Mechanical Atherothrombectomy (MATH) in the Treatment of Chronic Occlusions of the Popliteal Artery
Miroslav Bulvas
Cardiocenter, Third Faculty of Medicine, Charles University; Department of Surgery, University Hospital Královské Vinohrady, Division of Interventional Angiology, Prague, Czech Republic
*Corresponding author: Miroslav Bulvas, Cardiocenter, Third Faculty of Medicine, Charles University; Department of Surgery, University Hospital Královské Vinohrady, Division of Interventional Angiology, Prague, Czech Republic
Received: 19 August 2019; Accepted: 23 August 2019; Published: 26 August 2019
Article Information
Citation: Miroslav Bulvas. Mechanical Atherothrombectomy (MATH) in the Treatment of Chronic Occlusions of the Popliteal Artery. Cardiology and Cardiovascular Medicine 3 (2019): 248-257.
View / Download Pdf Share at FacebookAbstract
Mechanical atherothrombectomy (MATH) is rapid, effective and safe technique used for endovascular removal of occlusive material from peripheral vessels. It is widely used in patients with acute and subacute lower limb ischemia. Efficacy of the Rotarex catheter depends on the presence of fragmentable occlusive material and the treatment is successful also in patients with chronic ischemic symptoms. The technique reduces vessel barotrauma by lowering the frequency of balloon angioplasties and stenting procedures. Additionally, the need for surgical treatment can be also lowered.
The purpose of this review is to present an illustrative series of patients with popliteal artery occlusions and chronic ischemic symptoms, that may help the reader to learn about the technique potential in this challenging vascular location.
Keywords
<p>Mechanical thrombectomy; Acute limb ischemia; Arterial occlusion; Angioplasty; Stenting</p>
Article Details
Introduction
Elastic recoil, residual narrowing and dissections occurring after balloon angioplasty (PTA) significantly limit immediate technical success of the technique. In those cases, the stent implantation can be required to secure optimal blood flow through the treated segment. Barotrauma and the deep wall injury associated with PTA and/or stenting promote myointimal hyperplasia with early restenosis and thus can significantly reduce primary patency rate and clinical success of the treatment.
The popliteal artery is highly exposed to biomechanical stress resulting from repetitive vessel rotation, flexion, compression, torsion and extension occurring during walking and knee flexion. Therefore, the stents implanted into this challenging location can be prone to fracture(s), arterial wall damage, lumen reocclusions with subsequent patency loss. Furthermore, the stent(s) located in the popliteal artery can also restrict future surgical bypass options. These are the main reasons why the popliteal artery is considered as a “no-stenting zone” or as the location where the stent implantation should be highly restricted.
Removal of occlusive material (debulking) before PTA can be considered a part of vessel preparation, that leads to better drug uptake from drug-coated balloon (DCB) catheters and improved drug penetration into the arterial wall. The strategy “leave nothing behind” and the use of a debulking device, also excludes or minimizes the wall stress induced by inflation of the balloon catheter, reduces occurrence of post PTA dissections and the necessity to leave a foreign body (stent) in the vessel. Thus, the primary patency rate can be higher than with DCB angioplasty alone [1].
The Rotarex ®S (Straub Medical AG, Wangs, Switzerland) is a single lumen catheter equipped with a rotating head driven by a stainless-steel helix rotating at up to 60 000 rpm. The device has two superimposed stainless steel cylinders, each with two lateral apertures; the outer cylinder is connected to the rotating helix, and the inner cylinder to the catheter shaft. The shape of the head facilitates the detachment and fragmentation of the occlusive material together with a strong vortex created by rotation of outer cylinder. The displaced occlusion fragments are aspirated through head openings and are shredded again into debris that is transported to an external collecting bag [2]. The catheter can be currently used in vessels with diameter of 3 mm or more and its usage is dependent on ability to cross the lesion intraluminally with a guidewire. Efficacy of the Rotarex® S catheter depends on the presence of fragmentable occlusive material (atherosclerotic plaque – Figure 1, myointimal hyperplasia, fresh or old thrombus).
The following Figures (2-9) can document the technical success of Mechanical Atherectomy plus THrombectomy (MATH) with the Rotarex®S catheter in patients with chronic (non-stent) occlusions of the popliteal artery and clinically significant ischemic symptoms. The procedures either avoided completely the need for stenting, or enabled use of much a shorter stent length than the original length of the occlusion.
Existing literature contains Rotarex studies of which the majority of patients had subacute ischemia of lower limbs and milder acute symptoms. Target lesions were mostly located within the femoropopliteal segments, iliac arteries and femoropopliteal bypasses, including in-stent occlusions3-15. The only technical condition for MATH is the intraluminal passage of the guide wire through the occlusion which can be difficult in rigid and calcified chronic lesions.
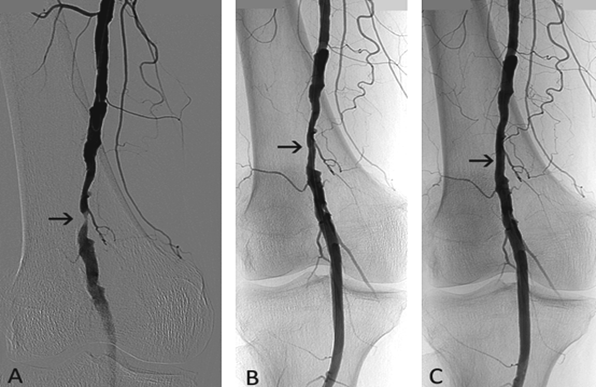
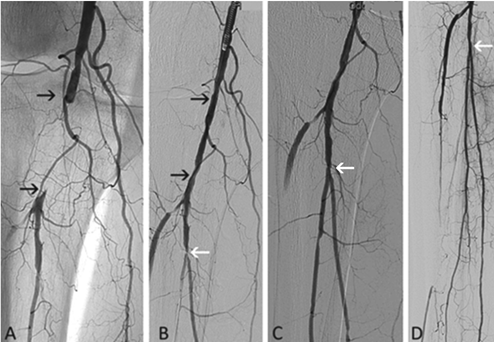
Figure 2: 64-year-old man, chronic lower limb ischemia, Rutherford category 3. (A) Occlusion of the popliteal artery is depicted by arrows. (B) Distal flow of the popliteal artery and tibial vessels. (C) Popliteal artery after debulking with the Rotarex®S catheter (originally occluded lumen is located between the arrows). Proximal arrow also depicts the Rotarex®S catheter head. (D) Angiography after low-pressure PTA with recanalized segment between the arrows.
Figure 4: 65-year-old man, chronic lower limb ischemia, Rutherford category 4. (A) Popliteal artery occlusion at its origin (black arrow). (B) Tibial anterior artery (double arrow) is filled via collaterals. (C) Angiography after debulking (segment between the arrows) with the Rotarex®S catheter and (D) balloon angioplasty (PTA) of tibioperoneal trunk (double arrow) and anterior tibial artery (arrow).
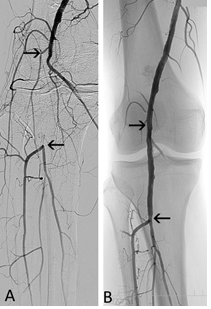
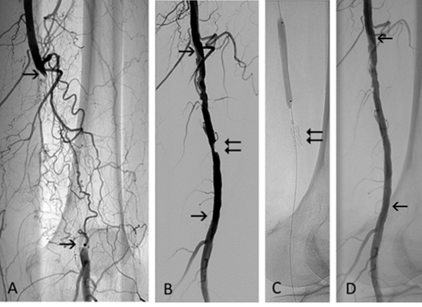
Figure 5: 68-year-old man, chronic lower limb ischemia, Rutherford category 4. (A) Occluded popliteal artery (between arrows), stenosed tibioperoneal trunk (double arrow) and stenosed anterior tibial artery (white arrow). (B) Angiography after the Rotarex®S debulking demonstrating the recanalized popliteal artery (double arrow) and after PTA of the tibioperoneal trunk and anterior tibial artery (white double arrow and black arrow).
Figure 6: 68-year-old man, chronic lower limb ischemia, Rutherford category 5. (A) Occlusion of the popliteal artery (depicted by arrows). (B) Angiography after the Rotarex®S debulking demonstrating a recanalized popliteal artery (between arrows). Stenosis of tibioperoneal trunk before PTA (white arrow). (C) Angiography after PTA (white arrows) with periphery (D).
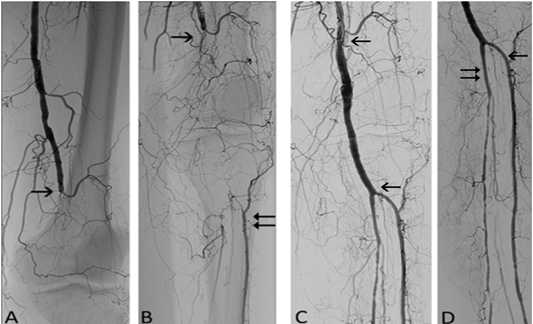
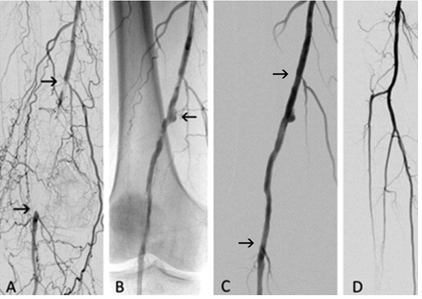
Figure 7: 71-year-old woman, chronic lower limb ischemia, Rutherford category 3. (A) Occluded popliteal artery (between arrows). The popliteal artery (distal arrow) is filled via collaterals. (B) Eccentric residual stenosis (double arrow) appeared after mechanical debulking with the Rotarex®S catheter. (C) Spot-stenting (double arrow) and PTA were necessary as adjunctive modalities. (D) Angiography following adjunctive balloon angioplasty with recanalized segment located between the arrows.
Figure 8: 69-year-old woman, chronic ischemia, Rutherford category 3. (A) The arrows depict the popliteal artery occlusion before the debulking. (B) Angiography after recanalization with the Rotarex®S catheter, residual short dilatation (arrow) left without intervention. (C) Residual wall irregularities were smoothed out with adjunctive low-pressure PTA; recanalized segment is located between the arrows. (D) Control popliteal and tibial angiography.
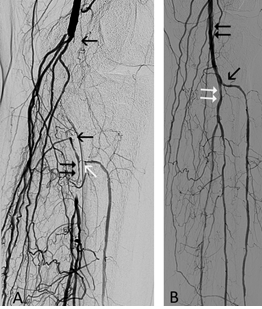
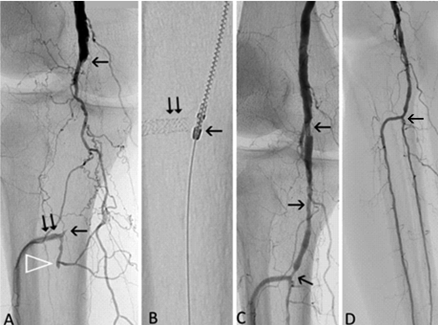
Figure 9: 76-year-old man, chronic ischemia, Rutherford category 5. (A) The arrows depict the popliteal artery occlusion before the debulking procedure. Stented (double arrow) anterior tibial artery is filled via collaterals. Arrowhead depicts occluded tibioperoneal trunk. (B) Final distal position of the Rotarex®S head (arrow) that is not in the contact with stent struts (double arrow) during debulking. (C) Angiography after debulking with the Rotarex alone. Residual narrowing is depicted by arrows. (D) Final angiography after additional PTA of the popliteal artery and tibioperoneal trunk (arrow).
Discussion
The above mentioned studies [3-15] reported mortality rates 0-1% at 30 days, technical success 92-100%, secondary surgical revascularization rates 0-5.3%. Complications associated directly with the debulking procedure were reported in 0-8% (vessel perforation) and 0-7% (peripheral embolization)4,6,8-10,14 and they were typically classified as minor with successful treatment by prolonged balloon inflation, stentgraft implantation and percutaneous aspiration thromboembolectomy.
Technically, it is important to monitor the blood flow through the catheter, follow the sound of the running Rotarex®S and be aware of pain reaction of the patient. The absence of the blood flow through the catheter reflects the existence of a significant proximal lesion or relatively large diameter of percutaneous introducer which causes a flow restriction. Rarely, the flow is stopped by wedged clot fragment (Figure 10). The sound frequency of the running catheter reflects the consistence of the plaque/vessel wall and increases with wall rigidity and the presence of calcification. Also, when there is no blood around the rotating head, a vacuum is created, the vessel wall comes into close contact with the head apertures and sound frequency increases together with the perforation risk. Pain can signal to the operator the closer contact of the catheter with the adventitial layer and thus increased risk of arterial perforation. Keeping this information in mind, the rotational, mechanical atherothromebctomy with the Rotarex®S device is a very effective and safe endovascular technique for vessel recanalization.
Figure 10: Catheter obstruction with the clot fragment (white arrow) can occasionally interrupt the blood flow through the catheter lumen and cause catheter dysfunction. Pulling the rotating head (white double arrow) out of the catheter in the direction of black arrow will uncover obstructing clot that can be removed with a puncture needle for example.
Conclusions
Mechanical atherothrombectomy (MATH) is a highly effective endovascular modality for removal of vascular occlusive masses. The advantage of its use is in the avoidance of stenting (Figures 1-6. 8,9) or to reduce the number and length of implanted stents (Figure 7) especially in such a challenging location as the popliteal artery. By recanalization of the popliteal artery, this technique also opens the way for catheter treatment of runoff vessels in patients with critical limb ischemia. Typical cases with angiograms are presented to bring closer those who are interested in newer aspects of endovascular therapy.
Conflict of interest
M.B. was proctoring for Straub Medical AG
References
- Sauget A, Philippart R, Honton B. Directional atherectomy with antirestenotic therapy for the treatment of no-stenting zones. J Cardiovasc Surg 60 (2019): 198-204.
- Schmitt HE, Jäger KA, Jacob AL, Mohr H, Labs KH, Steinbrich W. A new rotational thrombectomy catheter: system design and first clinical experiences. Cardiovasc Intervent Radiol 22 (1999): 504-509
- Duc SR, Schoch E, Pfyffer M, et al. Recanalization of acute and subacute femoropoplital artery occlusions with the Rotarex catheter: one year follow-up, single center experience. Cardiovasc Intervent Radiol 28 (2005): 603-610.
- Zeller T,Frank U,Bürgelin K et al. Early experience with a rotational thrombectomy device for treatment of acute and subacute infra-aortic arterial occlusions. J Endovasc Ther 10 (2003): 322-331.
- Wissgott C, Kamusella P, Richter A, et al. Mechanical rotational thrombectomy for treatment of acute and subacute occlusions of femoropopliteal arteries: retrospective analysis of the results from 1999 to 2005. Röfo 180 (2008): 325-331.
- Freitas B, Steiner S, Bausback Y et al. Rotarex mechanical debulking in acute and subacute arterial lesions: single-center experience with 525 patients. Angiology 68 (2017): 233-241.
- Bérczi V, Deutschmann HA, Schedlbauer P, et al. Early experience and midterm follow-up results with a new, rotational thrombectomy catheter. Cardiovasc Intervent Radiol 25 (2002): 275-281.
- Lichtenberg M, Käunicke M, Hailer B. Percutaneous mechanical thrombectomy for treatment of acute femoropopliteal bypass occlusion. Vascular Health and Risk Management 8 (2012): 283-289.
- Wissgott C, Kamusella P, Andresen R. Recanalization of acute and subacute venous and synthetic bypass-graft occlusions with mechanical rotational catheter. Cardiovasc Intervent Radiol 36 (2013): 936-942.
- Liu J, Li T, Huang W et al. Percutaneous mechanical thrombectomy using Rotarex catheter in peripheral artery occlusion diseases – experience from a single center (published online November 20, 2018). Vascular.
- Kronlage M, Printz I, Vogel B et al. A comparative study on endovascular treatment of (sub)acute critical limb ischemia: mechanical thrombectomy vs thrombolysis. Drug Des Devel Ther 11 (2017): 1233-1241.
- Stanek F, Ouhrabkova R, Prochazka D. Percutaneous mechanical thrombectomy in the treatment of acute and subacute occlusions of the peripheral arteries and bypasses. Vasa 45 (2016): 49-56.
- Heller S, Lubanda JC, Varejka P et al. Percutaneous mechanical thrombectomy using Rotarex S device in acute limb ischemia in infrainuinal occlusions. Biomed Res Int 2017:2362769
- Bulvas M, Sommerová Z, Van?k I, Weiss J. Prospective single-arm trial of endovascular mechanical debulking as initial therapy in patients with acute and subacute lower limb ischemia: one-year outcomes. J Endovasc Ther 26 (2019): 291-301.
- Milnerowicz A, Milnerowicz A, Kuliczkowski W, Protasiewicz M. Rotational atherectomy plus drug-coated balloon angioplasty for the treatment of total in-stent occlusions in iliac and infrainguinal arteries. J Endovasc Ther 26 (2019): 316-321.


 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 