Audio-Vestibular Alterations During the Phases of the Menstrual Cycle in Patients with Cochlear Implant
Pasqualina Apisa1, Gennaro Auletta1, Teresa Furia1, Pasquale Riccardi1, Carla Laria1, Rita Malesci1, Carmine Papa1, Angelo Sarnelli1, Elio Marciano1, Annamaria Franzé1,2*
1Unit of Audiology, Department of Neurosciences, Reproductive and Odontostomatologic Sciences, University of Naples “Federico II”, Via Pansini, Naples, Italy
2CEINGE- Advanced Biotechnologies, Naples, Italy
*Corresponding author: Annamaria Franzè, Unit of Audiology, Department of Neurosciences, Reproductive and Odontostomatologic Sciences, University of Naples “Federico II”, Via Pansini 5, 80131 Naples, Italy
Received: 07 October 2019; Accepted: 14 October 2019; Published: 11 November 2019
Article Information
Citation: Pasqualina Apisa, Gennaro Auletta, Teresa Furia, Pasquale Riccardi, Carla Laria, Rita Malesci, Carmine Papa, Angelo Sarnelli, Elio Marciano, Annamaria Franzé. Audio-Vestibular Alterations During the Phases of the Menstrual Cycle in Patients with Cochlear Implant. Archives of Clinical and Biomedical Research 3 (2019): 386-396.
View / Download Pdf Share at FacebookAbstract
Background: The female hormones fluctuations in woman’s life play a dominant role in homeostasis of the body and can influence the psycho-neurological processes in different body systems. In particular, in the auditory system seems that hormonal alterations during the menstrual cycle can compromise the homeostasis of the labyrinthine fluids altering balance and/or hearing.
Aims/Objectives: Our study aims to verify if hormonal changes during different menstrual phases may have an influence, in females with cochlear implants, on the trend of auditory and vestibular performances. Materials and methods: Tonal, speech audiometry, vestibular examination and a measure of cochlear implants electrodes impedances were realized in the follicular and luteal phases on a group of eight women who are at a fertile age, affected by profound sensorineural form of hearing loss and carrying a monolateral cochlear implant.
Results: Audio-vestibular alterations, as well as, an increase of impedance electrodes are reported in the luteal phase.
Conclusions: Our work suggests that audio-vestibular alterations are related to the different phases of the menstrual cycle with consequent variation also of the performances of the implant. It is very important to keep in mind this aspect to avoid negative results during rehabilitation.
Keywords
<p>Reproductive hormones; Cochlear implant; Auditory ability; Endolymphatic liquids; Vestibular apparatus</p>
Article Details
1. Introduction
The progesterone and estrogenic hormones, and their inter-dependent fluctuations in a woman’s life have always played a dominant role in their physiological development and in the homeostasis of the female body. The capacity that the reproductive hormones have to influence the psycho-neurological processes lead to different systems in the body interacting [1 (. The auditory system, as with every other part of our body, does not have a static function. On the contrary, it is continually controlled by specific demands, and it receives its input through hormones and neuro- transmitters [2]. The excitable quality of the sensorial receptors, the neuronal transmission, the ionic mechanisms and the synaptic plastic quality are all probably modulated by signals which reach the auditory system from the rest of the body and so make the best possible use of the auditory function. There are several papers in the international literature that support these hypotheses. Indeed, it has also been suggested that the hormonal alterations during the menstrual cycle, pregnancy and menopause can all compromise the homeostasis of the labyrinthine fluids [3], in that they can act directly on the enzymatic processes and on the effects of the neuro-transmitters. These changes can alter balance and/or hearing. Charitidi et al. [4] with data obtained by electro-physiological studies on animals have shown that the increase of the auditory plasticity and the capacity to communicate are increased by estradiol. Moreover, Lòpez-Grueso et al. [5], have shown that evidences come from physiological and behavioral experiments in animals that report estrogen- induced changes in auditory central auditory responses, in otoacoustic emissions and in understanding the meaning of words. In addition, Al-Mana et al. [6] and Adriztina et al. [7] by analyzing women after menopause have shown that the worsening of hearing loss is related to estrogen deprivation.
More recently, Da Silva et al. [8] have reported that the auditory system, in females, is sensitive to changes in the female hormones by influencing the latencies of auditory brainstem responses, the amplitudes of distortion product otoacoustic emissions, the pressure of middle ear, the function of eustachian tube, and low frequency hearing thresholds, supporting the data, above reported [4] previously obtained by in animals. Moreover, Emami et al. [9] observed that there are significant changes in hearing thresholds during the menstrual cycle phases: improved hearing thresholds were observed in the follicular phase (the hearing thresholds at high frequencies were better).
Also with regard to the vestibular system, there are studies in the literature that show a correlation between its functioning and female hormones. Gould et al. [10] and Ishii et al. [11] suggested that peripheral vestibular alterations can take place, in a pre-menstrual phase (luteal phase) because of liquid retention in this menstrual phase, which results by an increase in the release of estrogens, progesterone and aldosterone. The presence of vertigo has been reported in the days before the onset of menstruation in which the levels of estrogen, progesterone and aldosterone inside the ear are higher [12,13]. The effect of the increase in hormonal level results, in fact, in the hydrops of the labyrinth with symptoms similar to those which are found in Ménière’s Disease [13]. The scientific evidences mentioned above with a link between the auditory (peripheral / central) system and the hormonal fluctuations let us to focus our attention to the auditory/vestibular variations we observed in several women with cochlear implant (CI). In particular, for several women, we have detected a cyclical variability in the performances of the implants, suggesting a possible role of menstrual hormones in CI outcomes too.
Actually, very little information is reported on correlation between hormone alterations cochlear implant performances, but in one paper, it has been reported that aldosterone can reduce impedance changes of CI electrodes [14]. This is interesting because it is known that, normally, the electrode impedances should not undergo major changes during the life, in fact, it is reported that a reduction in the average electrodes impedance was observed only during the first weeks after implantation and although impedances tended to increase slightly with the use of the cochlear implant, no statistically significant differences were found between the following months [15]. Our study aims, then, to investigate, in women with CI, the possible relationship between different hormonal levels during the menstrual phases (follicular / late luteal phases) and their auditory performances. We focused our attention on the trend of electrodes impedance and auditory perceptual abilities, as well as on vestibular problems.
2. Materials and Methods
2.1 Patients’ selectionThe study was carried out by analyzing a group of women, at fertile age, recruited at the Unit of Audiology (University of Federico II, Naples). Only women with profound bilateral hearing loss and rehabilitated with mono-lateral CI from at least 5 years and a history of regular menstrual cycles were selected. Regular was defined as being foreseeable within three days and a normal cycle length (defined as 26-30 days) from at least a year before the study. Women were analyzed during both the follicular and luteal phases which represent menstrual phases with very different hormone levels: in the follicular phase the estrogen and progesterone are in the lowest level, while in the late luteal phase the progesterone is at its maximum level. We then selected eight female patients, aged between 18 and 45. None of the selected females smoked and had not taken, in the last six months, oral hormonal contraceptives. Seven of these had had their cochlear implant on their right hand side with only one of them having had it on the left side. The participants were evaluated between their 5th and 7th days (the follicular phase) and between the 24th and 27th days (late luteal phase) of their menstrual cycle. All of them reported, in anamnesis, particular symptoms in the days preceding the beginning of their menstrual cycle (late luteal phase).
Specifically, all eight women referred to a feeling of a lessening in their auditory ability, with four of them lamenting instability and a feeling of a thick head, two others referred to feelings of vertigo linked to neuro- vegetative symptomatology while the remaining two had an increase in pre-menstrual headaches. None of the eight patients lamented these symptoms in the follicular phase.
Follicular and luteal phases were confirmed by performing hormonal dosages of progesterone and estradiol. All the women in the study were followed for a year before starting the research, indicating they all had a normal ovulatory cycle.
2.2 Audiological evaluationAn audiological evaluation was performed, according to standard protocols, on all eight selected patients. In detail:
- Vestibular examination was realized by videonistagmoscopy. We evaluated: spontaneous- positional nystagmus with and without fixation, Vestibulo-Ocular Reflex (VOR) to slow rotation, Head shaking Test and Vibratory Test, with three stimulation trials of each mastoid, using 100 Hz with each stimulus lasting about 5-10 s.
- Pure tone audiometry at the frequencies of 250 Hz, 500 Hz, 1000 Hz, 2000 Hz, 4000 Hz and 6000
- Speech audiometry using single bi-syllabic words in common use, taken from an open list of words which were also phonetically
To measure the impedances of the electrodes, a digital cochlear implant directly linked to the sound processor was used. The measuring process was controlled by a computer and the interfaces were supplied by each cochlear implant companies. The measuring process was carried out by the software of the implant and, in the specific case the 3.0 Soundwave was used for the Advanced Bionics company implants, and 4.1 Custom Sound for the Cochlear company implants. All audiological tests were repeated in the follicular and luteal phases for every patient.
3. Results
3.1 Vestibular evaluationTo verify if the menstrual phases can have influence on vestibular working we performed vestibular examination, in the eight women selected, both during the follicular and luteal phases (Table 1). As shown in the Table 1, during the luteal phase all patients showed signs of peripheral labyrinthine suffering, while in the vestibular phase the signs of vestibular suffering are present only in some patients and with less severe symptoms.
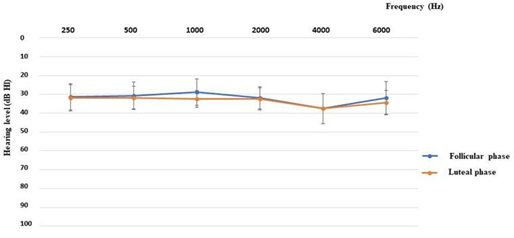
3.2 Evaluation of the auditory performancesIn the Figure 1 are shown the performances in the pure tone audiometry: for all patients they were similar in both the luteal and the follicular phases.
|
Patient |
Vestibular Examination in the Follicular Phase |
Vibratory Test in the Follicular Phase |
Vestibular Examination in the Luteal Phase |
Vibratory Test in the Luteal Phase |
|
R.F. |
Geotropic nystagmus on the left, persistent, stationary, partially inhibited. |
Re-enforces the left nystagmus |
Bipositional, bidirectional side,persistent, stationary geotropic nystagmus, inhibited. |
Re-enforces the left nystagmus |
|
Head Shaking Test: left nystagmus |
Head Shaking Test: left nystagmus |
|||
|
C.R. |
Omnipositional, persistent, inhibited down-beating nystagmus. |
Re-enforces the down nystagmus |
Persistent, stationary, inhibited nystagmus to the left. |
Re-enforces the left nystagmus |
|
Head Shaking Test: negative |
Head Shaking Test: left nystagmus |
|||
|
T.F. |
Absence of spontaneous nystagmus. |
Negative |
Up-right, omnipositional, persistent, inhibited nystagmus. |
Re-enforces the the right nystagmus |
|
Head Shaking Test: left nystagmus |
Head Shaking Test: left nystagmus |
|||
|
D.G. |
Negative |
Negative |
Bi-positional, bi- directional, persistent, stationary, inhibited nystagmus. Head Shaking Test: negative |
Negative |
|
T.C. |
Up-beating nystagmus, omnipositional, uninhibited. |
Negative |
Up-beating nystagmus, omnipositional, uninhibited. |
Re-enforces the up- beating nystagmus |
|
Head Shaking Test: negative |
Head Shaking Test: negative |
|||
|
D.V. |
Negative |
Evokes a right nystagmus |
Down-left, persistent, omnipositional, inhibited nystagmus. |
Evokes a right nystagmus |
|
Head Shaking Test: right nystagmus |
||||
|
P.L. |
Up-beating nystagmus in supine and side (left and right). |
Negative |
Up-beating nystagmus with rotational clockwise, omnipositional, persistent, inhibited nystagmus. |
Evokes an up- beating nystagmus |
|
Head Shaking Test: negative |
Head Shaking Test: negative |
|||
|
A.B. |
Left nystagmus in supine and left side, persistent, stationary, uninhibited |
Negative |
Down-beating, omnipositional, inhibited, stationary nystagmus. |
Re-enforces the left nystagmus |
|
Head Shaking Test: left nystagmus |
Head Shaking Test: left nystagmus |
Table 1: Synoptic table of the signs reported in the Vestibular Tests.
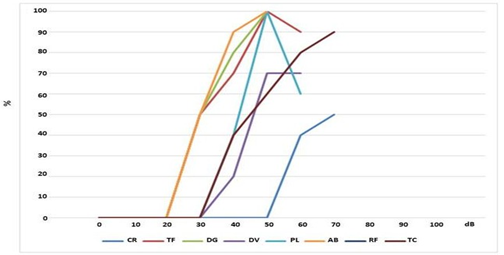
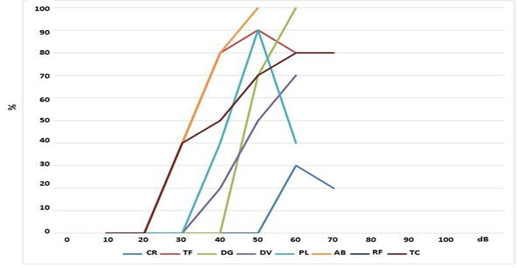
In Figures 2 and 3 are reported the speech audiometry results showing the qualitative response with a cochlear implant in the two phases of the menstrual cycle, respectively in the follicular and luteal phase. In particular, in the follicular phase (Figure 2) it can be
observed that 4/8 patients (50%) (AB, DG, TF, PL,) have a speech recognition threshold of 100% at the intensity of 50 dB; 1/8 patients (TC) 90% at 70 dB; 1/8 (DV) 70% at 50 dB; 1/8 patients (CR,) have 50% at 70 dB, 1/8 (RF) 0% at 60 dB.
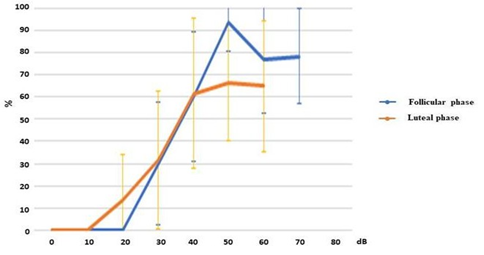
Instead, in the luteal phase (Figure 3) it can be observed that only 1/8 patients (AB) have a speech recognition threshold of 100% at the intensity of 50 dB and 1/8 patients (DG) 100% at 60 dB; 2/8 patients (TF and PL) 90% at 50 dB; 1/8 (DV) have a speech recognition threshold of 70% at 60 dB; 1/8 (TC) 80% at 70 dB; 1/8 (CR) 30% at 60 dB and 1/8 (RF) 0% at 60 dB. In Figure 4 it is reported the comparison between the means of the prosthetic functional response with IC above described.
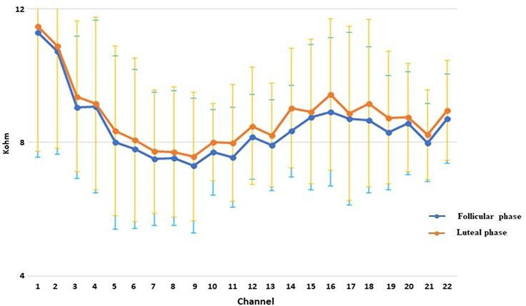
3.3 Evaluation of electrodes impedancesThe average impedance of the electrodes was calculated in the luteal and follicular phases (Figure 5).
The results showed a general increase in the impedances of the electrodes in the luteal phase: values reached a maximum increase of 2.55 KOhm.
4. Discussion
Several evidences [16, 17] suggested that the ovaric hormones influenced the metabolism of several electrolytes like calcium, magnesium, sodium and potassium during the different phases of the menstrual cycle. The coming together of these cyclical, electrolytic changes [16, 18-22] confirmed the datum that many women undergo variation in the balancing of their liquids and electrolytes in their pre-menstrual days. It is known that different types of electrolytes are physiologically involved in cochlear homeostasis to guarantee a balanced ionic cochlear environment and that, hormonal fluctuations are known to result in compromised homeostasis of the auditory system and indirect modulation of auditory functions because of the presence of receptors in the cochlea for estrogen and progesterone [8].
Thus, it is no surprise that the hormonal changes, which reverberated on the electrolyte balance, could be the cause of the signs and symptoms as reported by the patients in the luteal phase of the menstrual cycle. Experimental studies [23] have suggested that the aldosterone, which increased in the luteal phase, could increase the endolymphatic absorption inside the ear and, in particular, in the endolymphatic sac. The Na+/K+ATPasi pump, the Na+Cl- co-transporter and the epithelial Na+ canals seem to be controlled by the aldosterone and are expressed also in the endolymphatic sac [13]. Besides, the endolymphatic sac increased the Na+ permeability and the Na+/K+ATPasi activity, which potentially contributed to absorbing the endolymph. The numerous variations in internal ear homeostasis may then affect the auditory/vestibular system. In this study has been observed that, during the luteal phase, the examined patients reported an increase in audio-vestibular problems compared to the follicular phase.
First of all, as shown in Table 1, all patients showed signs of peripheral labyrinthine suffering during the luteal phase. There were evident signs which showed the canal-macular malfunction, as asymmetries where a side prevailed or/and an increase in the width of the nystagmus. The situation is different in the follicular phase (Table 1), the signs of labyrinthine suffering are not present in all patients and when present is less severe. As the literature confirmed [11, 24] labyrinthine suffering observed in the luteal phase could be a question of a probable expression of an endolymphatic hydrops caused by the electrolyte fluctuation because of a retention of sodium due to, as mentioned above, an increase in female hormones.
Moreover, for our patients, it was observed a lessening, in the luteal phase, in their auditory perceptive abilities (Figure 2-4), with a capacity perceptive reduced by 10% to the various intensities examined. In fact, they asked for the chance to carry out a fitting session for the cochlear implant during the luteal phase.
Our hypothesis is that the lessening in perceptive auditory abilities could be linked to the altered central auditory elaboration caused by an estrogenic fall and moreover to the increase in the electric impedances of the CI (Figure 5). About central auditory elaboration, recent studies [25, 26], have shown, in fact, that the fluctuations in female hormones can pilot the sound elaboration of the auditory cortex through modulations in the neuronal dopaminergic and cholinergic circuit in the cerebral cortex. Indeed the alterations of the cerebral substance during the menstrual cycle certainly play a role. As we mentioned previously, it's confirmed the increase in bilateral gray hippocampal volumes and the increase of the gray matter volumes of the right base ganglia after ovulation [4]. Moreover, a very recent study on 55 women has shown alterations of the brain substance during the menstrual cycle. In particular, it shows a significant estradiol- dependent pre-ovulatory increase in gray matter volumes of the bilateral hippocampus, but also a significant, progesterone- dependent increase in gray matter volumes of the right basal ganglia after ovulation [27]. The authors have shown that hormone driven menstrual cycle changes in human brain structure are small, but may be the underlying cause of menstrual cycle dependent changes in cognition and emotion. About electric impedances, our data showed, in the luteal phase, a homogeneous increase in the impedances of the electrodes over the entire sample which was studied (Figure 5). As has already been said, it is in this phase that the greatest hydro-electrolyte changes can be verified, among which is probably an endolymphatic hydrops. The hydro- electrolyte environment in which the electrodes were immersed was thus modified, in this phase, altering in part the electric transmission and thus probably auditory performances. It can be supposed that an increase in the K+ ions in the perilymph, which are normally scarcely represented, may perform a detergent function on the electrodes and may lead to a detachment of the subtle fibrotic area that keeps the electrodes in a stable position and thus an increase in impedances which negatively affects the hearing ability. With regard to this there can also be included an increase in the water through the aquaporin action which can determine the dilution of the endolymphatic liquids and so can lead to a reduction in its conduction capacity; it has been demonstrated in fact the influence of aquaporin in hydrops [28, 29].
Failure to alter hearing thresholds (Figure 1) despite the variation in electrode impedance (Figure 5) could be explained by the fact that electrical stimulation is within the micro-ampere range compared to nano-ampere in ion current, and would therefore not be relevant to hearing thresholds, but would instead affect the increase in volume and therefore the verbal perception (Figures 2-4).
The results showed how, during the late luteal phase, the symptoms the patients referred to corresponded to precise and objective signs. The work, therefore, add some suggestions on the role of hormones in corporal homeostasis and in particular, on the audio-vestibular performances. In fact, the audio-vestibular symptoms, reported by the patients with the cochlear implant, seemed to be amplified, in the luteal phase, and the lessening of their auditory ability is considered very annoying. Thus, it is important to bear in mind these features to avoid negative results during audiological rehabilitation.
It is clear that having studied a limited number of patients these data need further confirmation on a larger and statistically significant sample.
Conflict of Interest
The authors declare that they have no conflict of interest.
References
- Farage MA. Cognitive, sensory, and emotional changes associated with the menstrual cycle: a review. Arch Gynecol Obstet 278 (2008): 299- 307.
- Cherian KE, Kapoor N, Mathews SS, Paul TV. Endocrine Glands and Hearing: Auditory Manifestations of Various Endocrine and Metabolic Conditions. Indian J Endocrinol Metab 21 (2017): 464-469.
- Takeda T, Takeda S, Kakigi A, Okada T, Nishioka, R, et al. Hormonal aspects of Ménière's disease on the basis of clinical and experimental studies. ORL J Otorhinolaryngol Relat Spec 71 (2010): 1-9.
- Charitidi K, Meltser I, Canlon B. Estradiol Treatment and Hormonal Fluctuations During the Estrous Cycle Modulate the Expression of Estrogen Receptors in the Auditory System and the Prepulse Inhibition of Acoustic Startle Response. Neuroendocrinology 153 (2012): 4412-4421.
- López-Grueso R, Gambini J, Abdelaziz KM, Monleón D, Díaz A, et al. Early, but not late onset estrogen replacement therapy prevents oxidative stress and metabolic alterations caused by ovariectomy. Antioxid Redox Signal 20 (2014): 236-246.
- Al-Mana D, Ceranic B, Djahanbakhch O, Luxon LM. Alteration in auditory function during the ovarian cycle. Hear Res 268 (2010): 114-122.
- Adriztina I, Adnan A, Adenin I, Haryuna SH, Sarumpaet S. Influence of Hormonal Changes on Audiologic Examination in Normal Ovarian Cycle Females: An Analytic Study. Int Arch Otorhinolaryngol 20 (2016): 294-299.
- Da Silva Souza D, Luckwu B, De Andrade WTL, De Figueiredo Pessoa LS, et al. Variation in the hearing threshold in women during the menstrual cycle. International Archives of Otorhinolaryngology 21 (2017): 323-328.
- Emami SF, Gohari N, Ramezani H, Borzouei Hearing Performance in the Follicular- Luteal Phase of the Menstrual Cycle.International Journal of Otolaryngology (2018): 1-6.
- Gould E, Woolley CS, Frankfurt M, Mc Ewen BS. Gonadal steroids regulate dendritic spine density in hippocampal pyramidal cells in adulthood. J Neurosci 10 (1990): 1286-1291.
- Ishii C, Nishino LK, Campos CA. Vestibular characterization in the menstrual cycle. Brazilian Journal of Otorhinolaryngology 75 (2009): 375-380.
- Akdal, G, Ozge, A, Ergör, G. The prevalence of vestibular symptoms in migraine or tension- type headache. J Vestib Res 23 (2013):101- 106.
- Mori N, Miyashita T, Inamoto R, Matsubara A, Mori T, et al. Ion transport its regulation in the endolymphatic sac: suggestions for clinical aspects of Meniere's disease. Eur Arch Otorhinolaryngol 274 (2017): 1813-1820.
- Wilk M, Hessler R, Mugridge K, Jolly C, Fehr M, et al. Impedance Changes and Fibrous Tissue Growth after Cochlear Implantation Are Correlated and Can Be Reduced Using a Dexamethasone Eluting Electrode. PLoS One 11 (2016):
- Leone CA, Mosca F, Grassia R. Temporal changes in impedance of implanted adults for various cochlear segments. Acta Otorhinolaryngol Ital 37 (2017): 312-319.
- Szmuilowicz ED, Adler GK, Williams JS, Green DE, Yao TM, et al. Relationship between Aldosterone and Progesterone in the Human Menstrual Cycle. J Clin Endocrinol Meta 91 (2006): 3981-3987.
- Dullo P, Vedi N. Changes in serum calcium, magnesium and inorganic phosphorous levels during different phases of the menstrual cycle. J Hum Reprod Sci 1 (2008): 77-80.
- Pandya AK, Chandwani S, Das TK, Pandya KD. Serum calcium, magnesium and inorganic phosphorous levels during various phases of menstrual cycle. Indian J Physiol Pharmacol 39 (1995): 411-414.
- Pitkin RM, Renolds WA, William GA, Hargis GK. Calcium regulating hormones during the menstrual cycle. J Clin Endocrinol Metabol 47 (1978): 626-632.
- Solomon SF, Kurere MS, Callway DM. Menstrual cycle and basal metabolic rate in women. An J Clin Nutr 36 (1982): 611-616.
- James PK. Disorders of magnesium metabolism. In: Harrison TR and Fauci AS (eds). Harrison’s principles of internal medicines. USA: McGraw-Hill. P (1998): 2263-2265.
- Young MH, Jasani C, Smith DA. Some effects of ethinyl estradiol on calcium and phosphorous metabolism in osteoporosis. Clin Sci 34 (1968): 411-412.
- Andrews JC, Ator GA, Honrubia V. The Exacerbation of Symptoms in Ménière’s Disease during the Premenstrual Period Archives of Otolaryngology - Head and Neck Surgery 118 (1992): 74-78.
- Scheibe F, Haupt H. Differences in biochemical composition between perilymph, cerebrospinal fluid and blood plasma in the guinea pig. In: Ernst A, Marchbanks R, Samii M, editors. Intracranial and Intralabyrinthine Fluids. Springer, Berlin, Heidelberg (1996): 117-122.
- Hederstierna C, Hultcrantz M, Rosenhall U. Estrogen and hearing from a clinical point of view; characteristics of auditory function in women with Turner syndrome. Hear Res 252 (2009): 3-8.
- Price K, Zhu X, Guimaraes PF, Vasilyeva ON, Frisina RD. Hormone replacement therapy diminishes hearing in peri-menopausal mice. Hear Res 252 (2009): 29-36.
- Pletzer B, Harris T, Hidalgo-Lopez E. Subcortical structural changes along the menstrual cycle: beyond the hippocampus. Scientific Reports 8 (2018):
- Asmar MH, Gaboury L, Saliba I. Ménière's Disease Pathophysiology: Endolymphatic Sac Immunohistochemical Study of Aquaporin-2, V2R Vasopressin Receptor, NKCC2, and TRPV4. Otolaryngol Head Neck Surg158 (2018): 721-728.
- Jiang LY, He JJ, Chen XX, Sun XJ, Wang XZ, et al. Arginine vasopressin-aquaporin-2 pathway-mediated dehydration effects of electroacupuncture in guinea pig model of AVP-induced endolymphatic hydrops. Chin J Integr Med (2018): 1-7.


 Impact Factor: * 5.8
Impact Factor: * 5.8 Acceptance Rate: 71.20%
Acceptance Rate: 71.20%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 