Electrocardiography in the Assessment of Right Ventricular Dysfunction and related Clinical Outcomes in Patients with pulmonary embolism
Mohsen Sadeghi1, Negar Zarei1,2, Arda Kiani3, Babak Sharif-Kashani4, Kimia Taghavi1, Atefeh Abedini*1
1Chronic Respiratory Diseases Research Center, National Research Institute of Tuberculosis and Lung Disease (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran
2Department of Internal Medicine, Shahid Beheshti University of Medical Sciences, Tehran, Iran
3Tracheal Diseases Research Center, National Research Institute of Tuberculosis and Lung Diseases (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran
4Tobacco Prevention and Control Research Center, National Research Institute of Tuberculosis and Lung Diseases (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran
*Corresponding author: Atefeh Abedini, Chronic Respiratory Diseases Research Center, National Research Institute of Tuberculosis and Lung Disease (NRITLD), Shahid Beheshti University of Medical Sciences, Tehran, Iran
Received: 23 June 2019; Accepted: 16 August 2019; Published: 30 August 2019
Article Information
Citation: Mohsen Sadeghi, Negar Zarei, Arda Kiani, Babak Sharif-Kashani, Kimia Taghavi, Atefeh Abedini. Electrocardiography in the Assessment of Right Ventricular Dysfunction and related Clinical Outcomes in Patients with pulmonary embolism. Cardiology and Cardiovascular Medicine 3 (2019): 277-285.
View / Download Pdf Share at FacebookAbstract
Introduction: Several studies have been done on the relationship between right ventricular (RV) dysfunction in pulmonary embolism and electrocardiographic changes in the Electrocardiography (ECG). Confirmation of such relationships will help early detection of pulmonary embolism and, as a result, reduce the need for thrombotic and aggressive therapies. The aim of this study was to determine the relationship between ECG changes and the clinical outcomes of patients with pulmonary embolism.
Materials and Methods: Patients who referred to Masih Daneshvari Hospital during the year 2016 with acute dyspnea, chest pain and other symptoms associated with pulmonary embolism, and had embolism confirmed by Computed Tomography Angiography (CT angiography), in terms of symptoms Hemodynamic and echocardiographic disorders were studied. ECG results were compared with those of troponin I and previous uric acid found in the patient records, and the results were analyzed.
Results: In this study, 83 patients with mean age of 51.5 ± 15.8 and 25 to 83 years old were studied. Of these, 64 (77.1%) were male and 19 (22.9%) were female. The mortality rate in the study was 6% and 6% (7%) needed surgery. Among the findings of the ECG, the relationship between RAD, ST depression, T preordial, Qr with RV dysfunction was significant (p <0.001, p = 0.007, p = 0.003, p <0.001). Also, RAD, ST depression, T preordial, Qr
with secondary TR showed a significant relationship (p = 0.04, p = 0.03, p = 0.01, p = 0.001). There was a significant relationship between ST-depression and patient's mortality among ECG findings (p = 0.04). In addition, treatment with fibrinolytic and the need for surgery with RV dysfunction was significantly associated.
Discussion: Electrocardiographic changes were associated with right ventricular dysfunc
Keywords
<p>Right Ventricular Dysfunction; Computed Tomography Angiography; Pulmonary Embolism; Prognosis; Electrocardiography; Disease Outbreaks; Tachycardia</p>
Article Details
Introduction
Acute pulmonary embolism is a common and sometimes fatal illness [1]. Clinical evaluation and diagnostic testing are vital prior to the onset of anti-coagulane therapy. Since, avoiding of delay in the onset of treatment reduces the mortality and morbidity [2]. PE is associated with varying degrees of lung arterial obstruction, which may lead to an increase in arterial blood pressure. This increase in pressure increases the right ventricular (VE) velocity, which results in an increase in the RV wall, leading to dilatation and RV hypokinesis and secondary cirrhosis of the caspade. [3,4,5]. Acute pulmonary embolism may result in right ventricular (RV) enlargement and disorder, which is life-threatening [3]. Electrocardiogram (ECG) disorders are common in patients with suspected PE. The most common findings are tachycardia and nonspecific changes in the ST segment and (T) (70%). The disturbances that appear to suggest the PE (S1Q3T3, RV strain criteria, new incomplete RBBB) are rare (less than 13%). ECG disorders associated with poor prognosis in PE include atrial arrhythmias, bradycardia, tachycardia, S1Q3T3, Inferior Q waves (leads II, III and avf), new incomplete RBBB, ST-segment, and T-wave inversionAnterior [3]. In patients suspected of having PE, levels of troponin T and I have no diagnostic sensitivity and specificity. However, they can be used to evaluate prognosis in suspected PE patients.
In numerous studies on the association of ECG and cardiac biomarkers with right ventricular function in pulmonary embolism, variable results have been obtained, which in some cases showed a significant relationship with undesirable clinical outcomes [4]. However, the role of ECG in the diagnosing the RV dysfunction in APE is rather unclear.
Objective of the study
Early detection of high-risk patients in acute pulmonary embolism in a clinical trial is a challenge. Although none of the ECG symptoms are not sufficient for a specific diagnosis, but in cases of high clinical suspicion for PE, ECG findings help predict severe pulmonary embolism and avoid delay in treatment. The aim of this study was to determine the relationship between ECG changes and the clinical outcomes of patients with pulmonary embolism in Masih Daneshvari Hospital in Tehran.
Material and methods
Study Design
In a retrospective and analytical study, patients with clinical signs of pulmonary embolism, such as shortness of breath and chest pain who visited the Masih Daneshvari Hospital between January 2016 and December 2016, entered the study and had embolism confirmed by Computed Tomography Angiography (CT angiography). Exclusion criteria were: Electrolyte imbalance in the patient, renal impairment, Congestive Heart Failure (CHF), or Angina pectoris. People with previous MI or under anti-arrhythmic treatment have also been excluded from the current study.
At the beginning of the study, clinical manifestations, 12-lead ECG, and hemodynamics were studied [6]. The studied ECG markers were S1Q3T3 pattern, T wave inversion in V1 to V3, tachycardia sinus and right branch block.
T-wave inversion was interpreted by inspecting the pointed and symmetrical inverted T waves from leads V1 to V3. The S1Q3T3 pattern was defined according to the criteria of Mc Ginn and White10 (S wave in lead I and Q waves in lead III with a range> 0.15 mV [1.5 mm] associated with T-wave inversion in lead III). Right bundle branch blocks were defined according to common criteria. Sinus tachycardia was defined as an increasing in sinus rate to 2:100 beats/min.
RV function was measured using transthoracic echocardiography on the first day of hospitalization. In patients with 2: 1 of the following prescriptions, acute RV disorder was predicted: (1) paradoxical septal systolic sign, (2) RV/left ventricular end- diastolic diameter ratio >1 or end-diastolic diameter >30 mm in the 4-chamber (RV enlargement), and (3) RV/atrial gradient >30 mm Hg or Doppler pulmonary acceleration time <90 ms (pulmonary hypertension).
Changes in the ECG axis were initially evaluated and correlated with echocardiographic findings. Cardiac biomarkers (troponin I and uric acid) were also studied. In addition, treatment was prescribed and ultimately mortality was evaluated in patients.
Statistical analysis
Statistical analysis was performed using SPSS software version 7.0 (SPSS, Inc., Chicago, Illinois). While, continuous data was displayed under the mean + SD. dichotomous data was shown with numbers and percentages. Consecutive variables were studied using Student’s t-test. In addition, dichotomous variables were compared using Chi-square statistics. Mean values in different groups were analyzed by ANOVA. The value of p-value <0.05 reflected significant value and a 95% confidence interval was reported.
Results
In the current study, 83 patients with average age of 51.3±15.8 ranging from 25 to 83 years old were investigated. 64 patients (77.11%) were male and 19 (22.89%) were female. The mean heart rate was 92.7 ± 8.28, with a range of 52 to 130 beats per minute and an incidence of 26.63% tachycardia. The mean concentration of uric acid in patients was 1.9 ± 1.30 mg / dl with a range of 1.0-1.6 mg / dL and the mean arterial pressure was 96.2 ± 2.6 mg / dl with a range of 62 to 130 milligrams M Mercury. The mean arterial blood pressure (PAP) in patients was 15.8 ± 38.7 and 20 to 100, respectively.
ECG findings
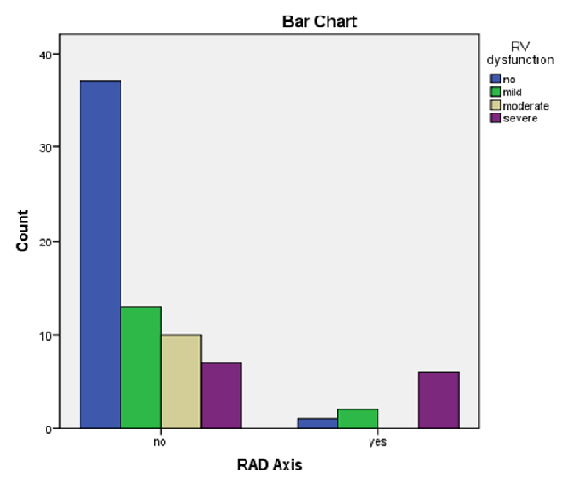
Out of 83 participants, only 23 patients (50%) showed normal electrocardiographic findings. However, severe RV dysfunction was seen in 11.11%, moderate RV dysfunction at 13.16%, and mild RV disorder in 19.27% of patients (table 1). 19.74% of the participants showed a secondary TR pattern, while 6.58%, 22.37%, 51.32% of patients had a severe, moderate, and mild secondary TR pattern, respectively. Regarding the natural rhythm outbreak, 76 patients (59.51%) had sinus rhythm and 7 patients (43.8%) had non sinus rhythm. 9 patients (10.84%) had RAD. The prevalence of ST depression was 6 (7.23%) in this study. The preordial T prevalence was 25 (12.30%) and the prevalence of inf Q was 22 (26.51%). The pattern of S1Q3T3 with the prevalence of 34 patients (40.96%) and inversion of the T wave in the findings of V1 to V3 with a frequency of 1.30%, as well as Inf Q, with the frequency of 26.51% were the highest ECG findings in pulmonary embolism.
|
Frequency and percentage of RV dysfunction and RAD Axis |
|||||
|
RAD Axis |
Total |
||||
|
no |
yes |
||||
|
RV dysfunction |
no |
Number |
37 |
1 |
38 |
|
% RAD Axis |
55.2% |
11.1% |
50.0% |
||
|
mild |
Number |
13 |
2 |
15 |
|
|
% RAD Axis |
19.4% |
22.2% |
19.7% |
||
|
moderate |
Number |
10 |
0 |
10 |
|
|
% RAD Axis |
14.9% |
.0% |
13.2% |
||
|
severe |
Number |
7 |
6 |
13 |
|
|
% RAD Axis |
10.4% |
66.7% |
17.1% |
||
|
Total |
Number |
67 |
9 |
76 |
|
|
% RAD Axis |
100.0% |
100.0% |
100.0% |
||
Table 1: Frequency and percentage of RV dysfunction and RAD Axis
Qr in v1 was reported in 11 patients (13.25%). Also, incomplete RBBB was observed in 2 patients (41.2%). The mean troponin level in 41.86% of patients was more than 0.02 mg / dl and in 58.14% it was less than or equal to 0.02 mg / dL. Five patients (6.02%) underwent fibriolysis and 6 patients (7.23%) underwent surgery. A massive PTE was reported in 11 patients (13.25%) and mortality rate was 6.02% in the present study.
Echo findings
Sinus tachycardia was significantly different from RV dysfunction (p = 0.008). Patients with ST depression pattern in the ECG had higher mean PAP in contrast to those with other patterns (p <0.0001).
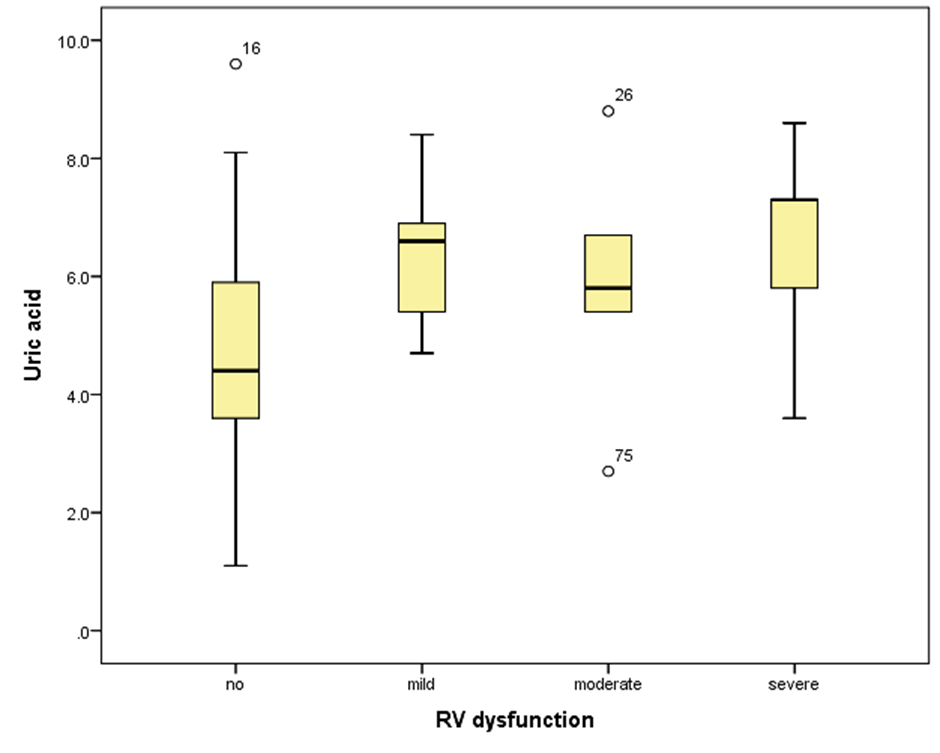
Uric acid and age were significantly higher in patients with S1Q3T3 pattern in the ECG than in those with other patterns (p ≤0.05). Patients with Inf Q pattern in ECG had a higher average WBC than those with other ECG patterns (14554.54±11953.23 vs 10670.49±3581.26, p = 0.02). Uric acid and pulmonary arterial pressure (PAP) in patients with Qr pattern in v1 in ECG were significantly higher than those with other ECG patterns (6.83±2.00 vs 5.10±1.79, p = 0.007) (55.45±17.81 vs 35.88±13.79, p <0.0001). Also, in T-preordial and RAD-qualified individuals, the two above mentioned parameters were high (p <0.0005).
In the current study, the RV dysfunction showed a significant relationship with Qr in v1, and the necessity for surgical treatment (p=0.006, p <0.0001 respectively). Additionally, RV dysfunction showed significant relationship with RAD Axis (p <0.001) (Diagram 1).
However, no significant relationships were seen between RV dysfunction and ST depression, RBBB, and incomplete RBBB (p = 0.009, p = 0.9, p = 0.2 respectively). T precordial and RV dysfunction showed significant relationship (p = 0.029). However, RV dysfunction did not show any correlations with inf Q or S1Q3T3 pattern (p = 0.1, p = 0.09). No substantial associations were appreciated among RV dysfunction and mean heart rate, mean arterial pressure, and troponin level (p >0.05, p >0.05, p = 0.05 respectively). On the other hand, RV dysfunction did not show significant correlation with level of uric acid (p=0.022) (Diagram 2).
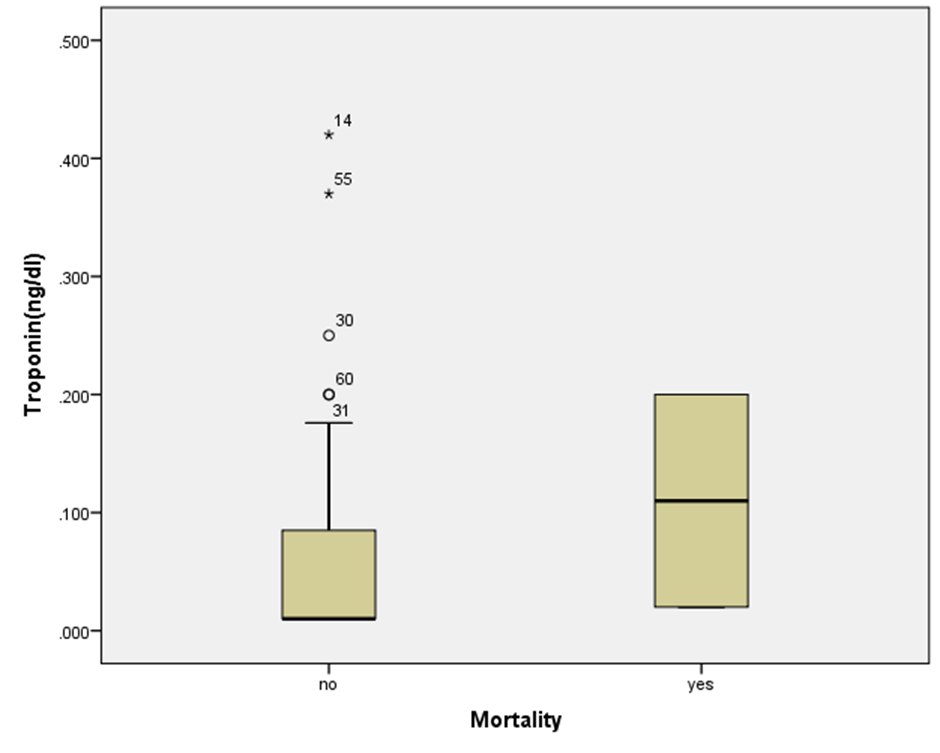
Statistical analyses showed that there was no significant difference in the mean PAP level, the presence of tachycardia, mean MAP level, mean troponin level, and mean level of uric acid between those without and with mortality (p=0.1, p >0.05, p >0.05, p >0.05, p >0.05). Moreover, there was no significant difference in the mean troponin level, between those without and with mortality (p >0.05) (Diagrams 3).
There was a significant correlation between mortality rate and either ST depression or necessity for surgical treatment (p=0.04, p=0.04 respectively). On the other hand, this correlation was not significant regarding the inf Q, Qr in v1, incomplete RBBB, RBBB, S1Q3T3 pattern, the presence of RAD Axis, the presence of T precordial p=0.5, p=0.8, p=0.2, p=0.06, p=0.4, p=0.1, respectively). Statistical analyses showed that there was a substantial association between Troponin level and Massive PTE (p=0.02).
Discussion
Acute pulmonary embolism is caused by several factors and can lead to right ventricular dysfunction (RV), which is fatal [7,8,9]. The aim of this study was to investigate the relationship between ECG changes with right ventricular disturbances and clinical findings in patients referred to pulmonary embolism clinic in Masih Daneshvari Hospital, Iran.
The definite frequency of PE is highly reliant on the clinical prospect for the manifestation of pulmonary embolism. The efficacy of the V/Q is improved when it is inferred beside the clinical similarity to PE. This was defined in the PIOPED study [10]. In a study by Rodger et al., on ECG of 264 patients only sinus tachycardia and incomplete RBBB were significantly more common in PE patients. The prevalence of RBBB in Rodger study was 6% [11]. Similarly, in the current study, the incidence of incomplete RBBB was significantly increased in the presence of the ST depression and T precordial and the necessity for prescribing fibrinolytic treatments.
The precordial outbreak in the current study was 3351%. Comparable with the study of Ferrari et al., in the current study, T precordial was correlated to RAD axis, S1Q3T3, elevated PAP, RV dysfunction, TR secondary, Qr in v1, incomplete RBBB, elevated Uric acid level, need for fibrinolytic treatment, massive PTE, as well as need for undergoing surgery [12].
Among Echo findings in the current study, RAD, ST depression, T precordial, Qr in V1 with RV dysfunction (p<0.001, p = 0.007, p = 0.003, p <0.001, and secondary TR (p = 0.04, p = 0.03, p = 0.01, p = 0.001) showed significant associations. In our study, common disorders were S1 Q3T3 patterns (41%), T precordial (30.1%) and inf Q (26.5%). In a study by Ryu et al., the most common ECG disorders were sinus tachycardia (39%), inverted T waves in V1-4 (39%) and S1Q3T3 (27%). Also, an average heart rate of ± 96 ± 20 / minute was reported. Complete RBBB was not obtained in any of the patients. Incomplete RBBB was reported in 10% and AF in 4% of patients [13]. Similarly, the mean heart rate in our patients was 92.7 ± 16.8, with a range of 52-130 beats per minute, with an outbreak of the of 26.33% tachycardia.
Increased arterial pressure in the lung due to obstruction of the embolus increases pressure on the right wall of the ventricle and leads to contraction and loss of the right ventricle. Afterwards, the right ventricle is dilated on the intervertebral septum or the coronary artery pushes to create right ventricular ischemia. This leads to the emergence of sinus tachycardia, atrial fibrillation (AF), creating a new wave in Line I and a new wave in Line III, with negative T wave in line III, the QRS axis deviation to the right and the inverted precordial T waves, which all of them can be attributed to structural changes in the right ventricle due to high arterial pressure [14,15,16].
The severity of such changes depends on the size of the embolus [15]. These changes may be due to myocardial ischemia, hypoxemia, and several chemical intermediates, such as catecholamines and histamines. Using an ECG scoring system, in the routine examination of patients with pulmonary embolism can be very effective [17].
Conclusions
In the present study, there was a significant relationship between ST depression of patients and their mortality. A complete review of ECG in determining the type of treatment and prognosis of mortality in patients with PTE can be valuable. Therefore, the use of ECG in routine clinical trials of patients with PTE is recommended.
Acknowledgments
The current study was obtained from a subspecialty thesis of pulmonology (IR.SBMU.NRITLD.REC.M.132) funded by Shahid Beheshti University of Medical Sciences. Hereby, we like to express our grateful appreciation.
Conflict of Interests
Authors have no conflicts of interest to declare. Authors give consent to submission and publication on the work. Authors disclose no relationship with any organization or industrial manufacture in any material discussed.
References
- Kukla P, McIntyre WF, Fijorek K, Mirek-Bryniarska E, Bryniarski L, Krupa E, etal. Electrocardiographic abnormalities in patients with acute pulmonary embolism complicated by cardiogenic shock. Am J Emerg Med 32 (2014): 507-510.
- Calkins H, Hindricks G, Cappato R, et al. HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation. Heart Rhythm 14 (2017): e275-e444.
- Skarlovnik A, Hrastnik D, Fettich J, Grmek M. Lung scintigraphy in the diagnosis of pulmonary embolism: current methods and interpretation criteria in clinical practice. Radiol Oncol 48 (2014): 113-119.
- Konstantinides S, Geibel A, Olschewski M, Heinrich F, Grosser K, Rauber K, etal. Association between thrombolytic treatment and the prognosis of hemodynamically stable patients with major pulmonary embolism: results of a multicenter registry. Circulation 96 (1997): 882-888.
- Lualdi JC, Goldhaber SZ. Right ventricular dysfunction after acute pulmonary embolism: pathophysiologic factors, detection, and therapeutic implications. Am Heart J 130 (1995): 1276-1282.
- Dielievska V, Kravchun P. Autoimmune Mechanisms of Cardiac Remodeling in Chronic Obstructive Pulmonary Disease. Acta Medica Bulgarica 44 (2017): 11-16.
- Yu S, Sun C, Wang Y, Yu C. Intracranial hematoma after stent treatment disappearance of ruptured anterior cerebral artery aneurysms with internal carotid artery stenosis. Biomedical and Biotechnology Research Journal (BBRJ) 1 (2017): 163-165.
- Tabarsi P, Moradi A, Marjani M, Baghaei P, Hashemian SM, Nadji SA, et al. Factors associated with death or intensive care unit admission due to pandemic 2009 influenza A (H1N1) infection. Ann Thorac Med 6 (2011): 91–95.
- Taghavi K, Fath PM, Hosseinkhani S, Mirzaei M, Behrooj H, Kiani A, etal. Construction and genetic improvement of copper bioreporter Escherichia Coli. Biomedical and Biotechnology Research Journal (BBRJ) 2 (2018): 26-30.
- Lakkakula S, Baraneedharan U, Lakkakula BV. Current perspectives on velocardiofacial syndrome. Biomedical and Biotechnology Research Journal (BBRJ) 1 (2017): 101-104.
- Rodger M1, Makropoulos D, Turek M, Quevillon J, Raymond F, Rasuli P, etal. Diagnostic value of the electrocardiogram in suspected pulmonary embolism. Am J Cardiol 86 (2000): 807-809.
- Stein PD, Dalen JE, McIntyre KM, Sasahara AA, Wenger NK, Willis PW 3rd. The electrocardiogram in acute pulmonary embolism. Prog Cardiovasc Dis 17 (1975): 247-257.
- Ryu HM, Lee JH, Kwon YS, Lee SH, Bae MH, Lee JH, etal. Electrocardiography patterns and the role of the electrocardiography score for risk
- Stein PD, Gottschalk A, Sostman HD, et al. Methods of Prospective Investigation of Pulmonary Embolism Diagnosis III (PIOPED III). Semin Nucl Med 38 (2008): 462-470.
- Wood P. Pulmonary Embolism: Diagnosis By Chest Lead Electrocardiography. Br Heart J 3 (1941): 21–29.
- Oshima Y1, Tachibana S, Hirota Y, Takeda Y, Kitajima I. Usefulness of arterial blood gas analysis and D-dimer measurement in the assessment of pulmonary embolism after orthopedic surgery. J Orthop Sci 11 (2006): 140-145.
- Daniel KR, Courtney DM, Kline JA. Assessment of cardiac stress from massive pulmonary embolism with 12-lead ECG. Chest 120 (2001): 474-481.


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 