Analgesic, Anti-inflammatory, and Antioxidant potential of S-adenosyl L-Methionine on Nitroglycerine induced Migraine in mice Models
Sharmila Jalgaonkar1, Raakhi Tripathi1, Nishtha Khatri2, Rachana Patankar2, Snehalata Gajbhiye3, Mohsin Sayyed2, Arun Shankar4
1Associate Professor, Seth GS Medical College and KEM Hospital, Parel, Mumbai, 400 012, India
2Pharmacology Resident, Seth GS Medical College and KEM Hospital, Parel, Mumbai, 400 012, India
3Associate Professor, All India Institute of Medical Sciences, Nagpur
4PhD student, Seth GS Medical College and KEM Hospital, Parel, Mumbai, 400 012, India
*Corresponding author: Raakhi Tripathi. Associate Professor, Seth GS Medical College and KEM Hospital, Parel, Mumbai, 400 012, India.
Received: 26 January 2023; Accepted: 02 February 2023; Published: 27 February 2023
Article Information
Citation: Sharmila Jalgaonkar, Raakhi Tripathi, Nishtha Khatri, Rachana Patankar, Snehalata Gajbhiye, Mohsin Sayyed, Arun Shankar. Analgesic, Anti-inflammatory, and Antioxidant potential of S-adenosyl L-Methionine on Nitroglycerine induced Migraine in mice Models.Journal of Pharmacy and Pharmacology Research. 6 (2023): 20-27.
View / Download Pdf Share at FacebookAbstract
S-adenosylmethionine (SAMe) has an antioxidant, anti-inflammatory and DNA methylation modulating effect. This study investigated the above effects of SAMe in Nitroglycerine (NTG) induced acute and chronic migraine mice model. Migraine was induced by injecting Nitroglycerine 10mg/kg. Mice were fed with SAMe in doses of 150 mg/kg and 250 mg/kg. Positive control (PC) groups received Sumatriptan (0.6 mg/kg) for acute model and propranolol (20 mg/kg) for chronic model. To assess the effect of SAMe, neurobehavioral tests (Hot plate test and 2-chambered box test) and biochemical tests (MDA & TNF-α and DNA methylation) were conducted. WithSAMe 250 mg/kg, there was a significant increase in withdrawal latency and increased time spent in light box versus disease control and it was comparable to sumatriptan (p>0.05) at 2hrs. In chronic migraine model, the improvement was consistent from Day 7 for both doses of SAMe. Treatment with SAMe with both doses recovered levels of MDA and TNF-α whereas 5-methylcytosine (5-Mc) levels showed significant rise only for SAMe 250 mg/kg group.Results of this study showed that SAMe 250 mg/kg dose has a potential to relieve pain for both acute and chronic migraine. The therapeutic mechanism for this effect could be due to its ability to decrease inflammatory markers like TNF-α, to decrease the oxidative stress by reducing MDA levels and restoring the DNA methylation changes.
Keywords
TNF-alpha, MDA, 5-mc, SAMe, Hot plate method, Two chambered box
Article Details
Abbreviations:
SAMe: S-adenosyl L-Methionine; NTG: Nitroglycerine; NMDA: N-methyl-D-aspartate; NSAID: Non-Steroidal Anti-Inflammatory Drugs; TCI: Tricyclic antidepressant; ACEI: Angiotensin-converting enzyme inhibitors; ARB: Angiotensin II Receptor Blockers; CGRP: Calcitonin Gene-Related Peptide; NO: Nitric Oxide; CPCSEA: Committee for the Purpose of Control and Supervision of Experiments on Animals; DNA: Deoxyribonucleic Acid
I.P: Intra Peritoneal; ANOVA: Analysis of Variance; MDA: Malondialdehyde; TNF-α: Tumor necrosis factor; DC: Disease control; NC: Normal control; SAMe 150: SAMe 150 mg/kg; SAMe 250: SAMe 250 mg/kg; Suma: Sumatriptan; Prop: Propranolol
1. Introduction
Migraine is a debilitating disorder and a common cause of reduced quality of life in patients. [1]. Migraine ranks among the top 20 causes of disability globally and is one of the forefronts causes of disease burden among women of 15-44 years of age [2]. On an average, migraine affects almost 1 in 10 individuals with a twice higher female preponderance as compared to males [3]. Evidence suggests that the prevalence of migraine in western countries is 11.4 % which closely mirrors the global prevalence of 11.6%. Understanding of complex migraine pathophysiology has improved significantly since last decade. Migraine is no longer thought of as primarily a vascular abnormality but instead as a disorder of neuronal excitability. Cortical spreading depression (CSD) is neural depolarization spreading across the cortex leading to a release of neurotransmitters and regional changes in blood flow, likely contributing to aura phase of migraine. Activation of the trigeminovascular system is considered as mechanism for headache. Input from the meningeal vessels (via adenylate cyclase-activating polypeptide and calcitonin gene-related peptide) pass through the trigeminal ganglion and eventually synapse onto thalamic neurons, contributing to migraine pain [4].
Apart from these pathogenic mechanisms, neuroinflammation and oxidative damage are now emerging as new theories for migraine pathogenesis. Various cytokines, including tumour necrosis factor (TNF), interleukin 1 (IL-1) and adiponectin, have been implicated in inflammation, modulation of the pain threshold, trigeminal nerve fibre sensitization, and ultimately the precipitation of migraine [5]. The metabolic changes in the cerebral cortex associated with intracellular calcium overload during CSD increase need of oxygen which could induce oxidative stress and change levels of oxidative biomarkers like glutathione (GSH), dismutase, catalase [6, 7]. Literature search also suggested that epigenetic mechanisms may contribute to CSD by changingneuronal plasticity and neuroprotection which is mainly associated with FHM mutation. Changes in episodic to chronic migraine can be associated with changes in the DNA methylation profile most strongly associated with CpG site at the SH2D5 gene, which is involved in the regulation of synaptic plasticity [8].
Pharmacological therapy is important for treatment of both acute attack and also as preventive therapy for chronic migraine [9]. Use of acute medications like ergotamine, triptans, NSAIDS, is thought to cause a rebound headache [10, 11]. Moreover, these drugs cannot be given in about 10-15% of patients on account of side effects contraindications. Most of the drugs used for prophylaxis of migraine are off label drugs as there are limited numbers of approved medications. Most widely used classes for prophylaxis of chronic migraine are beta blockers (propranolol), antidepressants (amitriptyline), Calcium channel blockers (verapamil, flunarizine) and drugs acting through other mechanisms (topiramate, gabapentin, eptinezumab etc) [12, 13]. These drugs lead to failure in the clinical practice due to poor efficacy and tolerability. Hence, researchers are looking for new alternatives targeting newer pathophysiological mechanisms in the treatment of migraine. Targeting inflammatory, oxidative and methylation pathway could be promising approach for development of new drugs.S-adenosylmethionine is final methyl donor in DNA methylation reaction.DNA methylation, as an epigenetic mechanism, plays a pivotal role in the regulation of gene transcription and establishment and maintenance of cellular identity, which is disrupted in many diseases including migraine. Due to its effect on DNA methylation, SAMe has been commissioned for use in diseases such as Parkinson’s disease, Alzheimer’s disease, depression, epilepsy,migraine and several other neurological diseases. In various studies SAMe have shown to exert anti-inflammatory, antioxidant effect apart from alternation of DNA methylation in various areas of brain.Due to its wide range of actionsi.e., antioxidant, anti-inflammatory SAMe can be a promising drug in migraine [14, 15]. DNA methylation is functionally involved in chronic pain and has possible implications for the development of future pain treatment strategies. SAMe have shown to have analgesic effect in neuropathic pain model.
The present study was planned to evaluate the antimigraine effect of SAMe in nitroglycerine (NTG) induced acute followed by chronic migraine in mice. The proposed underlying mechanism i.e., analgesic,anti-inflammatory, antioxidant and change in DNA methylation were explored by determining TNFα, MDA and 5 Methylcytosine levels in the whole brain.
2. Material and Methods
Institutional Animal Ethics Committee permission was taken before the commencement of the study under the reference number IAEC/12/2018. The study was conducted in accordance with the ARRIVE and Committee of Purpose of Control and Supervision of Experimentation in Animals (CPCSEA) guidelines. Equal numbers of male and female mice were randomly bred in the Central Animal House of Seth GS Medical College & KEM Hospital, Mumbai. A total of 48Swiss albino mice were used in the study. Equal numbers of male and female mice weighing 20-30 grams were randomly bred. The animals were housed in air-conditioned rooms with a temperature of 22 ± 3° C and relative humidity of 30-70 %. Each animal was housed in a single cage during the study. Cages had grilled stainless-steel top with food cabinet and could hold a polypropylene bottle for purified drinking water.
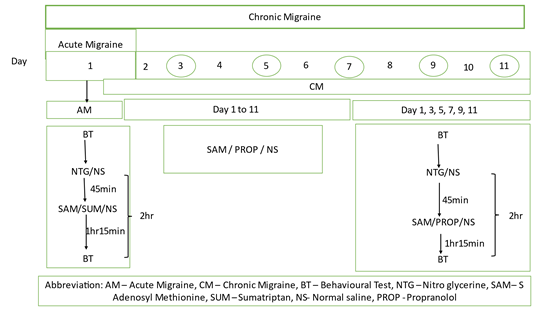
2.1 Experimental design [Fig:1]
For the experimental model of acute migraine i.e day 1 followed by chronic migrainei.e., day 1 to 11, there were five groups consisting of six mice in each group. The groups were as follows-
Group-1: Normal control: Received 0.9% normal saline, intraperitoneally on all days from day 1 to 11.
Group-2: Disease control: Received nitroglycerine(10mg/kg) intraperitoneally on day 1,3,5,7,9,11.
Group-3: Positive control: Received sumatriptan (0.6mg/kg) [16] intra peritoneally 45 minutes after nitroglycerine 10 mg/kg intraperitoneally on day 1 for acute migraine and received propranolol (20mg/kg) [17] intraperitoneally from day 2 to 11, 45 minutes after nitroglycerine(10 mg/kg) induction of chronic migraine( on the days when NTG was administered).
Group-4: Received S-adenosyl methionine (150mg/kg) [18] daily from day 1 to 11. On day 1,3,5,7,9,11 mice received S-adenosyl methionine (150mg/kg) 45 minutes after nitroglycerine (10 mg/kg).
Group-5: Received S-adenosyl methionine (250mg/kg) [18] daily from day 1 to 11. On day 1,3,5,7,9,11 mice received S-adenosyl methionine (250mg/kg) 45 minutes after nitroglycerine (10 mg/kg). The doses of all study drugs were selected from the previous literature.
The inducing agent, Nitroglycerine (NTG), was prepared in 30% alcohol, 30% propylene glycol and water. It was then freshly diluted in 0.9% saline to get a dose of 10 mg/kg. The chemical SAMe used was procured from Bangalore Sales Corporation, India and other chemicals were procured from Sigma-Aldrich. Injections were administered in 10mL/kg volume intraperitoneally.
2.2 Evaluation methods:
Behavioural assessment- Hot Plate test [19]:
Mice were placed on a heated metallic plate, which was set thermostatically at 52.5 ± 0.1 °C. Reaction time was measured in seconds (s) with a stopwatch just before (baseline) and 2 hrs after administration of NTG. The endpoint used was licking of the hind paws or jumping. A cut-off time of 45s was adopted to prevent injury to the paws of the animal.
Two chambered test [20]:
Light-aversive behaviour was examined in mice just before (baseline) and 2hrs after NTG injection. Before testing, mice were acclimatized at least 1 hr in their cages in the testing room with the lights on. Mice were placed on the dark side and the door connecting dark and bright sides were opened after 3 seconds. Door was used so that the mice do not enter lit chamber immediately with their motivation to escape from the experimenter. Time spent in the light chamber of the box was measured for a 10-min period and expressed in seconds (s).
Biochemical studies [21]:
All mice were sacrificed on day 11by administering 50 mg/kg Thiopentone sodium intraperitoneally. The whole brain of the mice was dissected out and used for biochemical estimation.
Malondialdehyde (MDA) estimation, Tumor Necrosis Factor-α (TNF- α) estimation:
Whole brain was used for biochemical analysis. The brain tissue was kept in cooled 0 .9% saline in a small container which was then kept on ice, blotted on filter paper and then weighed and homogenized in cold phosphate buffer (0.05 M, pH 7.4) using the Bead Mill instrument.The homogenates were centrifuged at 10,000 rotations per minute (rpm) for 10 min at 4ºC. The supernatant obtained was again centrifuged at 15,000 rpm for 1 hour at 4ºC. MDA was estimated in nmoles/ml, whereasTNF- α was estimated in pg/ml.
DNA methylation assay [22]:
Remaining volume of the supernatant sample from the above procedure was used for DNA elution. 20 mg of tissue was cut into pieces before lysing with proteinase K and buffer TG-A in 56 OC water bath, then TG-B was added and the solution was kept in a 70ºC water bath for 10 minutes. The reaction was stopped by adding 100% ethanol, and the solution was transferred to collection tubes. The tubes were centrifuged, the DNA was washed in buffers WAG and WB1, and the final DNA pellet was re-suspended in Tris-EDTA. The DNA was then checked for purity using the Nanodrop spectrophotometer. The extracted DNA was then used for DNA methylation assay. The MethylFlashTM Global DNA Methylation (5-mc) was estimated colorimetrically by ELISA kit. In this method, the DNA was bound to strip wells that are specifically treated to have a high DNA affinity. The methylated fraction of DNA was detected using capture and detection antibodies and then quantified colorimetrically by reading the absorbance in a microplate spectrophotometer at 450 nm.
2.3 Statistical analysis:
Data was analysed using SPSS version 25. Level of significance was set at p< 0.05. The results were expressed as mean (+/-) standard deviation. Repeated measures ANOVA followed by post-hoc Tuckey test was used for neuro-behavioral tests for chronic migraine model and one-way ANOVA followed by post-hoc Tuckey test for acute migraine model (neuro-behavioral tests) and analysis of biochemical tests.
3. Results
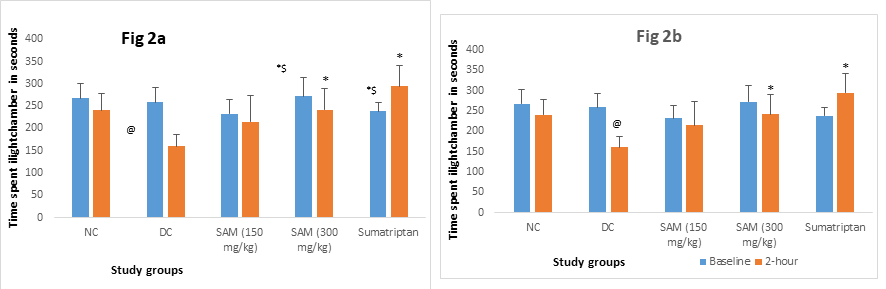
We evaluated the paw withdraw latencies (Fig 2a, hot plate test) and time spent in light chamber (Fig 2b, two chambered box test). In the acute migraine phase, injection of NTG significantly reduced paw withdrawal latencies and time spent in the light chamber in DC group at 2 hrs. With SAMe 250 mg/kg there was a significant increase in withdrawal latency (p<0.05) and increased time spent in light box (p<0.05) versus DC at 2hrs. In both these behavioral tests, the results of the above-mentioned variables for SAMe 250 mg/kg group were comparable to PC sumatriptan at 2hrs (p>0.05). There was no improvement in SAMe 150 mg/kg group in acute migraine phase on day 1.
Legendn=6, values expressed as mean ± SD.
Between group comparison for 2-hr readings: One way ANOVA followed by post hoc Tukey's test. @ - p = <0.001 vs Normal control;*-p = < 0.05 vs disease control, $-p = < 0.05 vs SAM 150 mg/kg,
NC: Normal Control, DC: Disease Control, SAM: S-adenosylmethionine
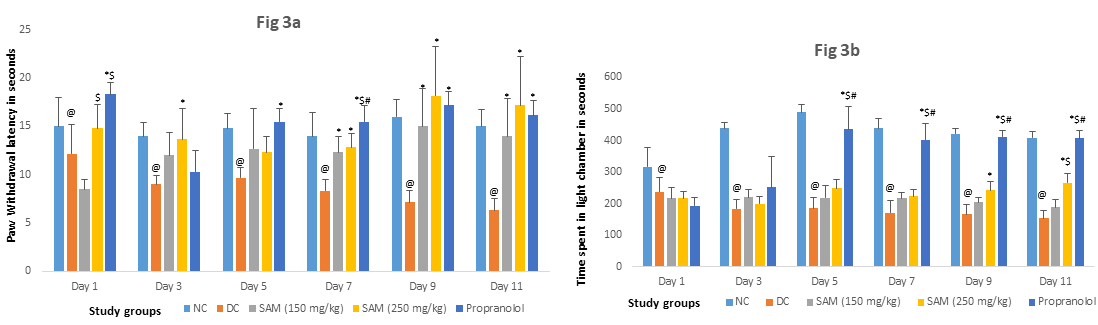
Repeated injection of NTG in the chronic migraine model significantly reduced the paw withdrawal latencies and time spent in the light chamber which suggested the development of thermal hyperalgesia and photophobia. However, following treatment with SAMe for 11 days, there was a significant improvement in paw withdrawal latencies compared to DC group in the hot plate test (p<0.005). In the two chambered box, there was significant increase in the time spent in light chamber for 250 mg/kg dose of SAMe vs DC group. (p<0.001). The improvement was consistent from day 3 to day 11. The results for 250 mg/kg dose of SAMe were comparable to propranolol for hot plate test (p>0.05). In chronic migraine model, the improvement was consistent from Day 7 for both doses of SAMe Fig:3.
Legend: Each day between group comparison: One way ANOVA followed by post hoc Tukey' s test.*-p = < 0.05 vs disease control, same day, $-p = < 0.05 vs SAM 150 mg/kg, same day,#-p = < 0.05 vs SAM 250 mg/kg, same day.Within group comparison: Repeated measures ANOVAp = <0.05 statistically significant over 11 days time within the group; NC: Normal Control, DC: Disease Control, SAM: S-adenosylmethionine
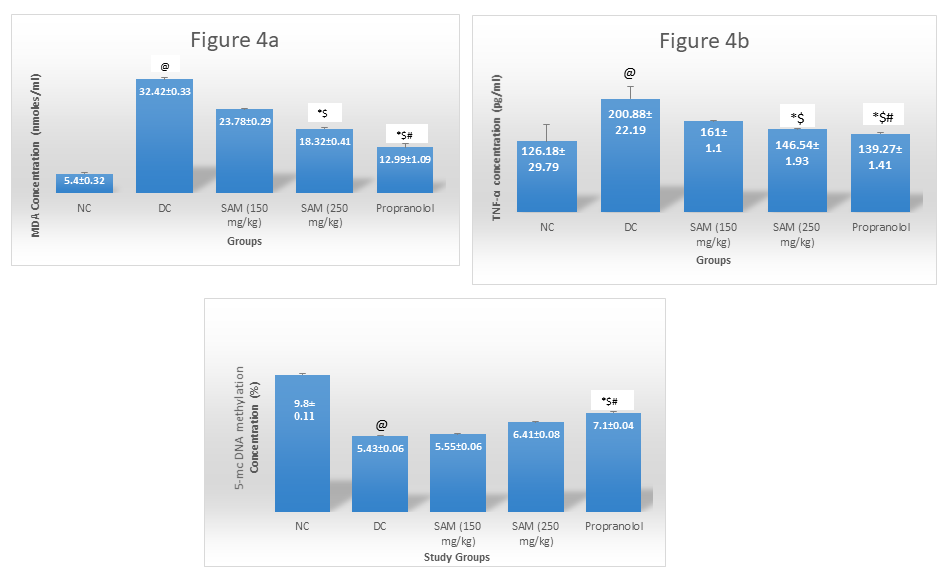
MDA and TNF-α were increased and 5 Methylcytosine levels were decreased in Disease Control group indicating general oxidative stress, inflammation and hypomethylationin chronic migraine model. Treatment with both doses of SAMerecovered the levels of MDA and TNF-αwhereas 5 Mc levels showed significant rise only for SAMe 250 mg/kg group, compared to DC group [Fig 4].
Legend: n=6, values expressed as mean ± SD. One way ANOVA followed by post hoc Tukey' s test
@ - p = <0.001 vs Normal control; *-p = < 0.05 vs disease control, $-p = < 0.05 vs SAM 150 mg/kg, #-p = < 0.05 vs SAM 250 mg/kg, NC: Normal Control, DC: Disease Control, SAM: S-
4. Discussion
A systematic review conducted by Woldeamanuel et al. found global prevalence 11.6% with pattern of rising global migraine prevalence. Migraine has complex pathophysiology with main focus on trigeminovascular system as the main structure causing migraine headache and its related symptoms. However, accumulative evidence also suggests oxidative stress playing significant role in migraine pathogenesis characterized by elevated MDA levels. Apart from oxidative stress, neuroinflammation with elevated levels of cytokines including TNF-α and epigenetic modification of certain genes may have role in pathogenesis of migraine [23]. The low methylation trend overall especially in RAMP1 gene have been observed in migraine patients. Migraine patient have shown high levels of homocysteine [24].
Management of migraine is multidisciplinary approach encompassing risk factor modification, effective treatment for acute attack followed by prophylactic treatment to reduce the headache frequency, severity and associated disability. Discontinuation rates are high for patients who initiate oral preventive therapy due to lack of efficacy and adverse events of drugs used for prophylaxis highlighting an unmet need for improved preventive therapies [25]. Nutraceuticals and vitamins like riboflavin are postulated to have role for the migraine treatment as they effectively relieve pain and are well tolerated compared to conventional pharmacotherapy. SAMe is one of the nutraceuticals currently for treatment of depression, osteoarthritis, hepatic disorders. It has shown to have an antioxidant and anti-inflammatory effects as it have shown to decrease oxidative and inflammatory markers like MDA, TNF-α in invitro as well as in in vivo studies. SAMe supplement reduces homocysteine levels and also corrects the methylation status of genes involved in pathogenesis of migraine headache [26].In the current study for acute migraine phase behavioral tests like hot plate test and 2-chambered box test were performed after administration of NTG. These tests determined pain and aura (pain due to aura induced by presence of light) respectively which was essential to determine analgesic property of our test drug SAMe in migraine headaches. The tests were performed for both acute and chronic migraine model, where recovery from migraine was assessed by latency in hind paw licking and time spent in light chamber for hot plate & 2-chambered box respectively. Animal models of NTG induced migraine are clinically relevant as the symptoms produced closely mimic the symptoms of migraine seen in human. NTG induced migraine model in previous studies have shown to induce pain, aura as well as elevated levels of MDA, TNF-α. Hence this model was considered to be appropriate to study therapeutic effect as well as to explore the intended mechanisms of SAMe.
The pain and aura induced by NTG in acute model were sustained till 2 hours. Similar results were seen in a study by Bates et al in which a dose of 10 mg/kg, NTG-induced thermal hypersensitivity developed within 30 min, peaked at 60 min, and subsided by 4 h after injection [16]. In this study authors also found that sumatriptan reduces thermal and mechanical hyperalgesia. This justifies the selection of sumatriptan as positive control for acute migraine model of our study. In our study as well, positive control sumatriptan decreased the thermal hyperalgesia. There was an increase in withdrawal latency and time spent in light chamber box at 2hr for high dose of SAMei.e., 250mg/kg. Results of the study indicated that SAMe in high dose may relieve pain and aura in the acute migraine.Chronic intermittent administration of NTG results in a progressive and sustained basal hyperalgesia along with acute hyperalgesia with each NTG dose which can be blocked by propranolol but not by Sumatriptan. Hence propranolol was used as a positive control for the current study. It has been recommended that chronic NTG model could be used as an additional tool for the relatively rapid screening of potential preventive compounds. The proposed compound SAMe in both doses have shown considerable improvement in basal hyperalgesia in chronic migraine model.
Migraine has complex pathophysiology and hence various genetic, neuromodulatory, inflammatory, neuroimaging biomarkers have been suggested for the diagnosis as well as prognosis of migraine. Among these MDA for oxidative stress and TNF-α for neuroinflammation [21] have been used previously in isoproterenol induced CHF experimental model. In migraine MDA as well as TNF-α levels are elevated. As SAMe possesses both antioxidant as well as anti-inflammatory property it has shown decrease in levels of both of these biomarkers.
DNA methylation/demethylation regulates gene expression and is required for normal brain development. Disturbance in DNA methylation are associated with a number of cardiovascular, autoimmune, neurological diseases and cancer. Epigenetic studies of migraine have shown the link between changes in DNA methylation in various genes involved in migraine pathophysiology. There are elevated homocysteine levels in patients with migraine with aura. In a randomised controlled trial conducted in patients having raised homocysteine levels and frequent attacks of migraine showed reduced level of homocysteine and decreased migraine attacks after supplement of Dietary folate and Vitamin B12 [27]. Studies have shown that supplement with SAMe bring back levels of homocysteine to normal level thus improving methylation status which is decreased if there is global hypomethylation. 5-methylcytosine (5-MC) is a validated biomarker for DNA methylation [26]. In our study we found decreased 5-MC levels in brain indicating global hypomethylation in rats with NTG induced migraine [28]. Study drug SAMe showed increase in 5 MC levels thus improving the methylation status in migraine.
Limitations of the study
The study has only 6 mice per groups and the study results are only exploratory in nature. Although we have tried to include important biomarkers involved in pathophysiology of migraine, other important biomarkers like CGRP could not be included in the study. Similarly, to explore epigenetic mechanism we have considered global methylation in the whole brain region, instead a specific region in the brainwith genes involved in migraine pathophysiology could be studied for more specificity. Further studies can be taken to explore this in-depth mechanism of SAMe for its beneficial effect in migraine.
Conclusion
Results of the study indicate that SAMe 250 mg/kg dose has a potential to relieve migraine pain due to its analgesic effect and is also potential candidate for prophylaxis of migraine. The therapeutic mechanism for its overall effect could be due to its ability to decrease inflammatory markers like TNF-α, to decrease the oxidative stress by increasing GSH levels. Another important mechanism for the drug SAMe could be to restore the DNA methylation changes in brain regions.
References
- Abu Bakar N, Tanprawate S, Lambru G, Torkamani M, Jahanshahi M, Matharu MS. Quality of life in primary headache disorders: A review. Cephalalgia 36(2016):67-91.
- Pressman A, Jacobson A, Eguilos R, Gelfand A, Huynh C, Hamilton L, et al. Prevalence of migraine in a diverse community—electronic methods for migraine ascertainment in a large integrated health plan. Cephalalgia 36(2016):325.
- Woldeamanuel YW, Cowan RP. The impact of regular lifestyle behavior in migraine: a prevalence case-referent study. J Neurol 263(2016):669-76.
- Mungoven TJ, Henderson LA, Meylakh N. Chronic Migraine Pathophysiology and Treatment: A Review of Current Perspectives. Front Pain Res (2021):52.
- Kursun O, Yemisci M, van den Maagdenberg AMJM, Karatas H. Migraine and neuroinflammation: the inflammasome perspective. J Headache Pain10 (2021):1-13.
- Togha M, Razeghi Jahromi S, Ghorbani Z, Ghaemi A, Rafiee P. An investigation of oxidant/antioxidant balance in patients with migraine: A case-control study. BMC Neurol 19(2019):1-10.
- Tripathi GM, Kalita J, Misra UK. A study of oxidative stress in migraine with special reference to prophylactic therapy. Int J Neurosci 128(2018):318-24.
- Khan J, Asoom LI Al, Sunni A Al, Rafique N, Latif R, Saif S Al, et al. Genetics, pathophysiology, diagnosis, treatment, management, and prevention of migraine. Biomed Pharmacother139(2021).
- Ashina M. Migraine. Ropper AH, editor. N Engl J Med. 383(2020):1866-76.
- Schulte LH, Jürgens TP, May A. Photo-, osmo- and phonophobia in the premonitory phase of migraine: mistaking symptoms for triggers? J Headache Pain 16(2015):1-5.
- Burstein R, Noseda R, Borsook D. Migraine: Multiple Processes, Complex Pathophysiology. J Neurosci 35(2015):6619.
- Estemalik E, Tepper S. Preventive treatment in migraine and the new US guidelines. Neuropsychiatr Dis Treat 9(2013):709.
- Ravishankar K, Tayade H, Mandlik R. Sublingual piroxicam in migraine without aura. Journal of The Association of Physicians of India 59 (2011):494-7.
- Jin Z, Liu Y. DNA methylation in human diseases. Genes Dis 5(2018):1-8.
- Greco R, Mangione AS, Sandrini G, Nappi G, Tassorelli C. Activation of CB2 receptors as a potential therapeutic target for migraine: evaluation in an animal model. J Headache Pain15(2014).
- Bates EA, Nikai T, Brennan KC, Fu YH, Charles AC, Basbaum AI, et al. Sumatriptan alleviates nitroglycerin-induced mechanical and thermal allodynia in mice. Cephalalgia 30(2010):170-8.
- Tipton AF, Tarash I, McGuire B, Charles A, Pradhan AA. The effects of acute and preventive migraine therapies in a mouse model of chronic migraine. Cephalalgia 36(2016):1048-56.
- Sharma A, Gerbarg P, Bottiglieri T, Massoumi L, Carpenter LL, Lavretsky H, et al. S-Adenosylmethionine (SAMe) for Neuropsychiatric Disorders: A Clinician-Oriented Review of Research. J Clin Psychiatry 78(2017):e656-67.
- Ghelardini C, Grazioli I, Uslenghi C. Indomethacin, caffeine and prochlorperazine alone and combined revert hyperalgesia in in vivo models of migraine. Pharmacol Res 46(2002):245-50.
- Markovics A, Kormos V, Gaszner B, Lashgarara A, Szoke E, Sandor K, et al. Pituitary adenylate cyclase-activating polypeptide plays a key role in nitroglycerol-induced trigeminovascular activation in mice. Neurobiol Dis 45(2012):633-44.
- Latif K, Khan AU, Izhar Ul Haque M, Naeem K. (2021) Bergapten Attenuates Nitroglycerin-Induced Migraine Headaches through Inhibition of Oxidative Stress and Inflammatory Mediators. ACS Chem Neurosci 12(2021):3303-13.
- Pfalzer AC, Choi SW, Tammen SA, Park LK, Bottiglieri T, Parnell LD, et al. S-adenosylmethionine mediates inhibition of inflammatory response and changes in DNA methylation in human macrophages. Physiol Genomics 46(2014):617-23.
- Bron C, Sutherland HG, Griffiths LR. Exploring the Hereditary Nature of Migraine. Neuropsychiatr Dis Treat 17 (2021):1183.
- Cacciapuoti F. Migraine homocysteine-related: old and new mechanisms. Neurol Clin Neurosci5(2017):137-40.
- Delussi M, Vecchio E, Libro G, Quitadamo S, De Tommaso M. Failure of preventive treatments in migraine: An observational retrospective study in a tertiary headache center. BMC Neurol 20(2020):1-12.
- Bottiglieri T. S-Adenosyl-L-methionine (SAMe): from the bench to the bedside--molecular basis of a pleiotrophic molecule. Am J Clin Nutr 76(2002).
- Menon S, Lea RA, Ingle S, Sutherland M, Wee S, Haupt LM, et al. Effects of dietary folate intake on migraine disability and frequency. Headache55(2015):301-9.
- Havliš J, Trbušek M. 5-Methylcytosine as a marker for the monitoring of DNA methylation. J Chromatogr B781(2002):373-92.


 Impact Factor: * 3.3
Impact Factor: * 3.3 Acceptance Rate: 74.39%
Acceptance Rate: 74.39%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 