Diagnostic Value of Procalcitonin in Patients with Heat Stroke
Xuan Song1, Xinyan Liu1, Huairong Wang2, Xiuyan Guo2, Maopeng Yang1, Daqiang Yang1, Yahu Bai3, Nana Zhang1, Chunting Wang4*
1Department of Intensive Care Unit, DongE Hospital Affiliated to Shandong First Medical University, Liaocheng, China.
2Education Department, DongE Hospital Affiliated to Shandong First Medical University, Liaocheng, China.
3Emergency Department, DongE Hospital Affiliated to Shandong First Medical University, Liaocheng, China.
4Department of Intensive Care Unit, Shandong Provincial Hospital Affiliated to Shandong First Medical University, Shandong, China.
*Corresponding Author: Chunting Wang, Department of Intensive Care Unit, Shandong Provincial Hospital Affiliated to Shandong First Medical University, 324 Jingwu Road, Jinan, Shandong Province, China, 250021
Received: 13 January 2020; Accepted: 23 January 2020; Published: 03 February 2020
Article Information
Citation: Xuan Song, Xinyan Liu, Huairong Wang, Xiuyan Guo, Maopeng Yang, Daqiang Yang, Yahu Bai, Nana Zhang, Chunting Wang. Diagnostic Value of Procalcitonin in Patients with Heat Stroke. Arch Microbiol Immunology 2020; 4 (1): 001-010.
View / Download Pdf Share at FacebookAbstract
Background: Heat stroke is a life-threatening disease, but there is currently no biomarker to accurately assess prognosis.
Objective: To study whether serum procalcitonin (PCT) is an effective biomarker for evaluating the prognosis for patients with heat stroke.
Method: Clinical data for 61 patients with heat stroke were retrospectively collected and analyzed. PCT, Acute Physiology and Chronic Health Evaluation (APACHE II) score and Multiple Organ Dysfunction Score (MODS) were calculated within the first 24 hours of admission to the ICU. The ability of PCT, APACHE II score, and MODS score to predict prognosis was assessed using a receiver operating characteristic curve (ROC) and area under the curve (AUC) analyses.
Result: There was no statistical difference in the PCT value on admission to the intensive care unit (ICU) (PCT1) between the survivors and non-survivors (3.27 [95% CI 0.15-13.55] vs 0.45 [95% CI 0.13-2.04], p=0.369). ROC curve analysis showed that AUC of APACHE II score and MODS score were 0.932 (95% CI 0.861-1.000), p<0.001 and 0.857 (95% CI 0.744- 0.970), p<0.001, respectively. The AUC of PCT1 was 0.599 (95% CI 0.353-0.846), p=0.370. The dynamic trend of PCT was also not statistically different between the survivor and non-survivor group (p=0.138).
Conclusion: PCT and its dynamic trend do not provide a good assessment of the prognosis for patients with heat stroke. An increase in PCT levels at ICU admission is not an indicator of bacterial infection, which should be confirmed by larger future studies.
Keywords
Heat Stroke; Procalcitonin; Prognosis; Biomarkers
Heat Stroke articles, Procalcitonin articles, Prognosis articles, Biomarkers articles
Article Details
1. Introduction
Heat stroke (HS) is a severe clinical syndrome caused by exposure to high temperature and high humidity, causing the body's core temperature to rise rapidly to over 40?. This causes sensations of skin burning, disturbance of consciousness (such as delirium, convulsions, and coma), and multi-organ system injuries [1]. HS is divided into classic heat stroke (CHS) and exertional heat stroke (EHS); CHS is usually caused by exposure to high temperatures, while EHS is caused by strenuous exercise [2]. The pathophysiological mechanism of HS is generally considered to be a hyperthermia syndrome, which is similar in many respects to sepsis. There is increasing evidence that endotoxin and cytokines may be involved in the pathogenesis. Progression to systemic inflammatory response leads to multiple organ dysfunction, which mainly manifests as central nervous system damage, so it is difficult to distinguish it from sepsis [3]. If HS is not treated with timely and rapid cooling, the patient's condition will rapidly deteriorate, which may cause systemic inflammatory response syndrome, multiple organ failure, disseminated intravascular coagulation (DIC), and other complications [4]. The in-hospital mortality rate is 14%-65%, and intensive care unit (ICU) mortality is >60% [5-7].
Procalcitonin (PCT) is a glycoprotein that is a precursor of calcitonin. Under normal conditions, all PCT is cleared by the body, so it is not detected in the serum of healthy individuals. In severe systemic infections, serum PCT can rise rapidly even to above 100 ng/mL [8, 9]. PCT has high sensitivity and specificity in the diagnosis of sepsis, and has been used as an important diagnostic indicator for sepsis and can predict its severity [10-12]. With the increasing research on PCT, we have reported that PCT is not only associated with sepsis, but also with systemic inflammatory responses to non-sepsis [10]. Additionally, it correlates with sepsis and non-sepsis SIRS severity [13, 14] and helps identify patients with SIRS or sepsis [15]. HS can progress to systemic inflammatory response syndrome, but there are few data used to assess disease severity, diagnose prognosis, and to distinguish between biomarkers with or without infection. At present, there are only three studies on the relationship between serum PCT and HS (including two CHS and one EHS), and it has been reported that the increase in serum PCT value is not associated with bacterial infection and that elevated PCT levels are associated with the severity of the disease, but not with mortality [16-18]. We investigated the diagnostic value of serum PCT in patients with HS, whether PCT is an effective prognostic biomarker for patients with HS, and any diagnostic markers that may be associated with infection.
2. Methods
This retrospective study included patients who were admitted to the ICU Department of DongE Hospital affiliated to Shandong First Medical University from July 1, 2016 to August 1, 2018 for either CHS or EHS. Exclusion criteria included age <18 years, irreversible conditions before ICU admission (malignant tumors, chronic renal insufficiency, etc.), and pregnancy. The study protocol was in accordance with the ethics standards and gained approval from the ethics committee (approval number: 2018-036). This was a retrospective study in which informed consent was exempted by the Ethics Committee. For our study, HS was defined as a rise in body temperature >40? due to exposure to high temperatures or strenuous exercise, accompanied by neurological abnormalities such as mental status changes, convulsions, or coma, and associated with multiple organ dysfunction. Positive cooling treatment was given after admission. The Acute Physiology and Chronic Health Evaluation (APACHE II) score and Multiple Organ Dysfunction Score (MODS) were calculated within the first 24 hours of admission to the ICU. Bacterial infection was diagnosed by two senior clinicians. Infection was defined as the growth of bacteria in blood cultures or sterile body fluids (such as urine specimens), or if there was a source of infection recorded in the medical record system (e.g., pulmonary infection is defined as new lung infiltration on the chest radiograph with acute respiratory symptoms). Finally, vital signs, comorbid disease, highest temperature at ICU admission, time required for the temperature to fall below 38.5°C, and other laboratory indicators were extracted from the electronic medical record system.
2.1 PCT Measurement
Serum PCT levels were determined by chemiluminescence using COBAS e 601 equipment (Hoffmann-La Roche, Inc, Basel, Switzerland). The reagents used in the assay were also developed by Roche, with normal values being 0-0.014 ng/ml. The serum PCT measured at ICU admission was used as the baseline value of procalcitonin and was recorded as PCT1. Serum PCT was measured again on day 2 (PCT2) and day 3 (PCT3). The trend of PCT dynamics on day 1 and day 2 were calculated using the following formula: (PCT1) – (PCT2)/( PCT1) * 100%, recorded as PCT1-2, and the same PCT change on day 1 and day 3 as PCT1-3.
2.2 Statistical Analysis
Data were analyzed using SPSS 19.0. According to the distribution of the data, statistical analysis was performed using t-test or chi-squared (X2) test. The relationship between PCT and both APACHE II and MODS scores was analyzed using bivariate correlation analysis. The ability of the PCT, APACHE II score, and MODS score to determine prognosis was assessed using a receiver operating characteristic curve (ROC) and area under the curve (AUC). AUC values were expressed as 95% confidence intervals (95% CI). p <0.05 was considered statistically significant.
3. Results
3.1 Patient clinical and demographic characteristics
The clinical and demographic characteristics of 61 patients were analyzed, including 41 cases of CHS and 20 of EHS. The overall mortality rate was 13.1% (8/61 patients). The general characteristics of patients in the survivor and non-survivor groups are shown in Table 1. APACHE II and MODS scores of non-survivors were significantly higher than that of survivors (p<0.05). There was no significant difference in the highest temperature (Tmax) between the two groups at ICU admission, but the time to fall below 38.5 °C (T1) was significantly higher in the non-survivor group (p<0.05). However, there was no significant difference in PCT1 between survivors and non-survivors. A total of 34 patients had a bacterial infection and received antibiotics, including 31 cases of pulmonary infection, 2 cases of urinary tract infection, and 1 case of catheter-related blood stream infection. Among these patients, 6 different bacteria were cultured, including Streptococcus pneumoniae, Escherichia coli, Klebsiella pneumoniae, Acinetobacter baumannii, and Clostridium.
3.2 Correlation analysis between PCT1 and apache ii and mods scores
To observe whether PCT1 was related to the severity of disease in patients with HS, the correlation analysis between PCT1 and APACHE II and MODS scores was performed, which showed that there was no correlation between PCT1 and APACHE II score (rs=0.213, p=0.111) or MODS score (rs=0.027, p=0.844). There was also no correlation between PCT1 and hospital length of stay (rs=-0.097, p=0.472) nor ICU length of stay (rs=-0.033, p=0.809). We then divided the patients into two subgroups of CHS and EHS, analyzed the correlation between PCT1 and APACHEII and MODS scores, and the same results were obtained.
3.3 Comparison of PCT1 between the infected and non-infected groups
As shown in Table 2, there was no difference in PCT1 between the infected group and non-infected group (0.66 [0.16-5.30] vs. 0.41 [0.13-1.53], p=0.20), indicating that elevated serum PCT at ICU admission in patients with HS are not associated with bacterial infection.
3.4 Comparison of PCT1 between the chs and ehs groups
Comparison of the CHS group and EHS group showed that there was no difference in APACHE II score, MODS score, nor the highest temperature at ICU admission. However, the PCT1 of the CHS group was significantly higher than that of the EHS group (p=0.011). Of note, there were significant differences in age and comorbid disease between the CHS and EHS groups (Table 3).
3.5 Predictive value of PCT1, apache ii, and mods scores on patient prognosis
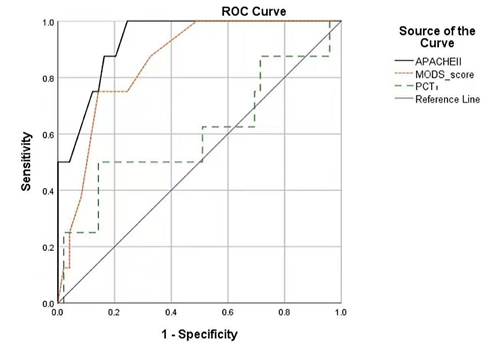
ROC curve analysis showed that both the APACHE II and MODS score had good predictive value for patients with HS (Figure 1); AUC was 0.932 (95% CI 0.861-1.000, p<0.001) and 0.857 (95% CI 0.744-0.970, p<0.001), respectively. AUC for PCT1 was 0.599 (95% CI 0.353-0.846, p=0.370), indicating that PCT at the time of ICU admission does not adequately predict the prognosis of patients with HS.
3.6 The relationship between dynamic trend of PCT and patient prognosis
We found no difference between PCT1-2 in the survivor and non-survivor groups (p=0.138). Moreover, through correlation analysis, it was found that PCT1-2 did not correlate with hospital length of stay nor ICU length of stay for patients with HS. Similar results were obtained for PCT1-3 (Table 4).
|
Parameters |
Non-Survivors (n=8) |
Survivors (n=53) |
P-value |
|
Gender |
0.270 |
||
|
Male |
4 (50) |
37 (70) |
|
|
Female |
4 (50) |
16 (30) |
|
|
Age (year) |
70.81 ± 13.47 |
62.13 ± 18.29 |
0.110 |
|
Comorbid disease |
0.462 |
||
|
Stroke |
2 (25) |
16 (30) |
|
|
Other |
1 (12) |
11 (21) |
|
|
No comorbid disease |
5 (63) |
26 (49) |
|
|
Type |
0.193 |
||
|
Classic heat stroke |
7 (87) |
34 (64) |
|
|
Exertional heat stroke |
1 (13) |
19 (36) |
|
|
APACHEII score |
23.40 ± 8.53 |
36.50 ± 4.63 |
<0.001 |
|
MODS score |
6.57 ± 3.11 |
10.38 ± 2.45 |
0.002 |
|
Tmax (?) |
40.43 ± 1.02 |
41.00 ± 0.86 |
0.141 |
|
T1 (min) |
90.00 (32.00 - 100.00) |
47.50 (30.00 - 60.00) |
0.021 |
|
HR (beats/min) |
142 (122.5 - 156.75) |
109 (93 - 135) |
0.042 |
|
SBP (mmHg) |
125.75 ± 26.68 |
77.50 ± 34.38 |
<0.001 |
|
DBP (mmHg) |
70.68 ± 18.8 |
41.13 ± 20.24 |
<0.001 |
|
Total white blood cell (×103/mm3) |
11.18 ± 5.12 |
9.47 ± 4.38 |
0.375 |
|
Neutrophil ratio (%) |
76.50 (69.00 - 81.43) |
83.70 (66.85 - 91.35) |
0.192 |
|
Platelet (X103/mm3) |
142.21 ± 60.54 |
113.25 ± 53.44 |
0.206 |
|
Hemoglobin (g/dl) |
11.81 ± 1.56 |
12.69 ± 2.77 |
0.188 |
|
Creatine kinase |
3255.00 (381.50 - 5706.25) |
935.00 (464.50 - 1856.00) |
0.257 |
|
PCT1 (ng/ml) |
3.27 (0.15 - 13.55) |
0.45 (0.13 - 2.04) |
0.369 |
|
CRP (mg/dl) |
0.85 (0.35 - 2.18) |
1.40 (0.60 - 5.10) |
0.166 |
|
Infection |
0.728 |
||
|
Yes |
4 (50) |
30 (57) |
|
|
No |
4 (50) |
23 (43) |
Data are expressed as mean ± SD, median (95% CI); Numbers in parentheses are percentages. Tmax: the highest temperature at ICU admission; T1: the time required for the temperature to fall below 38.5 °C; HR: Heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; PCT1: procalcitonin measured at ICU admission; CRP: C-reactive protein.
Table 1: Comparison between the survivor and non-survivor groups of heat stroke.
|
Indicator |
PCT1 |
P |
|
Infected group (n=32) |
0.66 (0.16 - 5.30) |
0.200 |
|
Non-infected group (n=25) |
0.41 (0.13 - 1.53) |
Table 2: PCT1 in patients with infection and non-infection.
|
Parameters |
Classic heat stroke (n=41) |
Exertional heat stroke (n=20) |
P-value |
|
Age (year) |
77 (71-84) |
61.5 (51 - 67.5) |
<0.001 |
|
Comorbid disease |
<0.001 |
||
|
Stroke |
13 (32) |
2 (10) |
|
|
Other |
18 (44) |
0 (0.00) |
|
|
No comorbid disease |
10 (24) |
18 (90) |
|
|
APACHEII score |
26.07 ± 8.86 |
23.15 ± 9.93 |
0.250 |
|
MODS score |
6.00 (5.00 - 10.00) |
7.50 (5.25 - 9.75) |
0.648 |
|
Tmax (?) |
40.50 (40.00 - 41.00) |
41.25 (39.78 - 41.93) |
0.055 |
|
T1 (min) |
50.00 (30.00 - 75.00) |
50.00 (26.25 - 68.75) |
0.753 |
|
PCT1 |
0.66 (0.16 - 5.30) |
0.41 (0.13 - 1.53) |
0.011 |
Data are expressed as mean ± SD, median (95% CI); Numbers in parentheses are percentages. Tmax: the highest temperature at ICU admission; T1: the time required for the temperature to fall below 38.5 °C; PCT1: procalcitonin measured at ICU admission.
Table 3: Comparison between classic heat stroke and exertional heat stroke.
|
Indicator |
Non-survivor group (n=8) |
Survivor group (n=53) |
P-value |
|
PCT1-2 |
23.07(-0.51~46.65) |
9.83(4.91~44.65) |
0.138 |
|
PCT1-3 |
8.26(-0.71~17.24) |
7.33(2.6~22.75) |
0.568 |
Table 4: Comparison of PCT dynamic trends between survivor group and non-survivor group.
4. Discussion
Our findings showed that PCT and its dynamic trend are poor predictors of prognosis for patients with HS, while APACHE II and MODS scores had good predictive value. Additionally, there was no difference in PCT in patients with bacterial infections. However, PCT was shown to be differ among those with CHS and EHS. HS is a life-threatening disease, and its pathophysiology has some similarities with sepsis [3]. There is increasing evidence that endotoxin and inflammatory factors may be involved in the pathogenesis, and HS causes a systemic inflammatory response that induces increased synthesis and release of acute phase proteins (such as PCT) [19, 20]. Numerous studies have shown that PCT is an effective biomarker for the diagnosis of sepsis and determinant of prognosis [21-23], but we have found that PCT is not an effective prognostic marker for patients with HS. Additionally, it is not a good indicator of whether patients with HS have a bacterial infection. The mortality rate in this study was significantly lower than published studies, which reported 40% to 60% mortality [7, 24]. The main reasons for this discrepancy are as follows: 1) The treatment of cooling was started in transit; 2) all patients were admitted to the ICU for standardized treatment; and 3) through the training of staff, the department had developed a system that conforms to the actual system and the HS diagnosis and treatment protocol. Repeated training and simulation exercises are conducted in order to make the whole staff aware and able to effectively implement it. We identified a statistically significant difference between the PCT of patients with CHS and EHS. The PCT of CHS was significantly higher than EHS, which was related to the age of CHS patients and presence of comorbid disease. Because patients with CHS present with many differences from EHS, [2] it is speculated that PCT is expressed differently in CHS and EHS, though the mechanism requires further study.The typical symptoms of HS are high fever with neurological symptoms. High fever is common in both infectious and non-infectious disease; therefore, it is necessary to determine whether patients with high fever are infected and whether they would benefit from antibiotic treatment. HS in patients is often associated with infections, which is more prevalent in older patients with CHS. Risk factors for concurrent infection with HS include changes in immune status, endotoxemia, and/or bacterial translocation due to intestinal ischemia and coma (which can cause aspiration pneumonia). In addition, infection may change the outcome of patients with HS by causing dehydration, fever, and multiple organ failure. Our study found no statistically significant difference in PCT between HS patients with and without infection. The explanation may be as follows: First, infected patients did not develop more severe systemic inflammation than non-infected patients. Second, HS itself can trigger a systemic inflammatory response that leads to elevated PCT release, which may mask the effects of infection on the induction of PCT synthesis and release. However, PCT exhibits a specific kinetic change during the inflammatory response, which begins to release into the blood 2-6 hours after the appearance of bacteria or bacterial products in the blood [25], and the infection diagnosis may occur after the elevation in PCT. PCT exhibits specific kinetic trends during systemic inflammatory response, peaking at 6-24 hours, with a half-life of about 24-35 hours, followed by a decline, which is almost unaffected by renal function [26]. Hence, dynamic monitoring of PCT trends can be an effective indicator of prognosis in patients with sepsis [27-30]. A retrospective study involving 156 sepsis patients in two ICU centers in the United States showed that PCT changes were independently associated with in-hospital mortality [31]. In this study, we analyzed the relationship between the kinetic trend of PCT and the prognosis of patients with HS, which was not mentioned in previous studies. These results indicate that there is no correlation between the PCT kinetics and the prognosis of patients with HS.
The main limitations of this study were the small sample size, uneven group sizes, and its single-center nature, meaning that the results may not be generalizable. Thus, further multi-center, prospective clinical trials are needed to confirm these results. In summary, PCT and its dynamic trend do not provide a good assessment of the prognosis of patients with HS, and PCT cannot accurately reflect severity. An increase in PCT levels at the ICU admission does not mean that a HS patient has a bacterial infection, although further large studies are needed to confirm these findings.
Declarations of interest
None
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Key Points
- Heat stroke is a life-threatening disease, but there is currently no biomarker to accurately assess prognosis.
- The pathophysiological mechanism of heat stroke is generally considered to be a hyperthermia syndrome, which is similar in many respects to sepsis.
- Procalcitonin (PCT) and its dynamic trend do not provide a good assessment of the prognosis for patients with heat stroke.
- An increase in PCT levels at ICU admission is not an indicator of bacterial infection, which should be confirmed by larger future studies.
Acknowledgments
None
Conflicts Of Interest
None
References
- People’s Liberation Army Professional Committee of Critical Care Medicine songqing3010301@ sina. com. Expert consensus on standardized diagnosis and treatment for heat stroke. Military Medical Research 3 (2016): 1-10.
- Wexler RK. Evaluation and treatment of heat-related illnesses. American family physician 65 (2002): 2307.
- Bouchama A, Knochel JP. Heat stroke. New England journal of medicine 346 (2002): 1978-1988.
- Leon LR, Bouchama A. Heat stroke. Comprehensive Physiology 5 (2011): 611-647.
- Al Mahri S, Bouchama A. Heatstroke. In Handbook of clinical neurology 157 (2018): 531-545.
- Argaud L, Ferry T, Le QH, Marfisi A, Ciorba D, Achache P, et al. Short-and long-term outcomes of heatstroke following the 2003 heat wave in Lyon, France. Archives of internal medicine 167 (2007): 2177-2183.
- Misset B, De Jonghe B, Bastuji-Garin S, Gattolliat O, Boughrara E, Annane D, et al. Mortality of patients with heatstroke admitted to intensive care units during the 2003 heat wave in France: a national multiple-center risk-factor study. Critical care medicine 34 (2006): 1087-1092.
- Assicot M, Bohuon C, Gendrel D, Raymond J, Carsin H, Guilbaud J. High serum procalcitonin concentrations in patients with sepsis and infection. The Lancet 341 (1993): 515-518.
- de Azevedo JR, Torres OJ, Beraldi RA, Ribas CA, Malafaia O. Prognostic evaluation of severe sepsis and septic shock: procalcitonin clearance vs Δ Sequential Organ Failure Assessment. Journal of critical care 30 (2015): 219.e9-e12.
- Becker KL, Snider R, Nylen ES. Procalcitonin assay in systemic inflammation, infection, and sepsis: clinical utility and limitations. Critical care medicine 36 (2008): 941-952.
- Schneider HG, Lam QT. Procalcitonin for the clinical laboratory: a review. Pathology 39 (2007): 383-390.
- Wacker C, Prkno A, Brunkhorst FM, Schlattmann P. Procalcitonin as a diagnostic marker for sepsis: a systematic review and meta-analysis. The Lancet infectious diseases 13 (2013): 426-435.
- Hensel M, Volk T, Docke WD, Kern F, Tschirna D, Egerer K, et al. Hyperprocalcitonemia in patients with noninfectious SIRS and pulmonary dysfunction associated with cardiopulmonary bypass. Anesthesiology: The Journal of the American Society of Anesthesiologists 89 (1998): 93-104.
- Mimoz O, Edouard AR, Samii K, Benoist JF, Assicot M, Bohuon C. Procalcitonin and C-reactive protein during the early posttraumatic systemic inflammatory response syndrome. Intensive care medicine 24 (1998): 185-188.
- BalcI C, Sungurtekin H, Gürses E, Sungurtekin U, Kaptanogvlu B. Usefulness of procalcitonin for diagnosis of sepsis in the intensive care unit. Critical Care 7 (2003): 85-90.
- Hausfater P, Hurtado M, Pease S, Juillien G, Lvovschi VE, Salehabadi S, et al. Is procalcitonin a marker of critical illness in heatstroke?. Intensive care medicine 34 (2008): 1377-1383.
- Nylen ES, Al Arifi A, Becker KL, Snider RH, Alzeer A. Effect of classic heatstroke on serum procalcitonin. Critical care medicine 25 (1997): 1362-1365.
- Tong HS, Liu YS, Wen Q, Tang YQ, Yuan FF, Su L. Serum procalcitonin predicting mortality in exertional heatstroke. Emergency Medicine Journal 29 (2012): 113-117.
- Bouchama AB, Parhar RS, el-Yazigi AD, Sheth KI, al-Sedairy SU. Endotoxemia and release of tumor necrosis factor and interleukin 1 alpha in acute heatstroke. Journal of Applied Physiology 70 (1991): 2640-2644.
- Lambert GP, Gisolfi CV, Berg DJ, Moseley PL, Oberley LW, Kregel KC. Selected contribution: Hyperthermia-induced intestinal permeability and the role of oxidative and nitrosative stress. Journal of applied physiology 92 (2002): 1750-1761.
- Hausfater P, Garric S, Ayed SB, Rosenheim M, Bernard M, Riou B. Usefulness of procalcitonin as a marker of systemic infection in emergency department patients: a prospective study. Clinical infectious diseases 34 (2002): 895-901.
- Hausfater P, Juillien G, Madonna-Py B, Haroche J, Bernard M, Riou B. Serum procalcitonin measurement as diagnostic and prognostic marker in febrile adult patients presenting to the emergency department. Critical care 11 (2007): R60.
- Jain S, Sinha S, Sharma SK, Samantaray JC, Aggrawal P, Vikram NK, et al. Procalcitonin as a prognostic marker for sepsis: a prospective observational study. BMC research notes 7 (2014): 458.
- Bouchama A, Dehbi M, Mohamed G, Matthies F, Shoukri M, Menne B. Prognostic factors in heat wave–related deaths: a meta-analysis. Archives of internal medicine 167 (2007): 2170-2176.
- Dandona P, Nix D, Wilson MF, Aljada A, Love J, Assicot M, et al. Procalcitonin increase after endotoxin injection in normal subjects. The Journal of Clinical Endocrinology & Metabolism 79 (1994): 1605-1608.
- Meisner M, Schmidt J, Hüttner H, Tschaikowsky K. The natural elimination rate of procalcitonin in patients with normal and impaired renal function. Intensive care medicine 26 (2000): S212-S216.
- Huang MY, Chen CY, Chien JH, Wu KH, Chang YJ, Wu KH, et al. Serum procalcitonin and procalcitonin clearance as a prognostic biomarker in patients with severe sepsis and septic shock. BioMed research international (2016).
- Lipi?ska-Gediga M, Mierzcha?a-Pasierb M, Durek G. Procalcitonin kinetics–prognostic and diagnostic significance in septic patients. Archives of medical science: AMS 12 (2016): 112.
- Liu D, Su L, Han G, Yan P, Xie L. Prognostic value of procalcitonin in adult patients with sepsis: a systematic review and meta-analysis. PloS one 10 (2015).
- Yu H, Qi Z, Hang C, Fang Y, Shao R, Li C. Evaluating the value of dynamic procalcitonin and presepsin measurements for patients with severe sepsis. The American journal of emergency medicine 35 (2017): 835-841.
- Schuetz P, Maurer P, Punjabi V, Desai A, Amin DN, Gluck E. Procalcitonin decrease over 72 hours in US critical care units predicts fatal outcome in sepsis patients. Critical Care 17 (2013): R115.


 Impact Factor: * 3.5
Impact Factor: * 3.5 Acceptance Rate: 71.36%
Acceptance Rate: 71.36%  Time to first decision: 10.4 days
Time to first decision: 10.4 days  Time from article received to acceptance: 2-3 weeks
Time from article received to acceptance: 2-3 weeks 
