How Do Anthocyanins Affect Peripheral Blood Flow: A Systematic Review
Eleanor Moore, Anna E Litwic*, Peter Belward, Pat Taylor, David Warwick, Elaine Dennison
MRC Lifecourse Epidemiology Unit, University of Southampton, Southampton General Hospital, Southampton, SO16 6YD, UK
*Corresponding Author: Anna E Litwic, MRC Lifecourse Epidemiology Unit, University of Southampton, Southampton General Hospital, Southampton, SO16 6YD, UK
Received: 12 January 2017; Accepted: 27 January 2017; Published: 29 January 2017
Article Information
View / Download Pdf Share at FacebookAbstract
Foods rich in polyphenols, a type of antioxidant, have many health benefits. The largest and most important subgroup of polyphenols are flavonoids, of which anthocyanins are one. In vitro studies have shown that anthocyanin induced vasorelaxation is dependent on Nitrous Oxide and a functional endothelium. The anthocyanin effect on change in blood flow could have potential therapeutic implications. However it is unclear if the results can be extrapolated to humans. The number of studies investigating the effects of anthocyanins within humans is fairly limited and the results are discordant. The conflicting evidence surrounding anthocyanins and blood flow highlights the need for a future research in this area.
Keywords
Anthocyanins; Vasodilation; Humans; Blood Flow; Systematic Review
Article Details
1. Introduction
Foods rich in polyphenols, a type of antioxidant, has many health benefits. They reduce the incidence of numerous diseases, including coronary heart disease and cancer [1]. There is also increasing evidence that these compounds reduce the incidence of neurodegenerative diseases associated with oxidative stress, such as Alzheimer’s disease and Parkinson’s disease [2]. Polyphenols can be divided into different sub-groups depending on the number of phenol rings and the chemical groups attached to the rings. The largest and most important sub-group of polyphenols are flavonoids [2]. Flavonoids are the most abundant type of antioxidant found in common diets, and have been shown to reduce oxidative stress. This is achieved by direct scavenging of oxygen free radicals, and by the inhibition of radical oxygen species producing enzymes [3]. Anthocyanins, a type of flavonoid, are a group of water-soluble pigments that give many fruits their blue, purple and red colour [4]. There are several hundred known types of anthocyanin. The chemical structure of anthocyanins consists of a C6-C3-C6 skeleton, with other functional chemical groups attached [5].
Numerous studies have shown that polyphenol rich foods, such as blackcurrants, strawberries and grapes, have vasodilatory effects [6-8]. Fruits containing anthocyanins also contain many other polyphenolic compounds. Identifying which compound is responsible for the associated health benefits with these fruits has been difficult. However, Bell et al investigated the vasodilatory properties of preparations containing only anthocyanins; they found that anthocyanins were responsible for relaxation of coronary vessels in animal models [9].
In vitro studies have shown that anthocyanin induced vasorelaxation is dependent on Nitrous Oxide (NO) and a functional endothelium [9-11]. For example a study performed by Xu et al showed that the anthocyanin cyanidine-3-glucoside exerts a vasodilatory effect by increasing production of NO40, by activation of eNOS34. Another study confirmed the vasodilatory properties of anthocyanins; it showed that blackcurrant anthocyanins caused phosphorylation of eNOS via the activation of the phosphatidylinositol-3 (PI3) kinase/protein kinase B (Akt) signalling pathway [12].
Scavengers of oxygen free radicals can also enhance NO mediated vasorelaxation [13]. Oxygen free radicals cause break down of NO, therefore reducing vascular smooth muscle relaxation caused by NO. Scavengers of oxygen free radicals reduce the breakdown of NO and so cause increased vascular smooth muscle relaxation. Ascorbic acid (Vitamin C) is one of the strongest reductants and radical scavengers [14]. Some studies have suggested that vitamin C supplementation may reduce the incidence of conditions in which vasomotor stability is impaired, such as Complex Regional Pain Syndrome (CRPS) [15]. In a randomised controlled trial 317 patients with 328 wrist fractures received either vitamin C or placebo, the incidence of CRPS in the vitamin C group was 2.8% compared with 10.1% in the control group (p=0.002) [16]. This study was subsequently supported by a meta-analysis, although this only included 4 trials [17]. A proposed hypothesis for the pathophysiology of CRPS is deep tissue microvascular pathology, where a series of events lead to ischaemia-reperfusion (I-R) injury [18]. An I-R injury is caused by endothelial damage via oxidants produced by neutrophils. The production of oxidants by neutrophils is reduced by the administration of vitamin C; vitamin C also protects the endothelium from direct injury from oxidants. Vitamin C increases NO production by the PI3 kinase/Akt signalling pathway [16]. Anthocyanins increase NO production via the same pathway [12] and they are also potent oxidant scavengers [19]. Therefore it can be hypothesised that anthocyanins may also be protective against CRPS.
Although there have been many in vitro experiments showing the vasorelaxant effects of anthocyanins, it is unclear if the results can be extrapolated to humans. The number of studies investigating the effects of anthocyanins within humans is limited. In a study by McLeay et al, an anthocyanin rich blueberry drink increased the rate of muscle recovery after exercise induced muscle damage [20]. The literature on anthocyanins effect on blood flow in humans is limited and the results are discordant; while a study by Matsumoto et al showed that anthocyanins increase forearm blood flow [21], another study showed that anthocyanins had no effect on peripheral blood flow [22]. We performed a systematic review of this subject.
2. Methods
An electronic search was carried out using the electronic databases MEDLINE (1996-2015) and EMBASE (1974-2015). The search terms used were ‘anthocyanin’ OR ‘anthocyanins’ AND ‘vasodilation’ OR ‘blood flow’ OR ‘vasorelaxation’ OR ‘vascularization’ OR ‘vascularisation’ OR ‘vasodilatation’. The final search date was 28th October 2015. Following the removal of duplicates, abstracts and citations were stored using the bibliographic software ENDNOTE.
3. Study selection
We evaluated each identified study against the following predetermined selection criteria:
Study population: human subjects
Intervention: anthocyanins, regardless of duration, frequency, or intensity
Comparator: placebo
Outcome measure: change in peripheral blood flow
Study design: randomized controlled trials.
Two reviewers independently screened the titles and abstracts to determine if a citation met the general inclusion criteria. The full text of citations classified as include or unclear was reviewed independently with reference to the predetermined inclusion and exclusion criteria. Finally, we hand-searched reference lists of any relevant conference abstracts and of the included trials for potentially relevant citation. Non-English full text citations were excluded. Disagreements between the two reviewers were resolved through consensus and by third-party adjudication, as needed.
4. Results
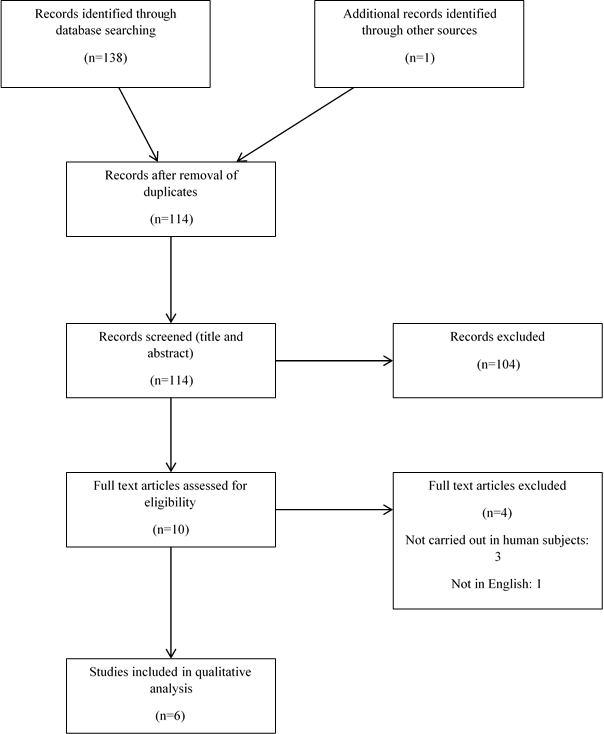
Of the 139 citations identified through electronic and hand searches, we included 6 trials enrolling a total of 308 participants (Figure 1) 22-27. Trials were published between 2005 and 2015. The outcomes relevant to peripheral blood flow included: total peripheral resistance, flow mediated vasodilatation, forearm blood flow and blood pressure. Only two trials did not measure flow mediated vasodilatation [23,27]. The key features of the included studies are outlined in Table 1.
| Reference | Type of Study | Duration | Participants | Outcomes * | Findings |
|---|---|---|---|---|---|
| Matsumoto H, et al. 2005 | RCT | Part 1: 4 hours Part 2: 2 weeks |
Part 1: 9 healthy individuals Part 2: 11 healthy individuals |
FBF and muscle O2 consumption | Part 1: increase in FBF at 2h Part 2: Increase in muscle O2 consumption |
| Dohadwala MM et al. 2011 | RCT | 10 weeks | 44 individuals with coronary artery disease | FMD, resting BP | No statistically significant change in FMD or blood pressure |
| Zhu Y, et al. 2011 | RCT | Part 1: 4 hours Part 2: 12 weeks |
Part 1: 12 hypercholesterolaemic individuals Part 2: 150 hypercholesterolaemic individuals |
FMD | Part 1: increase in FMD Part 2: no statistically significant change in FMD compared to placebo |
| Rodriguez-Mateos A, et al. 2013 | RCT | Part 1: 6 hours Part 2: 1 hour |
Part 1: 10 healthy individuals Part 2: 11 healthy individuals |
FMD and BP | Part 1: increase in FMD at 1,2 and 6hrs Part 2: increase in FMD |
| Draijer R, et al. 2015 | RCT | 10 weeks | 60 mildly hypertensive individuals | 24hr ambulatory BP, resting BP, FMD | Decrease in 24hr ambulatory BP due to grape-wine extract |
| Willems MET, et al. 2015 | RCT | 7 days | 13 healthy individuals (only 10 had TPR recorded) | TPR | Decrease in TPR |
Table 1: Randomised controlled trials examining the relationship between anthocyanins and peripheral blood flow
The included studies will be discussed in turn. A study performed by Matsumoto et al in 2005 consisted of two parts [23]. The first part (part 1) measured the effect of blackcurrant anthocyanin on peripheral circulation during rest. Firstly forearm blood flow was measured, and then blackcurrant anthocyanin was ingested. Forearm blood flow was then measured at hourly intervals for 4 hours. The second part (part 2) measured the effect of blackcurrant anthocyanin on peripheral circulation and muscle stiffness during typing work. The study used near infrared spectroscopy to measure forearm blood flow and muscle oxygen consumption. Neither the method of randomisation nor blinding is outlined in this study, so it is unclear if this is a potential source of bias. 9 healthy males with an average age of 29. 9 took part in part 1, and 11 healthy individuals (8 male and 3 female with an average age of 39.0) took part in part 2. The demographics of the placebo group and anthocyanin group were not disclosed in part 2 of the study. This could be a source of bias, as the groups may not have been adequately matched. The results of part 1 of this study show a significant (p<0.05) increase in forearm blood flow 2 hours after ingestion of blackcurrant anthocyanin compared to placebo. Part 2 of the study shows that blackcurrant anthocyanin intake prevented the decrease in oxygenated haemoglobin significantly, compared to placebo (p<0.05).
In a placebo-controlled crossover study subsequently performed by Dohadwala et al in 2011 the effects of cranberry juice consumption on vascular function in patients with coronary artery disease was assessed [22]. Treatment was received for 4 weeks with a 2-week rest period between beverages. The cranberry juice contained 94mg of anthocyanins per serving. As part of the vascular function testing brachial artery flow-mediated dilation was measured. Beverage order was randomized and both the participants and the investigators were blinded until after the statistical analysis had taken place. However the randomisation method was not detailed in the study so it is unclear as to whether randomisation was adequate. The two study groups were fairly well matched demographically. 22 participants from both groups completed the study. The juice first group and placebo first group had a mean age of 61 and 63 respectively. The sample size was modest (15 males and 7 females). Most of the subjects were on risk reduction therapy (ACE inhibitors, aspirin, clopidogrel or lipid lowering drugs). Of the 47 participants that started the trial, 44 completed the trial. The study reported a 98% compliance rate, however this ranged from 68% - 100%. Participants with a compliance rate less than 80% were removed and the data were reanalysed, but this made little difference to the result. The trial did not show a statistically significant change in brachial artery flow-mediated dilation, the pre-specified primary endpoint. Neither did it show a change in blood pressure. This may be because most of the participants were receiving risk reduction therapy, and so this interfered with the action of the anthocyanins.
A study by Zhu et al. in 2011 consisted of two parts; it investigated both the short and long term effects of anthocyanins on endothelial function in hypercholesterolaemic individuals [24]. This study measured blood flow by measuring brachial artery flow-mediated dilation. The short-term study measured flow-mediated dilation at baseline and at 1, 2 and 4 hours after ingestion of 320mg of anthocyanins. In the long-term study participants took 320mg of anthocyanin a day for 12 weeks, with measurements taken every 4 weeks. In the both the short and long term study the method of randomisation is not detailed. Blinding occurred, as anthocyanins and placebo were both provided in capsules. The short-term study had a small sample size of only 12 participants. However, a very large sample size (150 individuals), was used for the long-term study. The intervention and control groups had very similar demographics and so were adequately matched. 4 participants did not finish the trial, due to initiation of lipid-lowering therapy, or moving out of the study area. Compliance to the treatment was recorded by recalling empty capsule packets, but the compliance rate was not recorded in the study. The results of the short-term study showed that anthocyanin intake significantly improved (p<0.05) brachial artery flow-mediated dilation. In the intervention group of the long-term study, there was a statistically significant improvement in brachial artery flow-mediated dilation after 12 weeks of anthocyanin ingestion; no such change was observed in the placebo group (p<0.05).
A study by Rodriguez-Mateos et al. in 2013 consisted of two parts [25]. The first investigated how differing concentrations of anthocyanins affected vascular function over time. The second was a 6-arm study where participants consumed anthocyanin-containing drinks of differing concentrations or placebo. 21 healthy male volunteers took part in the study. 10 took part in the first and 11 took part in the second. In both parts of the study flow mediated dilation of the brachial artery was the main outcome. An independent researcher randomly generated treatment allocation. The method of randomization was adequate. The results of the first part of the study showed a biphasic increase in flow mediated dilation after ingestion of 310mg of anthocyanins after 1, 2 and 6 hours, with the largest increase after 1 hour. These results were statistically significant (p<0.05). The second part of the study showed a significant (p<0.05) increase in flow mediated dilation after ingestion of all concentrations of anthocyanins (129-724mg). There was no statistically significant change in blood pressure.
Draijer et al. carried out a randomized controlled double blind incomplete crossover study [26]. The study compared the effects of grape extract, grape-wine extract and placebo on blood pressure in mildly hypertensive subjects. All participants took a placebo for 2 weeks, followed by two 4-week intervention periods. Half of the subjects took grape-wine extract followed by grape extract; the other half took grape extract first. Measurements were taken at the end of the run in period, and at the end of the two 4 week intervention periods. Again, this trial does not outline the methods of randomization. All treatments were given in red gelatine capsules to ensure blinding. There were 60 subjects in this trial, half took grape extract and the other took grape-wine extract. The average age of the participants was 57.6. Of the 60 subjects that started, 5 dropped out during the placebo stage, however, these were replaced so that 60 people still completed the trial. However the circumstances of the new subjects joining the trial is not discussed in the methodology. It is unclear whether the new subjects took placebo for the same time as the original subjects or not. 70% of the participants were fully compliant with treatment, and the other 30% were 94-99% compliant. This high compliance rate was therefore unlikely to have an effect on the results. The flow- mediated vasodilation was similar in both intervention groups. The results of this trial showed that 24h-ambulatory blood pressure was significantly (p<0.05) lower after grape-wine extract compared with placebo (systolic blood pressure by 3mmHg and diastolic blood pressure by 2 mmHg). However there was no effect on blood pressure from the grape juice extract alone. This suggests that the effects on blood pressure were due to products in the wine rather than the grape juice. Grape juice is rich in anthocyanins and wine is rich in catechins and procyanidins. Therefore the change in blood pressure is most likely not due to anthocyanins.
Willems et al., in 2015, carried out a double-blind randomized placebo controlled crossover study, to investigate the effects of anthocyanins on blood lactate concentrations in triathletes during cycling and at rest [27]. However the study also investigated cardiovascular function as a secondary outcome, including total peripheral resistance. Therefore it is relevant to this review. The participants’ took blackcurrant drink daily, containing 138.6mg of anthocyanins, or a commercially available blackcurrant drink with low concentrations of anthocyanins (3-4mg) as placebo. The participants ingested anthocyanins for 7 days prior to testing.
Cardiovascular function (blood pressure, cardiac output, total peripheral resistance) was measured using a finger monitor beat-to-beat blood pressure monitoring system, a non-invasive way to measure haemodynamic parameters. The methods of randomisation are not detailed in this study. Both the participants and researchers were blinded to treatment allocation. The study was conducted on 8 males and 5 females, with an average age of 38 years (the demographics of the placebo and intervention groups are not detailed). Even though 13 participants took part in the trial, cardiovascular measurements could not be obtained for 3 of them, bringing into question the reliability of the method for measuring the cardiovascular parameters. It is also not detailed as to whether these participants were from the placebo or intervention group. Therefore this could be a serious source of bias in this study. The results of this trial show that at rest anthocyanins caused a 16% decrease in total peripheral resistance compared to the placebo (p<0.05). There was however no change to blood pressure at rest.
5. Discussion
The results of the 6 studies included in this review are discordant. Some studies found that anthocyanins do increase peripheral blood flow [23, 24, 27, 28]. However the other three studies showed no statistically significant change in peripheral blood flow due to anthocyanins [22, 26]. The potential reasons for these discrepancies include: difference in sample population, and differences in the duration of the studies.
In the studies that reported an increase in blood flow, most of the participants were healthy [23, 27, 28], apart from the study with hypercholesterolemic individuals [24]. The other studies, however, investigated the effect of anthocyanins on participants with hypertension and coronary artery disease. In vitro, anthocyanins have been shown to cause vasorelaxation, dependant on the presence of NO and functional endothelium [8, 9]. Coronary artery disease is characterised by endothelial damage; hypertension causes endothelial damage. Therefore these subjects may not have had fully functional endothelial cells and so anthocyanins did not cause statistically significant vasodilation. This may show that anthocyanins are ineffective at increasing peripheral blood flow in individuals with cardiovascular disease or risk factors.
Another reason for the difference in results may be the duration of the studies. All studies that measured the short-term effects of anthocyanins showed an increase in blood flow [23, 24, 28]. However, the trials carried out over a longer period of time did not show the same results. Therefore this may suggest that anthocyanins do not affect peripheral blood flow over a long period of time. However, because the trials that measured the chronic effects only used subjects with cardiovascular disease or risk factors, this may have caused the ineffectiveness of the anthocyanins in these studies. None of the trials in this review investigated the chronic effect of anthocyanins in healthy individuals; the longest trial was 2 weeks [23].
Furthermore, it is also important to discuss the sample sizes of the studies. Most studies that reported an increase in peripheral blood flow due to anthocyanins all had relatively small sample sizes (?21) so the conclusion that anthocyanins increase peripheral blood flow in healthy individuals lacks strength.
This review has some limitations. Due to the heterogeneity of the outcomes measured in the different trials, it was difficult to quantify the effects of anthocyanins on peripheral blood flow. The results of effect of anthocyanins on peripheral blood flow in studies included are conflicting and the potential explanations for these differences are only hypotheses.
This review shows that anthocyanins cause increased peripheral blood flow in healthy individuals. However, this
was not the case in individuals with cardiovascular disorders. This may be due to the lack of functional endothelial
cells in these individuals. There was no evidence that anothcyanins had a chronic effect on peripheral blood flow. The number of studies reviewed was fairly small. This, combined with the differing conclusions of the studies, highlights the need for further research into whether anthocyanins increase peripheral blood flow. It would be interesting to compare their effects in healthy individuals with those with cardiovascular risk factors. It would also be beneficial to investigate the long term effects of anthocyanins at increasing peripheral blood flow in individuals without cardiovascular disease or risk factors.
Acknowledgements
Funding and sponsorship: No external funding was received for this work.
Declaration of Interest
The authors have no relevant interests to declare.
References
- Xiang L, Xiao L, Wang Y, Li H, Huang Z, et al. Health benefits of wine: Don’t expect resveratrol too much. Food Chemistry156 (2014): 258-263.
- Sun AY, Wang Q, Simonyi A, Sun GY. Botanical phenolics and brain health. Neuromolecular medicine 10 (2008): 259-274.
- Siasos G, Tousoulis D, Tsigkou V, et al. Flavonoids in atherosclerosis: An overview of their mechanisms of action. Current Medicinal Chemistry 20 (2013): 2641-2660.
- Fang J. Classification of fruits based on anthocyanin types and relevance to their health effects. Nutrition (Burbank, Los Angeles County, Calif ) 31 (2015): 1301-1306.
- Clifford MN. Anthocyanins ? nature, occurrence and dietary burden. Journal of the Science of Food and Agriculture 80 (2000): 1063-1072.
- Edirisinghe I, Burton-Freeman B, Varelis P, Kappagoda T. Strawberry extract caused endothelium-dependent relaxation through the activation of PI3 kinase/Akt. J Agric Food Chem 56 (2008): 9383-9390.
- Edirisinghe I, Burton-Freeman B, Tissa Kappagoda C. Mechanism of the endothelium-dependent relaxation evoked by a grape seed extract. Clinical science 114 (2008): 331-337.
- Nakamura Y, Matsumoto H, Todoki K. Endothelium-dependent vasorelaxation induced by black currant concentrate in rat thoracic aorta. Japanese journal of pharmacology 89 (2002): 29-35.
- Bell DR, Gochenaur K. Direct vasoactive and vasoprotective properties of anthocyanin-rich extracts. Journal of applied physiology 100 (2006): 1164-1170.
- Andriambeloson E, Magnier C, Haan-Archipoff G, et al. Natural dietary polyphenolic compounds cause endothelium-dependent vasorelaxation in rat thoracic aorta. Journal of Nutrition 128 (1998): 2324-2333.
- Xu JW, Ikeda K, Yamori Y. Upregulation of endothelial nitric oxide synthase by cyanidin-3-glucoside, a typical anthocyanin pigment. Hypertension 44 (2004): 217-222.
- Edirisinghe I, Banaszewski K, Cappozzo J, McCarthy D, Burton-Freeman BM. Effect of black currant anthocyanins on the activation of endothelial nitric oxide synthase (eNOS) in vitro in human endothelial cells. J Agric Food Chem 59 (2011): 8616-8624.
- Taddei S, Virdis A, Ghiadoni L, Magagna A, Salvetti A. Vitamin C improves endothelium-dependent vasodilation by restoring nitric oxide activity in essential hypertension. Circulation 97 (1998): 2222-2229.
- Niki E. Action of ascorbic acid as a scavenger of active and stable oxygen radicals. The American journal of clinical nutrition 54 (1991):1119s-1124s.
- Zollinger PE, Tuinebreijer WE, Kreis RW, Breederveld RS. Effect of vitamin C on frequency of reflex sympathetic dystrophy in wrist fractures: a randomised trial. Lancet 354 (1999): 2025-2028.
- Zollinger PE, Tuinebreijer WE, Breederveld RS, Kreis RW. Can vitamin C prevent complex regional pain syndrome in patients with wrist fractures? A randomized, controlled, multicenter dose-response study. The Journal of bone and joint surgery 89 (2007): 1424-1431.
- Shibuya N, Humphers JM, Agarwal MR, Jupiter DC. Efficacy and safety of high-dose vitamin C on complex regional pain syndrome in extremity trauma and surgery--systematic review and meta-analysis. The Journal of foot and ankle surgery: official publication of the American College of Foot and Ankle Surgeons 52 (2013): 62-66.
- Coderre TJ, Bennett GJ. A hypothesis for the cause of complex regional pain syndrome-type I (reflex sympathetic dystrophy): pain due to deep-tissue microvascular pathology. Pain medicine 11 (2010): 1224-1238.
- Noda Y, Kaneyuki T, Mori A, Packer L. Antioxidant activities of pomegranate fruit extract and its anthocyanidins: delphinidin, cyanidin, and pelargonidin. J Agric Food Chem 50 (2002): 166-171.
- McLeay Y, Barnes MJ, Mundel T, Hurst SM, Hurst RD, et al. Effect of New Zealand blueberry consumption on recovery from eccentric exercise-induced muscle damage. Journal of the International Society of Sports Nutrition 9 (2012): 19.
- Matsumoto H, Takenami E, Iwasaki-Kurashige K, Osada T, Katsumura T, et al. Effects of blackcurrant anthocyanin intake on peripheral muscle circulation during typing work in humans. European Journal of Applied Physiology 94 (2004): 36-45.
- Dohadwala MM, Holbrook M, Hamburg NM, et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. American Journal of Clinical Nutrition 93 (2011): 934-940.
- Matsumoto H, Takenami E, Iwasaki-Kurashige K, Osada T, Katsumura T, et al. Effects of blackcurrant anthocyanin intake on peripheral muscle circulation during typing work in humans. European Journal of Applied Physiology 94 (2005): 36-45.
- Zhu Y, Xia M, Yang Y, et al. Purified anthocyanin supplementation improves endothelial function via NO-cGMP activation in hypercholesterolemic individuals. Clinical Chemistry 57 (2011): 1524-1533.
- Rodriguez-Mateos A, Rendeiro C, Bergillos-Meca T, et al. Intake and time dependence of blueberry flavonoid-induced improvements in vascular function: a randomized, controlled, double-blind, crossover intervention study with mechanistic insights into biological activity. The American journal of clinical nutrition 98 (2013): 1179-1191.
- Draijer R, de Graaf Y, Slettenaar M, de Groot E, Wright CI. Consumption of a polyphenol-rich grape-wine extract lowers ambulatory blood pressure in mildly hypertensive subjects. Nutrients 7 (2015): 3138-3153.
- Willems MET, Myers SD, Gault ML, Cook MD. Beneficial physiological effects with blackcurrant intake in endurance athletes. International Journal of Sport Nutrition and Exercise Metabolism 25 (2015): 367-374.
- Rodriguez-Mateos A, Rendeiro C, Bergillos-Meca T, et al. Intake and time dependence of blueberry flavonoid-induced improvements in vascular function: A randomized, controlled, double-blind, crossover intervention study with mechanistic insights into biological activity. American Journal of Clinical Nutrition 98 (2013): 1179-1191.


 Impact Factor:
* 5.8
Impact Factor:
* 5.8
 Acceptance Rate:
71.20%
Acceptance Rate:
71.20%
 Time to first decision: 10.4 days
Time to first decision: 10.4 days
 Time from article received to acceptance:
2-3 weeks
Time from article received to acceptance:
2-3 weeks
