Chronic Muscular Skeletal Pain Syndromes, New Models for an Old Problem. Specific and Nonspecific Pain- Time to Move on
Niemier K1,? , Schulz J1 , Emmerich J2, Liefring V2, Beyer L3
1Schmerz- und Rückenzentrum Westmecklenburg, Westmecklenburg Kliniken Helene, von Bülow, Parkstraße 12, 19230 Hagenow, Germany
2Consultant Pain Physician, Sanakliniken Sommerfeld, Klinik für Manuelle Medizin, Waldhausstraße 22, 16766 Kremmen, Germany
3Ärztehaus Mitte, Westbahnhofstraße 2, 07745 Jena, Germany
*Corresponding Author: Niemier K, Schmerz- und Rückenzentrum Westmecklenburg, Westmecklenburg Kliniken Helene, von Bülow, Parkstraße 12, 19230 Hagenow, Germany
Received: 15 January 2020; Accepted: 21 January 2020; Published: 17 March 2020
Article Information
Citation: Niemier K, Schulz J, Emmerich J, Liefring V, Beyer L. Chronic muscular skeletal pain syndromes, new models for an old problem. Specific and nonspecific pain- time to move on. Journal of Orthopaedics and Sports Medicine 2 (2020): 42-54.
View / Download Pdf Share at FacebookAbstract
Chronic muscular skeletal pain syndromes remain a common and poorly understood medical problem. The classification of specific and nonspecific pain has not proven valuable. Therefore is seems necessary to introduce a new model as the basis for further research and clinical practice. We would suggest the functional system of behavior developed by Anokhin as a basic model. It contains all relevant elements influencing the development of chronic muscular skeletal pain. The interaction of pathomorphological and psychosocial factors, somatic dysfunctions and neurophysiological changes in the peripheral and central nervous system are thought to be relevant for the development of chronic muscular skeletal pain syndromes. The complex interactions and the possibility to compensate impairments can be shown within the model. Furthermore, a subgrouping of patients might be possible. For this a standardized clinical assessment should be developed and scientifically evaluated.
Keywords
<p>Chronic muscular skeletal pain; Somatic dysfunction; Functional system; Subgrouping; Low back pain</p>
Article Details
1. Introduction
Chronic muscular skeletal pain syndromes (CMPS) are common, expensive and so far, not fully understood [1-7]. Low back pain (LBP) is divided in specific and nonspecific LBP. While specific LBP is defined by a clear pathomorphological cause for the pain (e.g. inflammation, fracture), for nonspecific LBP no cause has been defined. The diagnosis of nonspecific LBP results simply from the exclusion of a specific LBP [8]. Since the cause of the pain remains nonspecific, the treatments applied depend more on the treatment setting, on the specialty and the beliefs of the therapist and patient/therapist preferences than on clinical reasoning [9-11]. Scientific research on this basis remains somewhat nonconclusive [12]. Positive treatment outcome is only shown for multimodal treatment programs which target a variety of psychosocial, neurophysiological and functional problems [13-15]. So more than 20 years after the introduction of the “classification specific/nonspecific pain”, it must be concluded that it did not help to promote effective diagnostic or treatment for CMPS. It might be time to develop new models for the development and chronification of muscular skeletal pain in order to promote effective research and finally improve clinical outcome. For this we introduced an interdisciplinary working group consisting of physiatrist, orthopedic surgeons, psychiatrists, psychologists, general practitioners, pain specialists, rheumatologists, physiologists and manual therapists. The involved practitioners were working in out-and inpatient settings, in day units for chronic pain and at university hospitals. Over the last 2 Years the presented model was developed on the basis of literature review, clinical experience and intensive discussions. We concluded that CMPS is a multifactorial process. Pathomorphological changes, somatic dysfunctions, psychosocial factors and neurophysiological factors of nociception, transmission, perception and processing of afferences are thought to be relevant [12, 16-18]. It was found to be necessary to introduce a model to integrate all these different factors in order to explain, diagnose and treat CMPS effectively. In our opinion the functional system of behavior from Anokhin [19] might be a good model to comprehend the complexity of CMPS.
2. Functional System of Behavior
Human behavior is hedonistic; hence the main aim is the satisfaction of elementary needs, such as communication, alimentation and reproduction [20-22]. The locomotor system plays an important role for the satisfaction of these needs. Depending on the motivation, the sensory input and potential targets for a motoric action (Figure 1) [19]:
- Motoric programs are developed in the central nervous system (CNS).
- Efferent action potentials will be sent to the effector organs (e.g. muscle, cardio-pulmonary system) and
- Result in a motoric action.
- Via the afference from various receptors (e.g. muscle, tendons, joints)
- Feedback of the motoric action will be given to the CNS
Anokhin assumes a functional organ as an afference acceptor. The afference acceptor compares the result of the motoric action to the copy of the assumed result. If the afference and the assumed result differ,
the motoric program will be rectified and the process starts again (Figure 1). Today the afference acceptor is thought as a neuronal network [23]. The functional system of behavior contains all elements thought to be relevant for CMPS (pathomorphological factors, somatic dysfunction, psychosocial factors, neurophysiological changes of nociception, transmission, perception and processing of afferences). In our further discussion, we will concentrate on these different elements.
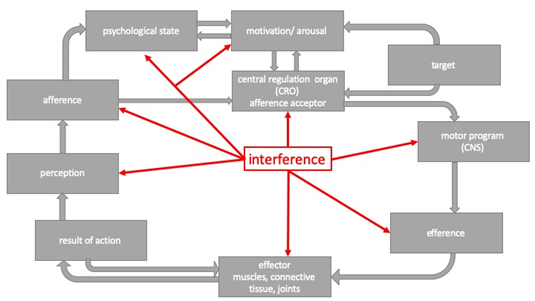
Figure 1: Model of the functional system and potential interference, adapted [20].
3. Adaptability, Compensation, Decompensation and Re-Compensation
The functional system is, as all physiological systems, not static but is constantly adapting to the needs and demands. Adpantability is therefore one of the main features of a functional system like the human body. If a functional system is not able to adapt, disease will develop [24]. Pathomorphological changes, psychosocial influences and somatic dysfunction are very common findings even in symptom free people.
Usually these findings are compensated within the functional system. If the adaptability does not suffice, or if the demands are to high, the system will decompensate, hence symptoms will develop. Reduced adaptability can result from all discussed elements of the functional system, or from other systems (e.g. cardio-pulmonary insufficiency). If treatment is successful, the system can regain balance, hence re-compensate. Prevention and
rehabilitation should focus on the adapatibility of the system and try to reduce negative influences like e.g. muscle weakness, poor coordination, poor psychological state etc., If the system can´t re-compensate chronic disease like CMPS develop. The treatment should reduce disturbances on the different influencing factors (see below) and improve adaptability.
4. Factors Responcible for the Development of CMPS
4.1 Pathomorphological factors
Degenerative changes are very common in patients with CMPS, but also in healthy subjects [25, 26]. Nevertheless, diagnosing and treating degenerative changes (e.g. medication, injections, operations) seems to be the main focus of medicine, at least in western countries [27-29]. While treating pathomorphological factors can be quite successful (e.g. hip replacement in hip arthritis, anti-inflammatory drugs in rheumatoid disease), it fails to show sustainable effects in chronic LBP and other CMPS. Furthermore, it is quite typical for CMPS to show at least initially intermittent rather than constant pain, while the degenerative changes remain stable. We would argue, that structureas a central role for normal function of the locomotor system. Structural changes influence the functional system and results in compensation mechanisms within the system. But we would also like to stress the known fact, that function determins structure. Poor function leads to poor structure as seen in many patients with CMPS, e.g. muscle wasting and osteoporosis due to inactivity. In order to evaluate the role of pathomorphological factors we would suggest dividing them according to the following scheme [30]:
- Pathomorphological changes causing
- Nociception
- Dysfunction
- Pathomorphological factors secondary to dysfunction/poor function
- Pathomorphological factors without importance for the clinical picture
Usually pathomorphological changes can be compensated within the functional system, hence adaptation of neuromodulation, motor control, psychosocial mechanisms etc. If compensation does not suffice, treatment might be necessary to improve the ability to adapt to the structural pathology and/or treat the structural pathology directly (e.g. hip replacement, surgical spine stabilization)
4.2 Somatic dysfunction
Function is the normal expression of life. The functional system of behavior includes all parts of normal function [31]:
- Biomechanical function
- Neurophysiological function
- Psychosocial function
- Interaction of the locomotor system with other systems (endocrine system etc.)
Therefore, dysfunction can be described on different levels. The international classification of function [32] describes psychosocial functioning, hence the interaction within society (participation). Sports medicine, rehabilitation etc. focus on function like walking, sitting, grip etc. as basis for a good participation. Physiotherapy, manual medicine and osteopathy diagnose and treat somatic dysfunction of structures (e.g. fascia, muscles, joints). Furthermore, pharmaceutical and physical methods influence biochemical/metabolic processes within and between cells. These different the levels are obviously connected, interact and are able to compensate dysfunction or pathomorphological changes. If the capacity for compensation is exceeded, dysfunctions become symptomatic. Typical symptoms are pain, impaired function (e.g. walking, sitting) and participation. If the dysfunctions are the central pathogenetic factor, we would suggest the term functional disorder (Figure: 3, [31]). Somatic dysfunctions are caused by a discrepancy between the loading of structureits endurance, hence the capacity to meet the actual demands. If the loading exceeds the endurance, dysfunctions or structural damages develop [31]. Frequent causes for a discrepancy between loading and endurance are:
- Input of massive force (accident)
- Morphological abnormalities (e.g. hip dysplasia)
- Morphological changes secondary to disuse (e.g. osteoporosis/weak connective tissue secondary to immobility or poor training)
- Muscle weakness (e.g. poor training condition, muscle wasting due to aging or disease)
- Deconditioning (muscles, cardiopulmonary)
- Motor control deficits
- Poor coordination of movement and posture
- Poor stabilization of movement and posture
Even a short nociceptive input can lead to changes in motor control and therefore lead to a vicious circle (Figure 2) [32-34].
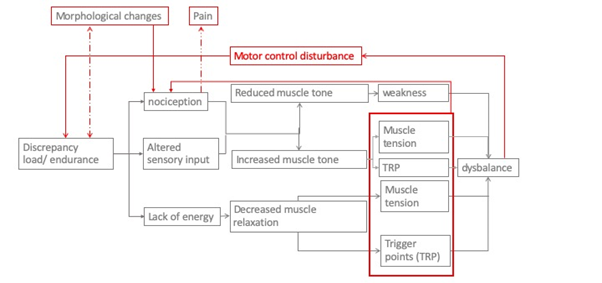
Figure 2: Model for the interaction of somatic dysfunction and development of a vicious circle sustaining functional disorder.
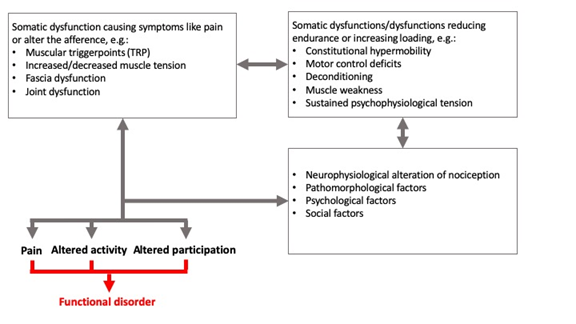
Figure 3: Model; Interaction of somatic dysfunction and other factors in the development of a functional disorder [31].
For systematic reasons we would suggest dividing somatic dysfunction into two categories [30, 31].
- Somatic dysfunctions/dysfunctions reducing endurance or increasing loading, e.g.
- Motor control dysfunction
- Deconditioning
- Constitutional hypermobility
- Sustained psychophysiological tension
- Somatic dysfunction causing symptoms like pain and/or alter the afference, e.g.
- Muscular triggerpoints (TRP)
- Increased/decreased muscle tension
- Fascia dysfunction
- Joint dysfunction (hyper-/hypomobility)
If not compensated, the interaction of these somatic dysfunctions and other factors lead to the suggested functional disorder (Figure 3) [4, 31, 35]. For patients with CMPS the role of somatic dysfunction for the disease should be assessed and be treated accordingly. It is important to include the different levels of function into the assessment.
4.3 Psychosocial factors
The influence of psychosocial factors on CMPS has been extensively explored. It has been shown, that psychological disorders like depression or anxiety, dysfunctional cognition and behavior and social factors like compensation claims/secondary gain from illness influence the development of CMPS and treatment outcome and vice versa. Within the model of the functional system, psychosocial factors can influence the development of CMPS in various ways [36-42].
- Altering muscle tension and posture and therefore causing somatic dysfunction, e.g.:
- Depression
- Anxiety
- Avoidance and endurance beliefs/behavior
- Altering processing of afferences and nociceptive input, e.g.:
- Depression
- Anxiety
- Decrease motivation for change, e.g.:
- Depression
- Compensation claims/secondary gain from illness
- External control beliefs
- Inability to participate on/comply with treatment, e.g.:
- Depression
- Anxiety
- Personality disorder
- Drug/medication addiction
- Transmission of psychological tension into somatic symptoms, e.g.:
- Somatoform pain disorder
- Somatization disorder
- Dissociative disorder
- Misinterpretation of afferences/somatic symptoms, e.g.:
- Panic disorder
- Hypochondria
As with pathomorphological factors, it is important not only to describe psychosocial factors, but to evaluate the influence on the disease for each individual patient.
4.4 Neurophysiological changes of nociception, transmission, perception and processing of afferences (neuromodulation)
It is well known, that neurophysiological changes of neuromodulation taking place early in the development of CMPS. Functional and structural changes have been demonstrated in the peripheral as well as in the central nerve system (PNS, CNS; [43-49]). It was postulated, that chronic pain may become independent from nociceptive afferences. While some pain syndromes are completely generated by the CNS (e.g. phantom pain), so far it is not possible to proof the existence of completely centrally generated CMPS.
Three different stages in the development of chronic pain have been postulated [50].While the first 2 stages are dependent from nociceptive input, stage 3 characterize a centrally produced pain independent from any nociceptive input (Figure 4). Clinically and neurophysiologically it is possible dividing the first two stages, stage 3 remains a hypothesis. In our view, it is rather unlikely to have a completely centrally generated CMPS. In the model of the functional system, changes in the PNS and CNS result from the altered sensory input including but not solely nociception. Functional and (reversible) structural alterations will not only affect the nociceptive system, but also the whole functional system including the sensorimotor system, behavior etc.
Therefore, we postulate that patients with CMPS can have various degrees of neurophysiological alteration of the PNS and CNS. The majority of these changes are functional and reversable, hence part of the discussed functional disorder. The assessment of neurophysiological alteration might be important to target the functional and medical treatment to patients.
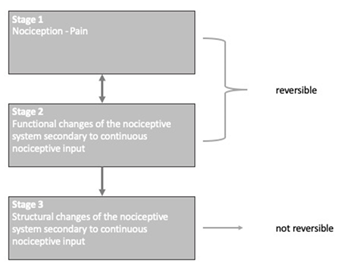
Figure 4: Stages of development of chronic pain; stage 3 hypothetical [51].
5. Subgroups Instead of Specific/Nonspecific Painh4>
On the basis of the model of the functional system, we would propose to develop a system to subgroup patients. The 4 described elements considered relevant for the development of CMPS ought to b included in the subgrouping prozess (Figure 5), [17. 51].
- pathomorphological factors
- somatic dysfunction
- psychosocial factors
- neurophysiological changes of nociception, transmission, perception and processing of afferences (neuromodulation)
The clinical and scientific focus should be shifted from simple one dimensional to a multidimensional view. The binding element within the whole system is the function on all levels. The different factors should be assessed for each individual patient. In order to obtain reliable and valid results. A standardized assessment system should be developed and scientifically evaluated. Attempts on such assessement systems have been made for tertiary medical care, but further development and research is required [51, 52].
On the basis of the subgroups, treatment can be targeted at patients with CMPS and research results for treatment methods become comparable. Furthermore, many patients present in daily practice with acute pain. Taking the history accurately, it reveals often one or more episodes of recurrent pain. In an acute episode, usually a triggering factor can be found, but underlying factors within the whole functional system are often overlooked. An early assessment and treatment of all elements might help to prevent the development of CMPS.
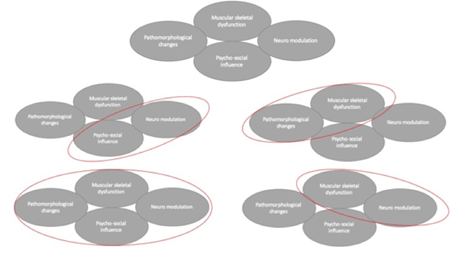
Figure 5: Elements of the development of CMPS and examples for possible subgroups.
6. Discussion and Conclusion
CMPS are multifactorial syndromes. The actual division in specific and nonspecific pain syndromes has not proven valuable. In our view it is important to move the function into center of attention. Function, from the cellular to the complex psychosocial functioning is the expression of life. Dysfunction and structural changes can be compensated by functional adaptation. If adaptation does not suffice disease develops. Improving function on all levels is central for prevention, rehabilitation and treating CMPS. On the basis of the model of the functional system of behavior and the main pathophysiological elements of CMPS, subgrouping of patients seems neccessary. For this, a standardized assessment procedure should be developed and introduced after scientific evaluation. On the basis of a valid assessment
- treatments and rehabilitation can be directed correctly
- prevention of CMPS is possible
- treatment method can be evaluated scientifically
An example for effective subgrouping for scientific research and clinical practice are the Criteria of the American College of Rheumatology for rheumatic diseases [52]. Therefore, further research should focus on subgrouping CMPS and its validation.
References
- Buchbinder R, van Tulder M, Öberg B, et al. Low back pain: a call for action. Lancet 391 (2018): 2384-2388.
- Hartvigsen J, Hancock MJ, Kongsted A, et al. Low back pain 1. What low back pain is and why we need to pay attention Lancet 391 (2018): 2356-2367.
- Foster NE, Anema JR, Cherkin D, et al. Low back pain 2 Prevention and treatment of low back pain: evidence, challenges, and promising directions. Lancet 391 (2018): 2368-2383.
- Buchbinder R, Blyth FM, March LM, et al. Placing the global burden of low back pain in context. Best Pract Res Clin Rheumatol 27 (2013): 575-589.
- Global Burden of Disease Study 2013 Collaborators. Global, regional and national incidence, prevalence and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. The Lancet 386 (2015): 743-800.
- Kohlmann T, Buchholz I. Epidemiologie von Schmerzerkrankungen des Bewegungssystems. In Niemier K, Seidel W, Psczolla M, Ritz W, Holtschmit, JH, Steinmetz A (Hrsg). Schmerzerkrankungen des Bewegungssystems Multimodale interdisziplinäre Komplexbehandlung. Degruyter 1 (2018): 12-31.
- Statistisches Bundesamt. Krankheitskosten - 2002, 2004, 2006 und 2008. Wiesbaden: Statistisches Bundesamt (2010).
- Deyo RA, Rainville J, Kent DL. What can the history and physical examination tell us about low back pain? JAMA 268 (1992): 760-765.
- Fullen BM, Doody C, David Baxter G, et al. Chronic low back pain: non-clinical factors impacting on management by Irish doctors. Ir. J. Med. Sci 177 (2008): 257-263.
- Fullen BM, David Baxter G, O`Donnovan BGG, et al. Doctors attitudes and beliefs regarding acute low back pain management: A systematic review. Pain 136 (2008): 388-396.
- Cherkin DC, Deyo RA, Wheeler K, et al. Physician variation in diagnostic testing for low back pain: who you see is what you get. Arthritis Rheum 37 (1994): 15-22.
- Niemier K. Multimodal, polypragmatisch und kostenintensiv - Rückenschmerzbehandlungen auf dem Prüfstand. Manuelle Medizin 50 (2012): 16-27.
- Nagel B, Korb J. Multimodale Schmerztherapie des Rückenschmerzes. Nachhaltig wirksam und kosteneffektiv. Orthopäde 38 (2009): 907-912.
- Kohles S, Barnes D, Gatchel RJ, et al. Improved physical performance outcomes after functional restoration treatment in patients with chronic low-back pain. Spine 15 (1989): 1321-1324.
- Hildebrandt J, Pfingsten M, Saur P, et al. Prediction of success from a multidisciplinary treatment program for chronic low back pain. Spine 22 (1997): 990-1001.
- Steinmetz A, Psczolla M, Seidel W, et al. Effect of subgroup-specificmultimodaltherapy on chronic spinal back pain and function-a prospective inpatient multicentre clinical trial in Germany Medicine 98(2019): 13825.
- Niemier K, Seidel W, Ritz W, et al. Sommerfeld Assessment System: Introduction and Evaluation of an Multiprofessional Assessment System for the Differential Diagnosis of Chronic Muscolosekletal Pain Syndromes. JOM 25 (2003): 21.
- Niemier K, Seidel W, Marnitz U. Chronische Rückenschmerzen – entzündlich, funktionell, psychosomatisch? OUP 5 (2016): 000-000.
- Anochin PK. Systemogenesis as a General Regulator of Brain Development. Progress in brain research 9 (1964): 54 -86.
- Jeannerod, M. The mechanism of self-recognition in human. Behavioral Brain Research 142 (2003): 1-15.
- Karsh K, Eitam B. I control therefore I do: Judgments of agency influence action selection. Cognition 138 (2015): 22-131.
- Haggard P. Conscious intention and motor cognition. Trends in cognitive science 9 (2005): 290-295.
- Von Heymann W, Locher H. Neurophysiologische Grundlagen von Schmerzen am Bewegungssystem. In Niemier K, Seidel W,Psczolla M,Ritz W,Holtschmit, JH, Steinmetz A (Herausgeber). Schmerzerkrankungen des Bewegungssystems Multimodale interdisziplinäre Komplexbehandlung. Degruyter 1 (2018): 61-77.
- Beyer LB, Niemier K. Funktionsstörungen am Bewegungssystems. Funktionelle Regiabilität als Grundlage eines optimalen Bewegungsresultats. Manuelle Medizin 4 (2018).
- Boden SD, Davis DO, Dina TS, et al. Abnormal -resonance scans of the lumbar spine in asymptomatic subjects. A prospective investigation.J Bone Joint Surg Am 72 (1990): 403-408.
- Deyo R, Mirza S, Turner Y. Overtreating chronic back pain: time to back off? J Am Board Fam Med 22 (2009): 62-68.
- Lang E, Eisele R, Jankowsky H, et al. Ergebnisqualität in der ambulanten Versorgung von Patienten mit chronischen Rückenschmerzen Der Schmerz 14 (2000): 146-159.
- Dietl M, Korczak D. Versorgungssituation in der Schmerztherapie in Deutschland im internationalen Vergleich hinsichtlich Über-, Unter- oder Fehlversorgung. Schriftenreihe Health Technology Assessment 111 (2011): 864-9645.
- Bitzer EM, Lehmann B, Bohm S, Pries H-W. Barmer GEK Report Krankenhaus 2015. Asgard Verlagsservice GmbH (2015).
- Niemier K. Hogräfe HC. Chronische cervicale Schmerzsyndrome. Vorstellungeines multimodalen interdisziplinären stationären Behandlungskonzepts (ANOA-Konzept). Akt Rheumatol 40 (2015): 1-9.
- Niemier K, Seidel W, Liefring V, et al. Von der Funktionsstörung zur Funktionskrankheit. Manuelle Medizin - Was ist der therapeutische Ansatzpunkt. Manuelle Medizin 56 (2018): 253-258.
- Summers SJ, Schabrun SJ, Hirata RP,et al. Effect of sustained experimental muscle pain on joint position sense. Pain Reports 4 (2019): 2471-2531.
- Qu N,Lindstrom R,Graven-Nielsen T, et al. Experimental cervical interspinous ligament pain altered cervical joint motion during dynamic extension movement Clinical Biomechanics 65 (2019): 65-72.
- Suda EY,Hirata RP, Palsson T, et al. Experimental knee-related pain enhances attentional interference on postural control European Journal of Applied Physiology 119(2019): 2053-2064.
- Niemier K, Seidel W, Psczolla M, etal. Schmerzerkrankungen des Bewegungssystems Multimodale interdisziplinäre Komplexbehandlung. Degruyter (2018).
- Ritz W. Psychosoziale, psychologische und psychosomatische Aspekte. In Niemier K, Seidel W, Psczolla M, Ritz W, Holtschmit, JH, Steinmetz A (Hrsg.). Schmerzerkrankungen des Bewegungssystems Multimodale interdisziplinäre Komplexbehandlung. Degruyter 1 (2018): 39-54.
- Gatchel RJ,Peng YB,Peters ML, et al. The biopsychosocial approach to chronic pain: scientific advances and future directions. Psychol Bull 133 (2007): 581-624.
- Hasenbring MI, Hallner D, Klasen B, et al. Pain-related avoidance versus endurance in primary care patients with subacute back pain: psychological characteristics and outcome at a 6-month follow-up. Pain 153 (2012): 211-217.
- McCracken LM, Vowles KE. Psychological flexibility and traditional pain management strategies in relation to patient functioning with chronic pain: an examination of a revised instrument.J Pain 8 (2007): 700-707.
- Plaas H,Sudhaus S,Willburger R,et al. Physical activity and low back pain: the role of subgroups based on the avoidance-endurance model. Disabil Rehabil 36 (2014): 749-55.
- Linton SJ, Bergbom S. Understanding the link between depression and pain. Scand J Pain 2 (2011): 47-54.
- Hasenbring M, Marienfeld G, Kuhlendahl D, et al. Risk factors of chronicity in lumbar disc patients. A prospective investigation of biologic, psychologic, and social predictors of therapy outcome. Spine 19 (1994): 2759-2765.
- Flor H, Braun C, Elbert T, et al. Extensive reorganization of primary somatosensory cortex in chronic back pain patients. Neurosci Lett 224 (1997): 5-8.
- Diers M, Koeppe C, Diesch E, et al. Central processing of acute muscle pain in chronic low back pain patients: an EEG mapping study. J Clin Neurophysiol 24 (2007): 76-83.
- Giesecke T, Gracely RH, Grant MA, et al. Evidence of augmented central pain processing in idiopathic chronic low back pain. Arthritis Rheum 50 (2004): 613-623.
- Baliki MN, Petre B, Torbey S, et al. Corticostriatal functional connectivity predicts transition to chronic back pain. Nat Neurosci 15 (2012): 1117-1119.
- Baliki MN, Geha PY, Apkarian AV, et al. Beyond feeling: chronic pain hurts the brain, disrupting the default-mode network dynamics. J Neurosci 28 (2008): 1398-1403.
- Flor H. The functional organization of the brain in chronic pain. In Sandkühler J, Bromm B, Gebhard GF (Hrsg) Nervous system plasticity and chronic pain, progress in pain research. ElsevierAmsterdam (2000): 313-322.
- Flor H, Ebert T, Kriet S, et al. Phantom limb painas a preceptual correlate of cortical reorganization following arm amputation. Nature 375 (1995): 482-48
- Treede RD. Entstehung der Schmerzchronifizierung. In Casser H-R, Hasenbrink M, Becker A, Baron R. (Hrsg.) Rücken- und Nackenschmerzen. Interdisziplinäre Diagnostik und Therapie, Versorgungspfade, Patientenedukation, Begutachtung, Langzeitbetreuung. Springer Berlin/Heidelberg 1 (2016): 27-38.
- Niemier K, Seidel W, Ritz W, et al. Sommerfeld Diagnostic System: Introduction and preliminary evaluation of an Multiprofessional Diagnostic System for die Differential Diagnosis of Chronic Muscoloskeletal Pain Syndromes. Proceedings of the International Society of Physical and Rehabilitation Medicine, Acta Fisiatrica 12 (2005).
- https://www.rheumatology.org/Practice-Quality/Clinical-Support/Criteria, access 14.02.2020.


 Impact Factor: * 5.3
Impact Factor: * 5.3 Acceptance Rate: 73.64%
Acceptance Rate: 73.64%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 