Does Emotion before a Chemotherapy Course Break a Heart? Cardiogenic Shock Complicating Takotsubo Cardiomyopathy
Taamallah K1*, Arous Y3, Haggui A1, Hajlaoui N1, Fehri W1, Msaddek F2
1Department of cardiology of the Military hospital of Tunis, Bab alioua, 1008 Montfleury, Tunisia
2Department of hemato-oncology of the Military hospital of Tunis, Bab alioua, 1008 Montfleury, Tunisia
3Department of Radiology of the Military hospital of Tunis, Bab alioua, 1008 Montfleury, Tunisia
*Corresponding author: Taamallah K, Department of cardiology of the Military hospital of Tunis, Bab alioua, 1008 Montfleury, Tunisia.
Received: 13 May 2022; Accepted: 13 May 2022; Published: 13 September 2022
Article Information
Citation: Taamallah K, Arous Y, Haggui A, Hajlaoui N, Fehri W, Msaddek F. Does Emotion before a Chemotherapy Course Break a Heart? Cardiogenic Shock Complicating Takotsubo Cardiomyopathy. Cardiology and Cardiovascular Medicine 6 (2022): 461-465.
View / Download Pdf Share at FacebookAbstract
Introduction: Takotsubo cardiomyopathy is characterized by a broad spectrum of clinical presentations. Despite its reversibility, it is associated with severe adverse in-hospital events. Recent studies have identified a high incidence of TTC in cancer patients.
Case Report: We report the case of a 59-year-old-man with TTC occurring just before injecting of chemotherapy drug complicated by cardiogenic shock and acute heart failure. Under medical treatment, a complete recovery of cardiac contractility was noted within three days. chemotherapy regimen has been stopped and then alleviated from RCHOP to the RCVD. No recurrence of cardiac symptoms was noted during the follow-up period. Left ventricle ejection fraction, BNP, and troponin levels were maintained normal.
Conclusion: Takotsubo cardiomyopathy can occur in cancer patients leading to interruption of chemotherapy because of left ventricular dysfunction, which may adversely affect the oncologic outcome because of interruption of cancer treatment. We report this case to remind clinicians that TTC should be considered in the differential diagnosis of chest pain in cancer patients who present with ECG findings characteristic of acute coronary syndrome. We hope to facilitate prompt diagnosis for an early intervention that allows rapid recovery of myocardial function, and resume cancer therapy early and may help lower mortality.
Keywords
<p>Cancer; Cardiotoxicity; Chemotherapy; Echocardiography; Stress; Takotsubo Cardiomyopathy</p>
Article Details
1. Background
Takotsubo cardiomyopathy (TTC), also known as stress-induced cardiomyopathy, is characterized by a broad spectrum of possible clinical presentations [1]. Despite its reversibility, it is associated with severe adverse in-hospital events, the acute stage of the disease can be complicated by hypotension, shock, arrhythmias, heart failure, and death [2]. Recent studies have identified a high incidence of TTC in cancer patients [3]. TTC has been linked to emotional stress due to the excessive release of catecholamines as a mechanism of response [1]. The emotional stress of a cancer diagnosis, physical stress, ongoing chemotherapy, the abnormal cancer-related wasting syndrome may increase the risk of developing TTC. There have been very few, and small studies were addressing the prevalence and outcomes of cancer patients with TTC [4]. We report the unusual case of a 59-year-old-man with Tako-Tsubo cardiomyopathy (TTC) occurring just before injecting of chemotherapy drug complicated by cardiogenic shock and acute heart failure, to highlight the importance of performing a differential diagnosis with cardiotoxicity, since TTC presents a rapid recovery allowing non-interruption of chemotherapy treatment.
2. Case Report
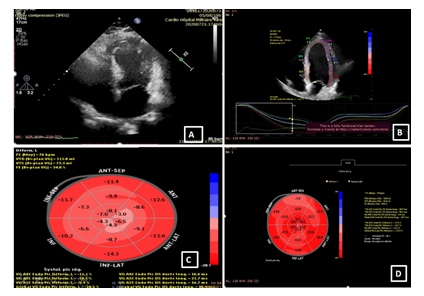
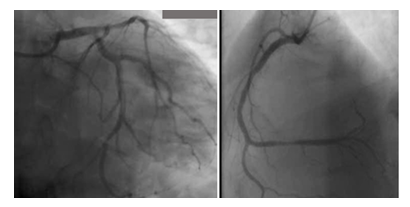
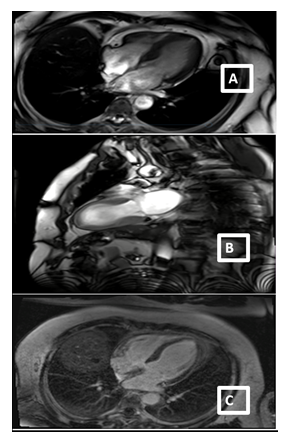
A 59-year-old man with a history of diabetes mellitus and systemic arterial hypertension was admitted to the department of hemato-oncology for chemotherapy for follicular non-Hodgkin’s lymphoma diagnosed in May 2020, He did not have any previous cardiac history; cardiac investigations done before starting chemotherapy were normal. Our patient had completed two cycles of chemotherapy of CHOP plus Rituximab regimen (doxorubicin, vincristine, cyclophosphamide, rituximab, and methylprednisolone), with the cumulated dose of anthracycline of 100mg/m2. Before the infusion of the third course of chemotherapy, the patient was stressed, he had pain because of a laborious arterial punction, he developed acute shortness of breath, discomfort, chest pain, and excessive sweating. He subsequently became tachycardic with a heart rate of 120 bpm, with rapid progression to cardiogenic shock requiring inotropic agents with blood pressure at 60/40 mm Hg and signs of cardio-circulatory failure and tissular hypoperfusion. The patient was then transferred to an intensive care unit. A 12-lead ECG showed a sinus tachycardia of 130 bpm, associated with ST-segment elevation in leads V1-V6. There was a rise in the level of serum troponin-I at 100 ng/L (normal range 0–40). An urgent transthoracic bedside echocardiogram revealed normal-sized but apical ballooning with hyperdynamic basal segments. The estimated left ventricular (LV) ejection fraction (EF) was 38%. There were no signs of tamponade and pulmonary embolism (figure 1). The 2D strain was decreased at -10.5% (figure 1c). A provisional diagnosis of anterior ST-elevation myocardial infarction was made, coronary angiography was performed, demonstrated a non-obstructive right coronary artery atheroma which did not explain the cardiogenic shock (figure 2). A subsequent cardiac MRI (figure 3) reported an akinetic left ventricle from the mid ventricle to the apex with impaired global ejection fraction, there were signs of myocardial edema in the LV walls in T2-weighted sequences, suggestive of Takotsubo cardiomyopathy and there was no evidence of myocardial inflammation, myocardial infarction, fibrosis, or infiltration in the late gadolinium enhancement sequences. Based on these findings, a diagnosis of TTC was made. Treatment with β-adrenoceptor antagonists: Bisoprolol 1.25 mg daily and ACE inhibitors: ramipril 2.5 mg daily (titrating the doses up to the maximum doses tolerated by the patient) was initiated. A remarkable improvement in LV function was noted in the echocardiogram performed three days after the event, LVEF and 2D strain became in the normal range (figure 1d). The chemotherapy regimen has been alleviated from RCHOP to the RCVD regimen which does not contain doxorubicin, known to be highly cardiotoxic; it was considered that its reintroduction in a patient with impaired myocardial function could be deleterious. The patient remains in follow-up in the onco-cardiology unit, he was examined weekly, for four weeks after the event, and reported no recurrence of his symptoms. in view of this favorable evolution, the standard regimen of chemotherapy (RCHOP) was resumed in combination with cardio-protective drugs. After each course of chemotherapy, troponin and brain natriuretic peptide (BNP) remained at normal levels, and the size and function of the left ventricle were maintained normal. The patient has completed his chemotherapy for six months, cardiological follow-up every three months has been maintained, no cardiac complications have been noted.
3. Discussion
This report illustrates the case of a male patient with lymphoma, treated with chemotherapy, presenting TTS occuring immediately before the starting of chemotherapy administration, manifzstzd as chest pain, diffuse anterior ST segment elevation and complicated by cardiogenic shock. Takotsubo cardiomyopathy, first described in the 1990s in Japan, is a form of reversible and transitory ventricular systolic dysfunction with a clinical and electrocardiographic presentation similar to acute coronary syndrome (ACS) or an acute heart failure [3]. In the general population, TTC is considered relatively uncommon with an incidence rate of 1% to 2%, this incidence is higher in cancer patients ( approximately at 10%). Many reports have been published linking emotional or physical stress induced by chemotherapy administration or healthcare-associated procedures to TTS. Sattler et al. [5] reported that the prevalence of malignancy was high amongst TTC patients and that cancer was a risk factor for worse outcomes. One of the hypotheses is that cancer and TTS share similar triggering mechanisms, which consist in the activation of the sympathetic nervous system [4]. This disease predominantly affects postmenopausal women [6], a similar incidence in men and women was noted in patients with TTC induced by cancer drugs [7]. Giza et al. [3] reported that the most common cancers associated with TTC were lymphoproliferative neoplasms (23.3%), followed by gastrointestinal (17%), ovarian (13.3%), and breast cancer (10%). The exact trigger for TTC is often challenging to identify. An emotional, physical, or combined trigger can precede the Takotsubo syndrome event, but this is not obligatory. The triggers which precipitate TTC in cancer patients are unknown, but a variety of factors have been described: chemotherapy, radiotherapy, radiofrequency ablation, and cancer pain [4]. Although our patient outwardly displayed little anxiety about his diagnosis and treatment approach, it is possible that he felt a heightened degree of internal emotional stress that may have contributed to the development of TTC. The pain during the arterial punction could have triggered a cardiomyopathy induced by the release of several inflammatory cytokines and metabolites related to this physical distress. Endothelial dysfunction in microvascular and epicardial coronary arteries might be a predisposing factor for the development of TTC, it occurs frequently in patients with cancer, especially during and after systemic chemotherapy or radiotherapy of the thoracic region, our patient had hypertension and diabetes mellitus and might have already endothelial dysfunction which might be worsened under chemotherapy [7]. Takotsubo cardiomyopathy remains a diagnostic challenge in cancer patients due to the difficulty in distinguishing TTC from chemotherapy-induced cardiomyopathy, as the two diseases differ in their management, reversibility, and long-term outcomes. Identifying reversible cardiomyopathy such as TTC in cancer patients is imperative, especially in patients who are undergoing curative cancer treatment. Indeed, recognition of chemotherapy-induced cardiotoxicity is important since repeated administration of the offending drug can potentially lead to irreversible cardiac damage. On the other hand, premature discontinuation of an effective anti-neoplastic agent due to co-existing cardiac events not directly related to therapy may increase oncologic morbidity and mortality. For these reasons, early diagnosis and appropriate management of TTC in patients with cancer are crucial. The classic wall motion abnormalities of the LV apical and mid segments which extend beyond a single coronary distribution or the typical appearance of “apical ballooning” provide a clue to the diagnosis of TTC. When LV contractile dysfunction is diffuse or global, it is challenging to distinguish Takotsubo syndrome from another type of chemotherapy-related cardiac dysfunction [8]. Certain chemotherapeutic agents have been implicated in triggering this form of cardiomyopathy. 5-fluorouracil is the most commonly reported anti-neoplastic agent associated with TTC [3,7]. Other chemotherapeutic agents were also implicated such as Bevacizumab, Capecitabine, Bevacizumab, Combretastatin, Tyrosine kinase inhibitors, Rituximab, Immune checkpoint inhibitors, Daunorubicin, Cytarabine, Trastuzumab, Ipilimumab, and Miscellaneous drugs. Our patient was taken Rituximab as part of the R CHOP regimen [7], the previous usage of this drug might have contributed to the development of myocardial dysfunction in our patient. Two postulated mechanisms explaining the pathophysiology of rituximab-induced TTC have been proposed: Coronary vasospasm and direct cardiotoxic effect [9], and the presence of diffuse reticulin fibers along in cardiac myocytes after treatment with rituximab with increased levels of transforming growth factors (TGF)–β, explaining cardiac events during later infusions of rituximab [10]. TTC related to Vincristine, Doxorubicin, Cyclophosphamide (administered in our patient) was not reported. In the case of TTC in cancer patients under chemotherapy, the question of whether the cancer drug is the offender, a mere bystander, or paved the way by impairment of the myocardial function, is a crucial question that needs to be examined in a case-by-case scenario. Rapid recovery of left ventricular function was observed in our patient, demonstrating the reversibility of this disease and the positive response to treatment. Recovery of systolic function typically occurs within one to four weeks [11]. In patients with TTC occurring under chemotherapy, the recovery of a normal left ventricular function was noted within a mean duration of one month (range five days to three months) [7]. Our patient was maintained under β-adrenoceptor antagonists and ACE inhibitors, whether medical treatment can be discontinued after normalization of LVEF remains debated. However, some authors supported the value of indefinitely continuing medical therapy by conducting a randomized trial, which assessed the effect of medical treatment withdrawal in patients who have recovered their LVEF after a dilated cardiomyopathy [12]. In a literature review conducted by Desai et al. [7] 44% of the patients in whom heart failure treatment was discontinued developed a relapse of cardiomyopathy during the first six months. The chemotherapy regimen was temporarily alleviated in our patient. Decision to change the patient's cancer treatment was taked after a thorough discussion between cardiologist and hematologist who weighed the benefits and risks of changing the chemotherapy regimen. Given the implications of suboptimal treatment (RCEOP, without anthracycline) on the risk of relapse, which are not trivial, and given the recovery of ventricular systolic function, we judged that resumption of the standard regimen after one month, in combination with cardio-protective drugs, is necessary. In patients who require further cancer treatment, rechallenging with the culprit anti-neoplastic therapy poses a significant clinical dilemma. There is limited data on the safety of reintroducing the chemotherapeutic agent. In a retrospective analysis of 30 patients with cancer and stress-induced cardiomyopathy treated at the MD Anderson Cancer Center [3], 21 patients required ongoing cancer treatment. Among them, 16 were able to safely resume chemotherapy after normalization of LVEF without recurrence of TTC. The median time to resume cancer treatment was 20 days after TTC. Given the worse outcomes in TTC patients with cancer and the difficulty in discriminating this cardiomyopathy from chemotherapy-induced cardiomyopathy and acute coronary syndrome, we believe that it is essential to develop an algorithm that includes imaging tests such as an echocardiogram, coronagraphy, MRI, to help make a timely diagnosis. The report of this case is an opportunity to stress the importance of pain control and emotional support in cancer patients.
4. Conclusion
In cancer patients, TTC should be considered in the differential diagnosis of chest pain, and electrocardiographic abnormalities without a culprit coronary artery lesion. Although the prognosis from TTC is generally good, this condition can lead to an interruption in chemotherapy, which may adversely affect the oncologic outcome. Early intervention may help lower mortality, rapid recovery of myocardial function, and resume cancer therapy early. Therefore, medical professionals need to recognize, monitor, and treat this condition appropriately.
Declarations
Conflict of Interest
The authors declare that they have no potential conflicts of interest related to this publication.
Funding
No funding to declare.
Authors’ Contributions
Taamallah Karima, Arous Younes: Wrote the first draft of the manuscript. Taamallah Karima: Bibliography research. Haggui Abeddayem, Hajaloui Nadhem: Contributed to the writing of the manuscript and bibliography research. Fehri Wafa, Msaddek Fehmi: Made critical revisions and approved the final version. All the authors reviewed and approved the final manuscript.
Availability of Data and Materials
Not applicable.
Ethics Approval and Consent to Participate
This case report was approved by the ethics committee of the Military hospital, Tunis, Tunisia. The patient gave written consent to use his clinical and paraclinical information to prepare this article.
Consent for Publication
The patient the patient allowed to publish her case, as well as relevant related workup and diagnostic images, presented in the medical literature.
Acknowledgments
No acknowledgments to declare.
References
- Pavin D, Le Breton H, Daubert C. Human stress cardiomyopathy mimicking acute myocardial syndrome. Heart Br Card Soc 78 (1997): 509-511.
- Singh K, Carson K, Shah R, et al. Meta-analysis of clinical correlates of acute mortality in takotsubo cardiomyopathy. Am J Cardiol 113 (2014): 1420-1428.
- Giza DE, Lopez-Mattei J, Vejpongsa P, et al. Stress-Induced Cardiomyopathy in Cancer Patients. Am J Cardiol 120 (2017): 2284-2288.
- Joy PS, Guddati AK, Shapira I. Outcomes of Takotsubo cardiomyopathy in hospitalized cancer patients. J Cancer Res Clin Oncol 144 (2018): 1539-1545.
- Sattler K, El-Battrawy I, Lang S, et al. Prevalence of cancer in Takotsubo cardiomyopathy: Short and long-term outcome. Int J Cardiol 238 (2017): 159-165.
- Templin C, Ghadri JR, Diekmann J, et al. Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med 373 (2015): 929-938.
- Desai A, Noor A, Joshi S, et al. Takotsubo cardiomyopathy in cancer patients. Cardio-Oncol Lond Engl 5 (2019): 7.
- Citro R, Okura H, Ghadri JR, et al. Multimodality imaging in takotsubo syndrome: a joint consensus document of the European Association of Cardiovascular Imaging (EACVI) and the Japanese Society of Echocardiography (JSE). Eur Heart J Cardiovasc Imaging 21 (2020): 1184-1207.
- Cheungpasitporn W, Kopecky SL, Specks U, et al. Non-ischemic cardiomyopathy after rituximab treatment for membranous nephropathy. J Ren Inj Prev 6 (2017): 18-25.
- Kanamori H, Tsutsumi Y, Mori A, et al. Delayed reduction in left ventricular function following treatment of non-Hodgkin’s lymphoma with chemotherapy and rituximab, unrelated to acute infusion reaction. Cardiology 105 (2006): 184-187.
- Sharkey SW, Lesser JR, Zenovich AG, et al. Acute and reversible cardiomyopathy provoked by stress in women from the United States. Circulation 111 (2005): 472-479.
- Halliday BP, Wassall R, Lota AS, et al. Withdrawal of pharmacological treatment for heart failure in patients with recovered dilated cardiomyopathy (TRED-HF): an open-label, pilot, randomized trial. Lancet Lond Engl 393 (2019): 61-73.


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 