Prophylactic Oropharyngeal Surfactant Administration before Cord Clamping in Very Low Birth Weight Newborns
Valentina Silveira1*, María Elena Machado1, Marianela Rodríguez-Rey1, Fernanda Blasina1, José Luis Díaz-Rossello1
1Departamento de Neonatología Hospital de Clínicas, Facultad de Medicina, Universidad de la República (Udelar), Montevideo, Uruguay
*Corresponding author: Valentina Silveira, Departamento de Neonatología Hospital de Clínicas, Facultad de Medicina, Universidad de la República (Udelar), Montevideo, Uruguay,
Received: 22 September 2022; Accepted: 30 September 2022; Published: 10 October 2022
Article Information
Citation: Valentina Silveira, María Elena Machado, Marianela Rodríguez-Rey, Fernanda Blasina, José Luis Díaz-Rossello. Prophylactic Oropharyngeal Surfactant Administration before Cord Clamping in Very Low Birth Weight Newborns. Archives of Clinical and Biomedical Research 6 (2022): 906-911.
View / Download Pdf Share at FacebookAbstract
Background: In the era of minimally invasive ventilatory strategies search, the prophylactic administration of surfactant with simple techniques, which do not require special skills, represents an innovative tool that, combined with delayed cord clamping, could reduce morbidity and mortality in premature babies.
Methods: In this descriptive restrospective study, 27 cases of premature newborns who received prophylactic surfactant through the oropharyngeal route before the first breath and with intact circulation of the umbilical cord are reported.
Results: After receiving the oropharyngeal surfactant 9 of 27 patients required endotracheal intubation in the first 24 hours. Only 2 of the 27 babies received 3 doses of surfactant by endotracheal way which corresponds to a total dose of 300 mg/kg. No major complications related to the oropharyngeal surfactant administration technique were registered. Seven patients did not require any other dose of surfactant after the administration of oropharyngeal surfactant. Bronchodysplasia was diagnosed in 2 of the 27 patients and the intraventricular hemorrhage was present in 3 patients. No patients with retinopathy of prematurity (ROP) were registered.
Conclusions: The experience described with this surfactant administration technique was conducted in a setting prepared for the care of the neonate during the third stage of labor. After the successful de-implementation of immediate cord clamping in all deliveries, including preterm ones regardless of the mode of delivery, oropharyngeal surfactant administration is a non-invasive, safe, feasible, and reproducible technique that can be performed in low-complexity settings, with the potential to improve premature outcomes.
Keywords
<p>Cord Clamping; Neonatal Care; Preterm; Surfactant</p>
Article Details
1. Introduction
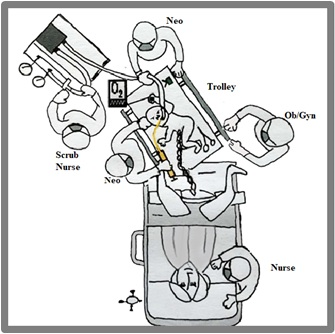
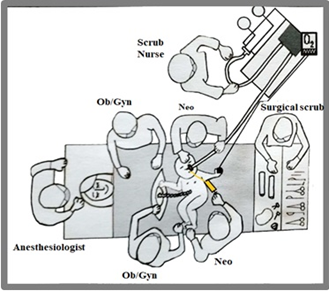
The classical endotracheal surfactant therapy in preterm infant respiratory distress syndrome (RDS) secondary to surfactant deficiency improves lung compliance and significantly reduces morbidity and mortality [1,2,3]. Although the benefits of surfactant are well demonstrated, controversies remain about the best strategy and timing of administration. Different studies reported that early surfactant administration is more effective if performed within the first 2 hours of life in ventilated preterm infants with RDS, and contributes to the reduction of mechanical ventilation and bronchodysplasia [4,5]. Preterm infant management in the first minutes of life carries a major impact on neonatal morbidity and mortality. Delayed cord clamping (DCC), maintenance of body temperature and ensuring the initiation and maintenance of spontaneous respiration with minimally invasive ventilatory support strategies have been successfully implemented to improve outcomes. All these strategies constitute a complex care bundle that facilitates physiological adaptation to extrauterine life that have been shown to reduce the short, medium and long term complications of premature infants [6,7,8]. Since routine endotracheal intubation of very low birth weight (VLBW) preterm infants has been abandoned as regular practice, early surfactant administration in non-intubated spontaneously breathing patients has led to intensive research and has become the standard care for many patients. Pioneering experiences with administering surfactant to spontaneously breathing newborns were readily adopted and are now part of our routine procedures. Recently, there has been a growing interest in exploring even less invasive surfactant administration techniques. For instance, oropharyngeal administration before the first inspiration represents a faster, easier and potentially more physiological administration route. Minimally invasive surfactant therapy combined with spontaneous breathing maintenance has been published internationally as Less Invasive Surfactant Administration (LISA) [9, 10, 11,12, 13]. Advances in minimally invasive surfactant administration techniques and studies on early surfactant administration have shown that the oropharyngeal surfactant application prior to the onset of the first inspiration reduces intubation requirements, improves oxygenation, and favors cardiovascular transition [14,15]; these procedures typically initiate post-cord clamping in VLBW newborns. In line with these observations, recent studies performed in experimental preclinical models report that the presence of exogenous surfactant in the lung at birth and before the first insufflation improves the spatial distribution of ventilation and reduces lung injury [16]. Since 2016, a European randomized controlled trial has been developed in newborns less than 29 weeks' gestation, given oropharyngeal surfactant in the first lung insufflations. These trials will determine whether the administration of oropharyngeal surfactant at birth reduces endotracheal intubation [17]. The availability of surfactant in the alveoli in early gestational ages is benefited by the induction generated by steroids, but some investigations have also shown that the administration of surfactant in the first inspiration, before the air enters the lung, can facilitate its distribution by creating a surfactant/water interface. The air/surfactant interface is established with the first breath [16]. Initial reports show no adverse events related to this form and timing of surfactant administration and open the possibility of exploring a minimally invasive intervention. This strategy is much more promising in settings already prepared to care for the newborn during the third stage of labor, before placental abruption and with the umbilical-placental circulation intact. In this ideal situation, in the time before initial inspiration the newborn will benefit from a fetal-like state, if it includes a troley to hold the baby at the level of the maternal perineum or on the mother's thighs in the Caesarean section (figure 1 and 2). Respiratory support pressure can also be done immediately after oropharyngeal surfactant administration. In accordance with the existing evidence, our health team has developed strategies for the initial care of premature infants that included minimally invasive management, in premature infants up to 32 weeks of gestational age, DCC, as well as the initiation of positive pressure prior to cord clamping [18]. Administration of exogenous surfactant to the oropharynx before the beginning of the first inspirations is a simple maneuver while the umbilicus-placental circulation still persists. We report 27 cases to assess the feasibility of this procedure by organizing the delivery room care and detect adverse events.
2. Methods
After approval by the Bioethics committee of the Hospital de Clínicas de la Universidad de la República, we collected the required information about the 27 cases. A descriptive retrospective single-center study was carried out. Patients born between October 2017 and October 2019 were enrolled in this study. The inclusion criteria were infants born by caesarean section or vaginal delivery with a gestational age less than or equal to 32 weeks and treated with nCPAP prior to DCC. Neonates with significant congenital abnormalities were excluded.
2.1 Intervention
Immediately after birth, as shown in figure 1, the newborn was placed on the maternal thighs in a plastic bag, maintaining the feto-placental circulation by one operator from the neonatal team. A second operator delivered a 4-8 ml bolus of Survanta® (100 mg/k) to the oropharynx through a silastic feeding tube through the mouth into the oropharynx, before the patient begins spontaneous breathing. In the meantime the first operator positioned the nose piece to deliver continuous positive airway pressure. Once the nostrils were placed at 5 cm H2O, positive pressure was applied at the end of expiration (PEEP) with 60% oxygen. Babies born vaginally were placed in the plastic bag and placed on the trolley platform, which was designed for the reception and stabilization of the newborn (Figure 2). The use of this platform allows the stabilization of the newborn while maintaining the placental circulation. After vaginal delivery the same respiratory stabilization protocol was used and the administration of oropharyngeal surfactant plus PEEP was the same as that described in the case of c-section. A member of the research team videotaped the birth until the third stage of labor was completed. From these recordings the temporal sequence of the maneuvers carried out was determined. An example is shown in the following video https://youtu.be/3MY-T9BxCFw
2.2 Data Collected and Statistical Analysis
Our main objective was to determine the feasibility of administering oropharyngeal surfactant at birth before the onset of the first inspiration and clamping of the umbilical cord. We collected and analyzed patient characteristics and short-term outcomes, including endotracheal intubation rate for respiratory failure within 24 hours of birth, endotracheal surfactant use, intraventricular hemorrhage, bronchodysplasia, and death. Each dose of endotracheal surfactant administered was 100 mg/kg (Survanta®). Descriptive statistics were calculated for the variables of interest.
3. Results
This study included 27 neonates born less than or equal to 32 weeks whose characteristics at birth and outcomes are presented in Table 1. The gestational age range of the patients was 26 to 32 weeks and the birth weight 711 g to 2046 g. In some births, the time of the maneuvers was recorded and the oropharyngeal surfactant was administered in the first four seconds of life (data not shown). A higher number of cesarean sections was evidenced with respect to vaginal delivery. The relative frequency (RF) of complete administration of prenatal corticosteroids was 0.85, and all the patients received at least one dose prior to birth. In most of the patients DCC was performed and these patients received nCPAP prior to the umbilical cord clamping. The results showed that after receiving the oropharyngeal surfactant 9 of 27 (RF 0.33) patients required endotracheal intubation in the first 24 hours. Only 2 of the 27 babies received 3 doses of endotracheal surfactant which correspond to a total dose of 300 mg/kg. In those patients who required a supplementary endotracheal dose of surfactant and did not require intubation, the surfactant was administered using the LISA technique. No major complications related to the oropharyngeal surfactant administration technique were registered. Seven patients did not require any other dose of surfactant after the administration of oropharyngeal surfactant (RF 0.25). Bronchodysplasia was diagnosed in 2 of the 27 patients (RF 0.03) and the intraventricular hemorrhage was present in 3 patients (RF 0.1). No patients with retinopathy of prematurity (ROP) were registered. Given that the use of inotropes is widely extended in critically ill and unstable patients, we wanted to know how many of our patients required their use in the first 24 hours of life, and only one patient received this kind of drugs in the first 24 hours. The administration of oropharyngeal surfactant proved to be a reproducible technique and there were no complications derived from its administration. Temperature at admission to the NICU was 35.9°C ∓ 0.5°C. This value is due to the lack of adequate thermoprotection measures, although plastic bags as well as heat chemical mattress were used, difficulty in controlling temperature of the environment and CPAP gas temperature evoked this result. The deaths recorded in this group of patients were 3 of 27 patients. We emphasize that the deceased were patients with a gestational age less than or equal to 26 weeks.
Table 1: Characteristics of the patients.
4. Discussion
Many preterm infants develop RDS and require respiratory support and exogenous surfactant. This study shows the feasibility of administering a first bolus of surfactant by the oropharyngeal route just at delivery while maintaining the umbilical placental circulation, using a non-invasive technique. With the paradigm shift in the initial care of premature infants, especially those more inmature babies, which implies accompanying the transition to extrauterine life guided by physiological events, various minimally invasive surfactant administration techniques have been reported [5,17 ,19,20, 21]. These techniques have sought to reduce morbidity and mortality in this population. Minimally invasive care measures in stabilization and care during the transition to extrauterine life are part of the regular practices in our service. These measures include DCC, nCPAP in the delivery room to ensure the initiation and maintenance of spontaneous respiration, and the use of minimally invasive techniques for the administration of surfactant (LISA). This bundle for initial newborn care responds to the guidelines of modern neonatology [22], which has led to a notable decrease in the invasiveness of treatments in recent years [23]. The administration of surfactant through the oropharyngeal route in preterm infants with a high risk of RDS has reduced the need for rescue intubation in those younger than 25 weeks, as well as the incidence of some of the complications derived from mechanical ventilation [12, 14,15]. One of the main objectives of the initial assistance strategies for premature infants is to avoid endotracheal intubation immediately after the delivery, since this determines better survival with less morbidity secondary to mechanical ventilator assistance [24, 25]. Our results show that the administration of surfactant into the oropharynx, just before the first spontaneous inspiration with the placental circulation ongoing, can be effective, feasible, safe and simple technique. This intervention showed a low rate of endotracheal intubation in the first 24 hours of birth and did not produce undesirable side effects. The administration of oropharyngeal surfactant is easy and avoids the adverse effects associated with endotracheal intubation. It could be done by non-experts because of the easy way to administer the surfactant, for this reason could be made in low resource or low risk maternities. We consider that hypothermia upon admission to NICU is not due to insufficient thermoprotection measures. Based on this result, our unit has incorporated strategies to protect the temperature of neonates. These strategies included polyethylene bags, chemical mattress, humidification and heating of gasses. Since many preterm deliveries are associated with the existence of an inflammatory environment and the inflammatory response may be the cause of preterm delivery, the earlier an exogenous surfactant is administered, the better the lung may be protected, as reported by evidence from animal research. Studies in lambs and premature rabbits showed that placing surfactant in the posterior pharynx before the first inspiration allows aspiration of surfactant at the start of ventilation and improves aeration of the lungs [26]. The administration of early surfactant and even prior to positive pressure ventilation could determine an anti-inflammatory effect shown by some studies carried out in newborn animals [27]. An ongoing trial is currently examining the efficacy of pharyngeal surfactant administration to premature infants less than 29 weeks gestation; the POPART study is an unblinded multicentre study that compares administration of poractant alfa in addition to CPAP versus CPAP alone. Interestingly, the amount of surfactant administered is decided upon gestational age and not on weight, which results in an approximated dose ranging from 150 to 267 mg/kg. The primary outcome is the incidence of respiratory failure in the first five days of life, but there are, to date, no results published [17]. The progress towards an improvement in the initial preterm care may require combining non-invasive and prophylactic actions that are guided by the transition physiology of this group of patients. We present this series of cases where, due to prematurity, spontaneous respiratory capacity is compromised and it is considered appropriate to administer surfactant when the lungs have not yet filled with air, thus avoiding the initial hemodynamic deterioration that could be an important decision to endure a critical moment during the transition, avoiding other deleterious events that trigger the onset and lead to major complications [28]. This report provides evidence about the possibility of introducing a practice that has not shown adverse effects, which involves the use of surfactant maintaining a stabilization of a preterm newborn while being transferred to an intensive care unit. The incorporation of this low complexity technique could contribute to the initial management and stabilization of preterms in developing countries as well as in low complexity centres all over the world.
Acknowledgments
We thank Dr. Jose Otero for his help in the writing of this manuscript.
Disclosure Statements
There are no conflicts of interest among the authors since there are no financial ties to products or other Institutions/Industries outside the University.
References
- Jobe AH, Ikegami M. Lung development and function in preterm infants in the surfactant treatment era. Annual Review of Physiology 62 (2000): 825-846.
- Jobe A, Ikegami M, Jacobs H, et al. Surfactant and pulmonary blood flow distributions following treatment of premature lambs with natural surfactant. The Journal of clinical investigation 73 (1984): 848-856.
- Rossello JD, Hayward PE, Martell M, et al. Hyaline membrane disease (HMD) therapy in Latin America: impact of exogenous surfactant administration on newborn survival, morbidity and use of resources. Journal of perinatal medicine 25 (1997): 280-287.
- Stevens TP, Harrington EW, Blennow M, et al. Early surfactant administration with brief ventilation vs. selective surfactant and continued mechanical ventilation for preterm infants with or at risk for respiratory distress syndrome. Cochrane Database Syst Rev (2007): CD003063.
- Abdel-Latif ME, Osborn DA. Pharyngeal instillation of surfactant before the first breath for prevention of morbidity and mortality in preterm infants at risk of respiratory distress syndrome. Cochrane Database Syst Rev (2011): CD008311.
- Knol R, Brouwer E, van den Akker T, et al. Physiological-based cord clamping in very preterm infants - Randomised controlled trial on effectiveness of stabilisation. Resuscitation 147 (2020): 26-33.
- Rabe H, Reynolds G, Diaz-Rossello J. A systematic review and meta-analysis of a brief delay in clamping the umbilical cord of preterm infants. Neonatology 93 (2008): 138-144.
- Martherus T, Oberthuer A, Dekker J, et al. Supporting breathing of preterm infants at birth: a narrative review. Arch Dis Child Fetal Neonatal Ed 104 (2019): F102-F107.
- Dargaville PA. Newer Strategies for Surfactant Delivery. In: Eduardo Bancalari, editors. The Newborn Lung 2 nd ed. Elsevier (2019): 221-238.
- Dargaville PA. Newer Strategies for Surfactant Delivery. In: Eduardo Bancalari, editors. The Newborn Lung.2nd ed. Elsevier (2019): 221-238.
- Kribs A. Minimally Invasive Surfactant Therapy and Noninvasive Respiratory Support. Clin Perinatol 43 (2016): 755-771.
- Herting E, Härtel C, Göpel W. Less invasive surfactant administration: best practices and unanswered questions. Curr Opin Pediatr 32 (2020): 228-234.
- Vento M, Bohlin K, Herting E, et al. Surfactant Administration via Thin Catheter: A Practical Guide. Neonatology 116 (2019): 211-226.
- Lamberska T, Settelmayerova E, Smisek J, et al. Oropharyngeal surfactant can improve initial stabilisation and reduce rescue intubation in infants born below 25 weeks of gestation. Acta Paediatrica 107 (2018): 73-78.
- Kattwinkel J, Robinson M, Bloom BT, et al. Technique for intrapartum administration of surfactant without requirement for an endotracheal tube. Journal of Perinatology 24 (2004): 360.
- Tingay DG, Wallace MJ, Bhatia R, et al. Surfactant before the first inflation at birth improves spatial distribution of ventilation and reduces lung injury in preterm lambs. J Appl Physiol 116 (2014): 251-258.
- Murphy MC, Galligan M, Molloy B, et al. Study protocol for the POPART study-Prophylactic Oropharyngeal surfactant for Preterm infants: A Randomised Trial. BMJ Open 10 (2020): e035994.
- Devera, Andrea. Uso de presión positiva durante la transición neonatal. Monografía de posgrado de Neonatología. Departamento de Neonatología Hospital de Clínicas Dr. Manuel Quintela. Escuela de graduados, Facultad de Medicina. Universidad de la República. Febrero (2018) https://drive.google.com/drive/folders/1-q11kEWISALEjm8Lex4wzycWhoLuXp1L.
- Aguar M, Vento M, Dargaville PA. Minimally invasive surfactant therapy: an update. NeoReviews 15 (2014): e275-85.
- Stevens TP, Blennow M, Myers EH, et al. Early surfactant administration with brief ventilation vs. selective surfactant and continued mechanical ventilation for preterm infants. Cochrane Database Syst Rev (2007): CD003063.
- Sweet DG, Carnielli V, Greisen G, et al. European Consensus Guidelines on the Management of Respiratory Distress Syndrome - 2019 Update. Neonatology 115 (2019): 432-450.
- Stoll BJ, Hansen NI, Bell EF, et al. Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Neonatal outcomes of extremely preterm infants from the NICHD Neonatal Research Network. Pediatrics 126 (2010): 443-456.
- Klebermass-Schrehof K, Wald M, Schwindt J, et al. Less invasive surfactant administration in extremely preterm infants: impact on mortality and morbidity. Neonatology 103 (2013): 252-258.
- Kribs A. Minimally invasive surfactant therapy and noninvasive respiratory support. Clinics in perinatology 43 (2016): 755-771.
- Abdel-Latif ME, Davis PG, Wheeler KI, et al. Surfactant therapy via thin catheter in preterm infants with or at risk of respiratory distress syndrome. Cochrane Database Syst Rev 5 (2021): CD011672.
- Enhörning G, Robertson B. Lung expansion in the premature rabbit fetus after tracheal deposition of surfactant. Pediatrics 50 (1972): 58-66.
- Nold JL, Meyers PA, Worwa CT, et al. Decreased lung injury after surfactant in piglets treated with continuous positive airway pressure or synchronized intermittent mandatory ventilation. Neonatology 92 (2007): 19-25.
- Bhatt S, Alison BJ, Wallace EM, et al. Delaying cord clamping until ventilation onset improves cardiovascular function at birth in preterm lambs. The Journal of physiology 591 (2013): 2113-2126.


 Impact Factor: * 5.8
Impact Factor: * 5.8 Acceptance Rate: 71.20%
Acceptance Rate: 71.20%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 