The Misuse of Clobetasol Propionate 0.05% Based Products as Skin Lightening Agents Among Sudanese Women
Monzer Farah1*, Mongi Farah2, Mohmmed Ali3, Mohamed Ibrahim4, Yassin Abedelraheem5
1Alneelain university, Sudan
2Khartoum ENT hospital, Sudan
3Galway university hospital, Sudan
4Modren care medical complex, Sudan
5Police hospital, Sudan
*Corresponding author: Monzer Farah, Alneelain university, Sudan.
Received: 03 May 2022; Accepted: 13 May 2022; Published: 19 May 2022
Article Information
Citation: Monzer Farah, Mongey Farah, Mohmmed Ali, Mohamed Ibrahim, Yassin Abedelraheem. The Misuse of Clobetasol Propionate 0.05% Based Products as Skin Lightening Agents Among Sudanese Women. Journal of Women’s Health and Development 5 (2022): 152-161.
DOI: 10.26502/fjwhd.2644-28840085
View / Download Pdf Share at FacebookAbstract
Despite the social, pathological, physiological, and psychological effects that topical corticosteroids (TCs) present, it has become a widespread practice among Sudanese women. This cross-sectional study involving the three localities of Khartoum state assessed the perceived risks and reasons behind the use of TC among Sudanese women. Data was collected from 217 women using a validated self-administered questionnaire with a Cronbach's alpha value of 0.773. The data were analyzed descriptively and inferentially. Findings showed that 151 (69.6%) of respondents had used at least one clobetasol propionate 0.05% based product, 22 (14.6%) of them used very potent TC for >6 months. 67(44.4%) have a negative perception of its risks and consequences. The reasons behind why clobetasol propionate 0.05% based product were superior is that: it has a very rapid result (54.2%), low price point (35.5%) and no prescription needed (10.3%), while the main motives for commencement were engagement\wedding\graduation party (societal pressure),33 (21.3%) to satisfy the community beauty standards and 49 (31.6%) commence because of their low self-esteem due to their colored skin [self-satisfaction]. There was a significant difference in the perception of the risks of very potent TCs based on institutional affiliations (p=.000). This study concluded that the misuse of very potent TC was very prevalent among the studied population and that the participants had a negative perception of its risks and consequences. The most frequently reported adverse effects are stretch marks (striae) 68.8% and facial redness 57%.
Keywords
<p>Clobetasol propionate; Topical corticosteroids; Sudanese women; Duration; Adverse effect</p>
Article Details
1. Introduction
Topical Corticosteroids (TC) have been used for more than six decades to treat various dermatological disorders due to their wide range of action [1, 2]. As several countries where potent TCs are easily available over-the-counter at a low price, misuse has been noticed among the general population, producing many adverse effects [3, 4]. TCs have been the cornerstone of treatment for non-infectious dermatologic illnesses associated with inflammation since their debut in the early 1950s [5]. These disorders include a wide variety of dermatoses, such as atopic dermatitis, eczema, contact dermatitis, psoriasis, seborrheic dermatitis, vitiligo, lichen sclerosis, and intertrigo [6]. The potency of TCs has been assessed by measuring their vasoconstrictive effect on the skin. Based on their potency, British National Formulary (BNF) divides the topical corticosteroids into four groups:
|
Class I: Very potent |
Class II: Potent |
Class III: Moderate |
Class IV: Mild |
|
Clobetasol propionate 0.05% |
Beclometasone dipropionate 0.025% |
Alclometasone dipropionate 0.05% |
Hydrocortisone 0.1%-2.5% |
|
Betamethasone valerate 0.1% |
Betamethasone valerate 0.025% |
Fluocinolone acetonide 0.0025% |
|
|
Betamethasone dipropionate 0.05% |
Clobetasone butyrate 0.05% |
||
|
Diflucortolone valerate 0.1% |
Fluocinolone acetonide 0.00625% |
||
|
Fluocinolone acetonide 0.025% |
Fluocortolone 0.25% |
||
|
Hydrocortisone butyrate 0.1% |
|||
|
Mometasone furoate 0.1% |
|||
|
Triamcinolone acetonide 0.1% |
Table 1: BNF classification of topical corticosteroids Class Potency Generic name and strength (De, 2019) [7].
Although it is a prevalent problem, no research has been conducted in Sudan on the misuse of very potent topical corticosteroid medications. This study was conducted among females living in different localities of Khartoum state\Sudan, the frequency of TC misuse, duration, number of products, the reasons behind it, and the knowledge of adverse effects resulting from it are the main focus of the study.
1.1. Why OTC clobetasol propionate based products are cheap in Sudan
The main reason is that; these products imported to Sudan from the neighboring countries in an illegal way without any regulations. Another reason is these products sold in cosmetic shops as fractions of a package (titrated by a small spoon i.e. a small spoon for only 35 SDG) which made their purchase easier for people on budget.
1.2. Preparation of women for marriage life
This is a month or two period before the women get married and move to her husband period. In this period there is high pressure from the society and the husband to achieve lighter flawless skin; and that what make very potent TCs a valid option due to their relatively cheap prices and rapid results.
2. Materials and Methods
2.1. Research setting and design
The study was conducted in Khartoum state females from different localities [Khartoum (Capital), Omdurman, Khartoum North, Sharq al Nil, Jabal Awliya, Om Badda , and Karari] were selected, and those who agreed to participate were included. This study utilizes the descriptive cross-sectional study design. The study was conducted on February 2022.
2.2. Target Population
The target population comprises all Sudanese females living in Khartoum state who are whiling to participate in the study.
2.3. Sampling Technique and Sample Size
The sampling was done by three stage cluster sampling technique .A number of 217 females where selected based on the willingness for participation.
2.4. Research Instrument
An on-line survey was done with Google forms using a semi-structured self-administered questionnaire containing participants’ bio data, education, the use of very potent TC including the name of the product, duration of use, reasons and knowledge of side effects. The questionnaire was advertised on Dr On Board platform with ? 80000 follower that provides day to day skin care advices and educate the general population about the use and side effects of unauthorized cosmetic products The products mentioned in the questionnaire form were selected based on a quick survey on 4000 females on a famous online Sudanese platform (Dr On Board; a platform with ? 80000 follower that provide day to day skin care advices and educate the general population about the use and side effects of unauthorized cosmetic products) asking about which product containing TCs they use the most. The products included are: ornavate gel\ornavate cream\civic cream\civic lemon\movate cream and newlight cream. The questionnaire was attached to the bio of the platform and the followers were asked to participate.
2.5. Ethical Consideration
An ethical approval from Sudan ministry of health was obtained. Also, informed consent was obtained from the participants before the commencement of the study. Participants were assured anonymity and confidentiality of the information and that they were at liberty to decide to discontinue in the study at any time.
2.6. Procedure for Data Collection
Preliminary visits were made to the localities to seek their permission for the conduct of the study. Participants’ consent was obtained, and they were given the questionnaire in a Google form which they filled Without any form of coercion.
2.7. Data Analysis
Data analyses were performed in Statistical Package for Social Sciences (SPSS) (v.20) using descriptive and inferential statistics. Statistical methods employed include frequency count, percentage and chi-square. Association
between the use of very potent TC and demographic characteristics of the respondents as well as actual use of very potent TC was explored using Chi-square.
3. Results
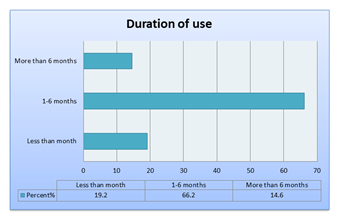
Graph 1: Duration of us of very potent TC [N=151].
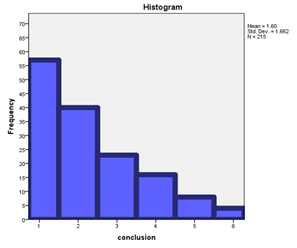
Graph 2: number of clobetasol propionate 0.05% based products tried [N=151]
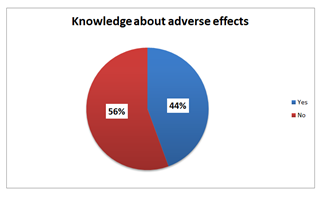
Graph 3: Knowledge about adverse effects among TC consumers [N=151].
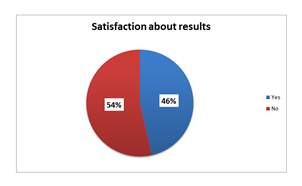
Graph 4: Satisfaction about results among TC consumers [N=151].
Table (1) present the participants age , marital status , level of education and if their faculty applied is it medical or non-medical school. The age distribution of the respondents showed that about (54.4%) were between 16 and 25 years , the mean of age is 25.58 and the standard deviation is 5.145. The prevalence of use and the used products names are mentioned in table (2). Table (3): shows distribution clobetasol propionate 0.05% based products by product name among medical and non-medical higher education participants.
|
No. |
% |
chi |
df |
P |
|
|
Age |
26.356 |
25 |
0.389 |
||
|
16-25 |
118 |
54.4 |
|||
|
26-35 |
89 |
41 |
|||
|
36-50 |
10 |
4.6 |
|||
|
Marital status |
6.599 |
5 |
0.252 |
||
|
Single |
142 |
65.5 |
|||
|
Married |
64 |
29.4 |
|||
|
Divorced |
11 |
5.1 |
|||
|
Level of education |
6.535 |
3 |
0.088 |
||
|
Primary |
7 |
3.2 |
|||
|
Secondary |
15 |
6.9 |
|||
|
Higher education |
195 |
89.9 |
|||
|
Medical\Non-medical education |
6.227 |
2 |
0.044 |
||
|
Medical |
|||||
|
Non-medical |
112 |
57.4 |
|||
|
85 |
43.6 |
Table 1: Age, marital status, level of education and faculty.
|
Ever use clobetasol propionate 0.05% based products (N=217) |
No. |
% |
|
Yes |
151 |
69.6 |
|
No |
66 |
30.4 |
|
Ornavate gel (clobetasol propionate0.05%) |
||
|
Yes |
92 |
42.4 |
|
No |
125 |
57.6 |
|
Ornavate cream (clobetasol propionate0.05%) |
||
|
Yes |
12 |
5.5 |
|
No |
205 |
94.5 |
|
Civic cream (clobetasol propionate0.05%) |
||
|
Yes |
101 |
46.5 |
|
No |
116 |
53.5 |
|
civic lemon (clobetasol propionate0.05%) |
||
|
Yes |
60 |
27.6 |
|
No |
157 |
72.4 |
|
Movate cream (clobetasol propionate0.05%) |
||
|
Yes |
55 |
25.3 |
|
No |
162 |
74.7 |
|
New light cream (clobetasol propionate0.05%) |
||
|
Yes |
23 |
10.6 |
|
No |
194 |
89.4 |
Table 2: Ever use Very potent TCs for non-medical purpose.
|
Medical field |
N = 112 |
Outside the medical field |
N = 85 |
|||
|
No. |
% |
No. |
% |
|||
|
Ornavate gel (clobetasol propionate0.05%) |
Ornavate gel (clobetasol propionate0.05%) |
|||||
|
Yes = |
45 |
59.8 |
Yes = |
38 |
44.7 |
|
|
No = |
67 |
40.2 |
No = |
47 |
55.3 |
|
|
Ornavate cream (clobetasol propionate0.05%) |
Ornavate cream (clobetasol propionate0.05%)) |
|||||
|
Yes = |
Yes = |
|||||
|
No = |
9 |
8 |
No = |
83 |
97.6 |
|
|
Civic cream (clobetasol propionate0.05%) |
103 |
92 |
Civic cream (clobetasol propionate 0.05%) |
2 |
2.4 |
|
|
Yes |
Yes |
|||||
|
No |
54 |
No |
||||
|
Ornavate lemon (clobetasol propionate0.05%) |
58 |
48.2 |
Ornavate lemon (clobetasol propionate0.05%) |
38 |
44.7 |
|
|
Yes |
51.8 |
Yes |
47 |
55.3 |
||
|
No |
35 |
No |
||||
|
Movate cream (clobetasol propionate0.05%) |
77 |
31.3 |
Movate cream (clobetasol propionate0.05%) |
|||
|
Yes |
68.7 |
Yes |
20 |
23.5 |
||
|
No |
No |
65 |
76.5 |
|||
|
New light cream (clobetasol propionate0.05%) |
33 |
New light cream (clobetasol propionate0.05%) |
||||
|
Yes |
79 |
29.5 |
Yes |
18 |
21.2 |
|
|
No |
70.5 |
No |
67 |
78.8 |
||
|
16 |
||||||
|
96 |
85.7 |
6 |
7.1 |
|||
|
14.3 |
79 |
92.9 |
||||
|
product name among medical and non-medical higher education participants |
||||||
Table 3: Distribution clobetasol propionate 0.05% based products by product name among medical and non-medical higher education participants.
|
Side effect |
Percentage |
|
Face |
|
|
Redness |
57 |
|
Acne |
38.2 |
|
Excessive facial hair |
21.7 |
|
Body |
|
|
Striae |
68.6 |
|
Telangiectasia |
15.5 |
|
Excess of body hair |
12 |
|
depigmentation |
5 |
Table 4: The percentages of the reported adverse effects of clobetasol propionate 0.05% based products.
3.1. Reason and psychology behind their use
Out of 155 who use at least one product containing clobetasol propionate 0.05% 55(35.5%) claim the reason was the price point [very cheap 1-2 USD],16 (10.3) because no prescription needed while the majority (54.2%) claim the reason behind their use is the rapidity of the results. When asking the exact 155 females mentioned above about the psychology behind their commencement of OTC very potent steroids as a lighting agent: 73 (47.1%) started because they have engagement\wedding\graduation party,33 (21.3%) to satisfy the community beauty standards {as they mention}; this highlights the importance of societal pressure on women to have lighter skin on social gatherings. 49 (31.6%) commence because of low self-esteem due to their colored skin [self-satisfaction].
3.2. Reported adverse effects
We divide the adverse effects into face & body adverse effects. The facial adverse effects include; redness (), acne() and excess growth of facial hair. The most frequently reported body adverse effect is striae which is the main reason for the consumers to seek professional health care advice. While adverse effects were the major reason for OTC potent TC discontinuation. Steroid induced withdrawal dermatitis is the most reported reason behind long term use despite the knowledge of adverse effects.
4. Discussion
Misuse of TC appears to be a common problem, as reported by many studies in India, China, Iraq, and Senegal. Like these countries in Madagascar, most TCs are available at cheap prices and they are mostly sold as over-the-counter products. As in many African countries, Sudanese women face pressure to lighten their skin due to the widespread social perception that light skin is considered more attractive and reflective of high social class [8, 9]. This study examined the perception of risk and factors associated with very potent TC among Sudanese women in Khartoum State. The findings revealed that there was a high prevalence (69.6%) of very potent TC practices among the participants. This prevalence is a little higher than that obtained by an earlier Ethiopian study (60%) conducted among Ethiopian TC consumers [Bilal, 2018]. The age distribution of consumers is almost the same (54.4% are from 16-25 years) compared to( 46.5% were from 18-28 years ) [Bilal, 2018]. The study confirms the previously mentioned claims that TCs are commoner among single females (65.5% ) compared to (54.5%), while higher education participants (89.9%) in relation to (56.6%) [Bilal, 2018]. Represent the majority of TC consumers [10].
The duration of use: less than 1 month (19.2%),1-6 months (66.2%) and greater than 6 months(14.6%) in contrast to (32.9%),(28.3%),(38.8%) respectively[Bilal, 2018] 10.This reveals that the rapid effect of TC and thinning effect on the skin which gives a perception of a lighter\softer skin is a strong motive to continue TC for longer periods.
Findings from this study are also in support that cheap price and rapid results are the main reasons to commence very potent TC while social gatherings (engagement. weddings and graduation parties) represent the most frequent motive (47.1%). Regarding the knowledge of side effects (44.4%) did know the possible adverse effects and yet commence TC use anyway, and this obviously indicates the preference of looks over health so this is a huge problem and needs to be deeply investigated from a psychological perspective.
5. Conclusion
Our study revealed that misuse of topical corticosteroids is a major public health problem where several sectors were involved. TC are sold in cosmetic retailers and do not need prescription in pharmacies. The over-the-counter use of TC has a psychological impact on the user; no restriction use of TC ensures users about the safety of this product. In addition, misuse of TC shows that the supply channel of this product is defective and illegal in Sudan. A task force against TC abuse should be formed in Sudan, which aims to raise public awareness, run media campaigns, form study for doctors, highlight the problem in journals, and meet with central and state authorities.
Reference
- MB Sulzberger, VH Witten. The Effect of Topically Applied Compound F in Selected Dermatoses. Journal of Investigative Dermatology 19 (1952): 101-102,.
- TH Rindani. Topical action of steroid hormones on inflammation,” Archives Internationales De Pharmacodynamie Et De Therapie 99 (1954): 467-473.
- B Ladizinski, N Mistry, RV Kundu. Widespread use of toxic skin lightening compounds: Medical and psychosocial aspects,” Dermatologic Clinics 29 (2011):111-123.
- H Lu, T Xiao, B Lu, et al. Facial corticosteroid addictive dermatitis in Guiyang City, China,” Clinical and Experimental Dermatology 35 (2010): 618-620.
- Saravanakumar RT. Study of prescribing pattern of topical corticosteroids in the department of dermatology of a multispecialty tertiary care teaching hospital in south India. International Journal of Research in Pharmaceutical Sciences 3 (2016): 685-687.
- Woodson JM. The Role of Low-Potency Topical Steroids in Day-to-Day Practice. Skin and Aging supplement of December (2009): 1-8.
- De A.BNF classification of topical corticosteroids (2019).
- del Giudice P, Yves P. The widespread use of skin lightening creams in Senegal: a persistent public health problem in West Africa.International Journal of Dermatology 41(2002): 69-72.
- Ahmed AE, Hamid ME. Use of Skin-Whitening Products by Sudanese Undergraduate Females: a Survey. Racial and Ethnic Health Disparities4 (2017):149-155.
- Bilal A, Shimels T, Tsegaye M. Prevalence of topical corticosteroids related adverse drug events and associated factors in selected community pharmacies and cosmetic shops of Addis Ababa, Ethiopia.Sudan Journal of Medical Sciences 13 (2018): 62.


 Impact Factor: * 3.4
Impact Factor: * 3.4 Acceptance Rate: 78.89%
Acceptance Rate: 78.89%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 