Concurrent Massive Pulmonary Embolism With Emboli in Transit and Anterior ST Elevation Myocardial Infarction with Increased Bleeding Risk Successfully Treated with Low-Dose Systemic Alteplase: A Case Report
Joseph Marc S. Seguban1*, Elaine Alajar1,2, Jessore Isidro1, Noemi Pestaño1, Felix Eduardo Punzalan1,2, and Rafael R. Castillo1,4,5
1Section of Adult Cardiology, Department of Internal Medicine, Manila Doctors Hospital, Manila, Philippines
2Cardiovascular Section, University of the Philippines-Philippine General Hospital, Manila, Philippines
3Department of Clinical Epidemiology, University of the Philippines Manila, College of Medicine, Manila, Philippines
4College of Medicine, Adventist University of the Philippines, Silang, Cavite
5CardioMetabolic Research Unit (CaMeRU), FAME Leaders Academy, Makati City, Philippines
*Corresponding author: Joseph Marc S. Seguban, Manila Doctors Hospital, United Nations Ave., Ermita, Manila, Philippines
Received: 27 August 2019; Accepted: 13 September 2019; Published: 17 September 2019
Article Information
Citation: Joseph Marc S. Seguban, Elaine Alajar, Jessore Isidro, Noemi Pestaño, Felix Eduardo Punzalan, and Rafael R. Castillo. Concurrent Massive Pulmonary Embolism With Emboli in Transit and Anterior ST Elevation Myocardial Infarction with Increased Bleeding Risk Successfully Treated with Low-Dose Systemic Alteplase: A Case Report. Cardiology and Cardiovascular Medicine 3 (2019): 352-359.
View / Download Pdf Share at FacebookAbstract
Massive pulmonary embolism (MPE) and acute myocardial infarction (AMI) are both life-threatening conditions, and the concomitant presence of both, though a rare occurrence, usually ends in a fatal outcome.
A 67-year-old male, hypertensive, smoker with recent surgery for prostate cancer presented with dyspnea, syncope and angina. His electrocardiogram revealed anterior wall STEMI; echocardiogram showed left ventricular segmental hypokinesia, with right atrial thrombus—likely an embolus in transit—and right ventricular dysfunction. Compression ultrasound revealed a left popliteal vein thrombosis. Coronary angiography showed left main coronary and left anterior descending artery stenoses for which angioplasty with stenting was performed. Pulmonary angiography showed filling defects in the main pulmonary arteries consistent with massive pulmonary embolism. Computed tomography pulmonary angiography showed another likely embolus in transit at the superior vena cava. Despite the coronary intervention, his dyspnea persisted even at rest, thus intravenous tissue plasminogen activator (tPA) was administered at a reduced dose (50 mg for 2 hours) due to high risk of bleeding. He improved dramatically after the thrombolytic infusion and was discharged improved on the 9th hospital day, and improved further clinically and by echocardiogram on 3-month follow up.
To our knowledge, this is the first reported case showing the efficacy and safety of low-dose alteplase in massive pulmonary embolism with documented emboli in transit at the superior vena cava and right atrium, in a setting of an acute anterior wall STEMI, who had just undergone PCI. This is also the first reported case in our country to manage such a complicated case of MPE with low-dose alteplase (50 mg) leading to a favorable&
Keywords
<p>Pulmonary embolism; STEMI; Acute coronary syndrome; Progressive dyspnea; Increased bleeding risk; Alteplase; Plasminogen activator; Thrombolytic therapy</p>
Article Details
The case
A 67-year-old Filipino male, with a 30 pack year smoking history, hypertensive, was transferred to our tertiary hospital from another hospital for progressive chest pains and difficulty of breathing. He recently underwent elective radical prostatectomy for Stage II prostate carcinoma in the other hospital. Intra-operatively, he had bleeding requiring transfusion of 10 units packed red blood cells and two units of whole blood. He had chest pains and mild dyspnea post-operatively which were relieved with oxygen supplementation, nitrates and other oral cardiac medications. He remained angina-free but still felt weak, until 18 days postoperatively, when he developed recurrence of his chest pains with severe dyspnea, tachycardia and syncope prompting transfer to our hospital for further diagnostic evaluation and management. Electrocardiogram (ECG) showed acute anterior wall STEMI, compared to his pre-operative ECG which only showed borderline sinus tachycardia, nonspecific ST-TW changes.
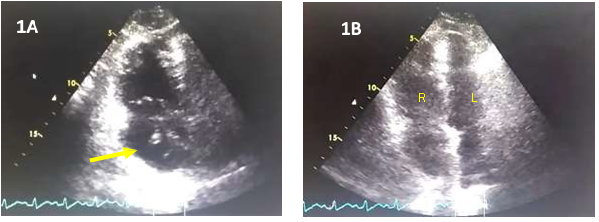
At the emergency room, he remained dyspneic and still complained of chest tightness; his systolic blood pressure (BP) was palpatory 90 mmHg, pulse rate of 110 bpm and respiratory rate of 25 cpm. He had distended neck veins. Auscultation revealed tachycardic rate with regular rhythm, distinct S1 and S2, no murmur; bibasal rales; no hepatomegaly, no pedal edema and with barely palpable pulses on both extremities. He had no neurologic deficits. He was immediately started on intravenous inotropic support and oxygen (O2) inhalation by face mask, which improved his BP to 110/70 mmHg and his O2 saturation to 99%. Troponin-I was elevated at 0.471 ng/ml (normal value of 0-0.034) The patient’s chest x-ray showed no pulmonary infiltrates nor congestion that could explain the severe dyspnea. Transthoracic echocardiography revealed concentric left ventricular remodeling with mild anterior wall hypokinesia, preserved systolic function and ejection fraction of 76% (done while on intravenous inotropic support). Grade I diastolic dysfunction was also noted. There was dilatation of right atrium with a suspicious mobile echogenic density (Figure 1A) suggestive of a right atrial embolus in transit. The right ventricle was also dilated (Figure 1B) with dyskinesia of the midwall region and relative sparing of the apex and base and reduced regional ventricular function. The pulmonary artery pressure by TR jet velocity was 53 mmHg consistent with moderate pulmonary hypertension. No interatrial or interventricular shunt was demonstrated. Given the overall clinical picture and results of the ancillary procedures, acute high risk pulmonary embolism on top of an acute STEMI was considered.
D-Dimer was elevated at 9635 ng/ml (normal value of 0-5000) and arterial blood gases showed uncompensated respiratory alkalosis with adequate oxygenation for age. Venous duplex scan of the lower extremities showed an acute thrombosis of the left popliteal vein with pulsatile venous flow pattern indicative of venous hypertension, pulmonary hypertension and right sided heart failure.
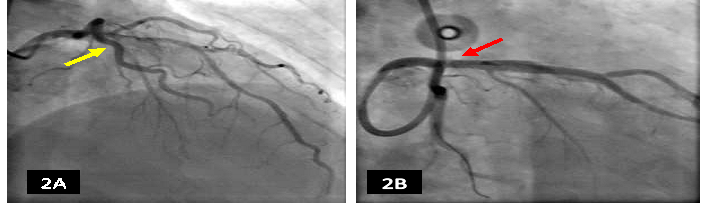
Since the patient was hemodynamically unstable, immediate coronary and pulmonary angiogram were done. Coronary angiography showed 20% distal stenosis of the left main coronary and 70-80% eccentric stenosis of the proximal left anterior descending arteries with soft thrombus (Figure 2A) for which angioplasty with stenting was performed (Figure 2B).
Figure 2: (A) Coronary angiogram showing eccentric 70-80% of the proximal left anterior descending artery (yellow arrow) and mild stenosis of the left main coronary artery. (B) Percutaneous coronary intervention with deployment of drug eluting stent at the ostial left main coronary artery (red arrow) up to the proximal and mid left anterior descending artery.
Pulmonary angiography showed large filling defects partially occluding the right main pulmonary artery and its branches as well as the left main pulmonary artery. Since the patient’s dyspneic episodes initially occurred immediately after the surgery, an acute on top of chronic pulmonary embolism was considered. A computed tomography pulmonary angiography confirmed the findings on pulmonary angiography and defined the age and extent of the embolus from main pulmonary artery down to its branches. It also revealed another possible embolus in transit at the superior vena cava and right ventricular enlargement.
After the percutaneous coronary intervention (PCI), the patient was maintained on aspirin and ticagrelor. However, despite relief of anginal symptoms and weaning off from inotropic support, he remained dyspneicat rest. Given the extensive embolization in both main pulmonary arteries and presence of possible emboli in transit at the superior vena cava and right atrium, and proximal deep vein thrombosis, systemic thrombolysis was given. Due to his age, recent surgery with multiple transfusions for major bleeding, malignancy, use of dual antiplatelet for his PCI, he was considered high risk to develop another major bleed. Thus, low-dose alteplase (50 mg to run for 2 hours) was administered, without any episode of bleeding. Post-thrombolysis, the dyspnea was dramatically relieved, and the rest of his hospital course was uneventful. He remained on dual antiplatelet therapy and enoxaparin at 1 mg/kg/dose b.i.d. He was discharged stable in functional class II on the 9th hospital day and remained on dual-antiplatelet therapy and extended anticoagulation with enoxaparin to prevent possible cancer-related venothrombosis.
On follow-up at 3 months, the patient improved further and was in NYHA functional class I, being able to tolerate more than ordinary physical activities. There was no recurrence of any bleeding. Repeat echocardiogram showed concentric left ventricular remodeling with good wall motion and preserved systolic function. The right ventricle and atrium were both normal in size, with normal right ventricular systolic function and normal pulmonary artery pressures.
Discussion
Massive pulmonary embolism (MPE) and acute myocardial infarction (AMI) individually carries a serious prognosis [1], and the concomitant presence of both in the same patient, though a rare occurrence, usually ends in a fatal outcome [2,3]. In this report, we present a case of a hemodynamically unstable patient presenting with both MPE and AMI, and highlight the value of prompt recognition, evaluation utilizing different diagnostic modalities, and managing the patient with a calibrated therapeutic strategy resulting in a favorable outcome. Although there are treatment guidelines outlining treatment strategies for acute ST elevation MI (STEMI) and high risk MPE, the presence of both in the same patient is beyond current guidelines and requires an individualized, calibrated approach, especially in the setting of an increased bleeding risk.
This patient is high risk to develop venous thromboembolic complications considering the presence of prostate cancer, recent surgery, multiple blood transfusions and multiple risk factors for coronary artery disease. The patient presented symptoms of chest pain, dyspnea, syncope and hemodynamic instability which can be the presenting symptoms in either MPE or extensive AMI [1], making it difficult to distinguish one from the other. A high index is needed to suspect PE when the findings on chest x-ray and other ancillary evaluation are not commensurate to the severity of dyspnea, as in this case. Cardiac troponin is a highly sensitive and specific marker in identifying myocardial injury but can also be elevated in massive pulmonary embolism thus it is not useful in differentiating the two. However, significant troponin elevation has a prognostic value for both and may indicate higher risk of complications and mortality [4].
Electrocardiographic changes indicative of right ventricular strain in MPE [4] includes T-wave inversion in V1-V4, a QR pattern in V1, S1Q3T3 pattern and incomplete or complete right bundle branch block—all of which were not present in the patient. Rather, the patient presented with anterior STEMI. ECG findings of MPE may be masked in the presence of an AMI.
Echocardiography is a rapid, practical and sensitive technique for detecting abnormalities associated with both coronary artery disease and MPE. In this case, the transthoracic echocardiogram confirmed a dilated right atrium, and in it, a likely mobile embolus in transit, and enlarged right ventricle with increased right-sided pressure indicative of right ventricular dysfunction and elevated pulmonary artery pressure.
Since in the majority of cases, pulmonary embolism originates from deep venous thrombosis (DVT) of the lower extremities, compression venous ultrasonography is considered to be the primary noninvasive method for diagnosing DVT. In this case, the lower extremity ultrasound identified an acute left popliteal DVT and presence of pulsatile venous Doppler flow pattern in both lower extremities, suggestive of venous hypertension, pulmonary hypertension and right sided heart failure [5]. Such findings are considered sufficient evidence to warrant anticoagulant treatment [5].
Anticoagulation is still the cornerstone of treatment for acute pulmonary embolism [6], and in high risk pulmonary embolism, intravenous systemic thrombolysis may be opted. Unlike in an AMI, effective use of thrombolysis for PE shows a wider “time window” of benefit, and those who receive thrombolysis up to 14 days after onset of new symptoms or signs may still derive benefit from this treatment [6]. Such was evident in this case. Although patient was also considered at high risk of recurrent bleeding, a calibrated approach to administer half the recommended dose of the tissue plasminogen activator alteplase (50 mg over two hours of intravenous infusion), proved to be effective and life-saving. Such approach may be preferred in those at high risk of bleeding, rather than using the standard dose of 100 mg of alteplase over two hours, which has been used and recommended in the majority of studies investigating systemic thrombolysis [7]. There is limited evidence to support a lower dose of thrombolytic therapy particularly in MPE. The Moderate Pulmonary Embolism Treated with Thrombolysis (MOPETT) trial evaluated the efficacy and safety of low-dose alteplase (50 mg) in patients with moderate or submassive PE [8]. The intervention group showed a significant early reduction in pulmonary artery systolic pressure, and this effect was maintained at 28 months [8].
To our knowledge, this is the first reported case showing the efficacy and safety of low-dose alteplase in MPE with documented emboli in transit at the superior vena cava and right atrium, in a setting of an acute anterior wall STEMI, who had just undergone PCI. This is also the first reported case in our country to manage such a complicated case of MPE with low-dose alteplase (50 mg) leading to a favorable outcome. The rare coexistence of these two vascular pathologies carries significant treatment implications requiring an individualized approach. When feasible, more aggressive modalities such as catheter-based therapy for the MPE may be considered. However, in a setting where expertise for catheter-based therapy is not available and the patient has considerable bleeding risks, it is rational to pursue an alternative treatment using low-dose thrombolysis, with dramatic improvement and safety as was shown in this case.
A repeat echocardiogram was done on follow-up after three months, to re-assess both right and left ventricular function. Those with persistent right ventricular dysfunction on echocardiography at 3 months after diagnosis have a four-fold increased risk of recurrent venous thromboembolism compared to those without right ventricular dysfunction [9]. The follow up echocardiogram done in this patient showed concentric left ventricular remodeling with good wall motion and preserved systolic function, as well as resolution of the right ventricular dysfunction and normalization of the pulmonary artery pressure indicating successful resolution of the DVT and MPE, and prevention of their recurrence.
Acknowledgement
The authors would like to acknowledge the following for their inputs on this case report, as well as their encouragement and moral support to complete and publish the paper: Dr. Bernadette Tumanan-Mendoza, Dr. Dante Morales, Dr. Nelson Abelardo, and Dr. Petrarch Bravo.
Conflict of interest
None
Source of funding
None
References
- Akgedik R, Günayd?n Z, Y?ld?r?m B, Da?l? C, Bekta? O. What should be done in the event of simultaneous massive pulmonary embolism and myocardial infarction with ST elevation? Turk Kardiyol Dern Ars 43 (2015): 734-738.
- Tomov I, Pelov R, Zlatareva N, Combined acute myocardial infarct and pulmonary thromboembolism with fatal outcome. Vutr Boles 24 (1985): 78-85.
- Perdigão C, Monteiro J, Andrade A, Ribeiro EC. Pulmonary embolism in acute myocardial infarct. Clinico-anatomic study of 19 cases. Acta Med Port 2 (1989): 65-71.
- Konstantinides SV,Torbicki A,Agnelli G,Danchin N,Fitzmaurice D,et al.Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC). 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J 35 (2014): 3033–3069.
- Abu-Yousef MM,Kakish ME,Mufid M.Pulsatile venous Doppler flow in lower limbs: highly indicative of elevated right atrium pressure. AJR Am J Roentgenol 167 (1996): 977-980.
- Mann DL, Zipes DP, Libby P, Bonow RO, Braunwald E (2015).Braunwald's Heart Disease: A textbook of cardiovascular medicine(Tenth edition.). Philadelphia, PA: Elsevier/Saunders
- Greco F, Bisignani G, Serafini O, Guzzo D, Stingone A, Plastina F. Successful treatment of right heart thromboemboli with IV recombinant tissue-type plasminogen activator during continuous echocardiographic monitoring: a case series report. Chest 116 (1999): 78-82.
- Sharifi M, Bay C, Skrocki L, Rahimi F, Mehdipour M. ‘‘MOPETT’’ Investigators. Moderate pulmonary embolism treated with thrombolysis (from the ‘‘MOPETT’’ Trial). Am J Cardiol 111 (2013): 273-277.
- Grifoni S,Vanni S,Magazzini S,Olivotto I,Conti A,et al. Association of persistent right ventricular dysfunction at hospital discharge after acute pulmonary embolism with recurrent thromboembolic events. Arch Intern Med 166 (2006): 2151.


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 