Post-Mortem Bronchopulmonary and Cardiovascular Histopathological Finding in Patients who Died of Interactable Asthma Attack with Synchronous Paecilomyces Infection
Zhana M Sizova1, Vasili M Akhunov1, Amir A Azimzadeh1*, Tatyana P Lavrentieva2
1Department of medical and social expertise, emergency and polyclinic therapy, Sechenov First Moscow State Medical University (Sechenov University), Moscow, Russian Federation
2Diagnostic Center of the Department of Health in Moscow
*Corresponding author: Amir A Azimzadeh, Department of medical and social expertise, emergency and polyclinic therapy, Sechenov First Moscow State Medical University (Sechenov University), 4 Bolshaya Pirogovskaya Street, Building 1, Moscow, Russian Federation
Received: 26 October 2021; Accepted: 05 November 2021; Published: 16 November 2021
Article Information
Citation: Zhana M Sizova, Vasili M Akhunov, Amir A Azimzadeh, Tatyana P Lavrentieva. Post-Mortem Bronchopulmonary and Cardiovascular Histopathological Finding in Patients who Died of Interactable Asthma Attack with Synchronous Paecilomyces Infection. Cardiology and Cardiovascular Medicine 5 (2021): 572-581.
View / Download Pdf Share at FacebookAbstract
Objective: The potential histopathological changes of Paecilomyces infection in bronchopulmonary and cardiovascular system of patients with bronchial asthma is evaluated on the postmortem tissue specimens.
Methods: Postmortem histopathological evaluation of cardiopulmonary system is performed in 15 patients (5 women and 10 men, aged between 18-77 years old). All the patient’s died as a result of intractable asthma attacks and acute cardiovascular insufficiency. Hematoxylin and Eosin-stained paraffin sections of bronchopulmonary and cardiovascular systems are examined. Special methods are employed to detect fungi and the tissue.
Results: Microscopic examination in the lungs revealed vascular congestion, extension of venules and capillaries, arteriolar spasm, endothelial inflammation with a tendency to desquamation, and increased permeability of the vascular wall with fungal cells in the perivascular space. Changes in vascular walls were characterized by a pattern of destructive-productive vasculitis at different stages of development from endovascular to perivascular space inflammation resulting in sclerosis. The same type of changes was found in vascular walls of other organs including the heart, resembling different stages of the process. Sclerosis of the vascular wall and the perivascular tissue may represent a protective inflammatory response to limit the spread of the pathogen.
Conclusions: Our data suggests that the complex vascular pathology in the cardiopulmonary system in patients with bronchial asthma may be triggered by a hematogenic infection caused by dimorphic Paecilomyces fungi.
Keywords
<p>Paecilomyces; Fungemia; Bronchial Asthma; Systemic Vasculitis; Sclerosis</p>
Article Details
1. Introduction
The issue of hemodynamic abnormalities in bronchial asthma is still challenging nowadays. According to recent studies, the presence of hypertension in patients with bronchial asthma increases the probability of coronary complications. At the same time, many aspects of their etiology and pathogenesis remain undetermined. Furthermore, associated diseases of the cardiovascular system dominate over the exacerbating factors of bronchial asthma [1-5]. Numerous hypotheses have led to believe that development of hypertension directly correlates to infectious nature of the disease. Recently, Paecilomyces fungi with dimorphism and potentially pathogenic properties were discovered as a new type of causative pathogen of chronic persistent blood fungal infection. These results contribute to a new way of addressing our current research. According to the literatures, Paecilomyces fungi cause the development of systemic fungal infection in individuals with both weak and intact immune systems [6-8]. Clinical manifestations of Paecilomyces infection vary [9-17], with dominating allergic symptoms and bronchial asthma [18]. The metabolic composition of the mycelium and liquid culture of Paecilomyces variotii taken from the patient's blood, was represented by free fatty acids and phospholipids, among which was the lysophosphatidylcholine. This has membrane toxicity properties resulting in cytopathogenic impact on host cells are shown [19, 20]. The experimental reproduction of Paecilomyces has also shown the development of chronic alterative and productive inflammation in the lungs of animals. Hence, vessels and bronchi get involved in the pathological process [21, 22]. The potential histopathological role of Paecilomyces infection in bronchopulmonary and cardiovascular systems of patients with bronchial asthma is the focus of this research.
2. The Process
The study was carried out at Sechenov University, Moscow City Clinical Hospital named after S.S. Yudina, and the Diagnostic Center (Center for Laboratory Research) of the Department of Health in Moscow. The confirmation process through the ethics board is achieved. All efforts including consents have been made to ensure confidentiality of any identifying information that was obtained in connection with this research. The tissue is achieved from 15 postmortem examinations of patients who died of intractable asthma who also had Paecilomyces infection (5 women aged between 18 and 68 years and 10 men aged between 43 and 77 years). Paecilomyces infection was established based on the results of microscopy of smears of peripheral blood and sputum. Analysis was carried out by the method of express diagnosis of Paecilomyces fungus with direct quantitative counting of mature spherules of the Paecilomyces fungus in 1 μl of blood together with isolating the culture of the fungus and serological studies. In addition, a complex of histological, histochemical, morphological and bacteriological studies was used. Additional methods were used to detect fungi in the tissue (Mac Manus, Gridley and Gram-Weigert) [23]. Paraffin sections from different tissues were stained with hematoxylin and eosin. The histopathological examinations of the bronchopulmonary and cardiovascular systems are performed by using a light microscope (Leica DM1000). All patients died as a result of profound hypoxemia caused by asthmatic attack triggered by Paecilomyces fungemia, induced by acute respiratory viral infection. In 13 patients (74%), coronary artery diseases were also present.
3. Results
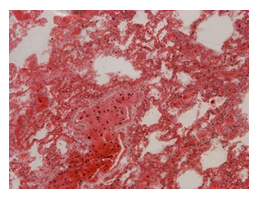
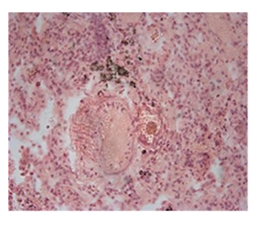
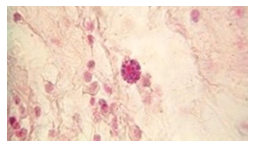
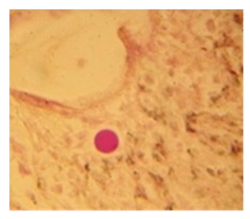
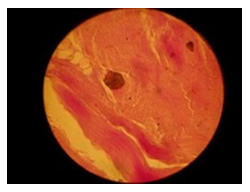
The histopathological findings were mainly in the blood vessel walls, perivascular and interstitial tissues. The bronchial wall appeared spasmodic or obturated with thick mucus in the form of mucous plugs. The swelling of the mucous membrane was detected with an exponential thickening of the basal membrane. Although there was a thick infiltration in all layers of the bronchial wall, around the areas of inflammation in the bronchi were also observed eosinophils, lymphocytes, histiocytes and plasma cells. Additionally, fungal cells were detected in thickness of the layers of the bronchial wall. Mucous plugs were contained cells of the desquamated bronchial epithelium, eosinophils, macrophages and spherules of the Paecilomyces fungus. In the same visual fields of pulmonary parenchyma, an uneven thickening of the interalveolar septum, along with pronounced proliferation and growth of pulmonary capillaries (Figure a) and foci of hemorrhage was found. Thickened vessel walls were also noted. There was evidence of swollen endothelium and desquamation of endothelial cell as well as fragmented internal elastic membrane. There was also evidence of increased permeability of vessel walls and perivascular edema (Figure b). Fungal cells were found in thrombi, vessel walls and the perivascular space. A pale pink transparent exudate with an admixture of desquamated epithelium and leukocytes was observed in the alveolar spaces. Minor macrophage-histiocytic infiltrates were noted in the perivascular soft tissue and bronchial wall, containing round-oval corpuscles with a two-contour capsule (measuring 5.0x5.0 to 29x30 μm and even larger) which stained positive to fungi (Figure c). In other visual fields, the interalveolar stroma appeared thickened, with more pronounced infiltration of inflammatory cells. Some foci show swelling and fibrinoid necrosis within the wall of small arteries and veins. The wall of small and medium size vessels showed a span of destructive-productive vasculitis due to the special resistance of the pathogen, from endovascular cells into perivascular space. This happened with increased permeability of their walls and efflux of fungal cells into the perivascular space. The cellular composition of infiltration around fungal cells was represented by activated eosinophils, lymphocytes, histiocytes and plasma cells. In three cases, a lympho-histiocytic infiltration with formation of a noncaseating granulomas was detected. The granulomas showed single giant multinuclear cells demonstrating cytoplasmic fungal particles or their cytoskeletons with granular contents. These were surrounded by epithelioid cells, macrophages and lymphocytes or by a thin layer of histiocytes and lymphocytes. In addition, foci of pulmonary parenchyma with expanded stroma were observed around the vessels in peribronchial space as well as in the alveolar walls. Cellular response was noticed in pulmonary tissue where fungal cells existed. This had a pattern of productive inflammation with a tendency to a protracted course, resulting in sclerosis in the affected area, limiting further spread of fungal cells (Figure d). It should be noted that along with pronounced histological changes in the lungs, there were areas where morphological disorders were minimal or completely absent, which indicated a disseminated nature of the process. The generalized spread of Paecilomyces caused dissemination of the fungus in the tissues of various organs through the involved vessel walls, which were mainly in small and medium sized vessels.
Examination of the sections of the cardiac muscle revealed chronologically different changes. In some visual fields, there was edema of the interstitial stroma with the separation of muscle fibers and necrosis of scattered muscle fibers. In the lumen of the vessels, fungal tissue cells at different stages of development were observed (Figure e). In other visual fields, focal infiltrates around small and medium size vessels with destruction of the vascular walls were evident in the form of fibrinoid swelling or fibrinoid necrosis and pronounced lympho-plasmocytic infiltration in the perivascular area. Additionally, some areas with the growth of fibrous tissue between the muscle fibers and vessels up to their complete obliteration were observed (Figure f).
4. Discussion
Arterial hypertension occurs in 65.1% of patients with bronchial asthma, which is significantly higher than in the total population. Studies of vascular structure and endothelial function in patients with arterial hypertension and bronchial asthma revealed signs of injury in the vascular wall, together with endothelial dysfunction which results in increased arterial stiffness when the disease becomes more severe [24, 25].
To date, the histopathological profile of changes in the tissues of various organs in Paecilomycosis has been poorly studied. According to Uys et al. [26], patients who passed away from septic endocarditis resulted from Paecilomyces variotii infection had also infarction foci in their brain, kidney and spleen. Branched mycelium and spores in the blood clot enveloping the mitral valve prosthesis were found in histological samples that were stained with hematoxylin and eosin after infarct. Some of the spores were attached to the mycelium branches. Spores and mycelium were not stained, but their outlines were marked. As a result of argyrophilia, they were stained with silver nitrate (Gomori). Isolated spores without argyrophilia were found near this area. In the place where the thrombus is attached to the heart tissue, a cluster of histiocytic and epithelioid cells with giant multinuclear cells in between was revealed. In the cytoplasm of giant multinuclear cells, phagosomes with elements of fungal cells were discovered. In smears of the affected tissue, oval gram-positive bodies resembling yeast cells were observed; however, it was without any budding signs.
The available data obtained during this experiment [21] revealed a picture of systemic mycosis with a predominant lesion of the bronchopulmonary system. Changes in the lungs were characterized by injuries in the vessels and bronchi histologically, accompanied by a response of the reticuloendothelial system clinically manifesting as lymphadenopathy, hepatomegaly, and splenomegaly. Paecilomyces, presenting as rounded-oval cells 2-5 microns in diameter, were located mainly in the interalveolar tissue, bronchial walls and blood vessels as part of cellular infiltrates. They were colored in dark pink to purple (Gridley) and in brown-black colors (Gomori-Grocott). These fungal tissue forms had a double layer shell. The mycelia phase of fungus has not been observed in tissues.
In the lungs, our results show the following: vascular congestion, extension of venules and capillaries, spasm of arterioles, swelling of endothelial cells with a tendency to desquamation and increased permeability of the vascular wall with the efflux of fungal cells in the perivascular space. Changes in vascular walls were characterized by a pattern of destructive and productive vasculitis at different stages of development from endovascular cells to perivascular space inflammation with a sclerotic outcome. Changes of the same type were found in vascular walls in heart tissues, which were distinguished only by the time of their occurrence. Sclerosis of the vessel walls and perivascular tissue were the manifestation of protective inflammatory reaction of the host organism, limiting further spread of the pathogen in the organ tissues.
5. Conclusions
Acknowledgement
Authors appreciate Dr. Navid Liaghati MD.FRCPC. for editing this paper.
Disclosure of Interest
None
Conflict of Interest
None
Ethics Approval and Consent to Participate
This study has been reviewed and received ethics clearance through the Sechenov First Moscow State Medical University (Sechenov University) Research Ethics Board and Moscow City independent Ethics Committee (Number 1476, 13.11.2019). The first phase of this study was permitted by the Academic Council of Sechenov University to be carried out on 15 autopsies of 5 women and 10 men. All efforts including consents have been made to ensure confidentiality of any identifying information that was obtained in connection with this research.
Studies Involving Animals
“Not applicable”
Consent for Publication
All authors provided consent for publication.
Data Availability Statement
The data analyzed during the current study are available from the corresponding author on reasonable request.
Competing Interests
The authors have no competing interests to declare.
Funding
The present work was supported by Sechenov First Moscow State Medical University (Sechenov University). The funding body had no role in the design of the study, collection, interpretation of data, analysis and writing of this manuscript.
Author Information
Affiliations
Department of medical and social expertise, emergency and polyclinic therapy, Sechenov First Moscow State Medical University (Sechenov University), Moscow, Russian Federation.
Zhana M. Sizova (MD, PhD, Professor, Head of department)
Amir A. Azimzadeh (MD, PhD, Assistant Professor)
Department of medical and social expertise, emergency and polyclinic therapy, Sechenov First Moscow State Medical University (Sechenov University), Moscow, Russian Federation. City Clinical Hospital named after S.S. Yudina
Vasili M. Akhunov (MD, PhD, Assistant Professor)
Diagnostic Center of the Department of Health in Moscow.
- Tatyana P. Lavrentieva (MD)
Contributions
Sizova Z.M designed the study and take responsibility for the integrity and accuracy of the data, data analysis and critical revision.
Akhunov V.M. contributed to the study design, data collection, data analysis, data interpretation, and the writing of the manuscript.
Azimzadeh A.A. contributed to the coordination, data analysis and interpretation, and the writing of the manuscript.
Lavrentieva T.P. contributed to laboratory diagnosis, data analysis and interpretation.
All the Authors read and approved the final manuscript.
References
- Mukharlyamov NM. systemic arterial hypertension and lung disease. Therapeutic archive. 1983.- Resolution 55 (1983): 54-60.
- Kozyrev AG. clinical and functional and biochemical characteristics of patients with bronchial asthma and systemic arterial hypertension. Abstract. Diss. PhD, Saint-Petersburg (2001).
- Yuldasheva IA. state of vascular endothelium in patients with bronchial asthma. Clin. Med 5 (2004): 36-49.
- Sun WX, Jin D, li Y, et al. Increased arterial stiffness in stable and severe asthma. // Respiratory Medicine 108 (2014): 57-62.
- Sun D, Li x, Heianzo Y, et al. A history of Asthma from childhood and Arterial Stiffness in Asymptomatic Young Adults: the Bogalusa Heart Study. Hypertension 71 (2018): 928-936.
- Ahunova AM, Shustova VI. Clinical and immunological characteristics of paecilomycosis. Russ Clin Med 69 (1991): 64-67.
- Jos Houbraken Paul E Verweij, Anthonius JMM Rijs, Andrew M Borman, et al. Identification of Paecilomyces variotii in Clinical Samples and Settings. Journal of Clinical Microbiology 48 (2010): 2754-2761.
- Danielly C Moreira, Manoel ME Oliveira, Cintia M Borba. Human Pathogenic Paecilomyces from Food//Microorganisms 6 (2018): 64.
- de Hoog S, Guarro J, Gene J, et al. Atlas of Clinical Fungi, 2nd edition (2000): 805.
- Aguilar C, Pujol I, Sala J, et al. Antifungal susceptibilities of Paecilomyces species. Antimicrob Agents Chemother 42 (1998): 1601-1604.
- Pastor FJ, Guarro J. Clinical manifestations, treatment and outcome of Paecilomyces lilacinus infections. Clin. Microbiol. Infect 12 (2006): 948-960.
- Swami T, Pannu S, Kumar M, et al. Chronic invasive fungal rhinosinusitis by Paecilomyces variotii: A rare case report. Indian J. Med Microbiol 34 (2016): 103-106.
- Bellanger AP, Cervoni JP, Faucher JF, et al. Paecilomyces variotii Fungemia in a Patient with Lymphoma Liver Transplant. Mycopathologia 182 (2017): 761-765.
- Heshmatnia J, Marjani M, Mahdaviani SA, et al. Paecilomyces formosus Infection in an Adult Patient with Undiagnosed Chronic Granulomatous Disease. J. Clin Immunol 37 (2017): 342-346.
- Kuboi T, Okazaki K, Inotani M, et al. A case of cutaneous Paecilomyces formosus infection in an extremely premature infant. J Infect Chemother 22 (2016): 339-341.
- Skiada ?, Pavleas I, Drogari-Apiranthitou ?. Rare fungal infectious agents: a lurking enemy. F1000 Res 6 (2017): 1917.
- Steiner B, Aquino VR, Paz AA, et al. Paecilomyces variotii as an Emergent Pathogenic Agent of Pneumonia. Case Reports in Infectious Diseases 2013 (2013): 273848.
- Akhunov VM, Sizova ZM. The prevalence of asthma of the Paecilomyces etiology and possible pharmacotherapy for outpatients. Biomedicine 5 (2010): 28-29.
- Nurtaev HS, Mirtalipov DT, Dehkan-Khodzhaeva NA. Lipid composition of the fungus Paecilomyces variotii Bainier var. Zaaminella. Modern problems of medical Parasitology in Uzbekistan. Proceedings of the research Institute of Parasitology of the Ministry of health of Uzbekistan 3 (1993): 130-131.
- Yulaev MF, Akhunova A. Some properties of the exogenous phospholipase A2 produced by the mycelium of the fungus Paecilomyces viridis. Probl. Med. Chem 45 (1999): 223-226.
- Khamidova RM, Giyasov ZA. Lung state in experimental zaaminellosis. In the book.: Sovr. Prob. Parasitol. Samarkand (1983): 220-222.
- Akhunov VM, Atakulov BM. Influence of hemotropic infection by dimorphic fungus of the genus Paecilomyces on the state of the cardiovascular system in the experiment. Clinical Pharmacology and Therapy 6 (2009): 253-254.
- Khmelnitsky O.K. Histological diagnosis of superficial and deep mycoses. Leningrad. Medicina Publishing House (1973).
- Roshina AA. Arterial hypertension in patient with bronchial asthma: Clinical-functional relationship and the possibilities of drug correction. Abstract. Diss. PhD. Saratov (2011).
- Nebieridze DV, Oganov RG. Endothelial Dysfunction as a risk factor for atherosclerosis, clinical significance of its correction. Cardiovascular therapy and prevention 3 (2009): 86-89.
- Uys CJ, Don PA, Schrire V. Endocarditis following cardiac surgery due to the fungus Paecilomyces. S. Afr. Med. J 37 (1963): 1276-1280.



 Impact Factor: * 5.6
Impact Factor: * 5.6 Acceptance Rate: 74.36%
Acceptance Rate: 74.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 