Role of Katankateryadi Kwatha in Insulin Secretion and Restoration of Biochemical Changes in Streptozotocin-Nicotinamide Induced Diabetes Mellitus Type 2 in Rats
Sanjay Kumar1*, Minshu Prashant1, B. Ram1, K. N. Dwivedi1, Pramod Kumar Gautam2
1Department of Dravyaguna, IMS, Banaras Hindu University, Varanasi, India., 2Department of Biochemistry, AIIMS Delhi, India.
*Corresponding author:Sanjay Kumar, Department of Dravyaguna, IMS, Banaras Hindu University, Varanasi, India
Received: 17 July 2021; Accepted: 27 July 2021; Published: 05 August 2021
Article Information
Citation: Sanjay Kumar, Minshu Prashant, B. Ram, K. N. Dwivedi. Role of Katankateryadi Kwatha in Insulin Secretion and Restoration of Biochemical Changes in Streptozotocin-Nicotinamide Induced Diabetes Mellitus Type 2 in Rats. International Journal of Applied Biology and Pharmaceutical Technology12 (2021): 362-379.
View / Download Pdf Share at FacebookAbstract
Diabetes Mellitus Type 2 is a metabolic disorder characterized by hyperglycemia with major and minor complications. The major cause of diabetes mellitus type 2 is lack of insulin secretion or insulin resistance. Experimentally the diabetes mellitus type 2 is induced by several chemicals like Streptozotocin (STZ). The nitrosourea moiety of STZ is responsible for producing beta cell toxicity which leads to development of diabetes mellitus type 2 in rats. The experimental study includes the formulation of Katankateryadi Kwatha, characterization of Kwatha, acute oral toxicity study of Kwatha, OGTT and antidiabetic effect of Katankateryadi Kwatha in STZNicotinamide induced diabetes in rats. Diabetic group treated with 200 mg/kg (189.34 ± 3.89 mg/dL FBG). Post prandial blood glucose (PPBG) level in Diabetic group treated with 200 mg/kg of Katankateryadi Kwatha shows better reduction in PPBG 159.35 ± 6.29 mg/dL in comparison to diabetic rats treated with 100 mg/kg (180.34 ± 2.45 mg/dL). Rats treated with 200 mg/kg Katankateryadi Kwatha significantly reduced the SGOT and SGPT level (133.34±2.25 IU/L and 170.89±3.76 IU/L). The group treated with Katankateryadi Kwatha (200 mg/kg) treated group increased the HDL level (23.56 ± 5.23) and decreases the TC (112.56 ± 3.24), TG (71.65 ± 5.26), LDL (52.41 ± 2.11) and VLDL (21.05 ± 4.62). C-Peptide data result shows that C-Peptide decreased in diabetic rats 0.36 ± 0.034 ng/dl. Katankateryadi Kwatha is useful in management of diabetes mellitus type 2.
Keywords
<p>Diabetes Mellitus Type 2; Streptozotocin; Nicotinamide; OGTT; Katankateryadi Kwatha; Glibenclamide; Gliclazide; C-Peptide</p>
Article Details
1. Introduction
Diabetes Mellitus is a metabolic disorder characterized by hyperglycemia followed by polyurea, polyphagia and polydypsia with vascular complications. The diabetes mellitus is broadly categorized in four types – Diabetes Mellitus Type I, Diabetes mellitus Type II, Gestational Diabetes and disease associated Diabetes Mellitus. Majority of people now a day suffered from diabetes mellitus type II. It is a major health issue that has been reached at alarming level [1]. Today nearly half a billion people are living with diabetes mellitus worldwide. The estimated projection of diabetes mellitus is about 700 million people suffer from diabetes mellitus. Two-thirds of people with diabetes mellitus type II are live in urban areas and three out of four are of working age [2]. Over four million people aged 20–79 years are estimated to die from diabetes-related causes in 2019. The countries with the largest numbers of adults with diabetes aged 20–79 years in 2019 are in China, India and the United States of America, and are anticipated to increase till in 2030. India is home to the second largest number (77 million) of adult population with diabetes worldwide. With 1.2 million deaths in 2019 (14.1% of all-cause mortality), the South East Asia Region has the second highest number of deaths attributable to diabetes in adults 20–79 years among the IDF Regions [3]. Many influences that affect the prevalence of disease throughout a country and identification of those factors is necessary to facilitate change when facing health challenges. Today modern medicine develops so many drugs for management of diabetes mellitus type II. The common drawback of modern medicine is drug resistance because the drugs are used for life long period. To avoid such types of problems with modern medicines the world now a day look towards the herbal medicines which are safe in use for long period of time. The medicinal plants play a pivotal role in the health care of ancient and modern cultures [4]. The utility of medicinal plants in the management of diabetes mellitus type 2 are rising in the developing countries worldwide. There is enhancement in scientific evidences that supports the claim towards the traditional healers. The research on herbal medicines and their natural products have significant importance in demands and use in few decades and increasing the demand of herbal drugs worldwide. The available ancient literatures show that there are more than 400 plant species showing anti-diabetic activity. Although some of the plants have great reputation in Ayurveda, the indigenous Indian system of medicine, many remain to be scientifically established [5]. Streptozotocin (STZ) is an antibiotic derived from species Streptomyces achromogenes and structurally it is a glucosamine derivative of nitrosourea. STZ first reported the diabetogenic activity in rats in 1963. Like alloxan [6], it also causes hyperglycaemia by cytotoxic action on beta cells of the pancreas. The nitrosourea moiety of STZ is responsible for producing beta cell toxicity, while deoxyglucose moiety enhances the transport across the cell membrane. The free radicals generation and alteration in the endogenous scavengers of the free radicals have been reported in STZ diabetogenecity [9]. In comparative to synthetic medicines, Ayurvedic formulations are effective, less side effects, broad range of action and relatively low cost, makes polyherbal formulation as a good choice in management of diabetes type 2 [10]. So many polyherbal formulations are reported in ancient Ayurvedic literatures but due to lack of scientific evidences such formulations are still hidden. Katankateryadi Kwatha is a polyherbal formulation of total five ingredients. The aim of this study is to scientifically prove the Katankateryadi Kwatha as antidiabetic polyherbal formulation for public welfare.
2. Material and Methodology
2.1 Experimental Animal Requirements
Albino rats (Charles foster strain of weight 150-200 g male and female) purchased from registered animal house of the IMS BHU. The proposal was approved from Central Animal Ethical Committee, Reg. No. 542/GO/ReBi/S/02/CPCSEA. Approval letter no is DEAN/2019/IAEC/1242 dated 26.05.2019. The rats were kept in polypropylene cages containing paddy husk bedding. The cages were stored inside a ventilated room of animal house in Department of Dravyaguna, IMS, BHU Varanasi. Each cage contains 6 rats and standard condition was maintained (temperature 20-30oC, humidity 65-70% and 12 hrs light & dark cycle). Rats were feed with standard pellet diet (Ashirwad trade,) and water.
2.2 Chemicals Requirements
Streptozotocin (STZ) Sigma, Nicotinamide (NAD) SRL, SGPT kit SRL, SGOT kit SRL, HDL kit Himedia, Total cholesterol kit Himedia, C-Peptidase kit Himedia, LDL kit, Katankateryadi Kwatha, Glibenclamide Himedia, Gliclazide Himedia.
2.3 Herbal crude drug requirement
Barberis aristata DC (Stem), Glycyrrhiza glabra Linn (Stem), Terminalia chebula Tetz(Fruit), Terminalia bellirica Roxb (Fruit), Emblica officinalis Gaertin (Fruit) and Plumbago zeylanica Linn (Root). All crude drugs was authenticated from Department of Dravyaguna, Faculty of Ayurveda IMS, BHU, Varanasi (Reference No: DG/21-22/313-318).
2.4 Preparation of decoction of Katankateryadi Kwatha
Katankatareyadi Kwatha (decoction) was prepared using six drugs i.e. Barberis aristata DC (Stem) [11], Glycyrrhiza glabra Linn (Stem) [12], Terminalia chebula Tetz (Fruit) [13], Terminalia bellirica Roxb (Fruit) [14], Emblica officinalis Gaertin (Fruit) [15] and Plumbago zeylanica Linn (Root) [16]. All the ingredients of Katankateryadi Kwatha were subjected for size reduction using the pulvarizer. Equal amount of all crude drugs was soaked in 4 times water in vessel and kept overnight for 12 hrs. After 12 hrs contents were boiled at 900 C – 950 C with stirring. Water was evaporated till 1/4th amount was remains and galenicals was filtered through cotton cloth. Filtrate was dried with rotatory evaporator and dried powder was used for their quality control standard test and Physico-chemical analysis [17].
2.5 Physicochemical evaluation of Katankateryadi Kwatha (Decoction)
Prepared Decoction of Katankateryadi Kwatha and dried powder of decoction was evaluated for their organoleptic properties, microbial contaminations, presence of heavy metals and presence of pesticide residue [18].
2.6 Phytochemical analysis of Katankateryadi Kwatha (Decoction)
The Phytochemical screening of different extracts of crude drugs and Kvatha (decoction) were performed according to the procedure mentioned in Ayurvedic Pharmacopoeia of India (API) [19].
2.7 Acute Oral Toxicity Study
Acute oral toxicity was performed as per OECD guidelines 423 for toxicity study and estimation of LD50 dose after treatment with Katankateryadi Kwatha at different doses within 24 hours for dose optimization. Rats were distributed into total four groups and each group contains 5 rats. Group 1 treated with Vidangadi Kwatha 5 mg/kg po, Group 2 treated with Vidangadi Kwatha 50 mg/kg po, Group 3 treated with Vidangadi Kwatha 300 mg/kg po, Group 4 treated with Vidangadi Kwatha 2000 mg/kg po. The rate of mortality and following parameters (Tremor, clonic convulsion, tonic convulsion, straub tail erection, catatonia, ataxia, loss of lighting reflex, sedation, hypnosis, lacrimation, diarrhea, writhing and rate of respiration) were observed for 24 hours [20].
2.8 Oral Glucose Tolerance Test (OGTT) in normal and diabetic Rats
The OGTT in normal rats was performed in overnight (18-h) fasted rats. Rats were randomly divided into following groups and each group contains 6 rats: Group 1: received only saline. Group 2 and 3: treated with Katankateryadi Kwatha 100 mg/kg po and 200 mg/kg po. Group 4: treated with Glibenclamide 600 µg/kg po. Similarly the OGTT in diabetic rats was performed and the rats were randomly divided into following groups: Group 1: normal control received only saline. Group 2: diabetic control received only saline. Group 3 and 4: treated with Katankateryadi Kwatha 100 and 200 mg/kg po. Group 5: treated with Glibenclamide 600 µg/kg po. Glucose (2 g/kg) was fed 30 min after the administration of the drugs. Blood sample was withdrawn at each 30, 60, and 120 min after glucose administration and blood glucose was estimated [21].
2.9 Effects of Katankateryadi Kwatha in STZ-NAD induced Diabetic Rats
2.9.1 Induction of diabetes and treatment
Streptozotocin (STZ) was freshly prepared in citrate buffer (pH 4.5) and Nicotinamide was prepared in normal saline. Hyperglycemia was confirmed by the elevated random blood glucose levels (>200 mg/dL) [22]. Diabetes mellitus type 2 in rats was induced administration of Nicotinamide 120 mg/kg ip followed by administration of 60 mg/kg Streptozotocin (Sigma, Germany) after 15 minute. The rats with fasting plasma glucose >126 mg/dL & post prandial blood glucose level > 200 mg/dL were used in the study. The rats were randomly grouped in to following groups and each group contains 6 rats: Group 1: normal control received normal diet and water. Group 2: per-see study treated with Katankateryadi Kwatha100 mg/kg po. Group 3: Diabetic control received normal diet and water. Group 4 and 5: Diabetic rats treated with Katankateryadi Kwatha100 and 200 mg/kg po. Group 6: Diabetic rats treated with gliclazide 4 mg/kg po. Biochemical analysis: During 15 days treatment protocol fasting and post prandial blood glucose was estimated and after 15 days blood were collected and allowed to clot formation. The blood serum was isolated by centrifugation technique at 2500 rpm for 15 min and analyzed for various biochemical parameters -SGOT, SGPT, HDL, LDL, VLDL, Total cholesterol, Triglycerides and C-Peptide level were measured [23].
3. Results
3.1 Physicochemical and Phytochemical evaluation of Katankateryadi Kwatha
Dried Katankateryadi Kwatha powder is dark brown in color, astringent-sweet taste and pungent smell. Heavy metals (Arsenic, Lead, cadmium and mercury) [24], microbial contamination [25] and pesticide residue [26] was absent in Katankateryadi Kwatha. Phytochemical screening of Katankateryadi Kwatha was performed and found that carbohydrate, flavonoids, alkaloids, proteins, tannins saponins and amino acids was present and volatile oils are absent in Katankateryadi Kwatha.
3.2 Acute oral toxicity study
The Acute oral toxicity shows that there was no mortality found at dose of 5, 50, 300, and 2000 mg/kg. So The LD50 dose was greater than 2000 mg /kg. Therefore the dose i.e. 100 mg/kg and 200 mg /kg were safe for treatment.
3.3 Blood glucose level during OGTT in normal and diabetic rats
Blood glucose level at different time intervals in OGTT treated with Katankateryadi Kwatha was significantly reduced after treatment compared to the diabetic control group and all treated groups [Table 1, 2 and Fig. 1,2].
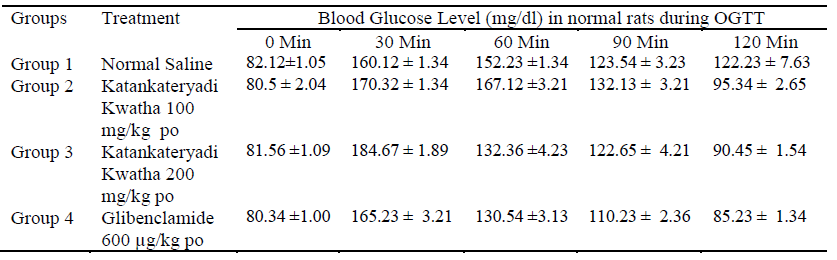
Table 1: Blood glucose level during OGTT in normal rats.
The values represent the mean ± SEM and analyzed by one way ANOVA followed by Dunnett’s test. Values are significantly different at the P < 0.05
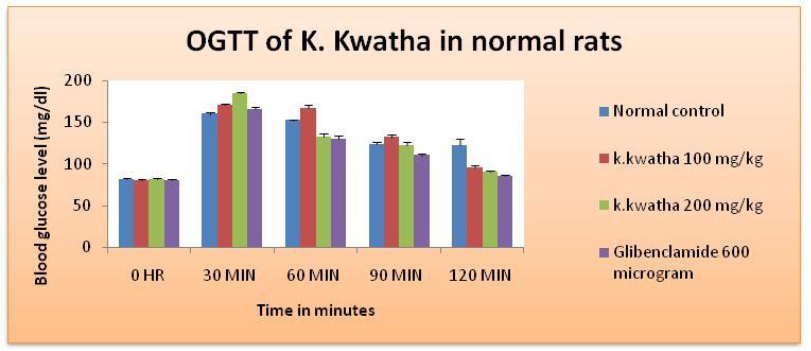
Figure 1: Blood glucose level during OGTT in normal rats after treated with Katankateryadi Kwatha and Glibenclamide.
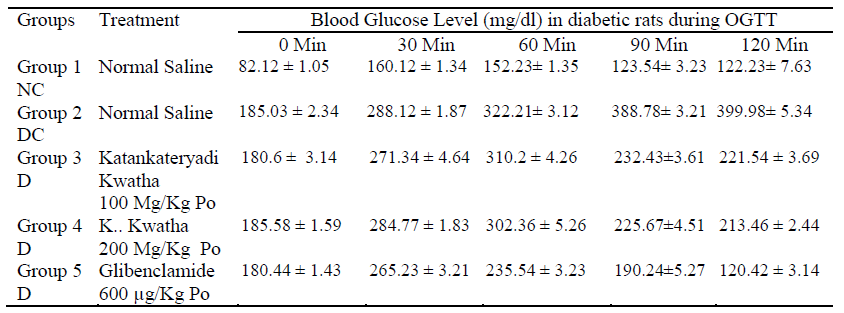
Table 2:Blood glucose level during OGTT in diabetic rats.
The values mean ± SEM with in the column and analyzed by one way ANOVA followed by Dunnett’s test are significantly different at the P < 0.05, NC= Normal control, DC = Diabetic control, D = Diabetic Group.
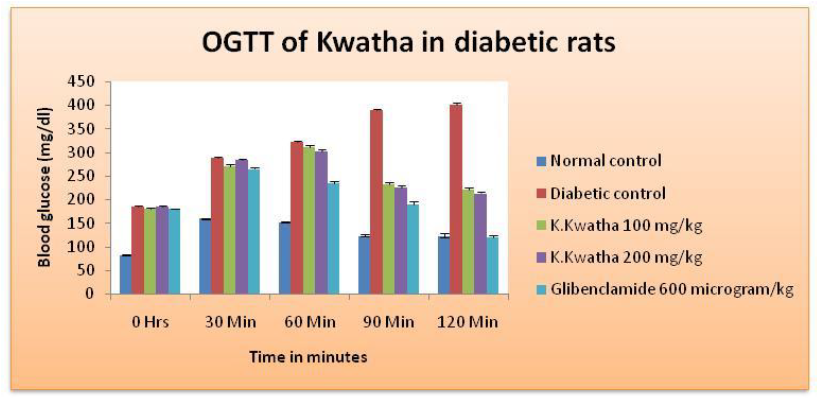
Figure 2: OGTT in diabetic rats after treated with Katankateryadi Kwatha and Glibenclamide.
3.4 Biochemical analysis after treatment in STZ-NAD induced diabetic rats
3.4.1 Estimation of fasting and post prandial blood glucose
During 15 days treatment protocol diabetic rats shows significant reduction in elevated fasting blood glucose level (FBG) and post prandial blood glucose level (PPBG) in Katankatareyadi Kwatha and Gliclazide treated groups. There were no significant changes in normal fasting blood glucose (FBG) level after treatment with Katankateryadi Kwatha in normal rats [28]. Diabetic group treated with 100 mg/kg shows mild reduction in fasting blood glucose level (210.34 ± 1.45 mg/dL)in comparison to rats treated with 200 mg/kg (189.34 ± 3.89 mg/dL FBG). Gliclazide group shows FBG level 140.32 ± 2.90 mg/dL after 15 days treatment while diabetic group shows 388.64 ± 2.53 mg/dL FBG level (Table 3, Fig. 3). Post prandial blood glucose (PPBG) level in diabetic group shows 401.54 ± 2.13 mg/dL and per-see group shows 125.89 ± 3.34 mg/dL. Diabetic group treated with 200 mg/kg of Katankateryadi Kwatha shows better reduction in PPBG 159.35 ± 6.29 mg/dL in comparison to diabetic rats treated with 100 mg/kg (180.34 ± 2.45 mg/dL). The Gliclazide treated group shows 140.32 ± 2.90 mg/dL PPBG (Table 4, Fig. 4).
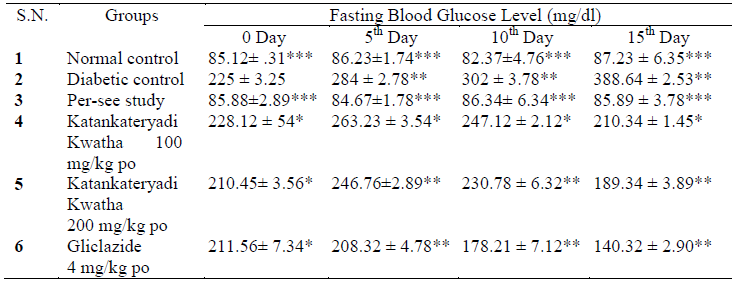
Table 3:Fasting Blood Glucose Level in STZ-NAD Induced Diabetic Rats during treatment.
Values represents in mean ±SEM (n = 6), analyzed by one way ANOVA, * P<0.05, ** P<0.001, ***P<0.0001 compared with diabetic control.
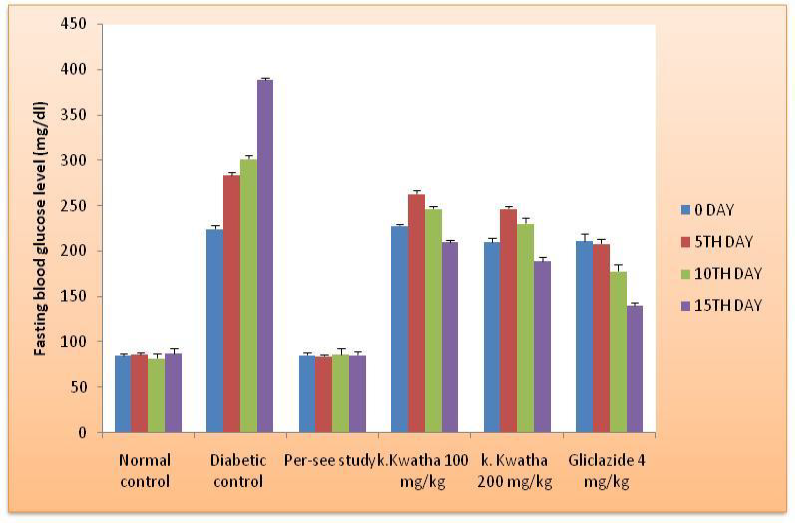
Figure 3: Fasting Blood Glucose Level in STZ-NAD induced Diabetic Rats during Treatment.
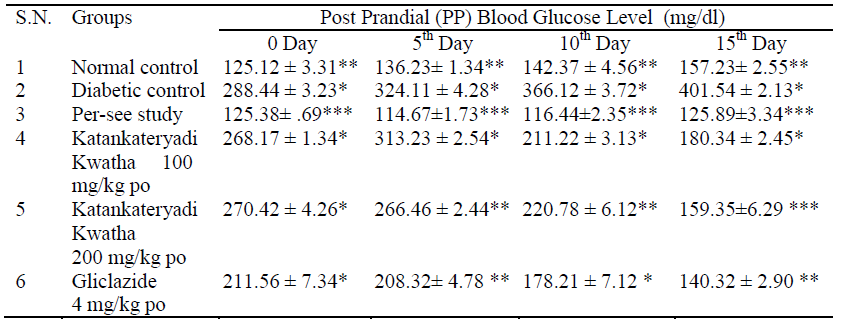
Table 4:Post Prandial Blood Glucose Level after treatment in STZ-NAD Induced Diabetic Rats during treatment.
Values represents in mean ±SEM (n = 6), analyzed by one way ANOVA, * P<0.05, ** P<0.001, ***P<0.0001 compared with diabetic control.
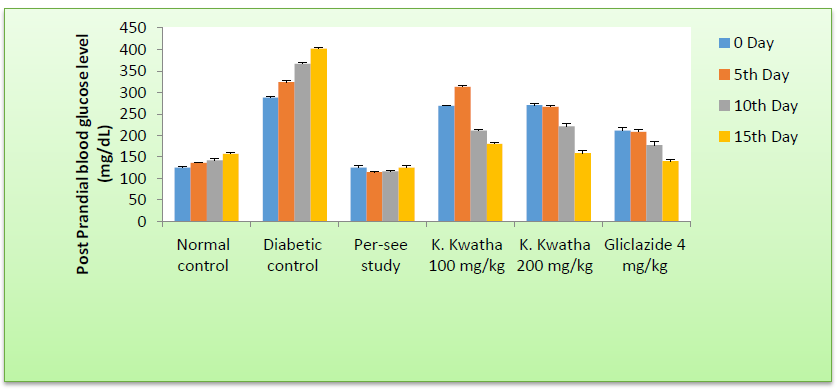
Figure 4: Post Prandial Blood Glucose (PPBG) Level in STZ-NAD induced diabetic rats
3.4.2 Estimation of SGOT and SGPT
The SGOT (serum glutamate pyruvate tansaminase) and SGPT (serum glutamaic oxaloacetic transaminase) are the liver markers. The increase level of SGOT and SGPT are the indication of hepatotoxicity [29]. The level of SGOT and SGPT reduced to its normal value after treatment with Katankateryadi Kwatha. Diabetic control group shows highest SGOT and SGPT level (143.60±3.94 IU/L and 232.02±4.39 IU/L). Rats treated with 200 mg/kg Katankateryadi Kwatha significantly reduced the SGOT and SGPT level (133.34±2.25 IU/L and 170.89±3.76 IU/L) when compared with rats treated with 100 mg/kg. Gliclazide treated group shows 110.12±3.65 IU/L SGOT and 125.23±2.36 IU/L SGPT level (Table 5, Fig. 5).
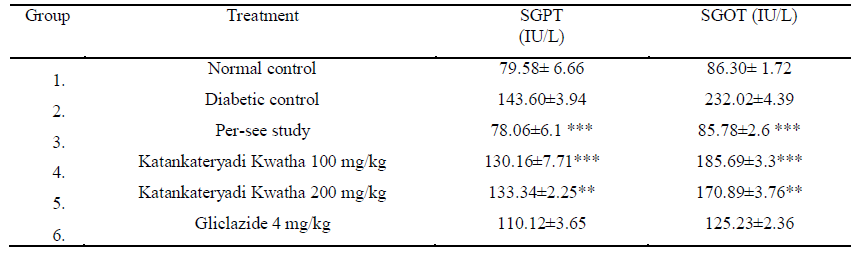
Table 5: Liver profile in diabetic rats after treatment with Katankateryadi Kwatha.
Values are Expressed as Mean ± S.E.M.,(n=6) analyzed by one way ANOVA followed by Dunnett’s test *P value<0.05, **P value <0.01,***<P value<0.001 compared with control group.
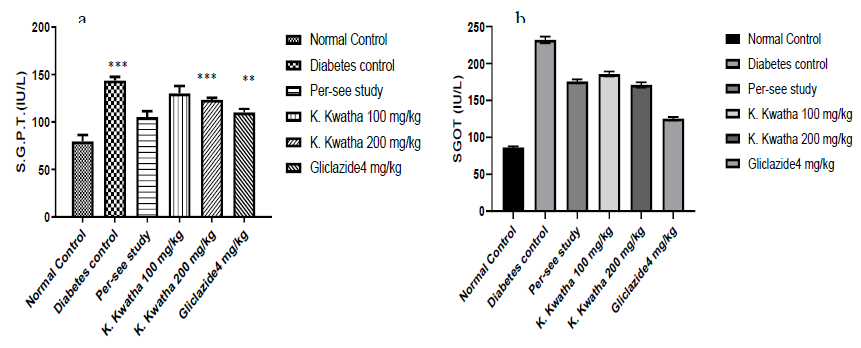
Figure 5: Shows the SGPT and SGOT level in STZ-NAD induced diabetic rats after treatment with Katankateryadi Kwatha and gliclazide
3.4.3 Estimation of HDL, LDL, VLDL, total cholesterol and triglycerides in STZ-NAD induced diabetic rats
In lipid profile HDL is good cholesterol and LDL is bad cholesterol [30]. In control group the normal value of TC, TG, HDL, LDL and VLDL are 85.6 ± 3.33, 75.6 ± 2.81, 31.63 ± 2.06, 38.68 ± 1.52 and 14.32 ± 0.39 mg/dL. While in diabetic group rats shows TC, TG, HDL, LDL and VLDL 170.23 ± 3.56, 165.47 ± 2.3, 25.32 ± 0.95, 185.56 ± 2.32 and 36.85 ± 0.65 mg/dL. The group treated with Katankateryadi Kwatha (200 mg/kg) treated group increased the HDL level (23.56 ± 5.23) and decreases the TC (112.56 ± 3.24), TG (71.65 ± 5.26), LDL (52.41 ± 2.11) and VLDL (21.05 ± 4.62) level significantly with Gliclazide treated rats (Table 6, Fig. 6).
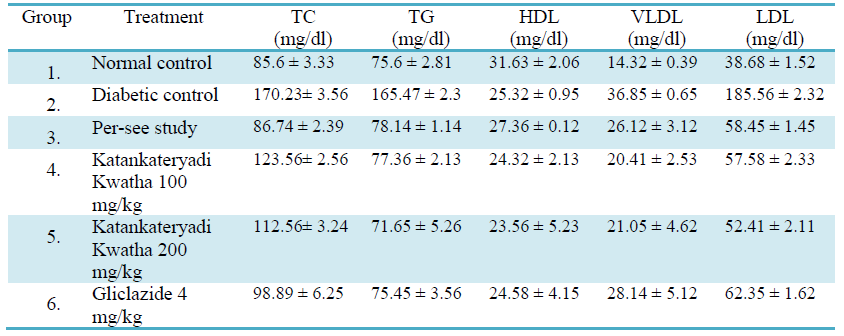
Table 6: Lipid Profile of diabetic rats after treatment.
Values are Expressed as Mean ± S.E.M.,(n=6) analyzed by one way ANOVA followed by Dunnett’s test * P value<0.05, **P value <0.01,***<P value<0.001 compared with control group.
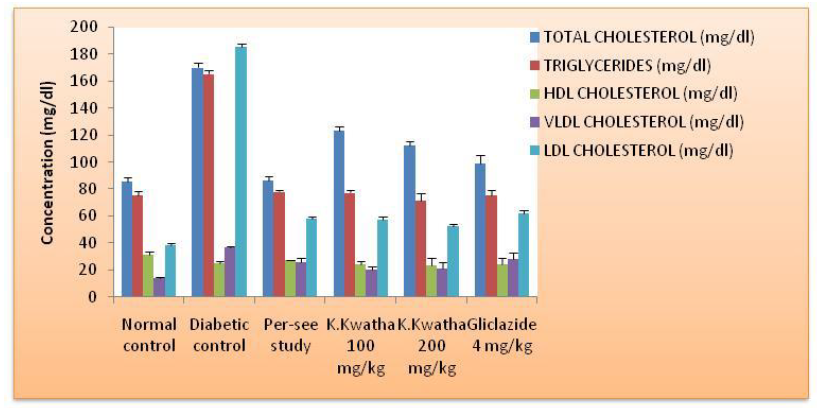
Figure 6: Lipid profile of treated diabetic rats.
3.4.4 Estimation of serum C-Peptide in STZ-NAD induced diabetic rats
After statistical analysis of C-Peptide data result shows that C-Peptide decreased in diabetic rats 0.36 ± 0.034 ng/dl when compared with normal group (0.68 ± 0.025) and treatments with Katankateryadi Kwatha 100 mg/kg, 200 mg/kg and Gliclazide 4 mg/kg significantly increases the C-Peptide level (0.42 ± 0.031, 0.46 ± 0.015, 0.58 ± 0.025) (Table 7, Fig. 8). The C-Peptide level is the marker of insulin secretion from beta cells of pancreas [31]. The Katankateryadi Kwatha at dose 200 mg/kg increases the C- Peptide level indicates that Katankateryadi Kwatha having the insulin secretary activity likewise Gliclazide.
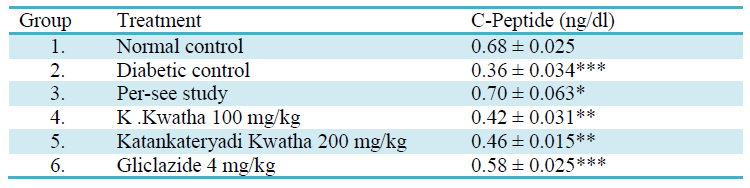
Table 7:C-Peptide level in rats after treatment
Values are expressed in mean ± SEM, *P<0.05 significant value when compared with control, **P<0.001 highly significant compared with diabetic control, ***P<0.0001 highly significant.
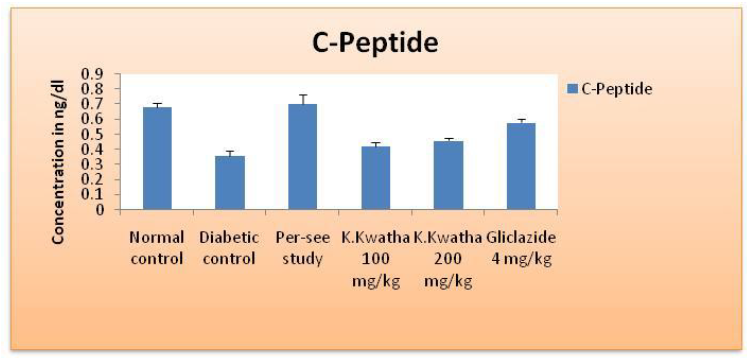
Figure 7:C-Peptide level in diabetic rats after treatment.
4. Discussion
Type 2 diabetes mellitus is a complex, heterogeneous, and polygenic disease characterized by hyperglycemia [32]. There are many factors that contribute to the high blood glucose levels in these type 2 diabetes patients. An important factor is the body’s resistance to insulin, and the second factor is the falling production of insulin by the cells of the pancreas. Lack of insulin plays a primary role in the metabolic derangements linked to diabetes, and hyperglycemia in turn plays a key role in the complications of the diabetes. Several other types of diabetes mellitus are caused by an interaction of genetics and environmental factors [33]. Diabetes type 2 is also refers to as Madhumeha in Ayurveda. Diabetes is associated with vascular and renal dysfunction characterized by hypertension, dyslipidemia, microalbuminuria, macroalbuminuria and glomerular messangial expansion. Diabetes mellitus may present with characteristic symptoms such as polyphagia, polydypsia, polyuria, blurring of vision and weight loss. In its severe forms, keto-acidosis or a non-ketonic hyperosmolar state may develop and lead to stupor, coma and in the absence of effective treatment to death. Diabetes patients are 25 times more prone to blindness, 2 times more prone to heart attacks, 2-6 times more prone to stroke and 17 times more prone to kidney damage as compared to non-diabetics [34]. The characteristic feature of type 2 diabetes mellitus is hyperglycemia that is reported by elevation in fasting plasma glucose [≥7.0 mmol/l (≥126 mg/dL)] and 2-hour postprandial glucose [≥11.1 mmol/l (≥200 mg/dL)] [35].
Diabetes mellitus type 2 is a syndrome of insufficient insulin secretion or insulin resistance or both. However, insulin secretary capacity is not the major determinant in the development of type 2 diabetes, as neither hyperglycemia nor glucose intolerance develops in insulin-resistant patients. Altered insulin secretion caused by either β-cell dysfunction or reduced β-cell mass or both. However, surgical and chemical reductions of β-cell volume induce functional adaptation of the normal β cell to prevent a rise in fasting glucose or reduction in the 1st phase of insulin secretion, suggesting that β-cell dysfunction is closely associated with the pathogenesis and pathophysiology of DM type 2 [36]. Normal dynamics of insulin secretion selective for glucose stimulation, especially the acute phase of the insulin response, is already reduced in the early stage of type 2 diabetes, patients with IGT, and their first-degree relatives. As the early phase of insulin secretion is determined by the RRP (readily releasable pools), a reduction in its size and/or impairment of signaling for exocytose of the insulin granules from the RRP (readily releasable pools) may well develop in IGT and the early stage of type 2 diabetes. The RRP (readily releasable pools) is completely depleted in completely developed stage of diabetes mellitus type 2 [37]. Type 2 diabetes, or its antecedent impaired glucose tolerance, is often associated with other disorders, particularly central (visceral) obesity, hypertension and dyslipidemia (characterized by elevated low-density lipoprotein (LDL), triglycerides and decreased high density lipoprotein (HDL) [38]. There is no specific recommendation on the percentage of calories that should come from carbohydrate, protein, and fat. The medications for the treatment of type 2 diabetes have the target to increase the insulin secretion (Sulfonylurea) or increase the peripheral utility of glucose or inhibit the metabolism of carbohydrate and their absorption [39]. The primary mechanism of action of the sulfonylurea drugs is to stimulate insulin release from pancreatic B cells. Sulfonylurea are used in patients with type 2 but not type 1 diabetes, since these medications require functioning pancreatic B cells to produce their effect on blood glucose. The Daruharidra has the Hypoglycemic, anti-cancer, gastro-irritant, anti fatigue; anticoagulant, Antipyretic, local-anesthetic, anti- protozoal, anti-tuberculosis, anti–bacterial, anti-tumor, hypotensive, anti-inflammatory, anti-trachoma, CNS-depressant action also [40].
The drug Yastimadhu is a prominent plant drug recommended in cough, asthma, bronchitis and throat affections e.g. hoarseness of throat and voice abnormalities (including laryngitis and pharyngitis) [41]. Haritaki contains Tanninic acid, Chebulic acid, anthraquinones and polyphenolic compounds. The fruit of Haritaki is bitter and astringent in taste. It is useful in dyspepsia, bilious headache and diarrhea; and applied to the eyes, piles and used as brain tonic [42]. Amalaki fruit is a good source of vitamin C, carotene, nicotinic acid, riboflavine, D-glucose, D-fructose, myoinositol and apectin with D-galacturonic acid, D-arabinosyl, D-xylosyl. L-rhamnosyl. D- glucosyl, D-mandosyl and D-galactosyl residues, embicol, mucic. Chitraka root contains Plumbagin, 3-chloroplumbagin, 3, 3’-biplumbagin, Chitranone, zeylinone, isozeylinone, elliptinone, droserone,chitranone, zeylinone, isozeylinone, isoshinanolone, maritinone, 4-naphthoquinone, plumbagic acid, seselin, 5-methoxyseselin, suberosin, xanthyletin, xanthoxyletin [43]. Chitraka roots and root bark are bitter, dry, stomachic, carminative, and astringent to the bowels, anthelmintic, alterative; they also cure intestinal troubles, dysentery, leucoderma, inflammation, piles, bronchitis, itching, liver disorders, consumption, ascitis, and anaemia [44].
Herbal medicines are popular because of low cost, minimum side effects and the abundant availability in India due to its varied climatic zones. India has around 45,000 species of plants, out of which 15,000–20,000 plants have proven medicinal value. The physicochemical properties of crude drugs including ash value, extractive value, foreign matter, loss on drying and the Phytochemical study was completed for the standardization of crude drugs [45]. The quality control testing of Katankatareyadi Kwatha was performed and shows there was no microbial contamination in the preparation, absence of heavy metals (lead, arsenic, cadmium and mercury) and the pesticide residue was absence. The Kwatha was dark brown in color, astringent taste and pungent smell due to presence of large amount of flavonoids and tannins. The acute oral toxicity was performed for finding of lethal dose (LD50) and effective dose (ED50) and found that Katankatareyadi Kwatha was safe and effective at maximum dose (>2000 mg/kg b.w.) in rats. Oral glucose tolerance test was performed in normal and diabetic rats and found that the 200 mg/kg of Katankatareyadi Kwatha was more effective than 100 mg/kg. The Katankatareyadi Kwatha reduced the mean value of 40 mg/dL within 2 hrs of treatment. In addition, diabetic rats have impaired glucose tolerance and the additional load of glucose is found to impair the tolerance further. Therefore, the present research investigated both short-term (i.e., FBG and OGTT) during Katankatareyadi Kwatha treatment. From the results obtained, it was clear that fasting blood glucose concentrations and post prandial blood glucose values were dramatically elevated together with oral glucose tolerance in diabetic rats. Katankatareyadi Kwatha at dose of 200 mg/kg produced a statistically significant decrease in FBG beginning from the 1 hrs and progressing till the end of 2 hrs in comparison to the normal and diabetic control group, which indicated Katankatareyadi Kwatha was more beneficial to the long term glucose blood control. From the data obtained in the OGTT, it was clear that Katankatareyadi Kwatha reduces blood glucose levels after 60 min and 120 min of glucose administration. The test suggested that Katankatareyadi Kwatha acts by increasing peripheral utilization of glucose. STZ-NAD induced Diabetes is reported with alterations in plasma blood glucose lipid and lipoprotein profile. In uncontrolled type 2 diabetes mellitus, there will be an increase in TC, LDL, VLDL and TC with decrease in HDL level which contributes to the coronary artery disease. Raised plasma free fatty acid (FFA) level plays a major role in the pathogenesis of insulin resistance and type 2 diabetes. In the present study marked increase in serum lipids and lipoprotein in diabetic rats was observed in diabetic rats and regular administration of the Katankatareyadi Kwatha for 15 days lowered the TC, TG, FFA, and LDL values while elevate the HDL value. The elevation of bad cholesterol and decreased good cholesterol were restored to near normal level during treatment. Results revealed that Katankatareyadi Kwatha was also effective in hyperlipidemia.
The previous studies show that insulin is the important hormone that regulates blood glucose homeostasis by stimulating the utilization of glucose by liver, muscle and adipose tissue, with stimulation of anabolic processes of glucose, protein and lipid synthesis. Insulin is initially synthesized in beta cells in the form of pro-insulin. In this form the alpha and beta chains of active insulin are linked by a third polypeptide chain called the connecting peptide, or C-peptide, which are assumed to primarily serve as a facilitator for the formation of active insulin by helping insulin to fold and be cleaved as necessary [47]. STZ-Nicotinamide injection caused diabetes mellitus type 2, which may be due to partial destruction of β cells of the islet of langerhans of the pancreas. Over-production (excessive hepatic glycogenolysis and gluconeogenesis) of glucose and decreased utility of glucose by the tissues are the fundamental basis of hyperglycemia in diabetes mellitus type 2 [48].
This study has revealed that Katankatareyadi Kwatha produced a marked decrease in blood glucose at 200 mg/ kg body weight in normal as well as in diabetic rats after 15 days of treatment protocol. Streptozotocin induces diabetes by free radical generation, which causes destruction of insulin secreting beta cells of the islets of langerhans, resulting in a decrease in endogenous insulin level. The free radical scavenging effect of Nicotinamide reduced the effect of STZ. NAD++ is the principal metabolite of Nicotinamide. NAD++ level in beta-cells in pre-diabetic and diabetic is significantly reduced and destruction of beta-cells may occur via oxidative stress. Increased levels of reactive oxygen species in beta-cells may result in oxidative damage to DNA resulting in DNA strand breaks. The enzyme poly ADP ribose polymerase or PARP is believed to play a role in DNA repair in beta cells and the PARP uses NAD++ as its substrate. In the context of a reduced level of NAD++ result in cellular apoptosis. Nicotinamide is an inhibitor of PARP and reduced the apoptosis. All of these effects of Nicotinamide play some important role in the possible anti-diabetogenic action of Nicotinamide against the STZ. Nicotinamide by the above mechanism minimize the effects of STZ and helps in partial destruction of beta cells leads to development of diabetes mellitus type 2 [49]. In the present study the data obtained clearly indicate that the administration of Katankatareyadi Kwatha lowers the hyperglycemia and increases the C-Peptide level means increases the synthesis and secretion of insulin. On the basis of the current investigation it was noted that the Katankatareyadi Kwathahas the antidiabetic activity and these results provide pharmacological evidence for its folklore claim as an antidiabetic agent.
5. Conclusion
The Katankateryadi Kwatha a polyherbal formulation reduces the blood glucose level significantly in diabetic rats. Kwatha reduces the enzymatic activity of alpha amylase, alpha glucosidase which are responsible for hyperglycemia. Kwatha also inhibit the glucose absorption from dialysis membrane, hence we can says that Katankateryadi Kwatha can be used as a potent antidiabetic formulation for the management of hyperglycaemia.
6. Acknowledgement
A very thankful Banaras Hindu University for providing the facilities for conducting this research and also thankful to all authors for their equal support in research work and paper writing. No grant for publication of this article.
7. Conflict of Interest
:No conflict of interest between the authors
8. References
- Qureshi SA, Asad W, Sultan V. The effect of Phyllantus emblica Linn. On type II diabetes, triglycerides and liver specific enzyme. Pak J Nutr 8 (2009): 125–8.
- “Diabetes Mellitus”, World Health Organization Fact Sheet 138 (1999).
- “Diabetes Mellitus”, American Diabetes Association - General Fact Sheet (1999).
- Khosla P, Bhanwra S, Singh J, Seth S, Srivastava RK. A study of hypoglycemic effects of Azadirachta indica (Neem) in normal and alloxan diabetic rabbits. Indian J Physiol Pharmacol 44 (2000): 69–74.
- Pillai NR, Santhakumari G. Hypoglycemic Activty of melia Azadirechta Linn (neem) Indian J Med Res74 (1981): 931–3.
- Edwin E, Sheeja E, Gupta VB, Jain DC. Fight Diabetes the herbal way. Express Pharma Pulse1 (2006): 41–2.
- Alam K, Mahpara S. Role of diet, nutrients, spices and natural products in diabetes mellitus. Pak J Nutr 2 (2003) :1–12.
- Chattopadhyay RR. Possible mechanism of antihyperglycemic effect of Azadirachta indica leaf extract. Part IV. Gen Pharmacol 27 (1996): 431–4.
- Chattopadhyay RR, Chattopadhyay RN, Nandy AK, Poddar G, Maitra SK. The effect of fresh leaves ofAzadirachta indica on glucose uptake and glycogen content in the isolated rat hemi diaphragm. BullCalcutta Sch Trop Med 35 (1987): 8–12.
- Dorababu M, Prabha T, Priyambada S, Agrawal VK, Aryya NC, Goel RK. Effect of Bacopa monnieraand Azadirachta indica on gastric ulceration and healing in experimental NIDDM rats. Indian J Exp Biol389 (2004): 389–97.
- Singh Jyotsna, Kakkar Poonam. Antihyperglycemic and antioxidant effect of Berberis aristata root extract and its role in regulating carbohydrate metabolism in diabetic rats. Journal of Ethnopharmacology 123 (2009): 22–26.
- Nisrat Jahan, Antioxidant, Antimicrobial and Antidiabetic Activities of Glycyrrhiza Glabra (Yastimadhu): A Review, Indo Am. J. P. Sci 3 (2016): 231-239.
- Murali YK, Ramesh Chander, Murthy PS. Antihyperglycemic effect of water extract of dry fruits of Terminalia chebula in experimental diabetes mellitus. Ind. J Clin Biochem 19 (2004): 202- 204.
- Latha RC, Daisy P. Insulin-secretagogue, antihyperlipidemic and other protective effects of gallic acid isolated from Terminalia bellerica Roxb. In streptozotocin-induced diabetic rats. Chemicobiological interactions189 (2011): 112-118.
- L Anila, N R Vijaylakshmi; Flavonoids from Emblica officinalis and Mangifera indica-effectiveness for dyslipidemia. Journal of Ethnopharmacology79 (2002): 81-87.
- Jain Paras et al. Pharmacological Profiles of Ethno-Medicinal Plant: Plumbago zeylanica- A Review, Int. J. Pharm. Sci. Rev Res24 (2014): 157-163.
- Aji Abraham, Sarala Samuel, Lizzy Methew. Phytochemical analysis of Pathyashadangam kwath and its standardization by HPLC and HPTLC. Journal of Ayurveda and Integrative Medicine 11 (2020): 153-158.
- Genovese T, Menegazzi M, Mazzon E, Crisafulli C, Di Paola R, Dal Bosco M, Zou Z, Suzuki H, Cuzzocrea S. Glycyrrhizin reduces secondary inflammatory process after spinal cord compression injury in mice. Shock 31 (2009): 367–375.
- Chen G, Zhu L, Liu Y, Zhou Q, Chen H, Yang. Isoliquiritigenin, a flavonoid from licorice, plays a dual role in regulating gastrointestinal motility in vitro and in vivo. J. Phytother Res 23(2009): 498–506
- Nakanishi T, Inada A, Kambayashi K, Yoneda K. Flavonoid glycosides of the roots of Glycyrrhiza uralensis. Phytochemistry 24 (1985): 339–341.
- Suresh Reddy Yanala D, Sathyanarayana K, Kannan A. Recent Phytochemical Review – Fruits of Tribulus terrestrisLinn, J. Pharm. Sci. & Res 8 (2016): 132-140.
- Halim EM. Lowering of blood sugar by water extract of Azadirachta indica and Abroma augusta indiabetes rats. Indian J Exp Biol 41 (2003): 636–40.
- Elias D, Prigozin H, Polak N, Rapoport M, Lohse AW, Cohen IR. Autoimmune diabetes induced by the b—Cell toxin STZ. Diabetes43 (1994): 992–8.
- Karunanayake EH, Hearse DJ, Mellows G. The metabolic fate and elimination of streptozocin. Biochemical Society Transactions 3 (1975): 410–14.
- Bhuyan BK, Kuentzel SL, Gray LG, Wallach D, Neil GL. Tissue distribution of streptozotocin (NSC 85998) Cancer Chemotherapy Report 58 (1974): 157–65.
- Zia T, Hasnain SN, Hasan SK. Evaluation of the oral hypoglycemic effect of Trigonella foenum-graecum in normal mice. J Ethanopharmacol 75 (2001): 191–5.
- Ribes G, Sauvaire Y, Da Costa C, Baccou JC, Loubatieres- Mariani MM. Antidiabetic effects of subfractions from fenugreek seeds in diabetic dogs. Proc Soc Exp Biol Med 182 (1986): 159–66.
- Abdel-Barry JA, Abdel-Hassan IA, Al-Hakiem MH. Hypoglycemic and antihyperglycemic effects of Trigonella foenum- graecum leaf in normal and alloxan induced diabetic rats. J Ethnopharmacol58 (1997): 149–55.
- Khosla P, Gupta DD, Nagpal RK. Effect of Trigonella foenum graecum (Fenugreek) on blood glucosein normal and diabetic rats. Indian J Physiol Pharmacol 39 (1995): 173–4.
- Eisenberg DM, Davis RB, Ettner SL, Appel S, Wilkey S, Van Rompay M, et al. Trends in alternative medicine use in the United States, 1990-1997: Results of a follow-up national survey. JAMA280 (1998): 1569–75.
- Ryan EA, Pick ME, Marceau C. Use of alternative medicines in diabetes mellitus. Diabet Med18 (2001): 242–5.
- Sabina AB, Williams A, Wall HK, Bansal S, Chupp G, Kartz DL. Yoga intervention for adults with mild to moderate asthma: A pilot study. Ann Allergy Asthma Immunol 94 (2005): 543–8.
- Gupta N, Khera S, Vempati RP, Sharma R, Bijlani RL. Effect of yoga based lifestyle intervention on state and trial anxiety. Indian J Physiol Pharmacol 50 (2006): 41–7.
- Sahay BK, Sahay RK. Lifestyle modification in management of diabetes mellitus. J Indian Med Assoc100 (2002): 178–80.
- Stevens DL. The use of complementary and alternative therapies in diabetes. Clin Fam Prac4 (2002): 911–28.
- Innes KE, Bourguignon C, Taylor AG. Risk indices associated with the insulin resistance syndrome, cardiovascular disease, and possible protection with yoga: A systematic review. J Am Board Fam Pract18 (2005): 491–519.
- Da Silva GD, Lorenzi-Filho G, Lage LV. Effects of yoga and the addition of Tui Na in patients with fibromyalgia. J Altern Complement Med 13 (2007): 1107–13.
- Elson DF, Meredith M. Therapy for type II diabetes mellitus. Wis Med J 97 (1998): 49–54.
- Field T, Hernandez RM, La Greca A, Shaw K, Schlanberg S, Kuhn C. Massage therapy lowers blood glucose levels in children with diabetes mellitus. Diabetes Spectrum 10 (1997): 237–9.
- Valtonen EJ, Lilius HG. Syncardial massage in diabetic and other neuropathies lower extremities. Dis Nerv Syst 34 (1973): 192–4.0.
- Cerasi E, Luft R. The plasma insulin response to glucose infusion in healthy subjects and in diabetes mellitus. Acta Endocrinologica 55 (1967): 278–304.
- Polonsky KS, Given BD, Hirsch LJ, et al. Abnormal patterns of insulin secretion in non-insulin-dependent diabetes mellitus. New England Journal of Medicine 318 (1988): 1231–1239.
- Rakieten N, Rakieten ML, Nadkarni MV. Studies on the diabetogenic action of streptozotocin (NSC-37917). Cancer Chemo ther Rep 29 (1963): 91-8.
- Ozturk Y, Atlan VM, Yildizoglu-Ari N. Effects of experimental diabetes and insulin on smooth muscle functions. Pharmacol Rev 48 (1996): 69-112.
- Junod A, Lambert AE, Stauffacher W, Renold AE. Diabetogenic action of streptozotocin. Proc Soc Exp Biol Med 126 (1967): 201-5.
- Ansari MN, Bhandari U. Antihyperhomocysteinemic activity of an ethanol extract from Embelia ribes in albino rats. Pharmaceutical Biology 46 (2008): 283-287
- Agarwal BB, Kumar A, Bharti AC. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res 23 (2003): 363-398
- Venkatesan P, Rao MN. Structure activity relationships for the inhibition of lipid peroxidation and the scavenging of free radicals by synthetic symmetric curcumin analogues. J Pharm Pharmacol 52 (2000): 1123-1128
- Weiss RB. Streptozocin. A review of its pharmacology, efficacy and toxicity. Cancer Treatment Report 66 (1982): 427–38.


 Impact Factor: * 3.0
Impact Factor: * 3.0 Acceptance Rate: 76.32%
Acceptance Rate: 76.32%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
