterioration [ 4, 5]. In our case the RV function worsened after upgrading form ICD to CRT-D and then recovered to normal parameters after switching off the biventricular stimulation, suggesting a specific detrimental effect of CRT on RV function, regardless the presence of the RV lead. Following the recovery of RV function, persisting poor LV function, we were able to implant a LVAD, obtaining a significant clinical improvement.
The mechanism underlying this response is unclear. The widening of QRS duration with biventricular stimulation is known to be associated to poor response or worsening in desynchronization of LV, but is uncommon to observe worsening of RV function. A recent study demonstrated that improving LV function can reduce RV function; specifically, LV and the RV might respond in an opposite manner to different programs of LV-RV interval and that LV pre-excitation improved LV contractility but decreased RV contractility, while RV pre-excitation might improve RV function but decrease LV contractility [6]. We hypothesized a negative effect on RV function induced by CRT increasing either interventricular dyssynchrony or secondary to rise in pulmonary arterial pressure induced by CRT, underlining the importance of RV afterload sensitivity and of LV contribution to RV function.
4. Conclusions
This anecdotal case shows, for the first time, an acute worsening of right ventricular function due to left ventricular pacing and subsequent recovery after CRT turn-off in a heart failure patient, underlining the importance of right afterload sensitivity and of left ventricular contribution to right ventricular function, the latter having a pivotal role in advanced heart failure therapy.
Conflicts of Interest:
Authors have no conflicts to disclose.
References
- Ponikowski P, Voors AA, Anker SD, et al. 2016 esc guidelines for the diagnosis and treatment of acute and chronic heart failure: The task force for the diagnosis and treatment of acute and chronic heart failure of the european society of cardiology (esc)developed with the special contribution of the heart failure association (hfa) of the esc. European heart journal 37 (2016): 2129-22002.
- Strauss D, Selvester R, Wagner G. Defining Left Bundle Branch Block in the Era of Cardiac Resynchronization Therapy. The American Journal of Cardiology (2011): 927-934.
- Daubert C, Behar N, Martins RP, et al. Avoiding non-responders to cardiac resynchronization therapy: a practical guide. European heart journal (2017): 1463-1472.
- Al-Bawardy R, Krishnaswamy A, Bhargava M, et al. Tricuspid regurgitation in patients with pacemakers and implantable cardiac defibrillators: a comprehensive review. Clinical Cardiology (2013): 249-254.
- Fazio S, Carlomagno G. The importance of tricuspid regurgitation and right ventricular overload in ICD/CRT recipients: beside the left, beyond the left. Pacing and clinical electrophysiology (2011): 1181-1184.
- Willemen E, Schreurs R, Huntjens PR, et al. The Left and Right Ventricles Respond Differently to Variation of Pacing Delays in Cardiac Resynchronization Therapy: A Combined Experimental- Computational Approach. Frontiers in Physiology 10 (2019): 17.


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 