Pathological and Surgical Outcomes of Retropubic Radical Prostatectomy in an Academic Center: A Contemporary Study
Thomas Prudhommea*, Mathieu Roumiguiéa, Sarah Péricartb, Nicolas Doumerca, Marine Lesourda, Pierre Marie Patarda, Xavier Gaméa, Michel Souliéa, Matthieu Thoulouzana
aDepartment of Urology, Andrology and Kidney Transplantation, University Hospital of Rangueil, Toulouse, France
bDepartment of Pathology, Institut Universitaire du Cancer, Toulouse, France
*Corresponding Author: Thomas Prudhomme, Department of Urology, Andrology and Kidney Transplantation, University Hospital of Rangueil, 31059 Toulouse Cedex 9, France
Received: 03 August 2020; Accepted: 14 August 2020; Published: 16 October 2020
Article Information
Citation: Thomas Prudhomme, Mathieu Roumiguié, Sarah Péricart, Nicolas Doumerc, Marine Lesourd, Pierre Marie Patard, Xavier Gamé, Michel Soulié, Matthieu Thoulouzan. Pathological and Surgical Outcomes of Retropubic Radical Prostatectomy in an Academic Center: A Contemporary Study. Archives of Clinical and Biomedical Research 4 (2020): 498-512.
View / Download Pdf Share at FacebookAbstract
Purpose: To report the peri-operative, pathological and oncological outcomes of a contemporary series of open retropubic radical prostatectomy (RRP), performed by one experienced surgeon on RRP technique developed by Barré C.
Methods: We analyzed data of consecutive patients who underwent an RRP as first-line treatment for localized prostate cancer, from January 2014 to December 2018, in single French academic center. a) Benign capsular incision (BCI) and b) Benign glandular tissue incision (BGTI) were defined as: presence of inadvertent intraprostatic incisions into benign glandular tissue in regions where capsule was a) present and b) absent.
Results: Overall, 312 patients were included. The rates of pT1c, pT2 and pT3 were 0.3% (n=1), 49.0% (n=153) and 50.6% (n=158), respectively. The rates of non-nerve sparing surgery (NSS), unilateral NSS and bilateral NSS were 20.5% (n=64), 31.4% (n=98) and 48.1% (n=150). The median length of hospital stay was 4.0 (3.0-5.0) days and the median estimated blood loss was 250.0 (150.0-180.0) milliliters. The total positive surgical margin (PSM) rate was 11.9% (n=37). The total pT2 PSM and pT3 PSM rates were 0% and 23.4% (n=37) and achieved a statistical difference (p<0.001). The total BCI and BGTI rates were 1.6% and 9.0 %. Biochemical recurrence occurred in 29 (9.3%) patients at median 1.0-year of follow-up.
Conclusion: Retropubic radical prostatectomy performed as previously described by Barré C., is reproductible and ensures optimal surgical results, in a predominantly population of intermediate-risk prostate cancer and high-risk prostate cancer.
Keywords
<p>Retropubic Radical Prostatectomy; Positive Surgical Margin; Nerve Sparing Surgery; Benign Capsular Incision; Benign Glandular Tissue Incision</p> <gdiv></gdiv>
Article Details
Introduction
Prostate cancer is the second most common solid malignancy in men (excluding keratinocyte cancers) behind lung cancer, with more than 1.200.000 cases diagnosed worldwide in 2018 [1].
Radical prostatectomy still remains the most common treatment for clinically localized prostate cancer. However, surgical practice patterns for localized prostate cancer have changed rapidly in last decades. Since the first reported robot-assisted laparoscopic prostatectomy (RALP) by Binder and Kramer [2] in 2001, rapid adoption of this surgical technique has been made such that in many countries (including France), RALP became the dominant surgical approach for radical prostatectomy [3].
Despite this rapid development, recent prospective multicenter study [4] reported similar functional and oncological outcomes between open retropubic radical prostatectomy (RRP) and RALP. However, main criticisms of open RRP were a higher mean operative blood loss and longer length of hospital stay, compared to RALP [5, 6].
In order to optimize surgical outcomes of RRP, Barré C. [7] developed a reproductible RRP surgical technique, based on the technique developed by Walsh P. [8]. In 2014, Barré C. et al [9] reported their results, with a pT2 positive surgical margin (PSM) rate of 2.3% which is the lowest pT2 PSM rate reported in the literature. However, opponents of retropubic radical prostatectomy highlight the significant surgical difficulty and the lack of reproducibility of the oncological results reported by Barré C. et al.
The aim of this study was to report the peri-operative, pathological and oncological outcomes of a contemporary series of RRP, performed by one experienced surgeon on RRP technique developed by Barré C.
Patients and Methods
1. Patients and Database
For the present prospective study, we analyzed data of consecutive patients who underwent an open retropubic radical prostatectomy (RRP) as first-line treatment for localized prostate cancer, from January 1th 2014 to December 31th 2018, in one French academic center.
2. Surgical Technique
All RRP were performed according to the previous described procedure by Barré C. [7], by one experienced surgeon (MT), who achieved more than 200 RRP prior to the beginning of this study. Key points were always (a) leaving a thin crown of striated fibers at the prostate apex, (b) removing the prostate fascia and Denonvilliers’ fascia which adjoins the prostatic capsule, and (c) resecting the bladder neck. There was no intrafascial dissection, the prostatic and Denonvilliers’ fascias were always recovered on the specimen.
Nerve sparing surgery (NSS) was performed by retrograde dissection after sphincter complex division. The bundle was released from the prostate with a microdissector by isolating connective tissue and small vascular micropedicles (which were controlled by extra-small 3.8 mm hemoclips (Ethicon Inc, Cincinnati, Ohio, USA)) and with pointed scissors which allows to release the neurovascular bundle from direct contact with the prostatic fascia and Denonvilliers’ fascia. NSS was performed in patients with pre-operative erectile function, based on pre-operative data (preoperative ISUP grade and preoperative pathological stage).
3. Histological assessment
According to the Stanford technique, developed by Mc Neal JE et al [10], a standard histological preparation of the prostate was performed. A positive surgical margin (PSM) was defined as presence of malignant glandular tissue at the inked surface of the resected specimen. A benign capsular incision (BCI) was defined as presence of inadvertent intraprostatic incisions into benign tissue in regions where capsule was present (mid-prostate and posterior base) [11, 12]. A benign glandular tissue incision (BGTI) was defined as presence of inadvertent intraprostatic incisions into benign glandular tissue in regions where capsule was absent (prostate apex and bladder neck, i.e. the anterior prostate base). The volume of the tumor was estimated by the pathologist as a percentage of the prostate that been infiltrated by the prostate cancer.
4. Study Variables and Outcomes
In the prospective database of consecutive patients, the patients baseline characteristics, intra- and post-operative outcomes data that were collected included: age (years), preoperative PSA (ng/ml), preoperative clinical stage, preoperative biopsy ISUP and Gleason grade, D’Amico risk classification, estimated blood loss (milliliters), operative time (minutes), length of hospital stay (days) and post-operative complications, according to the Clavien-Dindo classification [13]. NSS outcomes data collected were: NSS rate, pT2 and pT3 pathological stage, according to the NSS status. Pathological and oncological outcomes collected were: pathological stage, pathological ISUP and Gleason grade, prostate volume (cm3), tumor volume (%), lymph node dissection, number of lymph nodes removed, biochemical recurrence (BCR), BCR free survival in overall population and BCR free survival by pathological stage and surgical margin status. PSM outcomes collected were: total PSM rate, solitary and multiple PSM rate, location of solitary PSM, solitary and multiple PSM lengths, pT2 pathological stage PSM rate, pT3 pathological stage PSM rate, positive concordance between PSM and extracapsular extension, PSM status according to the pathological stage and PSM status according to the NSS status. BCI and BGTI outcomes collected were: total and solitary BCI rates, location of BCI, BCI length, total, solitary and multiple BGTI rates, location of BGTI, solitary and multiple BGTI lengths.
Biochemical recurrence (BCR) was defined as three postoperative PSA values of ≥0.2 ng/ml.
5. Statistical Analysis
Quantitative data were expressed as median (interquartile range) and qualitative data as number and proportion (%). The NSS rates, according to the pathological stage were compared using Student t test. PSM status according to the pathological stage and the NSS status were compared using Fisher’s exact test and Chi-square test, respectively. The postoperative BCR-free survival was estimated using the Kaplan-Meier method and compared with the log-rank test method. All reported p-values were two-sided with a significance level at p<0.05. A statistical analysis was performed using S Prism 7.0a (GraphPad Software Inc, La Jolla, CA, USA) and SPSS 20.0 (SPSS Inc., Arlington, Virginia).
Results
1. Patient Characteristics and Peri-Operative Outcomes
From January 1th 2014 to December 31th 2018, among the 1,084 radical prostatectomies performed in our Department, 312 (28.8%) were RRP (772 RALP versus 312 RRP). The baseline characteristics of these patients were summarized in Table 1. The median age was 65.2 (60.5-68.7) years. The median preoperative PSA was 8.0 (6.0-11.0) ng/ml. According to the D’Amico risk classification, the rate of patient who had of low-risk, intermediate-risk and high-risk prostate cancer was 14.4% (n=45), 64.1% (n=200) and 21.5% (n=67), respectively. The median estimated blood loss was 250.0 (150.0-180.0) milliliters and the median operative time was 160.0 (150.0-180.0) minutes. The median length of hospital stay was 4.0 (3.0-5.0) days. Early (post-operative day (POD) 30) major (Clavien-Dindo ≥III) post-operative complications was observed in 30 (9.6%) patients: 16 (5.1%) patients required a percutaneous drainage of a symptomatic pelvic lymphocele, 5 (1.6%) patients required a surgical re-exploration due to hematoma and 6 patients (1.9%) had an open or laparoscopic lymphocele marsupialization. 1 death occurred due to pulmonary embolism.
2. NSS outcomes
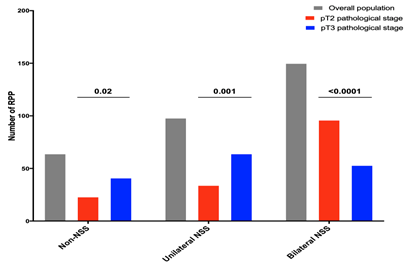
The rates of non-NSS, unilateral NSS and bilateral NSS radical prostatectomy were 20.5% (n=64), 31.4% (n=98) and 48.1% (n=150). The rates of non-NSS (25.9% vs 15.0%, p=0.02) and unilateral NSS (40.5% vs 22.2%, p=0.001) were statistically higher in pT3 than pT2 pathological stage. As expected, the rate of bilateral NSS was statistically lower in pT3 than pT2 pathological stage (33.5% versus 62.7%, p<0.0001), Figure 1.
3. Pathological and Oncological Outcomes
The rates of pT1c, pT2 and pT3 were 0.3% (n=1), 49.0% (n=153) and 50.6% (n=158), respectively. Among the 272 (87.2%) lymph node dissection performed, 38 (14.0%) patients had a positive lymph node disease. The median number of lymph nodes removed was 19 (14-24). Regarding the pathological ISUP grade, we observed 28 (9.0%) ISUP 1 grade, 154 (49.4%) ISUP 2 grade, 111 (35.6%) ISUP 3 grade, 8 (2.6%) ISUP 4 grade and 11 (3.5%) ISUP 5 grade. The median prostate volume (cm3) and the median tumor volume (%) were 45 (37-60) cm3 and 12 (7-25) %, respectively, Supplementary Table 1.
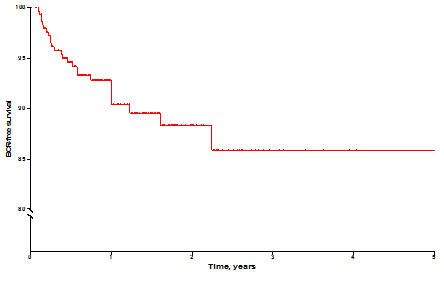
At a median follow-up of 1.0-year, biochemical recurrence (BCR) occurred in 29 (9.3%) patients. The BCR-free survival in the overall population was shown in Supplementary Figure 1.
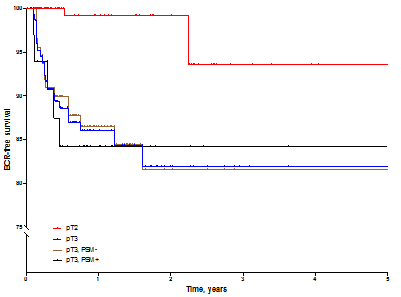
Extracapsular extension was associated with a poor BCR-free survival as compared to organ confined disease (p<0.0001). Intriguingly, in theses pT3 pathological stage tumor, PSM did not impact the BCR survival (HR: 1.1, 95% CI 0.4-3.1), Supplementary Figure 2.
4. PSM Outcomes
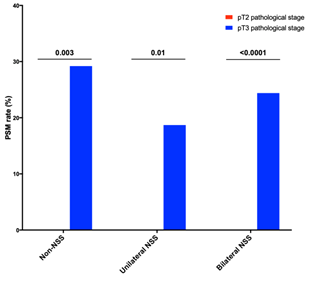
The positive surgical margin (PSM) rate was 11.9% (n=37). The total pT2 PSM and pT3 PSM rates were 0% and 23.4% (n=37) and achieved a statistical difference between two groups (p<0.001), Supplementary Table 2 and Supplementary Figure 3. The positive concordance rate between PSM and extracapsular extension was 84.6% (n=22) in case of solitary PSM and 54.5% (n=6) in case of multiple PSM. Twenty-six (70.3%) PSM were solitary while 11 (29.7%) PSM were multifocal. The median solitary and multifocal PSM length were 1.5 (1.0-2.3) mm and 6.0 (2.0-7.5) mm, respectively. The predominant location of solitary PSM was the prostate apex (46.2%, 12/37), followed by the bladder neck (38.5% 10/37) and then the posterolateral surface of the prostate (15.4%, 4/37). The rate of PSM in non-NSS, unilateral NSS and bilateral NSS patients was 18.8% (n=12), 12.2% (n=12) and 8.7% (n=13), Table 2. The PSM status was not correlated with the NSS status, Supplementary Table 3.
5. BCI and BGTI Outcomes
The benign capsular incision (BCI) and benign glandular tissue incision (BGTI) outcomes, according to the NSS status were summarized in Table 3. BCI has been observed in five patients (1.6%). The BCI was always located in the posterolateral surface of the prostate. The median BCI length was 2.0 (1.0-2.0) mm. The rates of BCI according to the NSS status were 1.6% (1/64) in non-NSS patients, 2% (2/98) in ipsilateral side of unilateral NSS patients and 1.3% (2/150) in bilateral NSS patients.
The total BGTI rate was 9.0% (28/312). Twenty-seven (96.4%) BGTI were unique and 1 (3.6%) BGTI was multiple. The predominant location of BGTI was bladder neck (58.6%). The medians solitary and multiple BGTI lengths were: 2.0 (1.0-3.0) mm and 8.5 (8.5-8.5) mm, respectively. The rates of BGTI according to the NSS status were 7.8% (5/64) in non-NSS patients, 4.1% (4/98) in unilateral NSS patients and 12.7% (19/150) in bilateral NSS patients.
Discussion
Currently, the retropubic approach is still used in Europe and represents for about 25% of RP in France, in 2015 [3 14]. Our results highlighted that retropubic radical prostatectomy performed as previously described by Barré C. is reproductible and ensures optimal surgical results, in a predominantly population of intermediate-risk prostate cancer (64.1%) and high-risk prostate cancer (21.5%).
Indeed, we reported the lowest pT2 PSM rate of the literature (0%) and pT3 PSM rate of 23.4% with a positive concordance between PSM and extracapsular extension of 84.6%. The pT2 PSM and PT3 PSM rates published in a recent randomized controlled trial were: 2% versus 3% in RRP and RALP group, for pT2 PSM rates, respectively, and 8% versus 11% in RRP and RALP, group, for PT3 PSM rates, respectively [4]. In 2016, Carlsson et al [15], in the LAPPRO trial, reported a pT2 PSM and pT3 PSM rates of 13.9% and 44.4%.
Furthermore, we evaluated all inadvertent incision into benign prostate tissue that might led to a PSM if tumor had been present. We report an overall incision rate into benign prostate tissue of 10.6% which is lower than the 11.4% reported by Barré et al [9]. In this study, we have distinguished not only between malignant (PSMs) and benign intraprostatic incisions, but also between benign incisions according to location in relation to the histological boundaries of the prostate (BCI and BGTI). This differentiation of benign intraprostatic incisions, which is systematically carried out by our uropathologist, allows to evaluate the quality of surgical treatment. Indeed, a small solitary BCI are inadvertent incisions of the external layer before recover the correct dissection plane. In contrast, multiple BCI are due to the development of an intracapsular dissection plane. Intracapsular plane dissection should be considered as a serious error in case of high-risk localized prostate cancer.
We reported a low median estimated blood loss (250 ml). Indeed, Yaxley et al [4] reported a mean estimated blood loss of 1338 ml in their RRP group and 444 ml in their RALP group. A target-controlled intravenous general anesthesia with cerebral function monitoring using bi-spectral index (BIS) was performed in order to achieve controlled hypotension, limited but stabilized to 65-70 mmHg mean blood pressure. This limited controlled hypotension reduces blood loss, especially during apex dissection and nerve sparing. Furthermore, we performed a ligation of the dorsal vein, according to the technique described by Barré C. et al [7], using instruments specially designed for RRP (B. Braun Aesculap, Tuttlingen, Germany), which allows to control blood loss.
Our median length of hospital stay was 4 days, lower than length of hospital stay reported by Haese et al [16], in their recent comparative study of RRP and RALP (7.6 days versus 8.0 days in RRP and RALP groups respectively, p<0.001). The establishment of an enhanced recovery after surgery (ERAS) program, from 2015, has reduced the length of hospital stay while providing low post-operative major complications rate. Thus, a multimodal analgesia and surgical site infiltration with local anesthetics was performed. An early mobilization was also systematically carried out. Patients went home with the urethral catheter and the catheter was removed by a home care nurse one week post-operatively.
Recently, several studies have reported the benefit or similar outcomes of RRP compared to RALP. Thus, Kretschmer et al [17] reported a better general health-related quality of life outcomes for RRP compared to RALP at 3 months postoperatively. In their comparative study of RRP and RALP, Haese et al [16] reported a higher 1-week continence rates after RRP and lower operative time.
In our academic Department, above 220 RP are performed annually with 70% of RALP. RALP are performed by three experienced surgeons. Our academic Department participates in the training on radical prostatectomy for young urologists from France and others countries. Training in the technique of RRP is essential because the persistent high cost of the Da Vinci ® robot (Intuitive Surgical Company, Sunnyvale, CA, USA) and disposable instrument prevented its adoption in all urological centers of western countries and in countries with limited health-care budgets [3]. Thus, in 2020, surgical robots remain scarce in Africa, Asia-Pacific and Eastern Europe. However, the monopoly situation of the Intuitive Surgical Company is likely to change, in the coming years, with the addition of new robotic machine manufacturers.
The retropubic radical prostatectomy technique, developed by Barré C [7] requires only instruments specially designed for this surgical procedure (B. Braun Aesculap, Tuttlingen, Germany), and can be carried out in any urological center.
Limitations of the present study were the omission of functional variables (IIEF5 and USP scores) but the analysis of the data will be carried out in the near future. However, to our knowledge, we report optimal surgical outcomes on a contemporary series of RRP performed by one experienced surgeon on RRP technique developed by Barré C.
Conclusion
Retropubic radical prostatectomy performed as previously described by Barré C., is reproductible and ensures optimal surgical results, in a predominantly population of intermediate-risk prostate cancer and high-risk prostate cancer. Despite the rapid development of RALP, RRP remains an optimal option for patients with localized prostate cancer.
Authors contribution
Thomas Prudhomme: Protocol development, Data Collection, Data Analysis, Manuscript writing; Mathieu Roumiguié: Protocol development, Data Collection, Data Analysis, Manuscript writing; Sarah Péricart: Data Collection, Data Analysis; Nicolas Doumerc: Data Collection, Data Analysis; Marine Lesourd: Data Collection, Data Analysis; Pierre Marie Patard: Data Collection, Data Analysis; Xavier Gamé: Data Collection, Data Analysis;
Michel Soulié: Protocol development, Data Collection, Data Analysis, Manuscript writing and editing; Matthieu Thoulouzan: Protocol development, Data Collection, Data Analysis, Manuscript writing and editing
Conflict of interest
The authors declare that they have no conflict of interest.
References
- GLOBOCAN 2018, Cancer incidence and mortality wolrdwide: IARC Cancer Base. https://gco.iarc.fr/today/home
- Binder J, Kramer W. Robotically-assisted laparoscopic radical prostatectomy. BJU International 87 (2001): 408-410.
- Costello AJ. Considering the role of radical prostatectomy in 21st century prostate cancer care. Nature Reviews. Urology 17 (2020): 177-188.
- Yaxley JW, Coughlin GD, Chambers SK, et al. Robot-assisted laparoscopic prostatectomy versus open radical retropubic prostatectomy: early outcomes from a randomised controlled phase 3 study. The Lancet 388 (2016): 1057-1066.
- Hudson ML, Bahnson RR, Catalona WJ. Clinical use of prostate specific antigen in patients with prostate cancer. The Journal of Urology 142 (1989): 1011-1017.
- Menon M, Tewari A, Baize B, et al. Prospective comparison of radical retropubic prostatectomy and robot-assisted anatomic prostatectomy: the Vattikuti Urology Institute experience. Urology 60 (2002): 864-868.
- Barre C. Open radical retropubic prostatectomy. Eur Urol 52 (2007): 71-80.
- Walsh PC: Anatomic radical retropubic prostatectomy. In Campbell’s Urology, Update 21. Edited by Walsh PC, Retik AB, Stamey TA, et al.: Philadelphia: WB Saunders (1997): 2565-2588.
- Barré C, Thoulouzan M, Aillet G, et al. Assessing the extirpative quality of a radical prostatectomy technique: categorisation and mapping of technical errors. BJU International 114 (2014): 522-531.
- Schned AR, Ernstoff MS, Wheeler KJ, et al. Prostate cancer volume. Am J Surg Pathol 21 (1997): 1392-1393.
- McNeal JE. Prostate. In: Mills SE, ed. Histology for the pathologist, 3rd ed. Philadelphia: Lippincott Williams & Wilkins (2007). Chapter 36: p 923–963.
- Epstein JI, Cubilla AL, Humphrey PA. Tumors of the Prostate Gland, Seminal Vesicles, Penis, and Scrotum. AFIP Atlas of Tumor Pathology Series 4.Washington, DC: ARP Press (2001): 1–21
- Clavien PA, Barkun J, De Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Annals of Surgery 250 (2009): 187-196.
- Salomon L, Rozet F, Soulie M. Surgery of prostate cancer: Technical principles and perioperative complications. Progres en urologie: Journal de l'Association Francaise d'Urologie et de la Societe Francaise d'Urologie 25 (2015): 966.
- Carlsson S, Jäderling F, Wallerstedt A, et al. Oncological and functional outcomes 1 year after radical prostatectomy for very-low-risk prostate cancer: results from the prospective LAPPRO trial. BJU International 118 (2016): 205-212.
- Haese A, Knipper S, Isbarn H, et al. A comparative study of robot-assisted and open radical prostatectomy in 10 790 men treated by highly trained surgeons for both procedures. BJU International 123 (2019): 1031-1040.
- Kretschmer A, Bischoff R, Chaloupka M, et al. Health-related quality of life after open and robot-assisted radical prostatectomy in low-and intermediate-risk prostate cancer patients: a propensity score-matched analysis. World Journal of Urology (2020): 1-9.
Supplementary Information
|
Age (years) Median (IQR) |
65.2 (60.5-68.7) |
|
Preoperative PSA (ng/ml) Median (IQR) |
8.0 (6.0-11.0) |
|
Preoperative clinical stage (n, %) T1c T2a T2b T2c T3 |
105 (33.7%) 59 (18.9%) 103 (33.0%) 27 (8.7%) 18 (5.8%) |
|
Preoperative biopsy ISUP and Gleason grade (n, %) ISUP 1 / Gleason 6 (3+3) ISUP 2 / Gleason 7 (3+4) ISUP 3 / Gleason 7 (4+3) ISUP 4 /Gleason 8 (4+4) ISUP 5 / > Gleason 8 |
69 (22.1%) 117 (37.5%) 90 (28.8%) 31 (9.9%) 5 (1.6%) |
|
D’Amico risk classification (n, %) Low-risk Intermediate-risk High-risk |
45 (14.4%) 200 (64.1%) 67 (21.5%) |
|
Estimated blood loss (ml) Median (IQR) |
250.0 (150.0-400.0) |
|
Operative time (minutes) Median (IQR) |
160.0 (150.0-180.0) |
|
Length of hospital stay (days) Median (IQR) |
4.0 (3.0-5.0) |
|
Post-operative complications (Clavien Dindo classification) Minor (I-II) (n, %) Major (≥III) (n, %) IIIa -Percutaneous drainage of symptomatic pelvic lymphocele IIIb -Surgical re-exploration due to hematoma -Marsupialization of lymphocele -Acute lower limb ischemia V -Death due to postoperative pulmonary embolism |
18 (5.8%) 30 (9.6%) 16 (5.1%) 5 (1.6%) 6 (1.9%) 2 (0.6) 1 (0.3) |
Table 1: Patients characteristics, intra- and post-operative outcomes (Quantitative variables were presented as median and Interquartile range while qualitative variable as number (n) and proportion (%)).
|
Positive Surgical Margin (PSM) |
Overall population (n= 312) |
Non-NSS (n= 64) |
Unilateral NSS (n= 98) |
Bilateral NSS (n= 150) |
|
PSM rate (n, % of NSS status) Total PSM Solitary PSM Multifocal PSM |
37 (11.9%) 26 (8.3%) 11 (3.5%) |
12 (18.8%) 6 (9.4%) 6 (9.4%) |
12 (12.2%) 9 (9.2%) 3 (3.1%) |
13 (8.7%) 11 (7.3%) 2 (1.3%) |
|
Location of solitary PSM (n, % of solitary PSM) Apex Posterolateral surface* Bladder neck |
12 (46.2%) 4 (15.4%) 10 (38.5%) |
2 (33.3%) 1 (16.7%) 3 (50%) |
4 (44.4%) 2 (22.2%) 3 (33.3%) |
6 (54.5%) 1 (9.1%) 4 (36.4%) |
|
PSM length (millimeters) Solitary PSM Median (IQR) Multifocal PSM Median (IQR) |
1.5 (1.0-2.3) 6.0 (2.0-7.5) |
1.0 (0.8-4) 4.0 (1.4-9.9) |
1.0 (1.0-2.0) 5.0 (3.0-7.0) |
2.0 (1.0-3.3) 15.5 (6.0-25.0) |
|
pT3 pathological stage PSM rate (n, % of pT3 pathological stage) Total PSM Solitary PSM Multifocal PSM |
37 (23.4%) 26 (16.5%) 11 (7.0%) |
12 (29.3%) 6 (14.6%) 6 (14.6%) |
12 (18.8%) 9 (14.1%) 3 (4.7%) |
13 (24.5%) 11 (20.8%) 2 (3.8%) |
|
Positive location concordance between PSM and extracapsular extension (n, % of PSM rate) Total PSM Solitary PSM Multifocal PSM |
28 (75.7%) 22 (84.6%) 6 (54.5%) |
8 (66.7%) 5 (83.3% 3 (50%) |
11 (91.7%) 8 (88.9%) 3 (100%) |
9 (69.2%) 9 (81.8%) 0 (0%) |
*Posterolateral surface: posterior, posterolateral, and lateral aspects of the mid-prostate and posterior prostate base
Table 2: Positive surgical margin (PSM) outcomes in overall population and pT3 stage disease according to the NSS status
|
Benign Capsular Incision (BCI) |
Overall population (n= 312) |
Non-NSS (n= 64) |
Unilateral NSS (n= 98) |
Bilateral NSS (n= 150) |
|
BCI rate (n, % of NSS status) Total BCI Solitary BCI |
5 (1.6%) 5 (1.6%) |
1 (1.6%) 1 (1.6%) |
2 (2.0%) 2 (2.0%) |
2 (1.3%) 2 (1.3%) |
|
Location of BCI (n, % of BCI) Posterolateral surface* |
5 (100%) |
1 (100%) |
2 (100%) |
2 (100%) |
|
BCI length (millimeters) Median (IQR) |
2.0 (1.0-2.0) |
2.0 (2.0-2.0) |
1.5 (1.0-2.0) |
1.5 (1.0-2.0) |
|
Benign Glandular Tissue Incision (BGTI) |
Overall population (n= 312) |
Non-NSS (n= 64) |
Unilateral NSS (n= 98) |
Bilateral NSS (n= 150) |
|
BGTI rate (n, % of NSS status) Total BGTI Solitary BGTI Multifocal BGTI |
28 (9.0%) 27 (8.7%) 1 (0.3%) |
5 (7.8%) 5 (7.8%) 0 (0%) |
4 (4.1%) 3 (3.1%) 1 (1.0%) |
19 (12.7%) 19 (12.7%) 0 (0%) |
|
Location of BGTI (n, % of BGTI) Apex Bladder neck |
12 (41.4%) 17 (58.6%) |
3 (60.0%) 2 (40.0%) |
2 (50.0%) 3 (75.0%) |
7 (36.8%) 12 (63.2%) |
|
BGTI length (millimeters) Solitary Median (IQR) Multiple Median (IQR) |
2.0 (1.0-3.0) 8.5 (8.5-8.5) |
1.0 (1.0-2.8) / |
1.0 (0.3-2.0) 8.5 (8.5-8.5) |
2.0 (1.0-3.3) / |
*Posterolateral surface: posterior, posterolateral, and lateral aspects of the mid-prostate and posterior prostate base
Table 3: Benign capsular incision (BCI) and benign glandular tissue incision (BGTI) outcomes, according to the NSS status
|
Pathological stage (n, %) pT1c pT2 pT2a pT2b pT2c
pT3 pT3a pT3b |
1 (0.3%) 153 (49.0%) 25 (8.0%) 6 (1.9%) 122 (39.1%) 158 (50.6%) 105 (33.7%) 53 (17.0%) |
|
Pathological ISUP and Gleason grade (n, %) ISUP 1 / Gleason 6 (3+3) ISUP 2 / Gleason 7 (3+4) ISUP 3 / Gleason 7 (4+3) ISUP 4 /Gleason 8 (4+4) ISUP 5 / > Gleason 8 |
28 (9.0%) 154 (49.4%) 111 (35.6%) 8 (2.6%) 11 (3.5%) |
|
Prostate volume (cm3) Median (IQR) |
45.0 (37.0-60.0) |
|
Tumor volume (%) Median (IQR) |
12.0 (7.0-25.0) |
|
Lymph node dissection (n, %) Total pN0 pN1
Number of lymph node removed Median (IQR) |
272 (87.2%) 234 (86.0%) 38 (14.0%) 19.0 (14.0-24.0) |
|
BCR (Median follow-up: 1.0 year) (n, %) |
29 (9.3%) |
Supplementary Table 1: Pathological and oncological outcomes (Quantitative variables were presented as median and Interquartile range while qualitative variable as number (n) and proportion (%)).
|
Variables |
Pathological stage |
Total |
||
|
pT2 |
pT3 |
|||
|
PSM status |
PSM + |
0 (0%) |
37 (23.4%) |
37 (11.9%) |
|
PSM - |
153 (100%) |
121 (76.6%) |
274 (88.1%) |
|
|
Total |
153 |
158 |
p<0.0001 |
|
Supplementary Table 2: Contingency table of PSM status according to the pathological stage (Fisher’s exact test).
|
Variables |
NSS status |
Total |
|||
|
Non-NSS |
Unilateral NSS |
Bilateral NSS |
|||
|
PSM status |
PSM + |
12 (18.8%) |
12 (12.2%) |
13 (8.7%) |
37 (11.9%) |
|
PSM - |
52 (81.3%) |
86 (87.8%) |
137 (91.3%) |
275 (88.1%) |
|
|
Total |
64 |
98 |
150 |
p=0.1 |
|
Supplementary Table 3: Contingency table of PSM status according to the NSS status (Chi-square test).
|
At risk |
0 |
1 year |
2 years |
3 years |
4 years |
5 years |
|
Overall population |
306 |
152 |
45 |
14 |
9 |
7 |
Supplementary Figure 1: Kaplan-Meier curves showing BCR-free survival after RRP in overall population
|
At risk |
0 |
1 year |
2 years |
3 years |
4 years |
5 years |
|
pT2 |
151 |
78 |
23 |
8 |
5 |
4 |
|
pT3 |
154 |
70 |
23 |
7 |
5 |
5 |
|
pT3, PSM- |
119 |
54 |
19 |
5 |
4 |
4 |
|
pT3, PSM+ |
35 |
17 |
5 |
3 |
2 |
2 |
Supplementary Figure 2: BCR-free survival by pathological stage and surgical margin (SM) status (SM+ versus SM-)


 Impact Factor: * 5.8
Impact Factor: * 5.8 Acceptance Rate: 71.20%
Acceptance Rate: 71.20%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 