Aqueous Solubility and Bioavailability of Albumin Conjugated Curcumin in Endothelial Cells Allege Potential of the Formulation for Anti-Inflammation Therapy
Deepa Sathee, Lissy Kalliyana Krishnan*
Division of Thrombosis Research, Biomedical Technology Wing, Sree Chitra Tirunal Institute for Medical Sciences and Technology, Thiruvananthapuram, Kerala, India
*Corresponding Author: Dr. Lissy K. Krishnan, PhD, Thrombosis Research Unit Biomedical Technology Wing SCTIMST, Trivandrum-695012, India
Received:17 December 2020; Accepted: 30 December2020; Published: 21 January 2021
Article Information
Citation: Deepa Sathee, Lissy Kalliyana Krishnan. Aqueous Solubility and Bioavailability of Albumin Conjugated Curcumin in Endothelial Cells Allege Potential of the Formulation for Anti-Inflammation Therapy. Cardiology and Cardiovascular Medicine 5 (2021): 86-105.
View / Download Pdf Share at FacebookAbstract
Endothelial cell (EC) activation and chronic inflammation can cause the initiation and progression of atherosclerosis. Curcumin (CM) is a potential candidate drug with minimal side effects. However, poor aqueous solubility, lack of bioavailability, and high hydrolytic degradation are limiting its clinical use. Conjugation of CM to Albumin (Alb) producing CM-Alb increases the solubility of the potential drug. This study evaluates an albumin-CM conjugate for cellular uptake demonstrating bioavailability, determining the non-cytotoxic concentrations and anti-inflammatory response in human umbilical vein endothelial cells (HUVECs). The maximum tolerated biocompatible dose determined in HUVEC (EC) culture is 30μM; accordingly, this study evaluates an anti-inflammatory response at non-toxic concentrations. The experiments using fluorescence microscopy and flow cytometry confirmed the endocytosis of fluorescent-tagged CM-Alb and showed comparable cellular uptake of native albumin. Cytokine- induced activation of inflammation in EC culture was standardized as in vitro model for evaluating the response to CM-Alb. Using quantitative real-time polymerase chain reaction (RT-PCR), the established upregulation of inflammatory markers in EC cultures stimulated with TNF-α proposes it as a suitable culture model to study the anti-inflammatory response of CM-Alb. Inflammatory markers were down- regulated by the non-cytotoxicconcentration (<30 μM) of CM-Alb on adding into cytokine-activated ECs. Expressions of markers at a protein level in cytokine-activated EC drop down to normalcy when treated with CM-Alb. In conclusion, increased aqueous solubility and receptormediated uptake of CM- Alb conjugate by ECs caused an anti-inflammatory response in the inflammation-induced culture model, advocating the potential for further study as an intravenous therapeutic formulation.
Keywords
Anti-inflammatory; Drugs; Endothelial cell activation; Pro-inflammation cytokines; Inflammation markers; Curcumin; Human serum albumin
Anti-inflammatory articles; Drugs articles; Endothelial cell activation articles; Pro-inflammation cytokines articles; Inflammation markers articles; Curcumin articles; Human serum albumin articles
Article Details
Abbreviations
AcLDL Acetylated Low density lipoprotein
Alb Albumin
ABS albumin binding sites
BSA bovine serum albumin
CVDs Cardiovascular diseases
cDNA Complementary deoxy-ribonucleic acid
COX-2 Cyclooxygenase-2; CM – Curcumin
CM-Alb Curcumin-Albumin
DAPI 4,6, diamidino-2- phenylindole
DMSO Dimethylsulfoxide
EC Endothelial cell
GAPDH Glyceraldehyde 3-phosphate dehydrogenase
FITC Fluorescein isothiocyanate
IL-6 Interleukin-6
IL-1β Interleukin-1β
MCs Mast cells
MCP-1 Monocyte chemoattractant protein-1
NKCs Natural killer cells
NSAIDs Non-steroidal anti-inflammatory drugs
NF-kB Nuclear factor-kB
PCNA Proliferating cell nuclear antigen
qRT-PCR quantitative real-time polymerase chain reaction
RNA Ribonucleic acid
SMC Smooth muscle cell
TNF-α Tumor necrosis factor-α
VCAM-1 Vascular cell adhesion molecule-1
vWF Von Willibrand factor
DMEM-F12 Dulbecco's modified Eagle's Medium
MTT - 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium
1. Introduction
Atherosclerosis is a consequence of chronic inflammatory responses resulting in cardiovascular diseases (CVDs). Classical proinflammatory monocytes are initiators of inflammation; they infiltrate into sub-endothelial space and get transformed into macrophages. The prolonged-expression of molecules like monocyte chemoattractant protein-1 (MCP-1) intensifies cellular responses triggering smooth muscle cell (SMC) activation [1]. In the initial phase of the disease, endothelial dysfunction initiated by chronic inflammation progresses into the accumulation offatty streaks/plaques beneath the SMC layer, causing obstructed blood flow and/or disruption of the plaque.The entry of pathogenic stimuli into the circulatory system induces cytokine release that acts on cell types such as mast cells (MCs), ECs, SMCs, natural killer cells (NKCs), etc., to effectively manage the inflammatory process[2]. In physiological defensive action, pro-inflammatory cytokines such as Tumor necrosis factor-α (TNF-α)/ Interleukin-6 (IL-6)/Interleukin-1β (IL-1β), released by activated lymphocytes and macrophages, up-regulate nuclear factor-kB (NF-kB), vascular cell adhesion molecule-1 (VCAM-1), MCP-1, and cyclooxygenase-2 (COX-2) in EC[3]. Since expression levels of these cytokines elevate during inflammation, these cytokines are valuable biomarkers for studying the anti-inflammatory effect of new drugs [4].
Statins make up an important class of drugs used for the treatment of atherosclerosis [5]. TNF-α antagonists, interleukin receptor antagonists, and methotrexate in low concentrations are proven anti-inflammatory drugs in experimental studies [5]. These non-steroidal anti-inflammatory drugs (NSAIDs) carry a risk of gastrointestinal complications, liver toxicity, heart failure, etc.[6]. As an alternative to this, pure CM from Curcuma longa possesses enormous therapeutic properties with potent activity against inflammatory responses [7]. The bioavailability, solubility, and rapid hydrolytic degradation are problems faced in the clinical use of CM [8]. Various methods attempted to increase bioavailability are nanoencapsulation, polymer encapsulations, and micelle formations. But these modified forms face the risk of a toxic carrier and insufficient drug delivery [9]. The safer technique of improving CM solubility by conjugating with bovine serum albumin (BSA) successfully exhibited anti-cancerous effects [10, 11]. Since cytotoxic action of CM-Alb cause anti-cancerous effects, identification of non-toxic concentration for safe use as an anti-inflammatory without non-detrimental action to cells is an important criterion to be studied for its use as an anti-inflammatory drug. The solubility of conjugated CM-Alb and its internalization by cells are unexplored areas. The internalization of the conjugated CM- Alb proposes the regulation of molecular pathways in the cytoplasm and nucleus. The main study focus is to establish solubility followed by a demonstration of its cellular uptake, identification of dose range showing minimal cytotoxicity, and anti-inflammatory response.
2. Materials andMethods
2.1 Preparation of Curcumin-Albumin (CM-Alb)conjugate
The study used commercially available CM (>95% Sigma-Aldrich, USA) and pharmacopeia grade human serum Alb (Intas Pharmaceutical Ltd, India) in all experiments. For conjugation, Alb (200mg ml-1 HSA) in physiological buffer was mixed with CM by adding small aliquots from a stock of 0.5M in DMSO to get a final concentration of 5mM. After 2h, the mixture was gel filtered using Sephadex G-25 beads (Sigma Chemicals, USA) and identified the conjugate based on A280:A420 ratio[11]. Pooled the selected fractions, filtered (0.22µm; Merck-Millipore), dispensed into 1 ml aliquots, and stored after freeze-drying (Edwards Modulyo, Edwards, UK).
2.2 Fluorochrome tagging of conjugate andalbumin
One aliquot of lyophilized CM-Alb was dissolved in a carbonate buffer of pH 9.0 and tagged with Fluorescein isothiocyanate (FITC) (50mM) using a standard method to track cellular uptake of the conjugate. The protein was first dialyzed against carbonate buffer and tagged with FITC. Unreacted FITC was removed by gel-filtration on a 5 ml column of Sephadex G-25 beads. Pooled the fractions with >2.0 A495: A280, sterile filtered (0.22µm), and freeze-dried as small aliquots for later use. The CM from replicate aliquots of conjugate and FITC-tagged conjugate were extracted into 9:1 DMSO-water mixture and quantified based on standard curve plotted using graded concentrations of CM in DMSO (Figure 1).
2.3 Assessing solubility ofCM-Alb
Estimated the CM content in the conjugate based on a calibration curve plotted using A420 of serially diluted CM in DMSO in the range of 20-80 µM. Briefly, extracted the CM using a 9:1 DMSO-water mixture) and estimated the concentration of CM in the conjugate. Estimated Alb in the conjugate using Lowry's protein assay. Assessed the solubility by adding a small aliquot in water to obtain a clear yellow-colored solution.
2.4 Isolation and characterization ofECs
The human umbilical cord was obtained aseptically (with informed consent) from patients undergoing C-section. Isolated the ECs from the vein as per the method described by Jaffe et. al, [12]. Briefly, used 0.5% type I collagenase (Gibco-Invitrogen) and cultured cells in Dulbecco's modified Eagle's Medium (DMEM-F12; Gibco-Invitrogen) supplemented with 10% fetal bovine serum. The isolated cells were grown on fibrin-coated plates as described by Chennazy and Krishnan [13]. Briefly, treated the culture plates with 5IU/ml thrombin for 1h and aspirated the excess enzyme. Air-dried the surface and deliver a thin layer (12.5 µl per cm2) of reconstituted and diluted fibrinogen (10mg ml-1). The fibrinogen composite comprises 20µg fibronectin and exogenous gelatin (0.2%; Sigma Chemicals, USA) was placed and allowed to clot, lyophilized (Modulyo 4K, Edwards, UK) in a sterile atmosphere, and stored at 4°C till used. Periodic analysis ensured typical cobblestone morphology, using phase-contrast microscopy (Leica DMIRB, Germany). Harvested cells upon reaching 70-80% confluency, using the standard trypsin digestion method. All experiments used cells from passage 3-5.
Before inflammatory experiments, the EC phenotype was confirmed by immune-staining the cells. Briefly washed and fixed the cells with 3.7% formaldehyde (Merck, India) and blocked with 1% BSA (Sigma Aldrich, USA); for Von Willibrand factor (vWF) cells were permeated with 0.1% Triton X-100 (Sigma Chemicals, USA). Stained for CD31 using 1:50 diluted, Alexa Flour- tagged primary antibody (Abcam, Cambridge, UK). Staining for vWF used 1:100 diluted primary antibody (Santa Cruz Biotechnology). Developed the antigen using1:1000 diluted Texas red-tagged anti-goat (Abcam, UK) antibody. Staining the nucleus used diluted 4,6, diamidino-2-phenylindole (DAPI; 1:5000) (Invitrogen, USA) in both cases. For determining EC function, incubated live cells with AcLDL (Invitrogen, USA) (1:100; 4h; in the dark) at 37°C followed by washing cells with 1x HBSS and captured images. All phase contrast and fluorescent images were acquired using a fluorescent microscope with a Leica Application Suite (LAS) camera system (Leica DMIRB, Germany).
2.5 Assay of the cytotoxic response of EC toCM-Alb
Cells were grown to 70-80% confluent stage in 24 well fibrin coated culture plates. Added CM and CM- Alb in 10 µM -100 µM incrementing concentrations into replicate wells; control was EC culture with no added curcumin or conjugate. At pre-determined time intervals of 24h and 48h, cells were incubated with 0.5mg/ml MTT reagent (Sigma Chemicals, USA) in the medium for two hours and lysed by 5 min incubation with 250µl DMSO (Merck India). The absorbance was measured at 595nm using Bio-Rad, iMark Microplate Reader. Cell viability calculation used the formula Absorbance of Test/Absorbance of Control x100.
2.6 PCNA Assay
Analyzed proliferating cell nuclear antigen (PCNA) as a marker to determine EC proliferation in the presence of CM-Alb. Seeded 5000 cells per cm2, and removed floating cells after two hours. Into the culture medium, added 5µM, 10µM, and 30µM concentration of CM-Alb and incubated the culture for 24h. Washed the cells and fixed with 3.7% formaldehyde and permeated with 0.1% Triton X-100 (Sigma Chemicals, USA). Then, incubated the cells at room temperature with a diluted (1:100) antibody against PCNA (Santa Cruz Biotechnology). Developed the antigen using Alexa fluor (Abcam) tagged secondary antibody (1:1000) for 1h at RT in the dark. Washed and prepared for quantitative fluorescence analysis using a flow cytometer (BD FACS Aria II). The acquired data were analyzed using Flow-jo software.
2.7 Cellular uptakeassay
For the assay, HUVECs were grown to 70-80% confluent stage in multi-well plates. Added CM-Alb conjugate tagged with FITC to replicate wells to get final CM-Alb concentrations of 30µM and incubated for a period of 8h. After taking images using a fluorescent microscope and LAS camera system (Leica DMIRB, Germany) after eight hours, harvesting of the cells used the standard trypsin digestion method. Washed cells were fixed and prepared for quantitative fluorescence analysis using a flow cytometer (Beckman Coulter Life sciences-Cytoflex). For comparing albumin uptake, incubated cultures with Alb- FITC conjugate for a period of 8h followed by microscopic imaging and flow cytometry analysis.
2.8 Standardization of cytokine activated EC
Evaluated anti-inflammatory response in EC culture after activating with 3 inducers: Tumor necrosis factor-α (TNF-α), Interleukin-6 (IL-6), and Interleukin-1β (IL-1β) (Sigma-Aldrich, USA). Incubated EC monolayers at near confluent stage (70-80%) with cytokines: TNF-α (2ng ml-1), IL-6 (10ng ml-1), IL-1β (10ng ml-1) for two different periods (8h and 24h); TNF-α (4ng ml-1), IL-6 (20ng ml-1), IL-1β (20ng ml-1) for a period of 24h. On termination, RNA was isolated using Trizol (Invitrogen, USA) and quantified using Nanodrop. For RT-PCR, 200ng of RNA was converted to cDNA using a cDNA synthesis kit (Origin Diagnostics, India) using Bio-Rad Thermal cycler. The expression of the following inflammatory markers: Nuclear factor-kB (NF-kB), monocyte chemoattractant protein-1 (MCP-1), endothelin-1 (ET-1), cyclooxygenase-2 (COX-2), and vascular cell adhesion molecule (VCAM-1) were determined by Quantitative Real-Time polymerase chain reaction (qRT-PCR) using 2X Master Mix (Origin Diagnostics, India) in Bio-Rad iQ5 Multicolour Real-Time PCR Detection System. The primers procured from Eurogentec, GeneX India Bioscience Pvt.Ltd is listed in the table below (Table 1). Calculated the cytokine-induced up-regulation of inflammatory genes, relative to normal ECs. Used Glyceraldehyde 3- phosphate dehydrogenase (GAPDH) as a house-keeping gene.
|
Genes |
Amplicon size |
Primer Sequence |
|
GAPDH |
120bp |
FP - GAAATCCCATCACCATCTTCCAGG RP – GAGCCCCAGCCTTCTCCATG |
|
MCP-1 |
120bp |
FP – CCGAGAGGCTGAGACTAAC RP – ATGAAGGTGGCTGCTATGA |
|
ET-1 |
96bp |
FP – TGAAGCCATAGCCTCCAC RP-AGTCAGGAACCAGCAGAG |
|
Cox-2 |
86bp |
FP -TCATCAACACTGCCTCAATTC RP –CTCTGGATCTGGAACACTGA |
|
VCAM-1 |
90bp |
FP – CCTCCTTAATAATACCTGCCATTG RP – TCTGTGCTTCTACAAGACTATATGA |
Table 1:List of primers and their sequences.
2.9 Analysis of anti-inflammatory response toCM-Alb
Analyzed CM-Alb conjugate for effect on activated ECs, after24h exposure of >70% confluent ECs with TNF-α (10ng ml-1) after treating with different doses of CM-Alb for 24h. For comparison, added free CM (5 µM, 10 µM, 20 µM, 30µM, 60µM) in cultures activated for 24h with TNF-α (10ng ml-1). Terminated the culture by extracting RNA and qRT-PCR was carried out as described above to estimate the effects of free and conjugated CM on relative expressions of NF-kB, MCP-1, ET-1, COX-2, VCAM-1 against EC treated with TNF-α (10ng ml-1) for 24h, as the baseline. Estimated the up- regulation of inflammatory genes in treated cultures relative to the ECs activated with TNF-α. The housekeeping gene was GAPDH.
2.10 Protein level assessment of inflammatorymarkers
Identified the inflammatory markers in TNF-α (10 ng/ml)-activated cells after treating with CM-Alb. Washed and fixed the treated cells with 3.7% formaldehyde (Merck India) and were blocked with 1% BSA (Sigma Aldrich, USA); and permeated with 0.1% Triton X-100 (Sigma Chemicals, USA). Incubated the with diluted (1:500) primary antibodies NF-kB, ET-1, COX-2 & VCAM-1 (Abcam, Cambridge, UK). After washing, Alexa fluor tagged anti-rabbit secondary antibody (Abcam, Cambridge, UK) in the dilution 1:1000 identified specific protein expressions. The staining of nucleus used 1:5000 diluted 4,6, diamidino-2-phenylindole (DAPI) (Invitrogen, USA). The fluorescent images were acquired using a Fluorescent microscope and LAS camera system (Leica DMIRB, Germany). Image J software estimated the fluorescence intensity by selecting fluorescent cells from different fields (20X) to avoid bias.
2.11 Statistical analysis
Replicate experiments confirmed both qualitative and quantitative analysis data. All the quantitative data presented are mean ± SD. A two-tailed t-test estimated the statistical significance determining differences among individual groups. P<0.05 was considered to be statistically significant for all experiments. P< 0.05 (*); P< 0.01 (**); P< 0.001 (***).
3. Results
3.1 CM-Alb conjugatesolubility
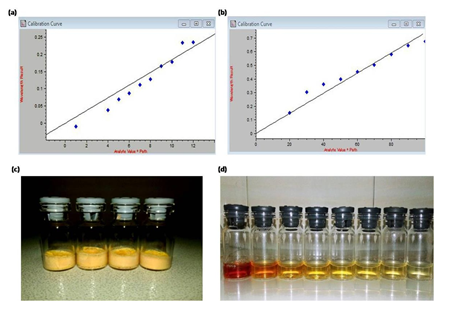
The free CM solubility in water was less than 12µM (Figure 1a) because, after 11µM concentration, the linearity of absorbance deviates. However, in DMSO, linearity was evident until 80 µM concentration (Figure 1b). In the conjugate, 0.2±0.05 mg CM was bound to 40±5mgAlb. The lyophilized powder of conjugate containing 0.2 mg CM (Figure 1c) dissolved within 1-2 min in 0.1 ml water. Therefore, the 12µM solubility of free CM increased to 5.4mM and show an intense yellow color of CM (Figure 1d) in the dissolved conjugate. The color intensity reduced upon serial dilution of the concentrate.
Figure 1:Representative images comparing solubility of free CM and conjugated CM-Alb. (a) Calibration curve of free CM dissolved in the water against A420; (b), a Calibration curve of free CM dissolved in DMSO against A420; (c), Lyophilised CM-Alb conjugate; (d), Lyophilised CM-Alb conjugate dissolved in water and serially diluted in the water showing color grading of diluted CM-Alb conjugate. In both a and b, replicate (n=3) confirmed the trend, and the correlation coefficient was >0.95.
3.2 Characteristics of EC
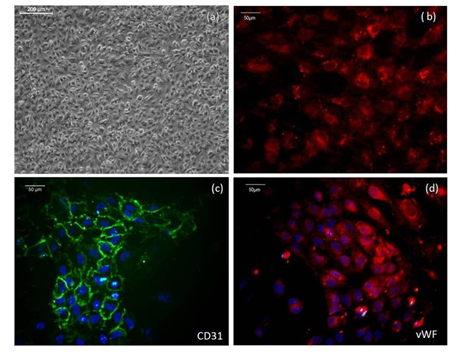
The ECs isolated from the umbilical cord and grown in culture showed typical cobblestone morphology (Figure 2a) under a phase-contrast microscope. The isolated cells showed uptake of AcLDL (Figure 2b), confirming the endothelial phenotype. The ECs were positive for both CD31 and vWF. CD31 (Figure 2c) is a membrane protein expressed by ECs that promotes interaction with platelets. vWF (Figure 2d) is a protein produced by Weibel Palade bodies present in the cytoplasm of ECs, which mediates interaction with other extracellular proteins like collagen.
Figure 2:Representative micrographs illustrating phenotype of EC (a) phase-contrast micrographs showing typical cobblestone morphology of EC; (b), a fluorescent micrograph of AcLDL taken up by ECs seen as red in the cytoplasm; (c), the fluorescent image of EC membrane protein CD31 immunostained as green; blue-DAPI stained nucleus; (d), a fluorescent micrograph of vWF in Weibel Palade bodies in ECs immunostained in red; blue-DAPI stained nucleus.
3.3 Cytotoxic response of EC to CM-Alb
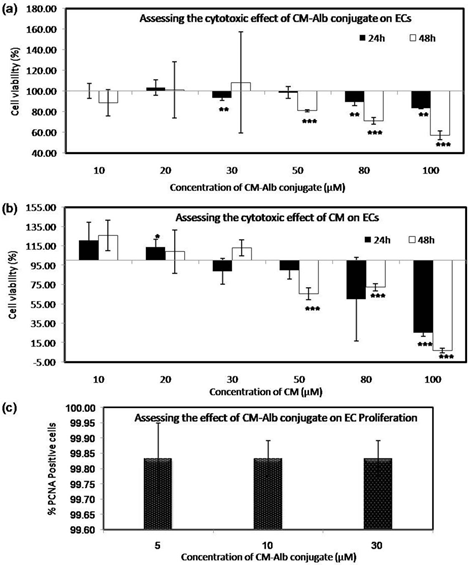
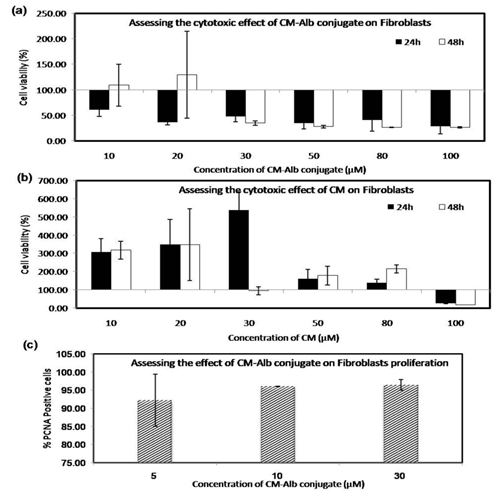
The ECs responded to CM-Alb in both dose-dependent and time-dependent manner. On exposure of CM-Alb conjugate at low concentrations (10 & 20µM),>90% ECs showed viability (Figure 3a). However,> 50µM concentrations resulted in a significant reduction in cell viability. There is a significant difference in viability between 24h and 48h exposure at all concentrations; the latter producing more nonviable cells. The ECs exposed to free CM (Figure 3b) at 10µM to 30µM concentrations showed comparable cytotoxic effect as compared to that seen with similar CM-Alb dose. At high concentrations (50 to 100µM), free CM also showed a significant reduction in cell viability (6 to 66%). The cytotoxic effect was not remarkably or consistently different between 24h and 48h exposure to free CM. The CM-Alb was significantly cytotoxic to fibroblasts cell lines (L929) (Suppl Figure 1a). On the other hand at all concentrations, free CM was not effective in inducing cytotoxic effect on fibroblast (Suppl Figure 1b). This difference in induction of cytotoxicity to EC and fibroblast by the free CM could be related to the albumin present in the culture medium. The EC culture requires 10% bovine calf serum supplement is for good EC growth; whereas, serum supplement is not needed in L929 cell line culture. In the EC culture medium, the added free curcumin could bind to the albumin in the medium which can transport CM into cytoplasm resulting in ctyto-toxic action, which is not the case in fibroblast culture. Apparently, there is a significant difference in cyto-toxicity of free CM in ECs and fibroblast. On the other hand, bothEC and fibroblast responded similarly to CM-Alb conjugate added in the medium. Upon comparison of time-dependent cytotoxic response, incubation of low concentrations (<20µM) of CM-Alb for 24h is found safe for testing the anti-inflammatory effect of CM-Alb in ECs.
Figure 3:Graphical representation of the cytotoxic response of EC to CM-Alb and CM. (a), Compiled data of dose-response combined with the effect of a period of exposure for 24h and 48h on ECs upon treating with different dose of CM-Alb; (b), compiled data of time-dependent and dose- dependent effect of free CM; (c), dose-response (5µM, 10µM & 30µM) of conjugated CM on EC proliferation quantified by flow cytometry of PCNA stained cells, after 24h incubation with the conjugate. Values are represented as Mean±SD; n=3.P< 0.05 (*); P< 0.01 (**); P < 0.001 (***).
The cell proliferation assay showed that varying concentrations (lower doses) of the conjugate (5, 10, 30µM) did not affect EC proliferation (Figure 3c). More than 99% of ECs were proliferative even after incubating with 10µM, 20µM, and 30µM of CM in the conjugated form for 24h. The observation suggests this range could be ideal for anti-inflammatory action in cells without affecting normal cellular activity. At the tested concentrations of CM-Alb, >95% of fibroblasts were PCNA positive and proliferating (Supplementary Figure 1c).
3.4 Endocytosis assay using FITC tagged CM-Alb conjugate
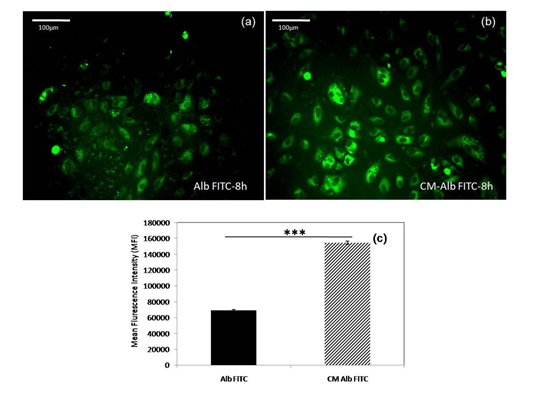
The endocytosis of FITC tagged Alb by ECs is evident in the micrographs (Figure 4a) similar to that seen in endocytosis of FITC tagged CM-Alb conjugate (Figure 4b). Mean fluorescence intensity (MFI) quantifies significant uptake of both Albumin and CM-Alb (Figure 4c). There was a significant increase in the uptake of FITC tagged CM-Alb (> 150000 MFI) compared to uptake of FITC tagged native Albumin (>60000 MFI). Both the microscopy and flow cytometry showed that the cells tend to uptake albumin in vitro. Specific interaction of albumin with cell-membrane receptors is indicated.
Figure 4:Data demonstrating endocytosis of CM-Alb conjugate by EC. (a) fluorescence micrograph showing uptake of FITC tagged Alb; and (b) fluorescence micrograph of FITC-tagged CM- Alb by ECs- both at 30µM concentration upon 8h incubation; (c) Quantification of FITC conjugated Albumin and CM-Alb conjugate based on MFI measured using flow cytometer. Values are represented as Mean±SD; n=3.P< 0.05 (*); P< 0.01 (**); P< 0.001 (***).
3.5 Standardization of inflammatory EC model
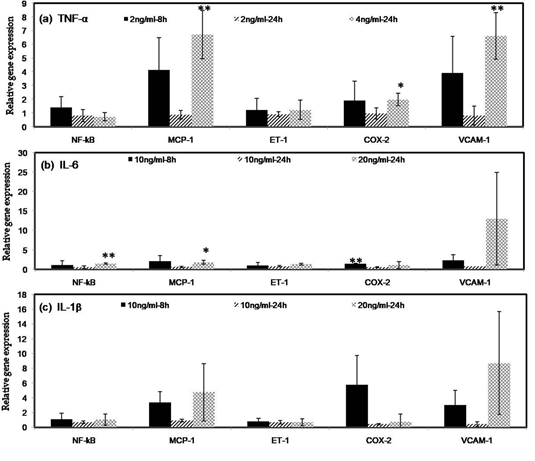
Cytokines TNF-α (2ng ml-1) and IL-6 (10ng ml-1) except IL-1β (10ng ml-1), when incubated with ECs for a period of 8h and 24h, respectively, showed time and dose-dependent variation in the expression of inflammatory markers (Figure 5). Upon 8h (Figure 5a) induction with an inflammatory inducer, TNF-α (2ng ml-1) showed expressions of all the inflammatory markers (NF-kB, MCP-1, ET-1, COX-2, VCAM-1). However, compared to control (cells not exposed to TNF-α), there was no significant increase in the inflammatory markers. Upon 24h (Figure 5a) induction with the same concentration, there was no upregulation observed in any of the inflammatory markers. But in 24h of induction using 4ng ml-1 TNFαresulted in upregulation of all inflammatory markers in ECs (Figure 5a). TNFα (4ng ml-1) treatment resulted in a significant increase in COX-2 (1.98-fold) expression compared to control; MCP-1(6.71-fold) and VCAM-1 showed a highly significant increase (6.61-fold) in gene expression compared to control. Upon IL-6 (10ng ml-1) treatment for a period of 8h (Figure 5b), except ET-1, all four inflammatory markers showed upregulation, and in particular, a significant increase in gene expression of COX-2(1.46). Upon 24h induction (Figure 5b) with the same concentration of IL-6, there was no upregulation observed in any of the inflammatory markers. Upon IL-6 (20ng ml-1) treatment for a period of 24h (Figure 5b) showed upregulation in terms of gene expression of all the five inflammatory markers but compared to control, a significant increase in gene expression was found only for NF-kB (1.82) and MCP-1 (1.34). Upon IL-1β (10ng ml-1) treatment for a period of 8h (Figure 5c), there is no significant upregulation of any of the inflammatory markers in terms of gene expression. Upon 24h induction, IL-1β at the same concentration failed to upregulate the expression of inflammatory markers. Upon 24h induction with IL-1β (20ng ml-1) also, all the inflammatory markers except ET-1 (0.67) and COX-2 (0.74) upregulate compared to control. However, on IL-1β (20ng ml-1) treatment of ECs for a period of 24h, there was no significant increase in gene expressions of inflammatory markers. Therefore, based on time and dose-dependent expression in ECs subjected to different concentrations of inflammatory inducers for different periods, TNF-α contributed significantly to the overexpression of inflammatory markers (MCP-1, COX-2, and VCAM-1) upon 24h incubation with ECs. The results confirmed the suitability of TNF-α as an inflammation inducer for further studies.
Figure 5:Graphical representation of data showing an inflammatory response upon induction of ECs with cytokines. Relative expression of inflammatory markers upon treating EC with: (a), TNF- α(2ng ml-1) for a period of 8h, 24h and (4ng ml-1) for 24h; (b), IL-6 (10ng ml-1) for a period of 8h, 24h and (20ng ml-1) for 24h; and (c), IL-1β (10ng ml-1) for a period of 8h, 24h and (20ng ml-1) for 24h. Values shown in the graph are mean±SD; n=3.P< 0.05 (*); P< 0.01 (**); P< 0.001 (***).
3.6 Anti-inflammatory action of CM-Alb in inflammatory EC
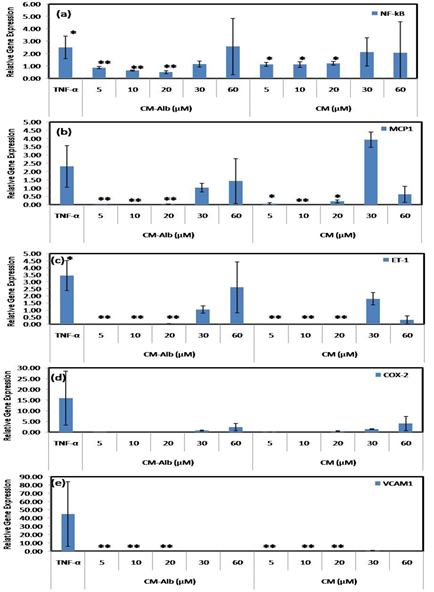
For demonstrating the anti-inflammatory effect of CM-Alb conjugate, TNF-α (10ng ml-1) induced EC to an inflammatory state in 24h. Following this, treated the inflammation-induced ECs with different concentrations of CM-Alb conjugate, as well as with free CM for 24h. The compiled relative gene expression data obtained via qRT-PCR confirms the anti-inflammatory action of CM-Alb (Figure 6). The gene expressions of NF-kB (Figure 6a), MCP-1 (Figure 6b), ET-1 (Figure 6c), and VCAM-1 (Figure 6e) significantly reduced when treated with CM-Alb conjugate at 5, 10, 20µM concentrations whereas, at 30 and 60µM, the conjugate upregulated most of the markers. In the case of COX-2, even though the gene expression levels lowered compared to the TNF-α treated ECs (control), there was no significant downregulation. For NF-kB, MCP-1, and ET-1, both CM-Alb and free CM, showed a slight increase in gene expression of inflammatory markers at 30 and 60µM, compared to their corresponding lower doses (Figure 6a to 6c). For ET-1 and VCAM-1, free CM at lower doses showed a significant reduction in gene expression but not in the case of NF-kB and MCP-1. However, for COX-2 (Figure 6d), both conjugate and free CM showed no significant effect in lowering the expression at both low and high doses. These results indicate that CM-Alb (5 to 20µM) down-regulate the gene expressions of inflammatory markers. Owing to its cytotoxic effect above 30µM, the inflammatory regulation is not much evident. Free CM also controlled the expression of inflammatory markers; but, the CM-Alb's effect seems higher.
Figure 6:Graphical representation of anti-inflammatory action of CM-Alb and free CM. Relative gene expression w.r.t control EC and EC treated with TNF-α(10ng ml-1) for 24h,estimated using qRTPCR for demonstrating effect of CM-Alb on: (a), NF-kB; (b), MCP-1; (c), ET-1; (d), COX-2; and (e), VCAM-1 expressions. Data represent dose response of CM-Alb upon treating with EC preexposed toTNF-α for 24h, followed by incubation with added conjugate at different cocentrations for additional 24h. Values are represented as Mean±SD; n=3.P<0.05 (*); P< 0.01 (**); P < 0.001 (***).
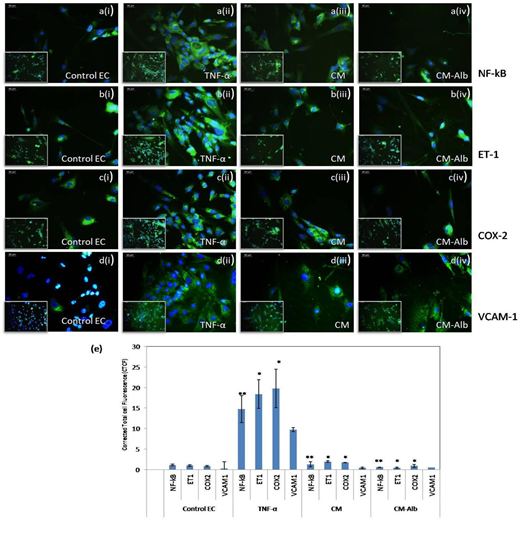
Confirming the results seen in qRTPCR, activated ECs immunostained positive for all the four inflammatory markers NF-kB, ET-1, COX-2, and VCAM-1 (Figures 7a to 7d). The control EC, CM-treated, and CM-Alb treated ECs stained positive for inflammatory markers. Upon quantifying fluorescence intensity, TNF-α activated ECs showed significantly higher expression of markers: NF-kB, ET-1, COX-2 and VCAM-1, compared to control ECs (Figure 7e). Also, on the exposure of the TNF-α treated ECs with 5µM CM-Alb or free CM, the frequency and intensity of immunostained EC decreased considerably. Thus, restoration of markers in CM-Alb treated inflammatory EC at protein level is indicated.
Figure 7:Representative micrographs for ECs treated with different conditions followed by immunostaining for inflammatory markers. (i), cells treated with medium (control); (ii), cells treated with TNF-α; (iii), cells treated with TNF-α+CM; (iv), cells treated with TNF-α+CM-Alb. a(i-iv) cells stained for NF-kB; b(i-iv) cells stained for ET-1; c(i-iv) cells stained for COX-2; d(i-iv), cells stained for VCAM-1. Green fluorescence represents cell cytoplasm and the nucleus is represented by blue color stained with DAPI, (e), Quantitative analysis of total cell fluorescence for each inflammatory marker in ECs subjected to different treatment conditions.
4. Discussion
Atherosclerosis is one of the inflammatory disorders marked by the dysfunction of ECs. Anti- inflammatory drugs with minimal side effects are of great demand to circumvent different inflammation- associated diseases. Curcumin is well-known for its anti-inflammation property. However, high hydrophobicity and related insolubility in an aqueous medium limit its bioavailability. Besides, it shows a tendency for quick hydrolytic degradation. Altogether the medicinal property of curcumin is not exploited in clinics [7]. This study focused on a method to improve the bioavailability of CM without affecting the functions of the molecule. The natural physiological drug carrier, human serum albumin was used to promote bioavailability and solubility by conjugating the drug molecule. In cancer treatment, the cytotoxic concentration of the CM affecting the cell-cycle is a requisite. But for inflammatory regulation, a non-cytotoxic CM dose is a necessity. This study compared cytotoxicity and anti-inflammatory properties of CM-Alb to CM. Also, the use of ECs as an in vitro model very well demonstrated the effect of the drug on inflammation. This study highlights the use of a non-cytotoxic dose of CM as an anti-inflammatory drug.
Other methods explored to improve bioavailability include: encapsulating or linking CM with other substances resulting in emulsions, and nanoformulations, etc. [9]. Even though these modifications have caused an increase in drug miscibility in aqueous medium, poor regulated release of the molecule from drug carrier and vehicle associated toxicity are major concerns limiting the clinical use. Degradation products curcumin glucuronide/curcumin sulfate were detected in blood plasma during clinical trials of administering these into patients [14]. But despite all these factors, attempts have successfully led to the development of a conjugate as an anti-cancer agent proven safe and efficient for cancer therapy by in vitro/in vivo animal studies [10, 11, 15]. When the conjugated CM reaches the tissue, Alb receptors on the cells could mediate internalization followed by a breakdown of the protein into amino acids, a normal physiological process releasing the drug. Inflammed tissues tend to accumulate Alb which might help inadequate localization of drug molecules in the concerned site [16]. Several studies have shown that the endothelial layer of microvessels present in different tissues expresses specific receptors such as albumin binding sites (ABS). One of the receptors is albondin (gp60), which facilitates the transcytosis of drug molecules across the cell membrane mediated by Alb [17,18]. However, if the drug conjugation process affects the albumin confirmation, it may not recognize the receptor to result in endocytosis. This study compared the endocytosis of both native and drug conjugated Alb. The CM-Alb endocytosis into EC is comparable or even better than native Alb. The solubility improved significantly upon conjugation of the drug and thus the bioavailability too. Fold increase of drug solubility in an aqueous medium is evident. The absence of cytotoxic response in L929 culture at <80uM concentration of free CM suggests may be due to instability and a lack of endocytosis. Better solubility and endocytosis of CM-Alb caused a toxic effect in L929 cells. Therefore, CM-Alb has a better opportunity for clinical viability. Also, since a low concentration (5µM) of CM-Alb is non-cytotoxic and anti-inflammatory, it may be safely administered intravenously.
Both IL-6/IL-1β failed to activate inflammatory genes consistently in normal EC culture even at 20 ng ml-1 doses and were found insufficient to produce inflammatory cells and test the effect of this conjugated drug. However, upon treating ECs with a 10ng ml-1 TNF-α dose, the inflammatory response was found sufficient to prove the anti-inflammatory effect of CM-Alb conjugate. Interestingly, the non- cytotoxic range of CM concentrations (5µM to 20µM) in the conjugate has down-regulated all inflammatory marker genes in 24h. Upregulated genes in the presence of >30µM indicate that thenecrosis-related inflammation could be a cause for the increase in molecular expressions [19, 20].
The transcription factor NF-kB is involved in regulating transcriptional stimulation of various genes during inflammation. Both TNF-α and NF-kB are mutually activating in unfavorable conditions [21]. Studies have reported that CM has an inhibitory effect on the functional activation of NF-kB. Usually, NF-kB is in association with the inhibitor of kB (IkB) in the cell cytoplasm.
COX-2 is an important enzyme that takes part in regulating inflammatory pathways. Normal healthy cells express COX-1 constitutively which is required for the smooth functioning of cellular activities. However, upon stimulation by a variety of inflammatory cytokines, cells begin to express another isoform COX-2. Both these forms convert arachidonic acid into prostaglandins during inflammatory pathways. And with the increase in the intensity of inflammatory stimuli, studies have shown that the expression of COX-2 also increases; meanwhile, the expression of COX-1 remains normal [3]. So cyclo-oxygenase being an important and critical enzyme in inflammatory pathways, the healthcare industry develops COX inhibitors to treat inflammation. This study demonstrates COX-2 inhibition by low concentrations of CM conjugated to Alb.
Recruiting monocytes and macrophages to the inflamed tissue is through MCP-1 [22]. In the current study, the expression of MCP-1 increases significantly upon treating ECs with TNF-α for a period of 24h. When the inflammation-induced ECs expose to 5 to 20µM concentrations of CM-Alb conjugate, MCP-1 expression down-regulated significantly, as compared to the TNF-α treated cells. During inflammation, there is an enhanced expression of adhesion molecules on vascular walls involving in the worsening of vascular diseases. This is accompanied by EC activation leading to the release of MCP-1 [23]. At the time of inflammation, the recruited monocytes differentiate into classical M1 macrophages and non-classical M2 macrophages resulting in cholesterol ingested plaques. These plaques serve as a reservoir for the release of several inflammatory cytokines that deteriorates the plaque and promotes inflammation in the affected region of the vascular wall [24]. The MCP-1 enhances the expression of VCAM-1 on ECs [23]. The observed control of MCP-1 by CM-Alb highlights the significance of the molecule for potential therapeutic applications.
Another major cytokine ET-1 released by ECs acts by promoting vasoconstriction of blood vessels during CVDs [25]. In the current study, ET-1 increases upon treatment with TNF-α and significantly reduced upon treatment with CM-Alb conjugate at 5, 10, 20µM concentrations. TNF-α and IL-6 are known to induce the expression of VCAM-1, ICAM-1, and other cell adhesion molecules on their surface due to their response to mediators of the inflammatory response [26]. A significant increase in the expression of VCAM-1 in EC upon incubating with TNF-α for a period of 24h gives a good model system to study the effect of the anti-inflammatory drug molecule. Further, the treatment of inflammation-induced ECs with CM-Alb conjugate at 5, 10, 20µM concentrations down-regulated the expression of VCAM-1 significantly compared to the TNF-α treated cells.
Several studies explored the anti-inflammatory property of CM-Alb nanoparticles and various such encapsulations [16]. However, studying the anti-inflammatory action of a highly water-soluble formulation is novel. The bioavailability of the conjugate reflects on endocytosis. It is interesting to note that the concentration range with no detrimental effect on EC viability/proliferation results in a significant anti-inflammatory response in cell culture. The study demonstrates the adverse effects of a high dose resulting in upregulation of genes, which may be due to proinflammatory changes linked to cell death.
An earlier study reported non-toxic, immunomodulatory, and anti-tumor functions of the conjugate using mice tumor model [10]. Further pre-clinical and clinical trials can determine a safe dose for use in vivo as an anti-inflammatory drug.
5. Conclusion
In the conjugated form, soluble CM is internalized and acts on cells causing a cytotoxic effect in a dose- dependent and time-dependent manner. The ECs isolated from the umbilical cord and grown in culture responded to TNF-α, activated inflammatory molecules, and proved the utility of inflammation-activated EC as a suitable model to study the anti-inflammatory effects of potential drug molecules. Very low concentration of 5µM CM-Alb restored the TNF-α activated markers in EC. With the cytotoxic concentration of CM (>30µM), the death-associated inflammatory response was evident. Therefore, the anti-inflammatory property of CM-Alb holds translational potential by the careful selection of doses. The study highlights the dose-based dual role of soluble curcumin in the albumin-conjugated formulation; as a cytotoxic anticancer drug at high concentration, and non-toxic anti-inflammatory cardiovascular medicine at low concentration.
Funding
The Technology development fund (TDF-6216) scheme of Sree Chitra Tirunal Institute for Medicals Sciences and Technology (SCTIMST), Trivandrum, India, provided financial support for conducting the study.
Author Contributions
Deepa S: Standardized the conjugation protocol; carried out the spectroscopic analysis of the conjugate; proved solubility of curcumin–albumin conjugate; carried out the cell culture experiments; proved the suitability of EC as inflammatory culture model; demonstrated endocytosis; cytotoxicity; anti- inflammatory property; compiled data; and, drafted the manuscript.
Lissy K Krishnan: conceived the idea, guided Deepa during the studies, analyzed and interpreted the data, and edited the manuscript.
Declaration of Competing Interest
The authors declare no competing interests (neither financial nor non-financial) in the publication of the data in this manuscript.
Acknowledgments
The authors acknowledge the Director SCTIMST and Head BMT Wing for providing valuable support and facilities. The authors are thankful to the Technology Development Fund (TDF-6216), and the remuneration of Deepa S. We acknowledge Dr. Anugya Bhatt, Scientist F and Ranjith S, Technical assistant of the division of Thrombosis Research for helping with conjugate endocytosis analysis by flow cytometry. Authors thank Dr. Rema, Senior Gyn & OBG, for providing umbilical cord aseptically from anonymous subjects undergoing C-section childbirth, with informed consent for isolating EC. The results presented are linked to continuation in a patent of US2016-0129127A1.
References
- Gonzalez-Quesada C, Frangogiannis NG. Monocyte Chemoattractant Protein (MCP-1)/CCL2 as a biomarker in Acute Coronary Syndromes. Curr Atheroscler Rep 11 (2009) 131-138.
- Sprague AH, Khalil RA. Inflammatory Cytokines in Vascular Dysfunction and Vascular Disease. Biochemical pharmacology 78 (2009): 539.
- Caughey GE, Cleland LG, Penglis PS. Roles of Cyclooxygenase (COX)-1 and COX-2 in Prostanoid Production by Human Endothelial Cells: Selective Up-Regulation of Prostacyclin Synthesis by COX-2. The Journal of Immunology 167 (2001): 2831-2838.
- Szmitko PE,Wang CH,Weisel RD. New markers of inflammation and endothelial cell activation: Part I. Circulation 108 (2003): 1917-1923.
- Klingenberg R, Hansson GK. Treating inflammation in atherosclerotic cardiovascular disease: emerging therapies. Eur Heart J 30 (2009): 2838-2844.
- Fendrick AM, Greenberg BP. A review of the benefits and risks of nonsteroidal anti- inflammatory drugs in the management of mild-to-moderate osteoarthritis. Osteopath Med Prim Care 3 (2009): 1.
- Gupta SC, Patchva S, Aggarwal BB. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS J 15 (2012): 195-218.
- Wang L, Lu N, Zhao L, et al. Characterization of stress degradation products of curcumin and its two derivatives by UPLC–DAD–MS/MS. Arabian Journal of Chemistry (2016).
- Prasad S, Tyagi AK, Aggarwal BB. Recent Developments in Delivery, Bioavailability, Absorption, and Metabolism of Curcumin: the Golden Pigment from Golden Spice. Cancer Res Treat 46 (2014): 2-18.
- Aravind SR, Krishnan LK. Curcumin-albumin conjugates as an effective anti-cancer agent with immunomodulatory properties. International Immunopharmacology 34 (2016): 78-85.
- Thomas C, Pillai LS, Krishnan L. Evaluation of Albuminated Curcumin as Soluble Drug Form to Control Growth of Cancer Cells in Vitro. Journal of Cancer Therapy 05 (2014): 723.
- Jaffe EA, Nachman RL, Becker CG, et al. Culture of Human Endothelial Cells Derived from Umbilical Veins. IDENTIFICATION BY MORPHOLOGIC AND IMMUNOLOGIC CRITERIA. J Clin Invest 52 (1973): 2745-2756.
- Chennazhi KP, Krishnan L. Effect of passage number and matrix characteristics on the differentiation of endothelial cells cultured for tissue engineering. Biomaterials 26 (2005): 5658-5667.
- Vareed SK, Kakarala M, Ruffin MT, et al.(2008).
- Krishnan LK, Christina A. Development of Soluble Albuminated Curcumin for Application in Cancer Therapy. US2016-0129127A1 issue date 12-01-2020.
- Kratz F. Albumin as a drug carrier: Design of prodrugs, drug conjugates, and nanoparticles. Journal of Controlled Release 132 (2008): 171-183.
- John TA, Vogel SM, Tiruppathì C, et al. Quantitative analysis of albumin uptake and transport in the rat microvessel endothelial monolayer. American journal of physiology. Lung cellular and molecular physiology 284 (2003) NaN-NaN.
- Tirupathi C, Song W, Bergenfeldt M, et al. Gp60 activation mediates albumin transcytosis in endothelial cells by the tyrosine kinase-dependent pathway. J. Biol. Chem. 272 (1997): 25968-25975.
- Li M, Carpio DF, Zheng Y, et al. An Essential Role of the NF-κB/Toll-Like Receptor Pathway in Induction of Inflammatory and Tissue-Repair Gene Expression by Necrotic Cells. The Journal of Immunology 166 (2001): 7128- 7135.
- Zhu K, Liang W, Ma Z, et al.(2018).
- Hewlings SJ, Kalman DS. Curcumin: A Review of Its’ Effects on Human Health. Foods 6 (2017).
- Cranford TL, Enos RT, Velázquez KT, et al. Role of MCP-1 on Inflammatory Processes and Metabolic Dysfunction Following High-Fat Feedings in the FVB/N Strain. Int J Obes (Lond) 40 (2016): 844-851.
- Yang J, Park Y, Zhang H, et al. Role of MCP-1 in tumor necrosis factor-α-induced endothelial dysfunction in type 2 diabetic mice. Am J Physiol Heart Circ Physiol 297 (2009): H1208-H1216.
- Chistiakov DA, Melnichenko AA, Myasoedova VAet al.(2017).
- Masaki T, Sawamura T. Endothelin and endothelial dysfunction. Proc Jpn Acad Ser B Phys Biol Sci 82 (2006): 17-24.
- Liao JK. Linking endothelial dysfunction with endothelial cell activation. J Clin Invest 123 (2013): 540-541.
Supplementary Material
Supplementary Figure 1:Effect of CM-Alb and Free CM on L929 Cell lines. (a), Graphical representation of the results of MTT assay in fibroblast (L929 culture) in the presence of CM-Alb; (b), graphical representation of the results of MTT assay in fibroblast in the presence of free CM. (c) dose- response (5µM, 10µM&30µM) of conjugated CM on EC proliferation quantified by flow cytometry of PCNA stained cells, after 24h incubation with the conjugate. Values are represented as Mean±SD; n=3.P< 0.05 (*); P< 0.01 (**); P < 0.001 (***).The cells were grown in DMEM F12 supplemented with 2% serum. Effect of conjugate and free CM on MTT was compared in the concentration range of 5 µM to 100µM. It is evident that free CM is not cytotoxic to fibroblast in the dose range of 5µM to 80µM and permits cell multiplication resulting in the increase in viable cells in culture by 24h/48h. The CM-Alb is cytotoxic to fibroblasts, however, being cell line, the viable cells multiplied in 48h at a lower dose (5 to 20 µM).


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 